Abstract
External feather rinses and homogenized whole-carcass tissue matrix from two hummingbird species found in California (Calypte anna and Archilochus alexandri) were analyzed for the presence of nine insecticides commonly used in urban settings. Using a liquid chromatography-high-resolution mass spectrometry (LC-HRMS) analytical method, samples were quantitatively tested for the following neonicotinoids: dinotefuran, nitenpyram, thiamethoxam, acetamiprid, thiacloprid, clothianidin, imidacloprid, and sulfoxaflor. This analytical method was also used to qualitatively screen for the presence of approximately 150 other pesticides, drugs, and natural products. Feather rinsates from both hummingbird species had detectable concentrations of carbamate and neonicotinoid classes of insecticides. Combined results of the rinsate and homogenized samples (n = 64 individual hummingbirds) showed that 44 individuals (68.75%) were positive for one to four target compounds. This study documented that hummingbirds found in California are exposed to insecticides. Furthermore, feather rinsates and carcass homogenates are matrices that can be used for assessing pesticide exposure in small bird species. The small body size of hummingbirds limits traditional sampling methods for tissues and whole blood to evaluate for pesticide exposure. Thus, utilization of this analytical method may facilitate future research on small-sized avian species, provide insight into pesticide exposure, and ultimately lead to improved conservation of hummingbirds.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The use of pesticides and other synthetic chemicals across the globe has increased rapidly over the last several decades and is a wide research topic across many disciplines (Bernhardt et al. 2017). Neonicotinoid insecticides were developed in the 1980s and currently includes the most widely used class of insecticides in the world (Goulson 2013). As of 2013, there were over 300 neonicotinoid products registered in California alone (Mineau and Palmer 2013). As systemic pesticides (meaning that the chemical is integrated in tissue throughout the entire plant), neonicotinoid pesticides offer long-lasting protection against insect herbivory (Tomizawa and Casida 2005). Compared to some other insecticides that neonicotinoids have replaced, such as organophosphates or carbamates, the acute toxicity of neonicotinoids to birds is relatively low (Mineau and Palmer 2013). Neonicotinoids act on insects by irreversibly binding to nicotinic acetylcholine receptors (nAChRs) located on the post-synaptic site of neuronal cells (Matsuda et al. 2001). Nicotinic acetylcholine receptors are activated by the endogenous neurotransmitter acetylcholine whereby low to moderate activation of these receptors cause nervous stimulation. Neonicotinoids, however, act as agonists and irreversibly bind at the receptor site causing overstimulation resulting in paralysis and death. Acetylcholinesterase, the enzyme which breaks down the endogenous ligand acetylcholine, cannot break down neonicotinoids.
Neonicotinoids can have effects on non-target species including bees and other invertebrates (Pisa et al. 2015) and vertebrates (Gibbons et al. 2015) in part because these chemicals are systemic and incorporated into the pollen and nectar of treated plants, where they come into contact with or are ingested by pollinating species. In neonicotinoid-treated plants, 11–24% of pollen and 17–65% of nectar is contaminated with the pesticide compound (Sanchez-Bayo and Goka 2014). For these reasons, pollinators are particularly vulnerable to neonicotinoid exposure (Hladik et al. 2018). Significant losses of bee colonies have been documented by beekeepers in many areas of the world and are an area of intense research due to the economic importance of pollination services by honeybees (Farooqui 2013). Experimental evidence suggests that exposure to neurotoxic pesticides can reduce survival and reproduction in honeybees (Farooqui 2013), induce immunosuppression in bees (Pamminger et al. 2018), and these sub-lethal effects can contribute to colony collapse disorder (Lu et al. 2014). While honeybee decline is a multi-faceted and synergistic issue involving habitat loss, pathogens, and agricultural pesticides (Goulson et al. 2015), three neonicotinoids (thiamethoxam, imidacloprid, and clothianidin) and two organophosphates (phosmet and chlorpyrifos) pose the biggest risk to honey bees overall (Sanchez-Bayo and Goka 2014).
Much research has been done on the exposure and effects of neonicotinoids on bees (Fairbrother et al. 2014) but less work has evaluated the exposure and risks of these compounds on other pollinating species overall (Hladik et al. 2018), especially vertebrate pollinators. However, there has been a distinct increase in papers published on the ecological and environmental effects of neonicotinoids overall (Giorio et al. 2017). Pollination is a valuable ecosystem service that is in decline on a global scale (Potts et al. 2010). In bats (ecologically important both as pollinators and insectivores), pesticide exposure is a possible contributor to population declines and susceptibility to white-nose syndrome, but long-term data is lacking (Bayat et al. 2014). Recent analyses suggest that the use of neonicotinoid pesticides is associated with declines in bird populations on a continental scale (Goulson 2014), and insectivorous birds are thought to be impacted the most through a pesticide-mediated reduction in food supply (Hallmann et al. 2014). Thus, neonicotinoid pesticides appear to impact non-target species at exposure concentrations well below the amount that would induce acute toxic effects such as death (Goulson 2014).
In addition to the effects of direct mortality, sub-lethal toxic effects of pesticides can affect the survival and reproduction of birds (Fry 1995; Stanton et al. 2018). Attempts have been made to include pesticide exposure into models of avian survival and reproduction, but are often limited by the availability of direct controlled toxicological studies (Bennett et al. 2007; Etterson and Bennett 2013). Compared to some other insecticides that neonicotinoids have replaced, such as organophosphates or carbamates, the acute toxicity of neonicotinoids to birds is relatively low (Mineau et al. 2001). However, sub-lethal effects documented in birds include debilitation such as ataxia which can be induced in birds given imidacloprid orally at an order of magnitude below the lethal dose (Callahan and Mineau 2008). Migratory ability was shown to be impaired in white-crowned sparrows given 10% of the LD50 for imidacloprid (Eng et al. 2017). Thus, establishing if birds are exposed is a crucial first step in evaluating the impacts of these chemicals on the survival and reproduction of wild birds.
Small birds are generally more sensitive to toxic chemicals than larger birds (Mineau et al. 1996), thus hummingbirds may offer great potential for monitoring bird exposure to pollutants. Since the diet of hummingbirds includes both insects and nectar from many sources, hummingbirds could be used as sentinel species for assessing environmental exposure to pollutants, including pesticides (Godoy et al. 2014). Given that bees and hummingbirds often forage in the same areas (Pritchard et al. 2017), it makes sense that these two ecologically similar groups of species may be exposed to the same suites of pollutants. However, testing for pesticide exposure is a challenge for individual small-bodied animals due to the constraints of the minimum sample mass required for many existing analytical methods. One gram or more of a single tissue type (i.e., liver) is commonly necessary for toxicological assays (Berny et al. 1999), which makes it difficult to use currently established analytical methods on samples from very small-bodied individual animals. North American hummingbirds generally weigh less than 4–5 g, and thus require alternative methods to evaluate individual chemical exposure.
The objective of this study was to evaluate external carcass feather rinsates and composite tissue samples from hummingbirds found in California for exposure to nine insecticides commonly used in urban environments using a previously established liquid chromatography-high-resolution mass spectrometry (LC-HRMS) assay method (Filigenzi et al. 2019).
Methods
Carcass sampling
Anna’s (ANHU; n = 21 Northern California; n = 22 Southern California) and Black-chinned (BCHU; n = 21) Hummingbirds that did not survive rehabilitation efforts were used in this study. Bird carcasses were obtained from wildlife care and rehabilitation centers in California. No birds were killed for this study. We investigated these two hummingbird species found in California because one species is migratory (Black-chinned Hummingbird, Archilochus alexandri) and the other species has both migratory and resident populations (Anna’s Hummingbird, Calypte anna). Black-chinned and some Anna’s Hummingbirds are known to migrate outside the USA (Phillips 1975) where pesticide regulations are different, so they may be exposed to a different suite of chemicals than are resident Anna’s Hummingbirds during their annual cycle.
Feather rinsate sample preparation
For the Northern California ANHU and BCHU, the intact carcasses were placed in a 50-mL plastic tube and enough acetonitrile was added to cover (approximately 40 mL) the sample. The tube was shaken for 10 min. The acetonitrile was then transferred to a glass tube, evaporated dry under nitrogen, and reconstituted for analysis in 200 μL of 20% methanol in water. For the Southern California ANHU, the carcasses were externally washed with a dilute solution of Alconox® detergent. The detergent solution used for the rinsates was not compatible with the existing liquid chromatography-high resolution mass spectrometry (LC-HRMS) analytical method and therefore these rinsates were not analyzed.
Homogenized tissue sample preparation
After the feather rinsate process, the same hummingbird carcasses were analyzed using a previously described LC-HRMS method (Filigenzi et al. 2019). After rinsing, carcasses were air dried. Individual carcasses were frozen in liquid nitrogen and ground in a Stein mill. One gram of the ground carcass was transferred to a 50-mL plastic tube and a mixture of isotopically labelled neonicotinoid pesticide internal standards was added. Then, 5 mL of water was added and the tube was vortexed to mix the contents. Acetonitrile (15 mL) was added and the tube was shaken for 5 min. The tube was centrifuged for 5 min and the supernatant decanted into a 50-mL tube containing magnesium sulfate and ammonium acetate. The tube was shaken by hand and then centrifuged. The acetonitrile layer was transferred to a tube containing magnesium sulfate and primary-secondary amine, shaken, and centrifuged. The solvent was then decanted into a glass tube, evaporated dry, and reconstituted for analysis in 200 μL of 20% methanol in water.
Analysis of feather rinsates and homogenized tissue samples
Feather rinsate and homogenized tissue extracts were analyzed by LC-HRMS using a Dionex Ultimate 3000 ultrahigh pressure liquid chromatograph interfaced with a Thermo Q-Exactive high-resolution mass spectrometer. The column used was a 100 × 2.1 mm and 1.7 μm Eclipse Plus C-18 from Agilent Corporation. The mass spectrometer was run in positive ion mode using electrospray ionization. The resolution was set at 70,000 (M/∆M at m/z 200). Neonicotinoid insecticides (dinotefuran, nitenpyram, thiamethoxam, acetamiprid, thiacloprid, clothianidin, imidacloprid, and sulfoxaflor) were analyzed in high-resolution MS/MS mode to provide enhanced specificity and quantitative data in carcass samples. While sulfoxaflor contains a sulfoximine group to make it chemically distinguishable from neonicotinoids (Zhu et al. 2011), sulfoxaflor acts on the same nicotinic acetylcholine receptors as neonicotinoids (Cutler et al. 2012). Thus, we included sulfoxaflor in our list of target neonicotinoids. Quantitation was performed on carcass samples using the isotopically labelled internal standards added prior to extraction (note that rinsate analysis was performed on a qualitative basis only). Five-point calibration curves ranging in concentration from 0.50 to 100 ng/g were used to establish instrument response and consistently showed r2 values > 0.99. The limits of detection and quantification for each analyte are listed in Supplemental Table 1.
Full-scan high-resolution mass spectrometry (MS) data from these analyses was used to screen for a list of approximately 150 other pesticides, drugs, and natural products including some carbamate and organophosphate insecticides (Supplemental Table 2). Laboratory-raised 1–2-day-old chicken carcasses were used for quality control analyses, including negative control samples and fortified control samples.
Data analysis
Due to the limited number of samples with quantifiable neonicotinoid concentrations, any sample with pesticide concentrations greater than the limit of detection (LOD) was deemed positive and those with concentrations below the LOD were deemed negative. An individual bird was considered positive for compound exposure if either the feather rinsate, homogenized tissue sample, or both were positive. Data were analyzed to determine exposure prevalence using generalized linear mixed models (GLMM) in R (version 3.4.3, R Core Team 2017) and odds ratios in Microsoft Excel (version 15.32, 2017). A p value of < 0.05 was considered significant. Kappa coefficients (Cohen 1960) were calculated in R (KappaGUI package) to determine the level of agreement between rinsate and homogenized samples.
Results
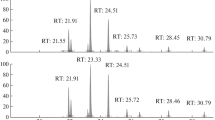
A total of 42 ANHU and 42 BCHU samples (homogenized carcass or rinsate) from 64 individual hummingbirds were successfully analyzed for the following neonicotinoids: dinotefuran, nitenpyram, thiamethoxam, acetamiprid, thiacloprid, clothianidin, imidacloprid, and sulfoxaflor. In addition, all samples were successfully screened for a variety of other compounds (Supplemental Table 2). Out of 84 total samples, only 3 had quantifiable concentrations above the LOD. One adult male ANHU carcass from Southern California contained a quantifiable level of imidacloprid (3.2 μg/kg). Two BCHU carcasses contained quantifiable levels of acetamiprid (3.1 and 3.4 μg/kg; adult male from Northern California and juvenile male from an unknown region, respectively). Of the 64 individual hummingbirds, combined results of the rinsate and homogenized samples showed that 44 individuals (68.75%) were positive for between one and four target compounds, while 20 individuals (31.25%) were negative for any target compounds.
Across all 64 individual hummingbirds screened, 42.2% (n = 27) tested positive for imidacloprid, 32.8% (n = 22) tested positive for carbaryl, 17.2% (n = 11) tested positive for acetamiprid, 4.7% (n = 3) tested positive for dinotefuran, 3.1% (n = 2) tested positive for thiacloprid, 3.1% (n = 2) tested positive for caffeine, 1.6% (n = 1) tested positive for thiamethoxam, and 1.6% (n = 1) tested positive for malathion (Tables 1 and 2). Imidacloprid, thiacloprid, acetamiprid, carbaryl, and caffeine were detected in the homogenized tissue samples. Imidacloprid, thiamethoxam, acetamiprid, carbaryl, malathion, and dinotefuran were detected in the feather rinsate samples. Nitenpyram, sulfoxaflor, and clothianidin were not detected in any hummingbird samples.
Agreement between homogenized carcass and rinsate samples in compound detections was found to have a kappa value of 0.38 for all compounds combined. Rinsate samples had more detections (n = 49) overall than did homogenized carcass samples (n = 30), although we were unable to screen for metabolites of target compounds which may have affected this observed pattern.
For compound prevalence and odds ratio calculations, ANHU were used as the referent category for species, females were used as the referent category for sex, hatch-year birds were used as the referent category for age, and northern California was used as the referent category for region.
Results of GLMMs are summarized in Table 3, where most odds ratio calculations did not reach the p < 0.05 threshold of statistical significance. However, some significant patterns were found for carbaryl, imidacloprid, and acetamiprid. BCHU were 0.23 times less likely to be positive for carbaryl (p = 0.036) and 0.20 times less likely to be positive for imidacloprid (p = 0.012) compared to ANHU. ANHU from the Southern California region were 0.26 times less likely to be exposed to imidacloprid (p = 0.018) compared to ANHU from the Northern California region. Adult birds were 0.14 times less likely to be exposed to acetamiprid (p = 0.006) compared to juvenile birds.
Discussion
This study showed that a sensitive analytical method can be used for evaluating feather rinsate and homogenized tissue samples for presence of insecticides in free-ranging hummingbirds. Imidacloprid, acetamiprid, and thiacloprid insecticides were detected in homogenized carcass samples while carbaryl, malathion, dinotefuran, imidacloprid, thiamethoxam, and acetamiprid were detected in feather rinsates from Anna’s and Black-chinned Hummingbirds found in California. This analytical method will also be useful for future studies as it provides a means for detecting carbamates, organophosphates, and neonicotinoids in animals with low body mass. Given the small sample mass and low pesticide concentrations, qualitative data predominated our current findings although three samples did contain quantifiable concentrations of target compounds (imidacloprid and acetamiprid).
To date, most studies evaluating insecticide exposure of pollinators have focused on bees. While direct insecticide residue data for free-ranging honeybees are sparse, compounds have been detected in honeybee tissue samples (and other bee products such as honey and pollen). A study in France found that 44.3% of honeybee samples contained between one and five insecticide compounds (of 42 screened for, including carbaryl) with concentrations ranging from 0.4 to 1545.6 μg/kg (Chauzat et al. 2011). Another study of declining bee populations in Greece detected imidacloprid in 60% of bee samples at concentrations of 14–39 ng/g of tissue, while other target insecticides were not detected (Bacandritsos et al. 2010).
Our results show that two avian pollinator species found in California (ANHU and BCHU), are exposed to a suite of insecticides. Moreover, greater numbers of insecticide compounds were detected in the external feather rinsate samples (n = 6 compounds) per bird than in the homogenized carcass samples (n = 5 compounds). Different compounds were also detected in different sample types; thiacloprid and caffeine were only detected in homogenized carcass samples, while thiamethoxam, malathion, and dinotefuran were only detected in feather rinsate samples, and imidacloprid, acetamiprid, and carbaryl were detected in both sample types. It is intriguing that more insecticides were detected externally, given that it has been previously reported that the greatest pesticide residue concentrations are generally found in the digestive tract contents of birds, at least in cases of acute poisoning (Mineau and Tucker 2002). The high number of external pesticide detections relative to internal detections overall raises the possibility that hummingbirds are exposed to pesticides through a mechanism other than dietary intake. However, due to a lack of commercial standards for the metabolites, our analytical method only evaluated for parent compounds. If analytical standards for the metabolites were available, we may have detected a greater number of positive samples and/or quantifiable concentrations of target compounds.
One challenge with this study was the external rinsate method for feather samples. After multiple rinses, it was unclear if the rinsate represented surface contamination or insecticides that had been incorporated into the feather during development and extracted during the feather rinsing process. Therefore, with the Southern California ANHU, the feather rinsate method was switched to detergent washing. However, the detergent-based rinsate was incompatible with the organic solvent used for the extraction method. In the future, it would be advantageous to further develop and validate optimal methods for feather rinsates.
Since hummingbirds are a highly mobile group of species with varying migration strategies, we cannot know where or how this exposure is occurring. However, hummingbirds may be exposed to these chemicals through dietary intake of contaminated flower nectar (Bishop et al. 2018) and/or more general exposure through contaminated air or water sources (Giorio et al. 2017). Some pesticides that are not approved by the EPA for use in the USA are approved for legal use in Mexico (US General Accounting Office 1992), thus exposing migratory birds (i.e., BCHU) to a different suite of chemicals than USA resident populations of birds (i.e., some ANHU). Public records of pesticide usage show that malathion is used commercially in the county where the hummingbird positive for malathion was obtained (Contra Costa County 2015). In addition, as these insecticides may degrade over time (Rouchaud et al. 1994) combined with the lack of analytical standards for metabolite compounds, our results likely underestimate total exposure in California hummingbirds.
The qualitative detection of neonicotinoids and other pesticide classes from avian tissue is a necessary first step for improving wildlife toxicology assays for new anthropogenic chemicals, which are often lacking in the rapidly changing world of chemical registration (Mineau and Tucker 2002). Even though only three carcasses had quantifiable concentrations of insecticide parent compounds (ranging from 3.1 to 3.4 μg/kg), our study establishes a method for future assessment of insecticide exposure in wild birds. For comparison of quantifiable concentrations of insecticides in free-ranging birds, a recent study of wild Eurasian Eagle Owls found one individual positive for imidacloprid at a concentration of 3.28 ng/mL in blood (Taliansky-Chamudis et al. 2017). A recent study of hummingbirds in an agricultural area in Canada found a combined concentration of imidacloprid, thiamethoxam, and clothianidin to be 3.63 ppb in pooled samples of cloacal fluid from many individuals (Bishop et al. 2018). Similar to our findings in hummingbirds, this demonstrates the relatively low concentrations that seem to dominate the results of wild bird exposure studies that are not cases of acute poisoning. Given the growing interest in the effects of pesticides on pollinators, which are declining on a global scale (Potts et al. 2010), toxicology studies on both pollinating and non-pollinating bird species are likely to be of increasing interest.
The analytical method applied here will be key to future work investigating the health and/or conservation impacts of chronic low dose pesticide exposure on wild birds. In addition, our studies provide first reported evidence that hummingbirds found in California are exposed to insecticides even though the location of exposure is unknown. This finding provides a glimpse into a necessary body of work that will examine the role hummingbirds play as a potential sentinel species for environmental contaminants. A similar sample preparation procedure combined with LC-low-resolution MS has been used for the analysis of pesticides in bees (Bargańska et al. 2014; García et al. 2018) so the method applied to the hummingbirds should also be applicable for LC-HRMS analysis of insects and other small bodied species.
Future research should attempt to resolve the observed differences in compound detection between homogenized carcass and feather rinsate samples. Assessing these differences of internal versus external insecticide exposure in wild hummingbirds in more detail will be important in determining the origin of exposure and to mitigate these practices. Future availability of metabolite compound analytical standards could also improve detection accuracy. Ultimately, the goal would be to understand the routes of exposure and establish best practices to protect hummingbird populations.
References
Bacandritsos N, Granato A, Budge G, Papanastasiou I, Roinioti E, Caldon M, Falcaro C, Gallina A, Mutinelli F (2010) Sudden deaths and colony population decline in Greek honey bee colonies. J Invertebr Pathol 105(3):335–340. https://doi.org/10.1016/j.jip.2010.08.004
Bargańska Ż, Ślebioda M, Namieśnik J (2014) Determination of pesticide residues in honeybees using modified QUEChERS sample work-up and liquid chromatography-tandem mass spectrometry. Molecules 19:2911–2924. https://doi.org/10.3390/molecules19032911
Bayat S, Geiser F, Kristiansen P, Wilson SC (2014) Organic contaminants in bats: trends and new issues. Environ Int 63:40–52
Bennett RS, Etterson MA, Bennett RS, Etterson MA (2007) Incorporating results of avian toxicity tests into a model of annual reproductive success. Integr Environ Assess Manag 3(4):498–507. https://doi.org/10.1897/IEAM
Bernhardt ES, Rosi EJ, Gessner MO (2017) Synthetic chemicals as agents of global change. Front Ecol Environ 15(2):84–90. https://doi.org/10.1002/fee.1450
Berny PJ, Buronfosse F, Videmann B, Buronfosse T (1999) Evaluation of the toxicity of imidacloprid in wild birds: a new high performance thin layer chromatography (HPTLC) method for the analysis of liver and crop samples in suspected poisoning cases. J Liq Chromatogr Relat Technol 22(10):1547–1559. https://doi.org/10.1081/JLC-100101750
Bishop CA, Moran AJ, Toshack MC, Elle E, Maisonneuve F, Elliott J (2018) Hummingbirds and bumble bees exposed to neonicotinoid and organophosphate insecticides in the Fraser Valley, British Columbia, Canada. Environ Toxicol Chem 37(8):2143–2152
Callahan J, Mineau P (2008) An evaluation of clinical sign data from avian acute oral toxicity studies. Appendix 11; Scientific opinion of the panel on plant protection products and their residues on risk assessment for birds and mammals. EFSA J 734:1–10
Chauzat M-P, Martel AC, Cougoule N, Porta P, Lachaize J, Zeggane S, Aubert M, Carpentier P, Faucon JP (2011) An assessment of honeybee colony matrices, Apis mellifera (Hymenoptera: Apidae) to monitor pesticide presence in continental France. Environ Toxicol 30(1):103–111. https://doi.org/10.1002/etc.361
Cohen J (1960) A coefficient of agreement for nominal scales. Educ Psychol Meas 20(1):37–46
Contra Costa County (2015) Pesticide use reports. http://www.co.contra-costa.ca.us/6243/Pesticide-Use-Data
Cutler P, Slater R, Edmunds AJF, Maienfisch P, Hall RG, Earley FGP, Pitterna T, Pal S, Paul V, Goodchild J, Blacker M, Hagmann L, Crossthwaite AJ (2012) Investigating the mode of action of sulfoxaflor: a fourth-generation neonicotinoid. Pest Manag Sci 69:607–619
Eng ML, Stutchbury BJM, Morrissey CA (2017) Imidacloprid and chlorpyrifoc insecticides impair migratory ability in a seed-eating songbird. Sci Rep 7:15176. https://doi.org/10.1038/s41598-017-15446-x
Etterson MA, Bennett RS (2013) Quantifying the effects of pesticide exposure on annual reproductive success of birds. Integr Environ Assess Manag 9(4):590–599. https://doi.org/10.1002/ieam.1450
Fairbrother A, Purdy J, Anderson T, Fell R (2014) Risks of neonicotinoid insecticides to honeybees. Environ Toxicol Chem 33(4):719–731. https://doi.org/10.1002/etc.2527
Farooqui T (2013) A potential link among biogenic amines-based pesticides, learning and memory, and colony collapse disorder: a unique hypothesis. Neurochem Int 62(1):122–136. https://doi.org/10.1016/j.neuint.2012.09.020
Filigenzi M, Graves E, Tell L, Jelks K, Poppenga R (2019) Quantitative analysis for neonicotinoid insecticides combined with broad range screening for other xenobiotics in small mass avian tissue samples using UHPLC-high resolution mass spectrometry. J Vet Diagn Investig. https://doi.org/10.1177/1040638719834329
Fry DM (1995) Reproductive effects in birds exposed to pesticides and industrial chemicals. Environ Health Perspect 19:165–171
García GMD, Martínez Galera M, Uclés S, Lozano A, Fernández-Alba AR (2018) Ultrasound-assisted extraction based on QuEChERS of pesticide residues in honeybees and determination by LC-MS/MS and GC-MS/MS. Anal Bioanal Chem 410(21):5195–5210. https://doi.org/10.1007/s00216-018-1167-7
Gibbons D, Morrissey C, Mineau P (2015) A review of the direct and indirect effects of neonicotinoids and fipronil on vertebrate wildlife. Environ Sci Pollut Res 22:103–118
Giorio C, Safer A, Sánchez-Bayo F, Tapparo A, Lentola A, Girolami V, van Lexmond MB, Bonmatin J (2017) An update of the Worldwide Integrated Assessment (WIA) on systemic insecticides. Part 1: new molecules, metabolism, fate, and transport. Environ Sci Pollut Res https://doi.org/10.1007/s11356-017-0394-3
Godoy LA, Tell LA, Ernest HB (2014) Hummingbird health: pathogens and disease conditions in the family Trochilidae. J Ornithol 155(1):1–12. https://doi.org/10.1007/s10336-013-0990-z
Goulson D (2013) An overview of the environmental risks posed by neonicotinoid insecticides. J Appl Ecol 50(4):977–987. https://doi.org/10.1111/1365-2664.12111
Goulson D (2014) Pesticides linked to bird declines. Nature 511:295–296. https://doi.org/10.1038/nature13642
Goulson D, Nicholls E, Botías C, Rotheray EL (2015) Bee declines driven by combined stress from parasites, pesticides, and lack of flowers. Science 347(6229):1255957. https://doi.org/10.1126/science.1255957
Hallmann CA, Foppen RPB, Turnhout C a M, Van Kroon H, De Jongejans E, van Turnhout C a M et al (2014) Declines in insectivorous birds are associated with high neonicotinoid concentrations. Nature 511(7509):341–343. https://doi.org/10.1038/nature13531
Hladik ML, Main AR, Goulson D (2018) Environmental risks and challenges associated with neonicotinoid insecticides. Environ Sci Technol 52:3329–3335
Lu C, Warchol KM, Callahan RA (2014) Sub-lethal exposure to neonicotinoids impaired honey bees winterization before proceeding to colony collapse disorder. Bull Insectol 67(1):125–130
Matsuda K, Buckingham SD, Kleier D, Rauh JJ, Grauso M, Sattelle DB (2001) Neonicotinoids: insecticides acting on insect nicotinic acetylcholine receptors. Trends Pharmacol Sci 22(11):573–580. https://doi.org/10.1016/S0165-6147(00)01820-4
Mineau P, Palmer C (2013) The impact of the nation’s most widely used insecticides on birds. The American Bird Conservancy. http://abcbirds.org/wp-content/uploads/2015/05/Neonic_FINAL.pdf
Mineau P, Tucker KR (2002) Improving detection of pesticide poisoning in birds. J Wildlife Rehabil 25(2):4–13
Mineau P, Collins BT, Baril A (1996) On the use of scaling factors to improve interspecies extrapolation of acute toxicity in birds. Regul Toxicol Pharmacol 24(1):24–29. https://doi.org/10.1006/rtph.1996.0061
Mineau P, Baril A, Collins BT, Duffe J, Joerman G, Luttik R (2001) Pesticide acute toxicity reference values for birds. Rev Environ Contam Toxicol 170:13–74
Pamminger T, Botías C, Goulson D, Hughes WOH (2018) A mechanistic framework to explain the immunosuppressive effects of neurotoxic pesticides on bees. Funct Ecol 32(8):1921–1930. https://doi.org/10.1111/1365-2435.13119
Phillips AR (1975) The migrations of Allen’s and other hummingbirds. Condor 77(2):196–205
Pisa LW, Amaral-Rogers V, Belzunces LP, Bonmatin JM, Downs CA, Goulson D, Kreutzweiser DP, Krupke C, Liess M, McField M, Morrissey CA, Noome DA, Simon-Delso J, Stark JD, Van der Sluijs JP, Van Dyck H, Wiemers M (2015) Effects of neonicotinoids and fipronil on non-target invertebrate. Environ Sci Pollut Res 22:68–102
Potts SG, Biesmeijer JC, Kremen C, Neumann P, Schweiger O, Kunin WE (2010) Global pollinator declines: trends, impacts and drivers. Trends Ecol Evol 25(6):345–353
Pritchard DJ, Tello Ramos MC, Muth F, Healy SD (2017) Treating hummingbirds as feathered bees: a case of ethological cross-pollination. Biol Lett 13:20170610. https://doi.org/10.1098/rsbl.2017.0610
R Core Team (2017) A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna. http://www.R-project.org/
Rouchaud J, Gustin F, Wauters A (1994) Soil biodegradation and leaf transfer of insecticide imidacloprid applied in seed dressing in sugar beet crops. Bull Environ Contam Toxicol 53:344–350
Sanchez-Bayo F, Goka K (2014) Pesticide residues and bees—a risk assessment. PLoS One 9(4):e94482. https://doi.org/10.1371/journal.pone.0094482
Stanton RL, Morrissey CA, Clark RG (2018) Analysis of trends and agricultural drivers of farmland bird declines in North America: a review. Agric Ecosyst Environ 254:244–254
Taliansky-Chamudis A, Gómez-Ramírez P, León-Ortega M, García-Fernández AJ (2017) Validation of a QuECheRS method for analysis of neonicotinoids in small volumes of blood and assessment of exposure in Eurasian eagle owl (Bubo bubo) nestlings. Sci Total Environ 595:93–100. https://doi.org/10.1016/j.scitotenv.2017.03.246
Tomizawa M, Casida JE (2005) Neonicotinoid insecticide toxicology: mechanisms of selective action. Annu Rev Pharmacol Toxicol 45:247–268. https://doi.org/10.1146/annurev.pharmtox.45.120403.095930
U.S. General Accounting Office (1992) Comparison of U.S. and Mexican pesticide standards and enforcement, pp 58. http://www.Gao.Gov/Assets/160/151984.Pdf
Zhu Y, Loso MR, Watson GB, Sparks TC, Rogers RB, Huang JX, Gerwick BC, Babcock JM, Kelley D, Hegde VB, Nugent BM, Renga JM, Denholm I, Gorman K, DeBoer GJ, Hasler J, Meade T, Thomas JD (2011) Discovery and characterization of sulfoxaflor, a novel insecticide targeting sap-feeding pests. J Agric Food Chem 59:2950–2957
Acknowledgements
The authors would like to thank the Lindsey Wildlife Museum and the California Wildlife Center for providing hummingbird carcasses for this study. This work is dedicated to Dr. Richard Melnicoe for his passion for birding and his expertise in integrated pest management.
Funding
This study was funded by the Western Hummingbird Partnership and the Daniel and Susan Gottlieb Foundation.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Roland Peter Kallenborn
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Graves, E.E., Jelks, K.A., Foley, J.E. et al. Analysis of insecticide exposure in California hummingbirds using liquid chromatography-mass spectrometry. Environ Sci Pollut Res 26, 15458–15466 (2019). https://doi.org/10.1007/s11356-019-04903-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-019-04903-x