Abstract
Many studies have investigated associations between maternal residential exposures to air pollutants and low birth weight (LBW) in offspring. However, most studies focused on the criteria air pollutants (PM2.5, PM10, O3, NO2, SO2, CO, and Pb), and only a few studies examined the potential impact of other air pollutants on LBW. This study investigated associations between maternal residential exposure to industrial air emissions of 449 toxics release inventory (TRI) chemicals and LBW in offspring using a case-control study design based on a large dataset consisting of 94,106 LBW cases and 376,424 controls in Texas from 1996 to 2008. Maternal residential exposure to chemicals was estimated using a modified version of the emission-weighted proximity model (EWPM). The model takes into account reported quantities of annual air emission from industrial facilities and the distances between the locations of industrial facilities and maternal residence locations. Binary logistic regression was used to compute odds ratios measuring the association between maternal exposure to different TRI chemicals and LBW in offspring. Odds ratios were adjusted for child’s sex, birth year, gestational length, maternal age, education, race/ethnicity, and public health region of maternal residence. Among the ten chemicals selected for a complete analysis, maternal residential exposures to five TRI chemicals were positively associated with LBW in offspring. These five chemicals include acetamide (adjusted odds ratio [aOR] 2.29, 95% confidence interval [CI] 1.24, 4.20), p-phenylenediamine (aOR 1.63, 95% CI 1.18, 2.25), 2,2-dichloro-1,1,1-trifluoroethane (aOR 1.41, 95% CI 1.20, 1.66), tributyltin methacrylate (aOR 1.20, 95% CI 1.06, 1.36), and 1,1,1-trichloroethane (aOR 1.11, 95% CI 1.03, 1.20). These findings suggest that maternal residential proximity to industrial air emissions of some chemicals during pregnancy may be associated with LBW in offspring.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Ambient air pollution (AAP), also known as outdoor air pollution, is ubiquitous (Polichetti et al. 2013) and has become a global public health problem (Brunekreef and Holgate 2002; Kampa and Castanas 2008). It has been well documented that exposure to AAP was associated with a number of adverse health outcomes, such as respiratory and cardiovascular diseases (Brook et al. 2004; Dominici et al. 2006; Polichetti et al. 2009), and even mortality (Hoek et al. 2013). Because developing fetuses are more vulnerable than children and adults, many studies have investigated possible adverse influences of AAP on birth outcomes since the mid-1990s (Li et al. 2017; Ritz and Wilhelm 2008; Shah et al. 2011; Srám et al. 2005), including low birth weight (LBW, newborn weighted less than 2500 g or 5.5 lb at birth), small for gestational age (SGA), preterm birth, and birth defects.
Because LBW infants may experience higher risk of mortality and/or morbidity in childhood (McCormick 1985; McIntire et al. 1999; Watkins et al. 2016), and are more susceptible for other diseases than normal-weight infants, including stroke (Lawlor et al. 2005), coronary heart disease, hypertension, type II diabetes (Whincup et al. 2008), delayed motor/social development (Hediger et al. 2002), and learning disabilities (Litt et al. 2005), LBW is considered as an important predictor of an infant’s health. Statistics suggested that 2.6% of live term singleton births were LBW births in the United States (U.S.) during 2000–2015 (U.S. CDC 2018).
Pollutants in the air can be divided into two categories: (1) six criteria air pollutants (CAPs) designated by the Clean Air Act and subsequent amendments (including particulate matter (PM2.5, PM10), ozone (O3), nitrogen dioxide (NO2), sulfur dioxide (SO2), carbon monoxide (CO), and lead (Pb)) (U.S. EPA 2017) and CAPs-related air pollutants, and (2) other air pollutants (non-criteria air pollutants). Based on publication records related to birth weight from the Web of Science database covering years 1904–2017 (Table 1), most studies reported in the literature focused on CAPs rather than non-criteria air pollutants (Wilhelm et al. 2012).
The associations between maternal exposure to the six CAPs and LBW have been investigated in a large number of studies (430 studies in total as of the end of 2017). For example, Salam et al. (2005) found that O3 exposure during the second and third trimesters and CO exposure during the first trimester were associated with increased LBW levels; Xu et al. (2011) discovered that exposure to PM10 during pregnancy might increase the risk of LBW. Ebisu and Bell (2012) identified associations between PM2.5 components and LBW in the northeastern and mid-Atlantic regions of the U.S. Dedele et al. (2017) showed that increased maternal exposure to NO2 tended to increase LBW risk in offspring. Around 12% of these studies have also examined components or combination of the CAPs (CAPs-related air pollutants), including total suspended particle (TSP) (Bobak 2000; Lee et al. 2002; Wang et al. 1997), black carbon (Brauer and Lencar 2008; Paciorek 2010), black smoke (Pearce et al. 2012; Stankovic et al. 2011), among others.
However, the examination of association between maternal exposure to non-criteria air pollutants and LBW in offspring has been reported in a limited number of studies (only 40 studies as of the end of 2017). Among these studies, 9 of them investigated benzene, toluene, ethyl benzene, and xylene (BTEX) and 11 of them studied polycyclic aromatic hydrocarbons (PAHs). For example, airborne benzene exposure was found to be associated with increasing odds of a LBW event (Slama et al. 2009; Zahran et al. 2012). Jedrychowski et al. (2017) found that exposure to PAH was inversely associated with birth weight. Aguilera et al. (2009) linked an increase in BTEX exposure levels to reductions in birth weight for women who spent < 2 h/day in nonresidential outdoor environments. However, other non-criteria pollutants were hardly mentioned.
One possible explanation for the unbalanced number of studies on the two categories of air pollutants was the lack of high-quality air monitoring data covering non-criteria air pollutants. The extensive monitoring network of CAPs with finer spatial-temporal sampling resolution has significantly facilitated the estimation of exposure to CAPs (Gong et al. 2016). Although 18 non-criteria air pollutants have been studied (Table 1), these pollutants only accounted for a limited portion of the total non-criteria pollutants released into the air. Based on information from the databases maintained by the toxic release inventory (TRI) program of the U.S. Environmental Protection Agency (U.S. EPA), there have been over 650 non-criteria pollutants from industrial facilities in the U.S. and 449 in Texas (U.S. EPA 2013). It is hypothesized that some of these pollutants (TRI chemicals) could possibly exert a negative impact on birth weight. Therefore, this study attempts to identify associations between maternal residential exposure to TRI chemicals during pregnancy and LBW in offspring using a case-control study design based on the analysis of massive georeferenced data over a 13-year period in Texas from 1996 to 2008.
Study area, data, and methods
Study area
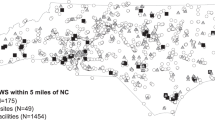
This study chose the state of Texas in the U.S. as the study area (Fig. 1). Among the 48 states in the contiguous U.S., Texas is the largest by area and second largest by population. Two datasets in Texas were used, including air emission data from industrial facilities and birth data.
Air emission data of industrial facilities
This study obtained air emission data of Texas industrial facilities from the U.S. EPA TRI program. The TRI program, a mandatory program established by Section 313 of the Emergency Planning and Community Right-to-Know Act (EPCRA), requires industrial facilities in the U.S. to report annual information about their names, locations, reporting years, types of chemicals released, and estimated quantity of a chemical released into the environment (U.S. EPA 2013). Based on reports during 1996–2008, 1286 to 1635 Texas industrial facilities reported air emissions to TRI program each year. Industrial facility addresses were geocoded and maintained at Texas State University (Zhan et al. 2015). During 1996–2008, 89.66% of the TRI facilities were successfully geocoded on average (Fig. 1). A TRI air emission geodatabase was constructed for the purpose of this study, containing both geographic locations of TRI facilities (Fig. 1) and non-spatial data summarizing the annual air emission amount of the 449 TRI chemicals released from industrial facilities in Texas during 1996–2008.
Birth data
This study obtained birth certificate data for all registered births during 1996–2008 in Texas from the Center for Health Statistics in the Texas Department of State Health Services (DSHS). Each birth certificate record contained information of maternal residential address at delivery; birth weight; birth year; plurality; child’s sex; gestational age in weeks; mother’s characteristics (age, race/ethnicity, education, marital status, and tobacco use during pregnancy); and father’s characteristics (age, race/ethnicity, and education). This study excluded births of weight > 5500 g or < 1000 g (0.1%), births with gestational age > 44 weeks or < 37 weeks (17.8%), births with incomplete location information (10.9%), plural deliveries (2.7%), or births given by non-Texas residents or occurred outside of Texas (0.2%). Only births with weight < 2500 g were considered as the LBW cases and used in this study. This study matched LBW cases and controls by year of birth (1996–2008) and selected four controls for each case to ensure sufficient study power. Control births from the same birth year were randomly selected from the birth certificate data. The maternal addresses of all cases and controls were geocoded to their exact geographic locations (latitudes and longitudes) by the Texas DSHS.
Air pollution exposure assessment
A modified version of the emission-weighted proximity model (EWPM) (Gong et al. 2016; Zou et al. 2009b) was used to estimate exposure intensities to a given TRI chemical at the location of a maternal residence for each case and control. This modified version of the EWPM model takes into account both the distances between the location of a maternal residence and industrial facilities within an effective distance as well as the rate and duration of a specific chemical released into the air by each facility annually. The formula for the EWPM model is given by Expression (1) below.
where Aθi is the estimated exposure intensity to chemical θ of a person at residence location i, represented by the quantity of chemical θ reaching location i from all emission sources (j, j = 1, 2, … …, m) within an effective distance (kθ); Eθij and Tθij are the emission rate and duration of emission of chemical θ from emission source j that is within the effective distance (kθ) of location i; Dij is the distance between location i and emission source j; kθ is the effective distance beyond which chemical θ is considered to have no harm on an individual (Gong et al. 2016; Zou et al. 2009b). This study used an effective distance (kθ) of 10 km.
Identification of the most likely potential risk factors
This study used odds ratios (ORs) from a logistic regression to assess the associations between exposure to different TRI chemicals and LBW in offspring first, and then identified chemicals with the highest positive statistically significant ORs as the most likely risk factors associated with LBW. For each TRI chemical, cases and controls were divided into two groups: exposed (cases and controls with estimated exposure intensity value to a given chemical greater than zero) or unexposed (cases and controls with estimated exposure intensity value to a given chemical equal to zero). The binary logistic regression analysis used the unexposed group as the reference group. The logistic regression analysis was adjusted for several covariates, including child’s sex, gestational weeks (37–44 weeks), maternal age (11–19, 20–24, 25–29, 30–34, 35–39, > 39 years), education (< high school, high school, > high school), race/ethnicity (non-Hispanic white, non-Hispanic black, Hispanic, other non-Hispanic), the public health service region of maternal residence (11 regions in Texas as 11 categories), and year of birth (1996–2008 as 13 categories). We selected these covariates for the analyses based on recommendations in the literature (Bell et al. 2007; Brender et al. 2014; Valero De Bernabé et al. 2004). In the next phase of the analysis, this study used values of adjusted odds ratios (aORs) to rank the TRI chemicals. Ten chemicals with the highest positive statistically significant aORs from the 449 TRI chemicals were selected for additional epidemiological analysis.
Additional epidemiological analysis
For each of the ten identified TRI chemicals, this study categorized the estimated exposure intensity values for all cases and controls into four levels: one unexposed group and three exposed groups. The cases and controls in the unexposed group were those with zero estimated exposure intensity for a given chemical. The ones with estimated exposure intensity value greater than zero were divided into three intervals in such way that each interval contained approximately the same number of controls. In epidemiology, it is common to categorize exposure levels based on the distribution of exposure levels in controls because controls are thought to most closely represent the underlying population. The four levels of exposure are called zero, low, medium, and high exposure in the rest of this article. In the next step, binary logistic regressions were used to estimate the associations (aOR and 95% confidence interval (CI)) between maternal residential exposure to these ten chemicals and LBW in offspring. The unexposed groups were considered as the reference groups in the analyses. The aORs were adjusted for the same covariates listed in the previous section. In addition, a multiple comparison correction was conducted to evaluate the results from the regression analysis.
Results
There were a total of 94,106 term LBW cases and 376,424 controls that were frequency-matched to cases by year of birth in this study. Table 2 compares the LBW cases and controls by child’s sex, mother’s age at delivery, mother’s race/ethnicity, gestational length, year of birth, mother’s education, and public health service region of maternal residence at the time of delivery. In this study population, LBW cases were more likely to be female and have shorter gestational length than controls. The mothers of the LBW cases were also more likely to be non-Hispanic black, younger at age of delivery, or less educated when compared to the control-mothers.
Estimated exposure intensities to the 449 TRI chemicals were calculated at the maternal residence location of each LBW case and control. To protect confidentiality, we use maps showing the estimated exposure densities at the locations of the centroids of census tracts, instead of maternal residential locations, to illustrate the geographic distribution of exposure intensity to a specific chemical. We used a four-step procedure to produce these maps (Fig. 2). First, centroids of the 4388 census tracts in Texas in 2000 were considered as hypothetical point receptors of air pollution exposure. Second, we used EWPM to estimate exposure intensities to a given chemical at the locations of these 4388 centroids. Third, the estimated exposure intensity at the centroid of a specific census tract was used to represent the exposure intensity of the whole census tract, assuming that the exposure distribution in the census tract is homogenous. Fourth, exposure intensities in the 4388 census tracts were categorized into seven levels (exposure intensity at zero and greater than zero divided into six equal-interval groups).
Figure 2 shows an example distribution of estimated exposure intensities to chemical 1,1,1-trichloroethane (chemical abstracts service (CAS) number 71556) among census tracts in Texas during 1996–2008. The highest exposure intensity level was observed in northeastern Texas (Dallas-Fort Worth area) in 1996. However, the exposure intensity level of this area decreased every year after 1996 and reached zero in year 2001. The southeastern Texas (Houston area) had a mid-level exposure intensity consistently during the 13 years. In 1996 and 1998, two areas in central Texas also showed mid-level exposure intensities.
Table 3 shows 78 TRI chemicals with statistically significant aORs greater than 1 when exposure intensities were dichotomized into exposed and unexposed groups. Compared with the unexposed reference groups, the LBW risks in exposed groups of the 78 chemicals increased by 2 to 60% (aOR 1.02–1.60). Then, 10 chemicals with the largest positive aORs were chosen from the 78 chemicals as the potential risk factors for LBW in additional epidemiology analysis. The ten chemicals were acetamide (aOR 1.60, 95% CI 1.09, 2.34), p-phenylenediamine (aOR 1.32, 95% CI 1.07,1.63), 2,2-dichloro-1,1,1-trifluoroethane (aOR 1.21, 95% CI 1.10, 1.34), 1,2-phenylenediamine (aOR 1.20, 95% CI 1.02, 1.41), resmethrin (aOR 1.14, 95% CI 1.01, 1.30), toluene-2,6-diisocyanate (aOR 1.14, 95% CI 1.02, 1.28), tributyltin methacrylate (aOR 1.14, 95% CI 1.05, 1.23), propetamphos (aOR 1.11, 95% CI 1.01, 1.23), 1,1,1-trichloroethane (aOR 1.10, 95% CI 1.05, 1.15), and creosote (aOR 1.09, 95% CI 1.02, 1.16).
Table 4 shows the results from the additional analyses of the above ten identified chemicals, where estimated exposure intensities were divided into four levels (zero, low, medium, and high exposure). Without multiple comparison correction, seven of the ten chemicals had statistically significant aORs larger than 1.0 in at least one of the three exposure levels. These seven chemicals are acetamide, p-phenylenediamine, 2,2-dichloro-1,1,1-trifluoroethane, resmethrin, toluene-2,6-diisocyanate, tributyltin methacrylate, and 1,1,1-trichloroethane. Five chemicals survived a multiple comparisons correction using the Benjamini-Hochberg procedure with false discovery rate (FDR) at the level of 0.05. These five chemicals are acetamide, p-phenylenediamine, 2,2-dichloro-1,1,1-trifluoroethane, tributyltin methacrylate, and 1,1,1-trichloroethane.
The largest aOR value was noted in the case of low exposure to acetamide (aOR 2.29, 95% CI 1.24, 4.20), indicating that mothers with low exposure to acetamide had a 129% higher chance of delivering LBW babies when compared with mothers who had no exposure to air emissions of acetamide from industrial facilities. Chemical 1,1,1-trichloroethane showed statistically significant aORs among mothers within two exposure levels (low and medium). Monotonically increasing trends were noted between maternal residential exposure to tributyltin methacrylate and LBW in offspring (highest aOR in high exposure: 1.20, 95% CI 1.06, 1.36). Statistically significant association was also observed in medium exposure to p-phenylenediamine and 2,2-dichloro-1,1,1-trifluoroethane (Table 4).
Discussions
Adverse health outcomes of exposure to the ten chemicals in Table 4 (acetamide, p-phenylenediamine, 2,2-dichloro-1,1,1-trifluoroethane, 1,2-phenylenediamine, resmethrin, toluene 2,6-diisocyanate, tributyltin methacrylate, propetamphos, 1,1,1-trichloroethane, and creosote) have been documented in various studies reported in the literature. Inhalation of tributyltin methacrylate and propetamphos might cause serve injury or death (U.S. NOAA 2016). Short-term exposure to the other eight chemicals might cause irritation to the eyes, skin, mucous membranes, and respiratory tract, while long-term exposure to these chemicals was reported to yield other adverse health effects, including skin sensitization, asthma, narcosis, cardiac disorders, anemia, dermatitis, and hyperpigmentation of skin (U.S. CDC 2015). Long-term exposure to p-phenylenediamine and 2,2-dichloro-1,1,1-trifluoroethane might also have effects on kidney and liver respectively (U.S. CDC 2015). Chemical 1,1,1-trichloroethane, also known as methyl chloroform, was reported to be associated with neural tube defects in a study conducted in Texas investigating the associations between chlorinated solvent exposures and birth defect in offspring (Brender et al. 2014). However, no study has examined the associations between maternal residential exposure to these chemicals and LBW in offspring. The identified association between LBW and the five TRI chemicals in this study created new opportunities for further epidemiological, biological, and toxicological research. It is important to note that some other significant pollutants from this exploratory analysis might also be important risk factors for LBW, despite that they did not survive multiple testing correction reported in this paper.
As shown in Table 4, with the increase of maternal residential exposure intensities to acetamide, p-phenylenediamine, 2,2-dichloro-1,1,1-trifluoroethane, and 1,1,1-trichloroethane, the risk of having LBW newborns did not necessarily increase monotonically in that the highest aORs were not noted in mothers who were categorized in the level of high exposure. Brender et al. (2014) also found similar trends in their studies examining associations between maternal chlorinated solvent exposures and birth defect in Texas. The non-monotonic trends may be due to the fact that data used in this study contained live births only. One possible explanation is that higher maternal residential exposure intensities to certain chemicals could have severely affected weight gain in the fetus during some pregnancies and resulted in induced terminations or fetal deaths. However, data about these pregnancies were not available in the present study. Therefore, the groups categorized as high exposure in this study might have odds ratios biased toward null.
The emission-weighted proximity model (EWPM) was used in this study to estimate maternal residential exposure to the TRI chemicals during pregnancy. Compared to other exposure assessment methods, the EWPM is simpler to implement and more cost-effective when many air pollutants are considered (Gong et al. 2016). Different types and magnitudes of exposure misclassifications are expected in different exposure assessment models, the EWPM might also introduce some degree of exposure misclassifications (Zou et al. 2009a). To assess the performance of EWPM in estimating exposure intensities, a study was conducted to examine how the EWPM estimated exposure intensities correlated with air monitoring data of 27 non-criteria air pollutants at 48 monitoring sites in Texas in 2005 (Gong et al. 2016). The study concluded that the EWPM is a valid and useful approach for exposure assessment when analyses involve data covering a large geographic area over multiple years (Gong et al. 2016). This study used a consistent effective distance (kθ = 10 km) for all chemicals, meaning exposure to a given chemical was only considered to be affected by sources within 10 km from the location of interest. However, the best effective distance (kθ) for each chemical may be related to chemical properties, meteorological conditions, and terrain surrounding the location of interest. Therefore, future studies should consider finding the best effective distance for a specific chemical before using the EWPM model in order to improve the accuracy of the estimated exposure.
Air emission data used in this study had several limitations. First, because only annual-level TRI air emission data were available, this study could only estimate annual exposure to the TRI chemicals. It was not possible to estimated exposure variations at finer temporal scales to reflect daily, monthly, and seasonal variations. Second, industrial facilities self-reported their annual air emission quantities to the TRI program, which may lead to some uncertainties in the data. Third, only industrial air emissions from point sources (stacks of TRI industrial facilities) were used for exposure assessment in this study, future studies should consider integrating more emission source types (linear, areal, and mobile sources) for more accurate measures. Fourth, based on the observation that a small portion of the study population resided in areas near the Texas state boundaries to other states or Mexico (8.92% (n = 41,957) within 10 km of the border, and 6.67% (n = 31,392) within 10–50 km of the border), we performed analyses to examine how the removal of the cases and controls within 10 km of the Texas state boundaries may affect the results. Among the five significant chemicals reported in Table 4, only acetamide did not survive the multiple comparison correction after those cases and controls were removed. The results are summarized in a table in the supplementary materials (Table S-1). The results suggest that the “edge effect” of geographic boundaries does affect the results to some degree, and it should be considered when interpreting the results of the type of analysis similar to the ones reported in this article.
Maternal residential addresses at delivery were used in this study to estimate exposure during pregnancy, assuming that each maternal residence was the same from conception to delivery. Therefore, exposure misclassifications might exist if there were maternal residential movements during pregnancy in the study population (Canfield et al. 2006; Lupo et al. 2010). To overcome this limitation, data with maternal residential history are needed to more accurately estimate and categorize exposure. For a large-scale study based on birth registry data such as the one reported in this paper, data with detailed residential history were simply not available. It is worth noting that the impact might be limited because the change of residences during pregnancy tends to involve only short distances in most cases (Lupo et al. 2010). In addition, there has been a number of studies suggesting an association between maternal food restriction (MFR) with reduced birth weight (Godfrey et al. 1996). It would be ideal to include data about MFR into the analyses of this study, but again data about MFR were simply not available for this large-scale study. We used maternal education level as a covariate in the analysis. Because education level is a good indicator for socioeconomic status, we hope this covariate will partly take care of this limitation.
Conclusion
This large population-based, case-control study examined whether maternal residential proximity to some of the 449 TRI air pollutants could be potential risk factors associated with LBW in offspring. This number of 449 chemicals far exceeded the number of non-criteria air pollutants examined by studies reported in the literature. Maternal residential exposure to the TRI chemicals during pregnancy was estimated using the EWPM model. The model takes into account reported quantities of annual air emission from industrial facilities and the distances between the locations of industrial facilities and maternal residence locations. This study concluded that maternal residential proximity to industrial air emissions of some TRI chemicals during pregnancy may be associated with LBW in offspring. These chemicals included acetamide, p-phenylenediamine, 2,2-dichloro-1,1,1-trifluoroethane, tributyltin methacrylate, and 1,1,1-trichloroethane. For most of these five pollutants, the exposure-response function does not seem to be monotonically increasing when the estimated exposure is categorized into low, medium, and high levels.
Abbreviations
- AAP:
-
ambient air pollution
- aOR:
-
adjusted odds ratio
- CAP:
-
criteria air pollutant
- CDC:
-
Centers for Disease Control and Prevention
- CI:
-
confidence interval
- DSHS:
-
Texas Department of State Health Services
- EWPM:
-
emission weighted proximity model
- LBW:
-
low birth weight
- NOAA:
-
National Oceanic and Atmospheric Administration
- OR:
-
odds ratio
- TRI:
-
toxic release inventory
- U.S. EPA:
-
United States Environmental Protection Agency
References
Aguilera I, Guxens M, Garcia-Esteban R, Corbella T, Nieuwenhuijsen MJ, Foradada CM, Sunyer J (2009) Association between GIS-based exposure to urban air pollution during pregnancy and birth weight in the INMA Sabadell cohort. Environ Health Perspect 117(8):1322–1327
Balsa AI, Caffera M, Bloomfield J (2016) Exposures to particulate matter from the eruptions of the puyehue volcano and birth outcomes in Montevideo, Uruguay. Environ Health Perspect 124(11):1816–1822
Bell ML, Ebisu K, Belanger K (2007) Ambient air pollution and low birth weight in Connecticut and Massachusetts. Environ Health Perspect 115(7):1118–1124
Berkowitz Z, Price-Green P, Bove FJ, Kaye WE (2006) Lead exposure and birth outcomes in five communities in Shoshone County, Idaho. J Hyg Environ Health 209(2):123–132
Bobak M (2000) Outdoor air pollution, low birth weight, and prematurity. Environ Health Perspect 108(2):173–176
Bobak M, Richards M, Wadsworth M (2001) Air pollution and birth weight in Britain in 1946. Epidemiology 12(3):358–359
Brauer M, Lencar C (2008) A cohort study of traffic-related air pollution impacts on birth outcomes. Environ Health Perspect 116(5):680–686
Brender JD, Shinde MU, Zhan FB, Gong X, Langlois PH (2014) Maternal residential proximity to chlorinated solvent emissions and birth defects in offspring: a case–control study. Environ Health 13(1):96
Brook RD, Franklin B, Cascio W, Hong Y, Howard G, Lipsett M, Luepker R, Mittleman M, Samet J, Smith SC, Tager I (2004) Air pollution and cardiovascular disease: a statement for healthcare professionals from the expert panel on population and prevention science of the American Heart Association. Circulation 109(21):2655–2671
Brunekreef B, Holgate ST (2002) Air pollution and health. LANCET 360(9341):1233–1242
Canfield MA, Ramadhani TA, Langlois PH, Waller DK (2006) Residential mobility patterns and exposure misclassification in epidemiologic studies of birth defects. J Expo Sci Environ Epidemiol 2006(16):538–543
Chen L, Yang W, Jennison BL, Goodrich A, Omaye ST (2002) Air pollution and birth weight in northern Nevada, 1991-1999. Inhal Toxicol 14(2):141–157
Cho S, Lee C-K, Kim B (2013) The impacts of air pollution on low birth weight. Appl Econ Lett 20(3):208–212
Coker E, Liverani S, Ghosh JK, Jerrett M, Beckerman B, Li A, Ritz B, Molitor J (2016) Multi-pollutant exposure profiles associated with term low birth weight in Los Angeles County. Environ Int 91:1–13
Currie J, Schmieder J (2009) Fetal exposure to toxic releases and infant health. Am Econ Rev: Pap Proc Annu Meet Am Econ Assoc 99(2):177–183
Darrow LA, Klein M, Strickland MJ, Mulholland JA, Tolbert PE (2011) Ambient air pollution and birth weight in full-term infants in Atlanta, 1994-2004. Environ Health Perspect 119(5):731–737
Dedele A, Grazuleviciene R, Miskinyte A (2017) Individual exposure to nitrogen dioxide and adverse pregnancy outcomes in Kaunas study. Int J Environ Health Res 27(3):230–240
Díaz J, Arroyo V, Ortiz C, Carmona R, Linares C (2016) Effect of environmental factors on low weight in non-premature births: a time series analysis. PLoS One 11(10):1–14
Dimitriev D, Dimitriev A, Konstantinova Y (2006) Association between ambient air pollution and birth weight in Novocheboksarsk, Russia. Epidemiology 17(6):S105
Dominici F, Peng RD, Bell ML, Pham L, McDermott A, Zeger SL, Samet JM (2006) Fine particulate air pollution and hospital admission for cardiovascular and respiratory diseases. J Am Med Assoc 295(10):1127–1134
Ebisu K, Bell ML (2012) Airborne PM2.5 chemical components and low birth weight in the northeastern and mid-Atlantic regions of the United States. Environ Health Perspect 120(12):1746–1752
Estarlich M, Ballester F, Aguilera I, Fernandez-Somoano A, Lertxundi A, Llop S, Freire C, Tardon A, Basterrechea M, Sunyer J, Iniguez C (2011) Residential exposure to outdoor air pollution during pregnancy and anthropometric measures at birth in a multicenter cohort in Spain. Environ Health Perspect 119(9):1333–1338
Geer LA, Weedon J, Bell ML (2012) Ambient air pollution and term birth weight in Texas from 1998 to 2004. J Air Waste Manage Assoc 62(11):1285–1295
Gehring U, Wijga A, Fischer P (2011) Traffic-related air pollution, preterm birth and term birth weight in the PIAMA birth cohort study. Environ Res 111(1):125–135
Ghosh JKC, Wilhelm M, Su J, Goldberg D, Cockburn M, Jerrett M, Ritz B (2012) Assessing the influence of traffic-related air pollution on risk of term low birth weight on the basis of land-use-based regression models and measures of air toxics. Am J Epidemiol 175(12):1262–1274
Gladen BC, Zadorozhnaja TD, Chislovska N, Hryhorczuk DO, Kennicutt MC, Little RE (2000) Polycyclic aromatic hydrocarbons in placenta. Hum Exp Toxicol 19(11):597–603
Godfrey K, Robinson S, Barker D, Osmond C, Cox V (1996) Maternal nutrition in early and late pregnancy in relation to placental and fetal growth. BMJ 312:410–414
Gong X, Brender JD, Langlois PH, Lin Y, Zhan FB (2016) Validity of the emission weighted proximity model in estimating air pollution exposure intensities in large geographic areas. Sci Total Environ 563–564(2016):478–485
Govarts E, Remy S, Bruckers L, Den Hond E, Sioen I, Nelen V, Baeyens W, Nawrot TS, Loots I, Van Larebeke N, Schoeters G (2016) Combined effects of prenatal exposures to environmental chemicals on birth weight. Int J Environ Res Public Health 13(5):1–19
Ha S, Zhu Y, Liu D, Sherman S, Mendola P (2017) Ambient temperature and air quality in relation to small for gestational age and term low birthweight. Environ Res 155:394–400
Harrath AH, Alwasel S, Khaled I, Mansouri L, Sirotkin AV, Faramawi MF (2015) Pregnancy exposure to atmospheric pollutants and placental weight in a Tunisian population. Fresenius Environ Bull 24(11C):4184–4189
Hediger ML, Overpeck MD, Ruan WJ, Troendle JF (2002) Birthweight and gestational age effects on motor and social development. Paediatr Perinat Epidemiol 16(1):33–46
Hoek G, Krishnan RM, Beelen R, Peters A, Ostro B, Brunekreef B, Kaufman JD (2013) Long-term air pollution exposure and cardio-respiratory mortality: a review. Environ Health 12(1):43
Hyder A, Lee HJ, Ebisu K, Koutrakis P, Belanger K, Bell ML (2014) PM2.5 exposure and birth outcomes use of satellite- and monitor-based data. Epidemiology 25(1):58–67
Jacobs M, Zhang G, Chen S, Mullins B, Bell M, Jin L, Guo Y, Huxley R, Pereira G (2017) The association between ambient air pollution and selected adverse pregnancy outcomes in China: a systematic review. Sci Total Environ 579:1179–1192
Janghorbani M, Piraei E (2013) Association between air pollution and preterm birth among neonates born in Isfahan, Iran. J Res Med Sci 18(10):875–881
Jedrychowski WA, Majewska R, Spengler JD, Camann D, Roen EL, Perera FP (2017) Prenatal exposure to fine particles and polycyclic aromatic hydrocarbons and birth outcomes: a two-pollutant approach. Int Arch Occup Environ Health 90(3):255–264
Kampa M, Castanas E (2008) Human health effects of air pollution. Environ Pollut 151(2):362–367
Laurent O, Hu J, Li L, Cockburn M, Escobedo L, Kleeman MJ, Wul J (2014) Sources and contents of air pollution affecting term low birth weight in Los Angeles County, California, 2001-2008. Environ Res 134:488–495
Lawlor DA, Ronalds G, Clark H, Davey Smith G, Leon DA (2005) Birth weight is inversely associated with incident coronary heart disease and stroke among individuals born in the 1950s: findings from the Aberdeen children of the 1950s prospective cohort study. Circulation 112(10):1414–1418
Lee BE, Park HS, Kim YJ, Hong YC, Kim H, Lee JT, Ha EH (2002) The effect of air pollution on low birth weight by gestational period in Seoul. Epidemiology 13(4):1–8
Li X, Huang S, Jiao A, Yang X, Yun J, Wang Y, Xue X, Chu Y, Liu F, Liu Y, Ren M, Chen X, Li N, Lu Y, Mao Z, Tian L, Xiang H (2017) Association between ambient fine particulate matter and preterm birth or term low birth weight: an updated systematic review and meta-analysis. Environ Pollut 227:596–605
Litt J, Taylor HG, Klein N, Hack M (2005) Learning disabilities in children with very low birthweight: prevalence, neuropsychological correlates, and educational interventions. J Learn Disabil 38(2):130–141
Lupo PJ, Symanski E, Chan W, Mitchell LE, Waller DK, Canfield MA, Langlois PH (2010) Differences in exposure assignment between conception and delivery: the impact of maternal mobility. Paediatr Perinat Epidemiol 24(2):200–208
Malmqvist E, Liew Z, Källén K, Rignell-Hydbom A, Rittner R, Rylander L, Ritz B (2017) Fetal growth and air pollution—a study on ultrasound and birth measures. Environ Res 152:73–80
McCormick MC (1985) The contribution of low birth weight to infant mortality and childhood morbidity. N Engl J Med 312(2):82–90
McIntire DD, Bloom SL, Casey BM, Leveno KJ (1999) Birth weight in relation to morbidity and mortality among newborn infants. N Engl J Med 340(16):1234–1238
Merklinger-Gruchala A, Jasienska G, Kapiszewska M (2017) Parity conditions the risk for low birth weight after maternal exposure to air pollution. Biodemography Soc Biol 63(1):71–86
Nieuwenhuijsen MJ, Dadvand P, Grellier J, Martinez D, Vrijheid M (2013) Environmental risk factors of pregnancy outcomes: a summary of recent meta-analyses of epidemiological studies. Environ Health 12(6):1–10
Paciorek CJ (2010) The importance of scale for spatial-confounding bias and precision of spatial regression estimators. Stat Sci 25(1):107–125
Pearce MS, Glinianaia SV, Ghosh R, Rankin J, Rushton S, Charlton M, Parker L, Pless-Mulloli T (2012) Particulate matter exposure during pregnancy is associated with birth weight, but not gestational age, 1962-1992: a cohort study. Environ Health 11(13):1–8
Perera FP, Rauh V, Tsai WY, Kinney P, Camann D, Barr D, Bernert T, Garfinkel R, Tu YH, Diaz D, Dietrich J, Whyatt RM (2003) Effects of transplacental exposure to environmental pollutants on birth outcomes in a multiethnic population. Environ Health Perspect 111(2):201–205
Polichetti G, Cocco S, Spinali A, Trimarco V, Nunziata A (2009) Effects of particulate matter (PM10, PM2.5and PM1) on the cardiovascular system. Toxicology 261(1–2):1–8
Polichetti G, Capone D, Grigoropoulos K, Tarantino G, Nunziata A, Gentile A (2013) Effects of ambient air pollution on birth outcomes: an overview. Crit Rev Environ Sci Technol 43(12):1223–1245
Quansah R, Jaakkola JJK (2009) Paternal and maternal exposure to welding fumes and metal dusts or fumes and adverse pregnancy outcomes. Int Arch Occup Environ Health 82(4):529–537
Ritz B, Wilhelm M (2008) Ambient air pollution and adverse birth outcomes: methodologic issues in an emerging field. Basic Clin Pharmacol Toxicol 102(2):182–190
Salam M, Millstein J, Li Y (2005) Birth outcomes and prenatal exposure to ozone, carbon monoxide, and particulate matter: results from the Children’s health study. Environ Health Perspect 113(11):1138–1644
Shah PS, Balkhair T, Knowledge synthesis group on determinants of preterm/LBW births (2011) Air pollution and birth outcomes: a systematic review. Environ Int 37(2):498–516
Siddiqui AR, Gold EB, Yang X, Lee K, Brown KH, Bhutta ZA (2008) Prenatal exposure to wood fuel smoke and low birth weight. Environ Health Perspect 116(4):543–549
Slama R, Thiebaugeorges O, Goua V, Aussel L, Sacco P, Bohet A, Forhan A, Ducot B, Annesi-Maesano I, Heinrich J, Magnin G, Schweitzer M, Kaminski M, Charles M-A, EDEN Mother-Child Cohort Study Group (2009) Maternal personal exposure to airborne benzene and intrauterine growth. Environ Health Perspect 117(8):1313–1321
Srám RJ, Binková BB, Dejmek J, Bobak M (2005) Ambient air pollution and pregnancy outcomes: a review of the literature. Environ Health Perspect 113(4):375–382
Stankovic A, Mitrovic V, Zivadinovic R (2011) Influence of air pollution on birth weight. Srp Arh Celok Lek 139(9–10):651–656
U.S. CDC (United States Centers for Disease Control and Prevention) 2015 International Chemical Safety Cards (ICSC). Available: https://www.cdc.gov/niosh/ipcs/default.html. last Accessed Nov 2017
U.S. CDC (United States Centers for Disease Control and Prevention) 2018 National environmental public health tracking network data explorer. https://ephtracking.cdc.gov/DataExplorer/#/. last Accessed May 2018
U.S. EPA (U.S. Environmental Protection Agency) 2013 Learn about the Toxics Release Inventory. http://www2.epa.gov/toxics-release-inventory-tri-program/learn-about-toxics-release-inventory. last Accessed Sept 2017
U.S. EPA (U.S. Environmental Protection Agency) 2017 Six Common Air Pollutants. http://www3.epa.gov/airquality/urbanair. updated 2017; last Accessed Sept 2017
U.S. NOAA (U.S. National Oceanic and Atmospheric Administration) 2016 CAMEO Chemicals, Database of Hazardous Materials. Available: https://cameochemicals.noaa.gov/. last Accessed Nov 2017
Valero De Bernabé J, Soriano T, Albaladejo R, Juarranz M, Calle ME, Martínez D, Domínguez-Rojas V (2004) Risk factors for low birth weight: a review. Eur J Obstet Gynecol Reprod Biol 116(1):3–15
Vassilev ZP, Robson MG, Klotz JB (2001a) Associations of polycyclic organic matter in outdoor air with decreased birth weight: a pilot cross-sectional analysis. J Toxic Environ Health A 64(8):595–605
Vassilev ZP, Robson MG, Klotz JB (2001b) Outdoor exposure to airborne polycyclic organic matter and adverse reproductive outcomes: a pilot study. Am J Ind Med 40(3):255–262
Veleminsky M, Hanzl M, Sram RJ (2016) The impact of air pollution in the southern bohemia region on fetuses and newborns. Neuroendocrinol Lett 37(2):52–57
Wang XB, Ding H, Ryan L, Xu XP (1997) Association between air pollution and low birth weight: a community-based study. Environ Health Perspect 105(5):514–520
Watkins WJ, Kotecha SJ, Kotecha S (2016) All-cause mortality of low birthweight infants in infancy, childhood, and adolescence: population study of England and Wales. PLoS Med 13(5):1–19
Whincup P, Kaye S, Owen C, Huxley R, Cook D, Anazawa S, Barrett-Connor E, Bhargava S, Birgisdottir B, Carlsson S, Rooij D, Dyck R, Eriksson J, Falkner B, Fall C, Forsén T, Grill V, Gudnason V, Hulman S, Hyppönen E, Jeffreys M, Lawlor D, Leon D, Minami J, Mishra G, Osmond C, Power C, Rich-Edwards J, Roseboom T, Sachdev H, Syddall H, Thorsdottir I, Vanhala M, Wadsworth M, Yarbrough D (2008) Birth weight and risk of type 2 diabetes: a systematic review. J Am Med Assoc 300(24):2886–2897
Wilhelm M, Ghosh JK, Su J, Cockburn M, Jerrett M, Ritz B (2012) Traffic-related air toxics and term low birth weight in Los Angeles County, California. Environ Health Perspect 120(1):132–138
Williams BL, Pennock-Roman M, Suen HK, Magsumbol MS, Ozdenerol E (2007) Assessing the impact of the local environment on birth outcomes: a case for HLM. Journal of Exposure Science and Environmental Epidemiology 17(5):445–457
Xu X, Sharma RK, Talbott EO, Zborowski JV, Rager J, Arena VC, Volz CD (2011) PM10 air pollution exposure during pregnancy and term low birth weight in Allegheny County, PA, 1994-2000. Int Arch Occup Environ Health 84(3):251–257
Zahran S, Weiler S, Mielke HW, Pena AA (2012) Maternal benzene exposure and low birth weight risk in the United States: a natural experiment in gasoline reformulation. Environ Res 112:139–146
Zhan FB, Brender JD, Langlois PH, Yang J 2015 Air Pollution-Exposure-Health Effect Indicators: Mining massive geographically referenced environmental health data to identify risk factors for birth defects. Final report (2011–2015; 325 pages), U.S. Environmental Protection Agency
Zou B, Wilson JG, Zhan FB, Zeng Y (2009a) Air pollution exposure assessment methods utilized in epidemiological studies. J Environ Monit 11(3):475–490
Zou B, Wilson JG, Zhan FB, Zeng Y (2009b) An emission-weighted proximity model for air pollution exposure assessment. Sci Total Environ 407(17):4939–4945
Acknowledgements
The research reported in this article was made possible in part by a U.S. EPA-STAR grant (#R834790). The data used in the analyses were obtained from the Center for Health Statistics in the Texas Department of State Health Services (DSHS) and United States Environmental Protection Agency (U.S. EPA). The contents are solely the responsibility of the authors and do not necessarily represent the official views of the U.S. EPA and the Texas DSHS. Furthermore, U.S. EPA, the Texas DSHS, and the authors do not endorse the purchase of any commercial products or services mentioned in this article. The authors wish to thank the support from the U.S. EPA and the Texas DSHS. Xi Gong and F. Benjamin Zhan appreciated the helpful discussions with Drs. Jean D. Brender and Peter H. Langlois about the case-control study design reported in the article. The authors greatly appreciate the helpful comments and suggestions from the editor and three anonymous reviewers.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interest
The authors declare that they have no competing interest.
Additional information
Responsible editor: Philippe Garrigues
Electronic supplementary material
ESM 1
(DOCX 54 kb)
Rights and permissions
About this article
Cite this article
Gong, X., Lin, Y. & Zhan, F.B. Industrial air pollution and low birth weight: a case-control study in Texas, USA. Environ Sci Pollut Res 25, 30375–30389 (2018). https://doi.org/10.1007/s11356-018-2941-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-018-2941-y