Abstract
The discharge of landfill leachate into the environment without effective treatment poses a serious threat for the aquatic ecosystems. This present study was undertaken to evaluate whether electrocoagulation process is efficient for treatment landfill leachate (LL) or not by using antioxidant biomarkers in Gammarus pulex. Glutathione peroxidase (GSH-Px), superoxide dismutase (SOD), and catalase (CAT) activities and malondialdehyde (MDA) and glutathione (GSH) levels in G. pulex exposed to untreated, treated, and diluted rates 1/10 and 1/20 in both LL during 24 and 96 h were tested. Physiochemical characteristics of leachate (chemical oxygen demand, electrical conductivity, pH, phosphate, turbidity, NH3, Cl−, and color) were determined pre and post treatment. All physiochemical characteristics of LL decreased after treatment process. GSH-Px and CAT activities and GSH and MDA levels were increased in untreated groups when compared to control (p < 0.05). After treatment by electrocoagulation, MDA and GSH levels and CAT activities were returned to control values. In conclusion, the abilities of LL to stimulate oxidative stress in G. pulex have been proven. The results revealed that antioxidant parameters are useful biomarkers for determining the treatment efficiency of the electrocoagulation process.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Landfill leachate is mostly generated due to biodegradation of the waste and the excess rainwater filtered through the solid waste layers. Migrating away from a landfill, small amounts of landfill leachate can cause critical pollution to the groundwater aquifer and adjacent surface waters (Li et al. 2006). Leachate includes many hazardous chemicals such as environmental persistence, toxicity, mobility, and lipophilicity, resulting in bioaccumulation in food webs (Fent 2004). Composition of leachates is usually characterized by a high level of organic matter. Leachates also contain some metal trace elements and different kinds of organic pollutants. Therefore, these cocktails of pollutants may have possible toxic and genotoxic effects (Olivero-Verbel et al. 2008).
There is crucial balance between reactive oxygen species (ROS) generation and their removal by antioxidant defense system in organisms. ROS are scavenged via antioxidant enzymes and non-enzymatic antioxidants (Hermes-Lima 2004; Halliwell and Gutteridge 2007). Lipid peroxidation is a chain reaction where oxidants create the breakdown of membrane phospholipids that have polyunsaturated fatty acids. Lipid peroxidation causes the damage to bio-membranes which can have important consequences for living organisms (Hermes-Lima 2004; Jemec et al. 2012). Pollutants could stimulate ROS production (Livingstone 2001). During oxidative challenge, cells mostly increase their levels of antioxidant enzymes such as glutathione peroxidase (GSH-Px), superoxide dismutase (SOD), and catalase (CAT) (Livingstone, 2001; Valavanidis et al. 2006). Lipid peroxidation and antioxidant defense mechanism have been successfully used as oxidative stress biomarkers in environmental studies and used in the assessment of effects of pollutants in aquatic environments (Livingstone 2001; Valavanidis et al. 2006).
Crustaceans are often used as bioindicators in many aquatic ecosystems (Rinderhagen et al., 2000). With their abundance in freshwater, their high ecological relevance, and their crucial role in the food chain, amphipods of the genus Gammarus are often employed in ecotoxicological studies (Kunz et al. 2010; Adam et al. 2010).
Electrochemical coagulation is an easy and effective method. It is the electrochemical production of destabilization agents that brings about charge neutralization for pollutant removal, and it has been utilized for wastewater treatment. Aluminum or iron plates are usually used as electrodes. Metallic ions that generated electrochemically from these electrodes can be hydrolyzed near the anode to produce a series of activated intermediates that can destabilize the finely dispersed particles present in the water or wastewater to be treated. The destabilized particles then aggregate to form flocks (Chen et al. 2002). Electrochemical coagulation (EC) is characterized for the removal of colloids, suspended solids, and other compounds (Oumar et al. 2016). One of the promising methods for treating wastewater is EC process. EC is applied to treat water containing dyes, oil wastes, food stuff wastes, organic matter from landfill leachates, chemical and mechanical polishing waste, synthetic detergent effluents, and mine wastes. Mechanism of electrocoagulation involves three steps: (a) creation of coagulants by electrolytic oxidation of the sacrificial electrode; (b) destabilization of the contaminants, particulate suspension, and refraction of emulsions, and (c) collection of the destabilized phases to form flocs (Saravanan et al. 2010).
In the present study, we aimed to investigate whether electrocoagulation process is efficient for treatment landfill leachate (LL) or not by using antioxidant biomarkers in G. pulex.
Materials and methods
Chemicals
Potassium chromate (K2CrO4), phenol (C6H6O), ammonium meta vanadate (NH4VO3), vanadate molybdate (NH4)6Mo7O24.4H2O, sulfuric acid (H2SO4), potassium dihydrogen phosphate (KH2PO4), sodium hydroxide (NaOH), and hydrochloric acid (HCI) were purchased from Sigma-Aldrich. Silver nitrate (AgNO3), trisodium citrate (Na3C6H5O7), and sodium hypochlorite (NaClO) were purchased from Carlo Erba. Sodium nitroprusside (Na2[Fe(CN)5NO]) was purchased from Merck. Chemical oxygen demand (COD) kits were purchased from HACH Lange, COD digestion vials 0–1500 ppm.
Characteristics of treated and untreated leachate
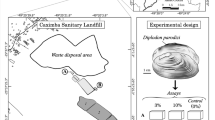
Landfill leachate was taken from a young municipal landfill site from Bingol Province of Turkey. All chemical measurements for both untreated and treated leachates were determined according to standard methods (SM). Chloride (CI−) was analyzed with the use of SM 4500 CI−. Ammonia nitrogen (NH3-N) was analyzed with the use of SM 4500-F. Phosphate (PO43−) was analyzed with the use of SM 4500-P (Apha and WPCF, 2005). The COD was measured using a thermoreactor (HACH, DRB200) with the closed reflux method (5220D) (Apha and WPCF, 2005). Physicochemical parameters such as conductivity and pH were analyzed using a multiparameter (Thermo Orion 420A), while turbidity was measured using a digital portable turbidimeter (HACH 2100P). Color was measured according to Res methods using a UV/VIS spectrophotometer (Shimadzu 1800 UV/VIS spectrophotometer).
Animals and experimental procedure
Individuals of G. pulex were collected with handnets in the Munzur River from Tunceli, Turkey (39.156820 N, 39.499640 E). The organisms were rapidly transferred in plastic bottles to the laboratory where they were stocked in aerated 20-L aquaria in a climate-controlled room at 18 °C and a 12:12 light:dark cycle and fed willow leaves for 15 days before they were used for experiments. Organisms which are similar, adult, healthy, and active were selected for the study (De Lange et al. 2006). Each aquarium consists of 1 L water consisted of three replicates with 10 individuals. Experimental conditions for the acute test (96 h) used static tests. Organisms were not fed during the experiments. The organisms were checked per 24 h and dead individuals were counted and removed from the experiment aquarium. Inactivity was accepted as the criterion for death.
Five experimental groups were designed as control group (C); X1, untreated leachate (diluted 1/10 with tap water); X2, untreated leachate (diluted 1/20 with tap water); Y1, treated leachate (diluted 1/10 with tap water) by electrocoagulation; and Y2, treated leachate (diluted 1/20 with tap water) by electrocoagulation.
Experimental organisms in similar inter molt stage with about 10 mm in length were selected for the study (De Lange et al. 2006). The study was repeated three times. In each 1-L aquarium, 10 individuals were used. A total of 300 individuals were used for the five groups, in two periods (24 and 96 h) and three repetitions. Since there was not enough homogenate in 1 individual, so 10 individuals were mixed and a single sample was formed.
Biochemical analyses
The samples were weighed and homogenized by adding PBS buffer (salt solution buffered with phosphate) at a rate of 1/5 w/v and using a homogenizer with ice to measure antioxidant parameters. The samples were centrifuged at 17,000 rpm for 15 min; the supernatants were kept in deep freeze at − 70 °C until their measurements were done. The concentration of GSH was performed by the method of Beutler et al. (1963) and expressed as nanomoles per gram tissue. The concentration of MDA was measured by the method of Placer et al. (1966) and expressed as nanomoles per gram tissue. SOD, CAT, and GSH-Px activities were conducted by using ELISA kit. The activities of SOD, CAT, and GSH-Px were determined by ELISA kits (catalog numbers, CAT 707002, SOD 706002, and GSH-Px 703102) purchased from The CAYMAN Chemical Company.
Electrocoagulation experiments
The design of the batch monopolar EC reactor was used in the present study. The electrolytic cell was made of plexiglass material and 1 L in volume, respectively. The electrode sets (anode and cathode) consisted of iron plates, with a dimension of 5 cm (width) × 6 cm (length) × 2 mm (depth), having a surface area of 30 cm2. The batch electrochemical cell consisted of iron electrodes that were all individually connected to the dc power supply (AA Tech ADC-3303D, 0–3 A, 0–60 V). A working volume of 900 mL of leachate was used for all experiments ensured with a magnetic stirrer. The magnetic stirrer was placed above the cell and set at constant 200 rpm. The experiments were performed at room temperature (25 ± 2 °C) and without any electrolyte addition. Operational time was selected to be 120 min. The performance of the electrocoagulation treatments of leachate was improved for 17 mA/m2 current density which was performed.
Statistical analyses
Data were analyzed using SPSS 18 software. One-way ANOVA and Duncan’s multiple range tests were employed to evaluate the statistical differences in each application group (X1, X2, Y1, Y2) in the same hours (abcp < 0.05). Two-tailed independent T test was used to compare the differences between exposure times (24 and 96 h) in the same application groups (*p < 0.05).
Results
In this study, physicochemical characteristics of LL were measured before and after electrocoagulation process and these parameters of LL were decreased after treatment (Table 1).
CAT, GSH-Px, and SOD activities and GSH and MDA levels of G. pulex exposed to all experimental groups (X1, X2, Y1, and Y2) during 24 and 96 h were illustrated in Fig. 1a–e.
a SOD (U/ml) activities, b GSH (nmol/g tissue) levels, c GSH-Px (nmol/min/ml) activities, d MDA (nmol/g tissue) levels, and e CAT (nmol/min/ml) activities of G. pulex exposed to different kinds of LL. Asterisk (*) shows statistical differences according to the two-tailed independent T test between different exposure time (24 and 96 h) in the same groups; *p < 0.05. Different letters on bar (a, b, c) show statistical differences of Duncan’s multiple range test among all application groups in the same exposure time; abcp < 0.05. Values represent mean ± SE; n = 10
MDA levels were higher in the untreated groups (X1, X2) (p < 0.05) than that in the control group for both 24- and 96-h exposure time. However, these levels in the treatment groups (Y1, Y2) were almost close to that in the control group (p < 0.05).
GSH levels for 24-h exposure time in all groups were lower than that in the control group (p < 0.05). These levels for 96 h in all groups were close to that in the control group (p > 0.05).
GSH-Px activities were decreased after treatment in the Y1 group for 24-h exposure time. The differences in GSH-Px activities of G. pulex between X1 and Y1 were found statistically significant for 24-h exposure time (p < 0.05) but these activities in all groups for 96-h exposure time were close to that in the control group (p < 0.05) except for the Y1 group.
CAT activities for 24-h exposure time in all groups increased compared to that in the control group except for Y2 (p < 0.05); however, for 96-h exposure time in all groups was close to that in the control group except for Y1 (p < 0.05).
SOD activities only decreased in the X1 group compared to that in the control group for 24-h exposure time (p < 0.05). Alterations in SOD activities in all groups were not statistically important for 96-h exposure time.
In the X1 group, there were no significant differences in CAT and SOD activities and GSH levels of G. pulex when exposure time was compared (p > 0.05); however, MDA levels and GSH-Px activities were observed to be statistically significant (p < 0.05). When exposure time was compared, no significant differences were found in all biomarkers in the X2 group (p > 0.05). In the Y1 group, there were no significant differences in GSH-Px activities and MDA levels of G. pulex when exposure time was compared (p > 0.05) while GSH levels and CAT and SOD activities were found to be statistically significant (p < 0.05). In the Y2 group, GSH-Px activities and GSH levels were statistically significant when exposure time was compared (p < 0.05).
Discussion
Landfill leachate is a liquid that is produced by the rain which falls on the solid waste. The leachate contains high concentrations of ammonium, organic matter, toxic compounds, and heavy metals. COD, heavy metals, and other substances need to be reduced during leachate treatment (Kurniawan 2011). COD is known as the amount of oxidant consumed when samples were treated by the oxidant. It has been suggested as a general index of the level of organic pollution (Xi et al. 1996). In the present study, COD levels and other physiochemical parameters were decreased after electrocoagulation process (Table 1). In our study, high NH3-N, conductivity, and chloride were found according to the EPA surface water quality criteria (EPA 2001). Studies have shown that NH4+-N promotes the methane production when the concentration of NH4+-N was lower than 0.4 g/L (Santos et al., 2004). NH3 is toxic to many organisms. The importance of ammonia in toxicity is not a surprise. The 96-h LCs0 range from 0.32 to 3.10 mg of un-ionized NH3-N/1 for several species of fish (Ruffler et al. 1981) and from 0.80 to 40 mg of un-ionized NH3-N/I for 10 species of macro-invertebrates (Jean, 1991). The particular importance of ammonia in toxicity of landfill leachates to fish was reported by Cameron and Koch (1980). Deneuvy (1987) found for his part a more or less good correlation between ammonia and landfill leachate toxicity to daphnids, micro-algae, and bacteria of Microtox.
There is a relationship between the conductivity and chloride levels. Conductivity is an indicator of the abundance of the total concentration of the ions or the abundance of the dissolved inorganic species (Banar et al., 2006; Tatsi and Zouboulis, 2002). High conductivity value of the leachate may be attributed to high dissolved salts and metals in the leachate, metals which may be responsible for toxicity of leachates (Aiyesanmi and Imois, 2011).
In our study, the dark color of landfill leachate is the presence of high concentrations of humic substances which represent the mostly organic compounds (Table 1) (Vedrenne et al., 2012; Qiu et al., 2016).
Recently, it has been demonstrated that leachates (complex chemical mixtures) are toxic and induced oxidative stress in aquatic organisms (Ali et al. 2004; Radetski et al. 2004). The toxicity level of leachate discharge to the aquatic environment can be determined by using aquatic organism as biological indicator (Raihana et al. 2014). Toxicological assessment of treated and untreated landfill leachate is essential to examine the effect of leachate discharged on the environment. Reduction in toxicity of the treated leachate helps in evaluating the effectiveness of the remediation strategy (Ganey and Boyd 2005). However, limited information is available that indicates oxidative stress inducing effect of LL. As far as what is known, there is no information in the literature on the effects of LL treated by electrocoagulation on SOD, CAT, and GSH-Px enzyme activities and GSH and MDA levels of G. pulex. In this study, a number of changes at the cellular level (SOD, CAT, and GSH-PX activities and MDA and GSH levels) of G. pulex after untreated, treated, and diluted rates 1/10 and 1/20 in both LL during 24 and 96 h were tested to reveal the electrocoagulation process efficiency in model organism (G. pulex).
Pollution in aquatic environments can enhance ROS production resulting from imbalance between ROS concentrations and antioxidant defense system, leading to toxic effects such as lipid peroxidation (Regoli et al. 2004). Key antioxidant enzymes and non-enzymatic antioxidants have been shown to be influenced by various single pollutants known to increase ROS levels (Valko et al. 2006; Ryter et al. 2007). The activities of oxidative stress, detoxification, and neurotoxicity biomarkers have been used in Gammarus (Demirci et al. 2017). We used antioxidant parameters in our study as a biomarker. Increment of glutathione and glutathione-dependent enzymes is defined as the primary response to exposure to toxic compounds with potential of inducing oxidative stress (Lindesjoo et al. 2002; Oruc and Uner 2002). The change in GSH-Px activity is often accompanied by changes in the level of GSH, which is the co-substrate for H2O2 decomposition by GSH-Px (Sies 1999). Kutlu and Susuz (2004) observed slightly inhibited activity of GSH-Px after exposure to lead acetate in invertebrate, G. pulex. In the present study, similar GSH-Px and GSH induction were determined in G. pulex exposed to different kinds of untreated leachate (Fig.1b, c).
Increased SOD, GSH-Px, and CAT activities were found in hearts, kidneys, and spleens of mice in response to leachate (Li et al. 2006). It was demonstrated that the pesticide mixture induces synergistic interactions leading to more oxidative stresses and thus induces CAT and SOD enzymes in Gammarus kischineffensis (Demirci et al., 2017). It was found that elevated antioxidant stress enzyme activities, e.g., superoxide dismutase (SOD) and catalase (CAT), were detected in Vicia root tissues even at the lowest tested leachate concentration (Radetski et al. 2004). Sub-lethal Cd exposure concentrations and durations significantly affect the levels of antioxidative defense enzymes (SOD, CAT, and GSH-Px), MDA, and protein content in G. pulex. Cd accumulation and MDA content increased depending on the exposure concentration and duration (Duman and Kar. 2015). Similarly in our study, CAT activities and MDA levels were also increased in untreated groups when compared to control (p < 0.05) (Fig. 1d, e). CAT was stimulated to scavenge the H2O2 due to the increased pollutant concentration at higher concentration of leachate. The increased antioxidant enzymes and MDA levels show that biological membranes were attacked by free radicals and the antioxidant enzymes were induced to prevent against hyperoxia and lipid peroxidation, which was possible metabolic adaptation to the exposure to LL and was defense against oxidative damage (Li et al. 2006).
Superoxide dismutase is the first enzyme of antioxidant defense system (Vijayavel et al. 2004) by accelerating the dismutation of superoxide (O2) to H2O2. It damages the membrane and biological structures. Decreased SOD activity was found in untreated groups compared to control; the decrease of SOD indicates that this antioxidant enzyme was inhibited; its protective effects against free radicals were reduced. After treatment by electrocoagulation, MDA and GSH levels were returned to control values (Fig.1b, d).
The landfill leachate toxicity to the Asian clam Corbicula fluminea was investigated by using biomarkers and variability in biomarker responses among leachate concentrations was observed (Oliveira et al. 2014). The toxicological effects of municipal landfill leachate on Vicia faba were studied in different seasons and toxicity study was performed via various antioxidant parameters. A dose-dependent elevation in the MDA level and inhibited antioxidant enzyme activities were observed (Gupta and Rajamani 2015). In the present study, we also found different biomarker responses depending on leachate concentrations and exposure times. Exposure time is important in determining cellular responses because the intracellular redox state is presumed to be altered according to the duration of the exposure (Ishihara and Shimamoto, 2007). Exposure concentration and duration have strong synergistic effects on antioxidant enzyme activity (Duman and Kar, 2015).
Conclusion
In conclusion, it has been demonstrated that LL stimulates oxidative stress and the positive correlations between antioxidant responses. Different LL concentration affected antioxidant biomarkers in different levels in G. pulex. Also, the biomarkers such as SOD, CAT, GSH-Px, MDA, and GSH clearly revealed metabolic changes after LL exposure but only MDA and GSH levels of G. pulex confirmed the LL treatment efficiency of electrocoagulation process.
References
Adam O, Degiorgi F, Crini G, Badot PM (2010) High sensitivity of Gammarus sp. juveniles to deltamethrin: outcomes for risk assessment. Ecotoxicol Environ Saf 73:1402–1407
Aiyesanmi A, Imois OB (2011) Understanding leaching behaviour of landfill leachate in Benin-City, Edo State, Nigeria through dumpsite monitoring. British J Environ Climate Change 1:190–200
Ali M, Parvez S, Pandey S, Atif F, Kaur M, Rehman H, Raisuddin S (2004) Fly ash leachate induces oxidative stress in freshwater fish Channa punctata (Bloch). Environ Int 30:933–938
Apha AWWA, WPCF, (2005) American Public Health Association, American Water Works Association, Water Pollution Control Federation, Washington
Banar M, Özkan A, Kürkçüoğlu M (2006) Characterization of the leachate in an urban landfill by physicochemical analysis and solid phase microextraction-GC/MS. Environ Monit Assess 121:439–459
Beutler E, Duron O, Kelly BM (1963) Improved method for the determination of blood glutathione. J Laboratory Clinical Medicine 61:882–888
Cameron RD, Koch FA (1980). Toxicity of landfill leachates. J. Water Pollut. Control Fed. 52(4):760–769
Chen X, Chen G, Yue PL (2002) Investigation on the electrolysis voltage of electrocoagulation. Chem Eng Sci 57:2449–2455
De Lange HJ, Noordoven W, Murk AJ, Lürling MFLLW, Peeters ETHM (2006) Behavioural responses of Gammarus pulex (Crustacea, Amphipoda) to low concentrations of pharmaceuticals. Aquatic Toxicol 78(3):209–216
Demirci O, Duve K, Asma D, Ogut S, Ugurlu P (2017) Effects of endosulfan, thiamethoxam, and indoxacarb in combination with atrazine on multi-biomarkers in Gammarus kischineffensis. Ecotoxicol Environ Saf 147(1):749–758
Deneuvy JP. 1987. Les lixiviats de d6charges; approche m6thodologique de leurtoxicity aigu en fonction de differents modes de traitement, These de DocteurIng6nieur INSA/ENTPE, 339 pp.
Duman F, Kar M (2015) Evaluation of effects of exposure conditions on the biological responses of Gammarus pulex exposed to cadmium. Int J Environ Sci Technol 12:437–444
Environmental Protection Agency (EPA) (2001). Parameters of Water Quality. Ireland, p133
Fent K (2004) Ecotoxicological effects of contaminated sites. Toxicology 205:223–240
Ganey PE, Boyd SA (2005) An approach to evaluation of the effect of bioremediationon biological activity of environmental contaminants: dechlorination of polychlorinated biphenyls. Environ. Health Perspect 113:180–185
Gupta A, Rajamani P (2015) Toxicity assessment of municipal solid waste landfill leachate collected in different seasons from Okhala landfill site of Delhi. J Bio Sci and Eng 8:357–369
Halliwell B, Gutteridge JMC (2007) Free radicals in biology and medicine. Oxford University Press, New York
Hermes-Lima M (2004) Oxygen in biology and biochemistry: role of free radicals. In: Storey KB (ed) Functional metabolism: regulation and adaptation. John Wiley & Sons, Inc., USA
Ishihara Y, Shimamoto N (2007) Critical role of exposure time to endogenous oxidative stress in hepatocyte apoptosis. Redox Rep 12(6):275–281
Jean G (1991). Evaluation de la toxicit6 aigu6 de solutions synth6tiques et d’effiuents partir d’un multi-test macroinvert6br6s: le MTM. Comparaison des r6ponses des macroinvert6br6s avec celles de P. Phosphoreum, Daphnia magna et Brachydanio rerio, Th~se de Doctorat en Gestion et Traitement des D6chets de I’INSA de LYON, 213 pp.
Jemec A, Tisler T, Erjavec B, Pintar A (2012) Antioxidant responses and whole-organism changes in Daphnia magna acutely and chronically exposed to endocrine disruptor bisphenol A. Ecotoxicol Environ Saf 86:213–218
Kurniawan F (2011) Analisis Ketepatan Pengukur Nilai Tegangan dan Arus RMS Jala-Jala Listrik Berbasis Mikrokontroler ATmega8535. Jurnal Angkasa, 3:2
Kunz PY, Kienle C, Gerhardt A (2010) Gammarus spp. in aquatic ecotoxicology and water quality assessment: toward integrated multilevel tests. Rev Environ Contam Toxicol 205:1–76
Kutlu M, Susuz F (2004) Effects of lead as an environmental pollutant on EROD enzyme in Gammarus pulex (L.) (Crustacea: Amphipoda). Bulletin of environmental contamination and toxicology, 72(4):750–755
Li G, Sang N, Guo D (2006) Oxidative damage induced in hearts, kidneys and spleens of mice by landfill leachate. Chemosphere 65:1058–1063
Lindesjoo E, Adolfsson-Erici M, Ericson G, Forlin L (2002) Biomarker responses and resin acids in fish chronically exposed to effluents from a total chlorine-free pulp mill during regular production. Ecotoxicol Environ Saf 53:238–247
Livingstone DR (2001) Contaminant-stimulated reactive oxygen species production and oxidative damage in aquatic organisms. Mar Pollut Bull 42:656–666
Oliveira LF, Silva SMCP, Martinez CBR (2014) Assessment of domestic landfill leachate toxicity to the Asian clam Corbicula fluminea via biomarkers. Ecotoxicol Environ Saf 103:17–23
Olivero-Verbel J, Padilla-Bottet C, De la Rosa O (2008) Relationships between physicochemical parameters and the toxicity of leachates from a municipal solid waste landfill. Ecotox Environ Safe 70:294–299
Oruc EO, Uner N (2002) Marker enzyme assessment in the liver of Cyprinus carpio (L.) exposed to 2,4-D and azinphosmethyl. J Biochem Mol Toxicol 16:182–188
Oumar D, Patrick D, Gerardo B, Rino D, Ihsen BS (2016) Coupling biofiltration process and electrocoagulation using magnesium-based anode for the treatment of landfill leachate. J Environ Man 181:477–483
Placer ZA, Cushmann LL, Johnson BC (1966) Estimation of products of lipid peroxidation (as malondialdehyde) in biochemical systems. Anal Biochem 16:359–364
Qiu A, Cai Q, Zhao Y, Guo Y, Zhao L (2016) Evaluation of the treatment process of landfill leachate using the toxicity assessment method. Int J Environ Res Public Health 13(12):1262
Radetski CM, Ferrai B, Cotelle S, Masfaraud JF, Ferard JF (2004) Evaluation of the genotoxic, mutagenic and oxidant stress potentials of municipal solid waste incinerator bottom ash leachates. Sci Total Environ 333:209–216
Raihana AR, Sharifah Norkhadijah SI, Emilia ZA, Praveena SM (2014) Landfill leachate toxicity analysis with Orechromis mossambicus (Mozambique tilapia): a review. Int J Sci: Basic Applied Res 18(2):198–216
Regoli F, Frenzilli G, Bocchetti R, Annarumma F, Scarcelli V, Fattorini D, Nigro M (2004) Time-course variations of oxyradical metabolism, DNA integrity and lysosomal stability in mussels, Mytilus galloprovincialis, during a field translocation experiment. Aquat Toxicol 68:167–178
Rinderhagen M, Ritterhoff J, Zauke GP (2000) Crustaceans as bioindicators. In: Gerhardt, (Ed.), Biomonitoring of polluted water—reviews on actual topics, Environmental Research Forum. Trans Tech Publications—Scitech Publications, Zürich, pp. 161–194
Ruffler PJ, Boyle WC, Kleinschmidt J (1981) Short-term acute bioassays to evaluate ammonia toxicity and effluent standards. J. Water Pollut. Control Fed. 53:367–377
Ryter SW, Kim HP, Hoetzel A, Park JW, Nakahira K, Wang X, Choi AM (2007) Mechanism of cell death in oxidative stress. Antiox Redox Signal 9:49–89
Santos ABD, Bisschops IAE, Cervantes FJ, Lier JBV (2004) Effect of different redox mediators during thermophilic azo dye reduction by anaerobic granular sludge and comparative study between mesophilic (30 °C) and thermophilic (55 °C) treatments for decolourisation of textile wastewaters. Chemosphere 55:1149–1157. https://doi.org/10.1016/j.chemosphere.2004.01.031
Saravanan M, Sambhamurthy NP, Sivarajan M (2010) Treatment of acid blue 113 dye solution using iron electrocoagulation. Clean–Soil, Air, Water 38(5–6):565–571
Sies H (1999) Glutathione and its role in cellular functions. Free Rad Biol Med 27:916–921
Tatsi AA, Zouboulis AI (2002) A field investigation of the quantity and quality of leachate from a municipal solid wastelandfill in a Mediterranean climate (Thessaloniki, Greece). Adv Environ Res 6(3):207–219
Valavanidis A, Vlahogianni T, Dassenakis M, Scoullos M (2006) Molecular biomarkers of oxidative stress in aquatic organisms in relation to toxic environmental pollutants. Ecotoxicol Environ Saf 64:178–189
Valko M, Rhodes CJ, Moncol J, Izakovic M, Mazur M (2006) Free radicals, metals and antioxidants in oxidative stress induced cancer. Chemico-Biol Int 160:1–40
Vedrenne M, Vasquez-Medrano R, Prato-Garcia D, Frontana-Uribe BA, Ibanez JG (2012) Characterization and detoxification of a mature landfill leachate using a combined coagulation–flocculation/photo Fenton treatment. J Hazard Mater 29:208–215
Vijayavel K, Gomathi RD, Durgabhavani K, Balasubramanian MP (2004) Sublethal effect of naphthalene on lipid peroxidation and antioxidant status in the edible marine crab Scylla serrata. J Mar Poll Bull 48:429–433
Xi DL, Sun YS, Liu XY (1996) Environment monitoring. Higher Education Press, Beijing, pp 389–391
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Philippe Garrigues
Rights and permissions
About this article
Cite this article
Serdar, O., Yildirim, N.C., Tatar, S. et al. Antioxidant biomarkers in Gammarus pulex to evaluate the efficiency of electrocoagulation process in landfill leachate treatment. Environ Sci Pollut Res 25, 12538–12544 (2018). https://doi.org/10.1007/s11356-018-1491-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-018-1491-7