Abstract
Pot and greenhouse trials were conducted for the management of root-knot nematode, Meloidogyne javanica, infestation in tomato. Growth parameters, gall index, soil, and root nematode populations were measured to assess the effect of a novel bio-pesticide (Dazitol®), made from mustard oil and oleoresin of Capsicum, on plant growth and nematode reproduction. Data generated within the pot experiment showed that the tested bio-pesticide did not improve plant growth, but it reduced significantly root-knot nematode damage resulting in a decrease in gall index and root (91%) and soil (62%) population of M. javanica compared with untreated plants. The greenhouse experiment showed that Mocap® and Dazitol® decreased nematode incidence significantly (P < 0.05) on tomato. The result of this study suggested that the best nematode control was obtained by combining soil solarization with chemical or botanical nematicides as an integrated pest management approach.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Root-knot nematodes (RKN), Meloidogyne spp., are the most economically important plant-pathogenic nematodes affecting vegetable crops in all regions of the world and cause both considerable losses in crop production and quality (Sikora and Fernandez 2005; Moens et al. 2009). The yield losses caused by the nematode to tomato in Tunisia ranged from 12 to 60% (Horrigue-Raouani 2003). Although tomato yield suppression caused by root-knot nematodes could reach up to 100% (Seid et al. 2015).
Effective management practices of root-knot nematodes have been and still are the objective of several studies and chemical nematicides are widely used for the control of plant-parasitic nematodes (Talavera et al. 2012; Mekete et al. 2015). Additionally, the efficiency of chemical nematicides is for a limited period only besides being expensive and environmentally hazardous (Abd-Elgawad 2008).
Biofumigation, a sustainable management method considered as an alternative to methyl bromide, was reported efficient in controlling several pests including nematodes (Salem 2014). Biofumigation through the toxic volatile isothiocyanates (ITCs), the released biocidal compounds from brassicaceous plants, suppresses various soil-borne pathogens and plant diseases (Thoden et al. 2011).
Botanicals and plant extracts are perceived by the public as natural products safer than synthetic chemicals and thus have been proposed as tools for integrated RKN management because they provide nematode control as well as reduced risks of pesticide residues, thereby boosting production in sustainable agriculture (Caboni and Ntalli 2014).
Soil solarization, a non-chemical and soil thermal disinfection method for controlling diseases, nematodes, and weeds, was developed as thermic disinfection through mulching soil in hot seasons (Katan et al. 1976). This management tool was also successful in controlling plant-parasitic nematodes (Stapleton and Heald 1991).
Currently, the trend is to use control measures alternative to chemical pesticides. The global focus on sustainability in the agricultural environment is increasing in order to produce healthy, safe and good-quality food. This focus includes the implementation of integrated pest management (IPM). The most sustainable approach to root-knot nematodes control will integrate several tools and strategies, including reasonable use of nematicides, crop rotation, resistant cultivars, and soil solarization (Stoddard et al. 2010).
The combination of two or more management methods are encouraged in an IPM context. The use of biofumigation, botanicals, and solarization are particularly interesting for organic agriculture. The current investigation focused on determining the effect of a mixture of botanicals, Dazitol®, made from mustard and pepper extract plus soil solarization to control root-knot nematodes on tomato under pot and greenhouse conditions.
Materials and methods
Nematode inoculum
Egg masses of a monoxenic population of Meloidogyne javanica extracted from tomato galled roots were shaken with diluted 1% sodium hypochlorite for 4 min following the Hussey and Barker (1973) method. The eggs were washed three times with distilled water and filtered through 100 and 20 μm sieves. Eggs retained by the 20 μm sieve were collected by washing the sieve with distilled water over a container. The distilled water-egg suspension was incubated at 25 ± 3 °C during 3 days in a Baermann funnel to allow egg-hatching. The number of freshly emerged juveniles per ml of suspension was determined three times using a counting chamber under light microscope. The solution was adjusted to1500 J2 per 1 ml.
Pot experiment
Tomato seeds (Lycopersicum esculentum Mill. cv. Riogrande, susceptible to Meloidogyne) were sterilized by immersion in 5% hypochlorite sodium for 10 min soon followed by three washes by distilled water and sown in alveolus plates filled with peat. Germinated, four leaves and uniform seedlings were transplanted into plastic pots (12-cm diameter) filled with an autoclaved (during 1 h at 120 °C) mix of soil (1 l), sand, and peat at rates of 1:1:1, v/v. One day after transplanting, plants were inoculated with 1500 freshly emerged juveniles (J2) by pouring 1 ml of root-knot nematode suspension into two holes around the plant root. The tested nematicide (Dazitol®, Champon Millennium Chemicals Inc., Northvale, NJ, USA) (0.42% w/v capsaicin + 3.7% w/v allyl-isothiocyanate) was applied at recommended label rates of 10 l/ha, corresponding to 0.75 ml per pot, 5 days before seedlings transplantation by pipette at top of soil. The commercial nematicide Vydate®, with active chemical substance oxamyl (DuPont de Nemours South Africa (Pty) Ltd.; 10% oxamyl) was applied, 2 days after nematode inoculation, at rates of 20 kg/ha, corresponding to 0.15 g per pot by soil application. Controls (positive and negative) were non-inoculated pots and inoculated but non-treated pots. Nine replicated pots per treatment were arranged in a completely randomized design and the whole experiment was repeated twice. All pots were maintained under greenhouse conditions with controlled temperature at 25–30 °C and humidity at 60–75%. Pots were watered regularly and fertilized with nutrient solution according to Pharand et al. (2002). Sixty days after inoculation, tomato plants were uprooted carefully and washed free of soil. Fresh and dried shoot weight and root length and weight were measured for each plant. Nematode damage was rated on a scale of 0 to 5 for gall index according to Hussey and Janssen (2002). Number of galls and egg masses per root system were counted. Root-knot nematodes (second stage juveniles and eggs) were extracted from 5 g roots by blender centrifugal method (Coolen 1979) and soil (500 cm3) by flotation-centrifugation technique according to the modified method by De Grisse (1969) and counted under a stereo microscope.
Greenhouse experiment
A field experiment was carried out in two adjacent greenhouses in Chatt-Mariem, Sousse, Tunisia from October to June 2015–2017. The greenhouse soil was naturally infested with Meloidogyne javanica at the level of 107 J2 s per 100 cm3 of soil, as assessed by the extraction technique previously described. Soils in both greenhouses had similar soil texture (loamy sandy) and pH 7.4. One greenhouse was soil solarized during the hot season (July–August) while the other was not. Two weeks before solarization, the soil was disked three times. Then, the soil was irrigated up to field capacity and covered with transparent polyethylene film (100 μm thickness) for 6 weeks. The recorded soil temperature reached more than 50 °C (nearly 55 °C at 15–25 cm soil depth). Each plot was planted with four plant rows, 8-m long, with 1-m row spacing. Blocks were separated by a 1.2-m alley. The experiment was arranged in a randomized complete block design in each greenhouse, using three plots per treatment, which were Dazitol, Mocap, and a control (soil naturally infested with Meloidogyne javanica and not treated). Each treatment had 20 replicates. At the end of the soil solarization period and 5 days prior to tomato planting, Dazitol® was applied through the drip irrigation system. Mocap (10% ethoprophos; BAYER Cropscience, Rhone-Poulenc, Inc.) was applied at the recommended application rate of 50 kg/ha incorporated into the soil in 30-cm wide bands at planting time. At the end of experiment, plant growth parameters were assessed (plant height, fresh and dried shoot weight, length and weight of root). Evaluation of nematode damage on tomato roots was assessed by visually rating gall indices and counting extracted nematodes from soil and roots, as described before in the pot trial.
Data analysis
Data derived from pot and greenhouse experiments were analyzed for significance by analysis of variance and Duncan’s multiple range test (P < 0.05), using the software SPSS 20 for windows.
Results
Pot experiment
Nematode infection caused a significant decrease in plant growth (Table 1). All growth parameters were reduced significantly in the inoculated control compared with non-inoculated control. The aerial part of tomato plants showed no significant differences in dried and fresh shoot weight between Dazitol treatment and non-inoculated plants. Dazitol did not improve plant growth compared to inoculated control, while Vydate treatment increased fresh and dry weight of the plants when compared to Dazitol treatment.
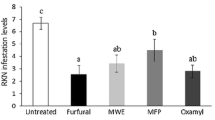
Both Vydate and Dazitol decreased significantly Meloidogyne javanica development. All treatments significantly reduced root galling index, galls, and egg masses numbers per g of root compared to the untreated inoculated control (Table 2). Moreover, both treatments significantly reduced RKN populations in soil and roots (Table 2, Fig. 1).
Greenhouse experiment
Soil treatment with Mocap and Dazitol had no clear significant effect on tomato growth with or without soil solarization, when compared to the non-treated controls. Although, all solarized treatments (S) showed greater shoot length than their respective non-solarized treatments (NS) (Table 3).
Mocap and Dazitol reduced root-knot nematode reproduction (Table 4) and the greatest reductions in gall index, and number of nematodes per root system or g of soil were obtained by combining soil solarization and a nematicide treatment.
Discussion
The results of the present study prove that Dazitol has the potential to contribute as a successful eco-friendly component in the integrated nematode management package for the tomato. Our results complement those of Ploeg (2008) and Reddy (2011) reporting that isothiocyanates, the active ingredient of brassica crop, had a broad pesticidal activity against weeds, bacteria, fungi, and nematodes. Dazitol was as effective as the synthetic nematicides ethoprophos and oxamyl. This agrees with the results by Fleming et al. (2006) and Martin et al. (2007) who found that isothiocyanates from brassicas are as effective as fenamiphos and fosthiazate (active ingredient of Nemathorin). Additionally, Dazitol, in combination with soil solarization, can be used for root-knot nematode control in organic farms or conventional farming systems (Reddy 2011). For conventional farming systems, soil solarization, alone or in combination with organic amendments, was found effective towards Meloidogyne javanica and Globodera pallida infecting potato crop as it reduced nematodes multiplication and improved plant growth and yield (Elhajji and Horrigue-Raouani 2012). Moreover, combination of solarization and biofumigation was reported to be efficient in reducing pathogenic fungus severity (Gamliel and Stapleton 1993). Finally, in organic farms, the combination of soil solarization with organic amendments has notable potential for controlling M. javanica and M. incognita (Oka et al. 2007).
References
Abd-Elgawad MM (2008) The current status of phytonematode management in Egypt with special reference to applicable nematicides. Egypt J Agron 6:33–46
Caboni P, Ntalli NG (2014) Botanical nematicides, recent findings. In: Gross AD, Coats JR, Duke SO, Seiber JN (eds) Biopesticides: state of the art and future opportunities. American Chemical Society, Washington DC, pp 145–157
Coolen WA (1979) Methods for the extraction of Meloidogyne spp. & other nematodes from roots and soil. In: Lamberti F, Taylor CE (eds) Root-knot nematodes (Meloidogyne Species) systematics, biology and control. Academic Press, London, pp 317–329
De Grisse AT (1969) Redescription ou modification de quelques techniques utilisée dans l’étude des nématodes phytoparasitaires. Mededelingen Rijksfaculteit der Landbouwetinschap, Gent, pp 351–369
Elhajji L, Horrigue-Raouani N (2012) Efficiency of solarization and incorporation of cattle manure in management of Meloidogyne javanica (Treub.) Chitwood and Globodera pallida (stone) Behrens associated with potato. Pest Technol 6:75–78
Fleming CC, Turner SJ, Hunt M (2006) Management of root knot nematodes in turfgrass using mustard formulations and biostimulants. Commun Agric Appl Biol Sci 71(3):653–658
Gamliel A, Stapleton JJ (1993) Characterization of antifungal volatile compounds evolved from solarized soil amended with cabbage residues. Phytopathology 83:899–905
Horrigue-Raouani N (2003) Variabilité de la relation hôte parasite dans le cas des Meloidogyne spp. (Nematoda: Meloidogynidae). Thèse de docteur d’état. Université Tunis-El Manar, Faculté des Sciences de Tunis, Tunis, pp 222
Hussey RS, Barker KR (1973) A comparison of methods of collecting inocula of Meloidogyne spp. including a new technique. Plant Dis Rep 57:1025–1028
Hussey RS, Janssen GJW (2002) Root-knot nematode: Meloidogyne species. In: Starr JL, Cook R, Bridge J (eds) Plant resistance to parasitic nematodes. CAB International, Wallingford, pp 43–70
Katan J, Greenberger A, Alon H, Grinstein A (1976) Solar heating by polyethylene mulching for the control of diseases caused by soilborne pathogens. Phytopathology 66:683–688
Martin TJ, Turner SJ, Fleming CC (2007) Management of the potato cyst nematode (Globodera pallida) with bio-fumigants/stimulants. Commun Agric Appl Biol Sci 72(3):671–675
Mekete T, Decraemer W, Wesemael WML, Seid A, Fininsa C (2015) Tomato (Solanum lycopersicum) and root-knot nematodes (Meloidogyne spp.)—a century-old battle. Nematology 17:1–15
Moens M, Perry RN, Starr JL (2009) Meloidogyne species—a diverse group of novel and important plant parasites. In: Perry RN, Moens M, Starr JL (eds) Root knot nematodes. CAB International, Wallingford, p 483
Oka Y, Nurit S, Pinchas F (2007) Control of root-knot nematodes in organic farming systems by organic amendments and soil solarization. Crop Prot 26(10):1556–1565
Pharand B, Carisse O, Benhamou N (2002) Cytological aspects of compost-mediated induced resistance against Fusarium crown and root rot in tomato. Phytopathology 92:424–438
Ploeg A (2008) Biofumigation to manage plant-parasitic nematodes. In: Ciancio A, Mukherjee KG (eds) Integrated management and biocontrol of vegetable and grain crop nematodes. Springer, Dordrecht, pp 239–248
Reddy PP (2011) Biofumigation and solarization for management of soil borne plant pathogens, vol 431. Scientific publishers, Jodhpur
Salem MF (2014) Innovative approaches through modified biofumigation in controlling soil-borne pathogens and root-knot nematode. Aspects of Applied Biology 126, 5th International Symposium of Biofumigation, UK
Seid A, Fininsa C, Mekete T, Decraemer W, Wesemael W (2015) Tomato (Solanum lycopersicum) and root-knot nematodes (Meloidogyne spp.): a century-old battle. Nematology 17(9):995–1009
Sikora RA, Fernandez E (2005) Nematode parasites of vegetables. In: Luc M, Sikora RA, Bridge J (eds) Plant parasitic nematodes in subtropical and tropical agriculture. CAB International, Wallingford, pp 319–392
Stapleton JJ, Heald CM (1991) Management of phytoparasitic nematodes by soil solarization. In: Katan J, JE DV (eds) Soil solarization. CRC Press, Boca Raton, pp 51–59
Stoddard FL, Nicholas AH, Rubiales D, Thomas J, Villegas-Fernández AM (2010) Integrated pest management in faba bean. Field Crop Res 115(3):308–318
Talavera M, Sayadi S, Chirosa-Ríos M, Salmerón T, Flor-Peregrín E, Verdejo-Lucas S (2012) Perception of the impact of root-knot nematode-induced diseases in horticultural protected crops of South-Eastern Spain. Nematology 14:517–527
Thoden TC, Korthals GW, Termorshuizen AJ (2011) Organic amendments and their influence on plant parasitic and free living nematodes: a promising method for nematode management. Nematology 13:133–153
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
This study does not involve any human and/or animal participant.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Responsible editor: Philippe Garrigues
Rights and permissions
About this article
Cite this article
Hajji-Hedfi, L., Rebai, E., Larayedh, A. et al. Biological control of Meloidogyne javanica on tomato with Dazitol® and soil solarization. Environ Sci Pollut Res 25, 17278–17282 (2018). https://doi.org/10.1007/s11356-018-1962-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-018-1962-x