Abstract
Aflatoxin is among the natural toxins that cause serious side effects on living things. Diosmin is also one of the compounds with broad pharmacological effects. In this study, the effects on the oxidant/antioxidant system of 50 mg/kg body weight/day dose of diosmin, aflatoxin (500 μg/kg body weight/day), and combined aflatoxin (500 μg/kg body weight/day) plus diosmin (50 mg/kg body weight/day) given to the stomach via catheter female adult Wistar Albino rats is examined. Forty rats were used in the experiment, and these animals were randomly allocated to four equal groups. The test phase lasted 21 days, and blood samples and tissue (liver and kidney) samples were taken after this period was over. Some biochemical parameters (glucose, triglyceride, cholesterol, blood urea nitrogen, creatinine, uric acid, lactate dehydrogenase, aspartate aminotransferase, alanine aminotransferase, alkaline phosphatase, total protein, albumin) and levels of malondialdehyde, nitric oxide, and 4-hydroxynonenal and activities of superoxide dismutase, catalase, and glutathione peroxidase were analyzed in the samples. The aflatoxin administered over the period indicated a significant increase in levels of malondialdehyde (MDA), nitric oxide (NO), and 4-hydroxynonenal (4-HNE) in all tissues and blood samples. Therewithal, the activity of antioxidant enzymes showed a change in the decreasing direction. Biochemical parameters of the group in which aflatoxin were administered alone changed unfavorably. Parallel effects were also observed in the histopathological findings of this group. The results showed that aflatoxin changed antioxidant/oxidant balance in favor of oxidant and eventually led to lipid peroxidation. Diosmin administration to aflatoxin-treated animals resulted in positive changes in antioxidant enzyme activities while the levels of MDA, NO, and 4-HNE were reduced in all tissues and blood samples examined. Diosmin alleviates the oxidative stress caused by aflatoxin. Similar improvement was observed in biochemical parameters of this group as well as in liver and kidney histopathology. No significant change was observed in the group treated with diosmin alone in terms of the parameters examined and histologic findings. As a result, diosmin may be included in compounds that can be used as a therapeutic and prophylactic agent in the event of the formation of aflatoxin exposure and poisoning in animals.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Aflatoxins (AF) are mycotoxins produced by some Aspergillus strains, mainly A. flavus and A. parasiticus. Aflatoxin B1 is expressed as one of the most toxic natural carcinogens and is also the most common natural toxic compound in foods/feed (Pitt 2000; Bennett and Klich 2003; Wu et al. 2009). Cytochrome P-450 (CYP) 1A2 and 3A4 are the main enzymes that convert aflatoxin B1 into aflatoxin B1-8,9-epoxide (AFBO) in liver (Roze et al. 2013; Dohnal et al. 2014). Four major metabolic pathways, namely, O-dealkylation, ketoreduction, epoxidation, and hydroxylation, are described in the biotransformation of AFB1 (Wild and Turne 2002; Larsson et al. 2003; Wu et al. 2009). The AFBO responsible for the mutagenic activity of aflatoxin B1 is formed by cytochrome P450. Aflatoxin B1-8,9-epoxide is connected with the guanine N7 position in DNA and RNA to form aflatoxin B1-N7-guanine (Aiko and Mehta 2015). Thus, the particular metabolite that binds to nucleic acids and other cellular macromolecules also ultimately promotes carcinogenic effects (Wild and Turne 2002; Larsson et al. 2003; Wu et al. 2009). In other words, in summary, these toxic metabolites of aflatoxin are the main step for all toxic effects that inhibit DNA, RNA, and protein synthesis (Wang and Groopman 1999).
Flavonoids contain a tricyclic molecule linked to hydroxyl (OH) groups that share the same basic chemical structure (Anon 2004). Flavonoids have a number of biological functions, especially anti-tumoral, anti-lipoperoxidant, anti-ischemic, anti-thrombocytic, anti-allergic, and anti-inflammatory effects (Tanrikulu et al. 2013). Diosmin is a flavonoid glycoside which is found in a natural structure in the pericarps of different citrus fruits and is called 30,5,7-trihydroxy-40-methoxyflavone-7-rhamnoglucoside (Senthamizhselvan et al. 2014). In fact, this organic compound is obtained by dehydrogenation of hesperidin. Flavonoid is rapidly converted and absorbed by the intestinal flora in the form of aglycone diosmetin and then rapidly dispersed in the body, and the half-life of the compound is 26–43 h (Jain et al. 2014). Diosmin is molecularly separated from hesperidin since there are double bonds between the two carbon atoms in the central carbon ring. Diosmetin is reduced to conjugated derivatives of phenolic acids or glycine and is removed by the urine. The unabsorbed diosmin or diosmetin is removed in the stool (Anon 2004).
Oxidative stress occurs due to high free radical production and deterioration of the antioxidant defense system in the physiological system (Halliwell and Chirico 1993; Gutteridge 1995; Guéraud et al. 2010; Pisoschi and Pop 2015). Major antioxidant enzymes, such as superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GSH-Px), play an important role in the living organism (Gutteridge 1995; Halliwell 1999). Reactive oxygen species (ROS) are produced in oxidative metabolism. It is important to determine malondialdehyde (MDA), nitric oxide (NO), and 4-hydroxynonenal (4-HNE) levels in the estimation of the degree of cell damage caused by oxidant radicals produced in the biological structure (Opara 2006; Vladimirov and Proskurnina 2009; Pisoschi and Pop 2015). The activities of antioxidant enzymes also give an idea of the intensity of lipid peroxidation and the severity of oxidative stress (Vladimirov 1998; Opara 2006; Vladimirov and Proskurnina 2009). Aflatoxin leads to impaired pro-oxidant/antioxidant balance (Eraslan et al. 2013; Li et al. 2013; Abdel-Hamid and Firgany 2015; Karabacak et al. 2015). Oxidative stress is a highly effective mechanism for the occurrence of aflatoxin toxicity (Ingawale et al. 2014). Flavonoids, which are polyphenolic compounds due to their strong antioxidant effects and also because of their negligible side effects, were used as preventive and curative in previous studies against aflatoxin poisoning (Souza et al. 1999; Choudhary and Verma 2005; Preetha et al. 2006; Choi et al. 2010; El-Agamy 2010; El-Nekeety et al. 2011; Nili-Ahmadabadi et al. 2011; Sirajudeen et al. 2011; Gupta and Sharma 2011; Vipin et al. 2017).
In studies conducted with diosmin, the effect of this compound against oxidative stress was particularly emphasized. However, there are very limited data on these cases at present (Villa et al. 1992; Srinivasan and Pari 2012; Rehman et al. 2013; Senthamizhselvan et al. 2014; Arab et al. 2015). Nevertheless, according to the author’s knowledge, there are no studies on the use of diosmin in the oxidative damage caused by aflatoxins. Therefore, the reason for planning this study is that no significant data have been collected on the protective, curative, and preventive effects of diosmin against oxidative stress caused by aflatoxin so far. In accordance with this purpose, rats were given aflatoxin for 21 days and the effects of aflatoxins on the liver and kidney tissue were assessed taking into account oxidative stress markers. Based on the analyzed parameters/histopathological findings, the effect of diosmin was investigated. Thus, some results have been obtained regarding the therapeutic and prophylactic use of diosmin in aflatoxin poisoning.
Material and method
Different chemicals and kits used
Glutathione (reduced) and 2-thiobarbituric acid were purchased from Merck KGaA, Darmstadt, Germany. Bovine serum albumin, β-nicotinamide adenine dinucleotide 2′-phosphate reduced tetrasodium salt, glutathione reductase, nitroblue tetrazolium chloride, nitrate reductase, xanthine, xanthine oxidase, 5,5′-dithiobis-(2-nitrobenzoic acid), and diosmin were purchased from Sigma-Aldrich, St. Louis, MD, USA. Kits used in biochemical analyses were purchased from Cobas, Roche Diagnostics, Rotkreuz, Switzerland. Total aflatoxin competitive enzyme immunoassay kit was purchased from R-Biopharm Ridascreen AG, Darmstadt, Germany. 4-Hydroxynonenal adduct enzyme immunoassay kit was purchased from OxiSelect™, Cell Biolabs, San Diego, USA. All other chemical substances used in the current study were of the highest quality and analytical standard.
Animal material
Forty adult female Wistar Albino rats of 150–200 g live weight were selected as experimental material for use in this study. All rats were separated into four groups with equal mean body weights consisting of 10 animals. Up to four animals in each polyethylene cage were positioned. Animals in the experiment were kept in plastic cages under room temperature (22–24 °C) for 12 h day and 12 h night. During the test period, rat pellet feed (3100 kcal/kg, 5% crude cellulose, and 23% raw protein) and drinking water were available at all times. Research protocol was approved by Erciyes University experimental animals’ ethical board.
Aflatoxin production
Aflatoxin was produced in rice medium by using the Aspergillus parasiticus (NRLL 2999) strain based on the protocol reported by Shotwell et al. (1966) and with the modified Demet et al.’s method (1995). Determination of total aflatoxin content in the rice was carried out in an ELISA reading device using the R-Biopharm Ridascreen® aflatoxin total assay kit and the procedure described in the kit. The extraction and subsequent refinement of the aflatoxin content from the rice flour and the ultimate determination of the aflatoxin content as well as type and ratio analysis of aflatoxin were carried out according to the method as designed by Roberts and Patterson (1975) and Nabney and Nesbitt (1965), respectively. After extraction of aflatoxin from rice followed by purification and evaporation of the solvent, the remaining purified aflatoxin residue was dissolved in dimethyl sulfoxide. The type of analysis of this natural toxin reveals that the total aflatoxin was composed of AFB1, AFB2, AFG1, and AFG2, and their proportions were approximately 80%, 10%, 6%, and 4%, respectively.
Animal test
When the first group was designed as a control and only dimethyl sulfoxide at a concentration of 20% was given as a vehicle, the second group was given diosmin (in dimethyl sulfoxide/water, 20%) at the dose of 50 mg/kg body weight/day. The third group received only aflatoxin (in dimethyl sulfoxide/water 20%) at a dose of 500 μg/kg body weight/day. The fourth group received 500 μg/kg body weight/day aflatoxin with diosmin at a dose of 50 mg/kg body weight/day. The daily total volume of liquid administered to the animals in the vehicle was calculated as 2 ml/kg body weight for each animal. All the substances given to all groups were administered orally directly into the stomach by catheter. The dosages of aflatoxin (Kanbur et al. 2011; Karabacak et al. 2015; Eraslan et al. 2013, 2015) and diosmin (Srinivasan and Pari 2012; Senthamizhselvan et al. 2014; Jain et al. 2014; Arab et al. 2015) were determined according to previous studies. Applications for aflatoxin and diosmin were conducted continuously for 21 days between 10: 00–11: 00 and 15: 00–16: 00, respectively.
Sampling and preparations for analysis
When the 21st day of treatment is over, the blood of all animals was taken into heparin and no additive tubes under light ether anesthesia. The liver and kidneys of each rat were removed following the animal’s death. Blood samples collected with heparinized and no additive tubes were centrifuged at 6000×g for 10 min at + 4 °C. Plasma and serum of blood samples were separated. The red blood cells remaining in heparinized tubes were washed three times with the same volume of phosphate-buffered saline (PBS, pH 7.4), and then an equal volume of PBS was added into washed erythrocytes and suspension was vortexed gently. Erythrocytes suspension were hemolyzed with ice-cold PBS during the analysis (Winterbourn et al. 1975). Extracted tissues from all the animals were washed with ice-cold PBS, and then blood and fat/collagen tissues were extracted. Tissue homogenization was performed with PBS used to clean the tissues, and then the homogenate was subjected to centrifugation at 30000×g for 45 min (+ 4 °C) and the supernatants were separated by Eppendorf tubes. All obtained samples (serum, plasma, hemolysate and supernatant) were employed in the measurement of levels/activities of some biochemical parameters, protein, hemoglobin, MDA, NO, and 4-HNE as well as CAT and GSH-Px. The remaining supernatant and hemolysate were extracted with ethanol and chloroform mixture solution (6/10, v/v), and the resulting extract was then used to determine SOD activity. Until the end of analyses, all samples taken from the experimental animals and ready for analysis were stored at − 80 °C.
Oxidative stress analysis
Measurement of hemoglobin in erythrocytes was performed by the method of Fairbanks and Klee (1987), and the unit of measurement was expressed as milligrams of hemoglobin per milliliter of hemolysate. Tissue protein level was determined using the method of Miller (1959) with modification of Lowry et al.’s (1951) method, and the results were calculated as milligrams of protein per milliliter of homogenate. For the measurement of plasma MDA level, Yoshioka et al.’s (1979) method was preferred. In addition, tissue MDA level was measured by the following method of Ohkawa et al. (1978). The unit for calculating the results is nanomoles per milliliter or nanomoles per milligram of protein. In the analysis of CAT activity in the samples, the method developed by Luck (1965) was used and all the data were evaluated as katal per gram of hemoglobin and katal per gram of protein. In the determination of SOD activity, the method of Sun et al. (1988) was used. Results were computed as units per milligram of hemoglobin and units per milligram of protein. In the analyses of GSH-Px, the method reported by Paglia and Valentine (1967) was used. Obtained data were calculated in units per gram of hemoglobin and units per gram of protein. NO level was measured on the basis of the method reported by Tracey et al. (1995). The results are given in units of nanomoles per milliliter and nanomoles per milligram of protein. Serum 4-HNE contents were done using OxiSelect™ HNE adduct competitive enzyme immunoassay kit. The results are expressed in micrograms per milliliter.
The measurement of some biochemical parameters
The measurement of levels of serum glucose, triglyceride, cholesterol, blood urea nitrogen (BUN), creatinine, and uric acid and activities of lactate dehydrogenase (LDH), aspartate aminotransferase (AST), alanine aminotransferase (ALT), alkaline phosphatase (ALP), total protein, and albumin were analyzed using Roche Cobas label commercial kit and the same label of automatic biochemistry analyzers.
Histopathological evaluations
The kidneys and livers of the animals in each group were removed. For histopathological examinations, tissue samples were fixed in 10% neutral buffered formalin, embedded in paraffin wax, then sectioned at 5–6 μm thickness, mounted on glass slides, and stained with hematoxylin and eosin (H&E). The prepared sections were evaluated by light microscopy (Olympus BX51, Tokyo, Japan) and then photographed with the attached camera (Olympus DP71) via digital programmers (DP Inspector and DP Manager).
Statistical analysis
In the statistical calculations of research data, SPSS 13.0 statistics program was utilized. All data in the study were implied as arithmetic average and standard deviation of mean. In the identification of inter-group significance, one-way analysis of variance and Duncan’s multiple-range test were employed. In the measurement of significances, p < 0.05 levels were taken.
Results
Oxidative stress/lipid peroxidation parameters
Erythrocytes/plasma/serum
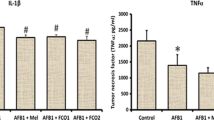
When plasma/serum MDA, NO, and 4-HNE levels and erythrocyte antioxidant enzyme activities were evaluated, values in the diosmin-administered group were closer to the control group at the indicated time and dose. There was no statistically marked difference between the control group and the diosmin-applied group in terms of all analyzed parameters. On the other hand, plasma/serum MDA, NO, and 4-HNE levels were higher in the aflatoxin-receiving group than the control group. On the contrary, the entire antioxidant enzyme activities examined were lower than the control group in the aflatoxin-treated group. The difference between MDA, NO, and 4-HNE levels/antioxidant enzyme parameters is statistically significant when compared with the control group in the aflatoxin-administered group. MDA, NO, and 4-HNE levels were closer to the control group in aflatoxin-diosmin-co-administered groups, but some meaningful differences were found between the control group and the aflatoxin-diosmin-co-administered groups. In animals receiving both compounds, significant changes occur in terms of antioxidant enzyme activities and MDA, NO, and 4-HNE levels compared to the aflatoxin-receiving group, and these values approached towards the control group. However, there is no statistically important difference in the group of two compounds taken together in terms of SOD, CAT, and GSH-Px activities comparing the control group (Table 1).
Liver tissue
Considering liver SOD, CAT, and GSH-Px activities and MDA and NO levels, no statistically meaningful changes were observed between the control group and the diosmin alone-group. On the other hand, MDA and NO levels were higher in the aflatoxin-treated group compared to the control group and statistical significant differences were observed between these groups. There is a statistically significant difference in SOD, CAT, and GSH-Px activity in the aflatoxin-exposed group compared to the control group. That is, the activities of antioxidant enzyme examined in the liver of animals receiving aflatoxin showed a statistically marked decrease compared to the control. In the group receiving aflatoxin with diosmin, the values for all parameters analyzed were closer to the control group. As a consequence of this convergence, there was no significant change in SOD and CAT activities and NO levels, but there was a statistically important change between the two groups in terms of MDA levels and GSH-Px activity (Table 2).
Kidney tissue
Contents of kidney MDA and NO and activities of kidney SOD and CAT in the diosmin-alone group are close to the control group. However, only the GSH-Px activity values in the diosmin-receiving group statistically increased compared to the control group in the diosmin-given group. The MDA and NO levels of the aflatoxin alone-receiving group showed a significant increase in comparison with the control group. SOD, CAT, and GSH-Px activities of the aflatoxin-alone-administered group observed a statistically remarkable decreasing tendency compared to the control group. In the group in which both compounds were given together, there was a marked improvement in this parameter and as a result of these developments, compared with the control group in all markers. From these changes, levels of MDA and activity of GSH-Px were statistically insignificant compared to the control group as a result of getting closer to each other. However, a statistically marked difference was still present between the two groups for NO levels and activities of SOD and CAT (Table 3).
Biochemical parameters
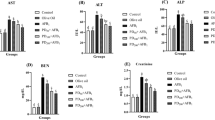
No statistically important differences were found in the serum biochemical parameters examined by the diosmin-alone group compared to the control group. Conversely, there was a statistically meaningful decrease in glucose, triglyceride, total protein, and albumin levels and a significant increase in cholesterol, BUN, creatinine, uric acid, LDH, AST, ALT, and ALP levels/activities in the aflatoxin-alone-receiving group compared to the control group. Most of the evaluated parameters were shown to be close to the control values in the groups receiving aflatoxin together with diosmin. When compared with the control group, there are statistically meaningful alterations in the levels of total protein and albumin in the co-administered group (Table 4).
Histopathological findings
Liver
Liver sections showed normal histo-architecture in the control group (Fig. 1a), but significant histopathological lesions were observed in the aflatoxin-treated group. That is, the most important finding of this study in the aflatoxin-alone group was hepatocellular hydropic/vacuolar degeneration and necrotic and dysplastic changes of hepatocytes. Hydropic (vacuolar) degeneration of hepatocytes was observed in the liver, especially in the periacinar regions. In some regions, the nuclei of some cells are pyknotic or eukaryotic and the cytoplasm is either lysed or eosinophilic. The nuclei and cytoplasm of some degenerative hepatocytes were very large, and this appearance was recorded as hepatocellular hypertrophy. The remark cords were fragmented due to severe degenerations (Fig. 1b–f). There was no histopathogical finding in the liver of rats treated with diosmin alone compared with the control group (Fig. 1g). Treatment with AF and diosmin showed marked improvement in liver histopathology (Fig. 1h).
Histological/histopathological findings in the rat liver tissue of control and experimental groups. a Control group depicts normal liver parenchyma with dilated central vein and hepatocytes. b–f Histopathological changes in the liver after administration of aflatoxin alone. b Vacuolization of hepatocytes. c Higher magnification of b. d, e Hepatocellular hypertrophy with eosinophilic granular cytoplasm degeneration, necrosis. f Cytoplasmic alteration (arrows); parenchymatous degeneration characterized by granular appearance of hepatocytes cytoplasm was observed. g The diosmin-treated group shows normal hepatocellular structure. h Combination with diosmin and aflatoxin in rats shows focal regeneration, stained with H&E. Bars × 20 (a, c, d–g), × 50 (b, h)
Kidney
Hematoxylin and eosin-stained sections prepared from the kidney tissue of the control group showed normal histological appearance (Fig. 2a). Significant histopathologic changes were observed in the kidney tissue of the aflatoxin-treated group. Hydropic and vacuolar degeneration, coagulation necrosis associated with degeneration of tubular architecture, moderate parenchymal tubular degeneration, manifested by tubal obstruction, epithelial swelling and blabbing, and fine granular appearance of the cytoplasm were fairly apparent in aflatoxin-treated rat (Fig. 2b–f). Pathologic findings were not detected in rats treated with diosmin alone in the kidney compared to the control group (Fig. 2g). Significant improvement was observed kidney histopathology of the animals in the diosmin group with AF. (Fig. 2h).
Histological/histopathological findings in the rat kidney tissue of control and experimental groups. a Kidneys of the control group showing normal histological structure of renal parenchyma. b–f Representative photomicrographs of sections from kidney samples given aflatoxin alone. Kidneys revealed dilation, congestion of renal blood vessels, proteinaceous casts in tubular lumen (yellow arrows), mild to moderate tubular necrosis, especially in the proximal tubule, tubular necrosis (green arrow with star), and vacuolization of epithelial lining renal tubule (orange arrow). g Normal histology of rat kidneys (glomeruli, tubules, interstitia, and blood vessels) was determined in the diosmin-alone-treated group. h The diosmin associated with aflatoxin-treated rat alleviates aflatoxin-induced renal histopathologic injury. Stained with H&E. Bars × 50 (c, d), × 100 (a, b, e–h)
Discussion
Aflatoxins are known to be one of the most dangerous natural pollutants due to their widespread exposure through food and feed (Marin et al. 2013; Aiko and Mehta 2015). Aflatoxins are widely distributed in tissues due to their toxicokinetic characteristics and cause various organ damages as well as dysfunctions/imbalance (Corcuera et al. 2012; Maren et al. 2013). The ability of oxidative stress formation among the toxic effect mechanisms of aflatoxin is also very important. In fact, these natural high toxic metabolites not only have the potential to create free radicals directly in the cells but also lead to the imparity of the cellular antioxidant defense mechanisms that indirectly cause such effects (O'Brien 1988; Hou et al. 2013). In fact, it was emphasized in previous studies that aflatoxin is effective in producing severe lipid peroxidation/oxidative stress in the biological system (Eraslan et al. 2013, 2015; Li et al. 2013; Abdel-Hamid and Firgany, 2015; Karabacak et al. 2015). Highly effective free radicals are formed during the metabolic processing of aflatoxins by cytochromal liver enzymes. The main reactions of aflatoxin metabolism in the liver are hydroxylation, oxidation, and demethylation (Wu et al. 2009). Most of the metabolites of AFB1 are able to be transformed to further metabolites. In other words, AFB1 is metabolized by cytochromes P450 to AFB1-8,9-exoepoxide and AFB1-8,9-endo-epoxide (Wild and Turne 2002). Cellular oxidative damage is formed when the produced ROS is above the antioxidant potential of the cell (Choi et al. 2010; El-Agamy 2010). AFB1-8,9-epoxide, an aflatoxin metabolite, binds tightly first with nucleic acids and other macromolecules, thus causing various undesirable damages as mentioned above (Guengerich et al. 1992; McLean and Dutton 1995; Dohnal et al. 2014). These highly reactive intermediate products can be detoxified in the form of reduced glutathione (GSH) conjugation by reactions mediated by glutathione-S-transferase (GST) (Larsson et al. 2003; Wu et al. 2009; Rawal et al. 2010).
A meaningful elevation in all sample materials on the MDA, NO, and 4-HNE level which is one of those often used as a marker (Michel et al. 2008; Guéraud et al. 2010; Csala et al. 2015) manifests that aflatoxin causes imbalance of the oxidant/antioxidant system in the group that received aflatoxin alone. As briefly mentioned above, the toxicity mechanisms that play a curial role in the formation of oxidative stress/lipid peroxidation are strongly related not only to the fact that aflatoxin is involved in the emergence of free radicals but also to the fact that the imbalance of cellular enzymatic/non-enzymatic antioxidant defense mechanism is effective. Free radicals formed at higher levels bind to unsaturated fatty acids on the cell membrane, thus triggering the formation of secondary products such as lipid hydroperoxides and hydroperoxides. This changes the activity of membrane enzymes and reduces the fluidity of the cell membrane. Meanwhile, MDA, NO, and 4-HNE are also generated as intermediates or final product (Ferrari et al. 1991; Michel et al. 2008; Niki 2009). On the other hand, aflatoxin-induced mitochondrial free radical formation is also one of the effective mechanisms (Mary et al. 2012). In addition, aflatoxin also directly causes the formation of high levels of nitric oxide and hydrogen peroxide (Asare et al. 2007; Ma et al. 2015). Within the scope of this study, an increase in MDA, NO and 4-HNE levels was detected in the samples analyzed. The increase in MDA, NO, and 4-HNE levels in tissue/plasma can be expressed by the formation of high-level free radicals through different mechanisms, or indirectly by the aflatoxin in cells, weakening cellular antioxidant defense mechanisms (Pimpukdee et al. 2004; Jodynis-Liebert et al. 2006; Li et al. 2013). Furthermore, there are changes in the activities of antioxidant enzyme in terms of inhibition in the aflatoxin-receiving group compared to the control group. Indeed, the reduction in antioxidant enzyme activity may be due to the depletion of antioxidant enzymes during free radical conversion to harmful, less harmful, and harmless compounds. Previous studies showed an increase in MDA, NO, and 4-HNE levels in animals treated with aflatoxin, while a decrease/increase in antioxidant enzyme activities was observed (Asare et al. 2007; Kanbur et al. 2011; Eraslan et al. 2013, 2015; Karabacak et al. 2015; Ma et al. 2015).
The natural flavonoid glycoside, diosmin, is formed by the conversion of hesperidin extracted from various citrus fruits (Anon 2004; Bogucka-Kocka et al. 2013). Diosmin prevents lipid peroxidation due to its effect of free radical scavenging (Kanaze et al. 2009; Campanero et al. 2010). Research data show that diosmin administration is a mitigating effect of aflatoxin-induced oxidative stress. It is understood that SOD, CAT, and GSH-Px activities and MDA, NO, and 4-HNE levels in the diosmin-treated group with aflatoxin are closer to the control group values. The main mechanism for reducing the oxidative stress of diosmin is presumably by reducing the level of free radicals by inducing the enzymatic antioxidant mechanism or neutralizing the free radicals by binding (Kanaze et al. 2009; Campanero et al. 2010). In addition, diosmin also has a protective effect against peroxidation by inhibiting the oxidation of the phospholipids of the membrane (Villa et al. 1992). Hence, the results of existing researches are parallel to those of other studies beforehand conducted with diosmin (Srinivasan and Pari 2012; Queenthy and John 2013; Arab et al. 2015; Germoush 2016). In addition, there was insignificant alteration in MDA, NO, and 4-HNE levels and antioxidant enzyme activities except for GSH-Px of kidney tissue compared with the control group in the group in which diosmin alone was given, suggesting that this compound alone did not show the effect of oxidative damage for the period and dose indicated as in other studies (Srinivasan and Pari 2012; Arab et al. 2015; Germoush, 2016).
Furthermore, there is no significant change in biochemical parameters in the group in which diosmin is given alone compared with the control group. Essentially, one of the essential mechanisms of the biochemical parameters changing the effect of aflatoxin involves the formation of a high amount of free radicals, which in turn causes damage to tissues and organs (Yang et al. 2000; Corcuera et al. 2012; Mary et al. 2012). In this study, changes in the activities of AST, ALT, ALP, and LDH and levels of cholesterol and triglyceride in the group receiving aflatoxin alone indicate that liver functions are impaired (El-Nekeety et al. 2011; Hathout et al. 2011; Karabacak et al. 2015). The tendency of total protein and albumin level to decrease in the living organism is also indicative of a deficiency in protein metabolism/synthesis (Hussain et al. 2009; Karabacak et al. 2015). Changes in BUN, creatinine, and uric acid levels may be a sign of damage to the kidney tissue (El-Nekeety et al. 2011; Nili-Ahmadabadi et al. 2011; Abdel-Hamid and Firgany 2015; Karabacak et al. 2015). A decrease in blood glucose concentration is usually expected when exposed to aflatoxin (Karabacak et al. 2015). It was observed that diosmin has a positive effect on the changes in some biochemical parameters and that these data are approaching to the data of the control group when compared to the aflatoxin-alone group in this group in which both substances are given together. This effect is directly or indirectly related to the radical scavenging activity of diosmin. All histopathological findings support these results. In addition, other studies using aflatoxin and other flavonoids achieved similar histopathologic findings (Souza et al. 1999; Preetha et al. 2006; El-Nekeety et al. 2011; Gupta and Sharma 2011; Vipin et al. 2017).
Conclusion
To summarize, in the current study, the antioxidant defense system is impaired in aflatoxin-treated rats. Oxidative stress/lipid peroxidation was alleviated in groups that received aflatoxin in combination with diosmin, so the possible toxicity that aflatoxin would cause was suppressed. This assessment supports the improvement in biochemical parameters and histopathological findings approaching the control group with oxidative stress indicators such as MDA, NO, and 4-HNE levels and antioxidant enzymes. For this reason, it has been concluded that diosmin used in this experimental study can remove, prevent, or alleviate certain adverse effects observed in aflatoxin poisoning. In the case of aflatoxin poisoning, diosmin may be used as a supportive agent in addition to other treatment options. If there is a risk of aflatoxin exposure, diosmin can be among the substances that may be preferred as a food supplement.
References
Abdel-Hamid AA, Firgany A-D (2015) Vitamin E supplementation ameliorates aflatoxin B1-induced nephrotoxicity in rats. Acta Histochem 117:767–779
Aiko V, Mehta A (2015) Occurrence, detection and detoxification of mycotoxins. J Biosci 40:943–954
Anon (2004) Monograph. Diosmin Altern Med Rev 9:308–311
Arab HH, Salama SA, Omar HA, Arafa e-SA, Maghrabi IA (2015) Diosmin protects against ethanol-induced gastric injury in rats: novel anti-ulcer actions. PLoS One 10:e0122417
Asare GA, Bronz M, Naidoo V, Kew MC (2007) Interactions between aflatoxin B1 and dietary iron overload in hepatic mutagenesis. Toxicology 234:157–166
Bennett JW, Klich M (2003) Mycotoxins. Clin Microbiol Rev 16:497–516
Bogucka-Kocka A, Woźniak M, Feldo M, Kockic J, Szewczyk K (2013) Diosmin-isolation techniques, determination in plant material and pharmaceutical formulations, and clinical use. Nat Prod Commun 8:545–550
Campanero MA, Escolar M, Perez G, Garcia-Quetglas E, Sadaba B, Azanza JR (2010) Simultaneous determination of diosmin and diosmetin in human plasma by ion trap liquid chromatography-atmospheric pressure chemical ionization tandem mass spectrometry: Application to a clinical pharmacokinetic study. J Pharm Biomed Anal 51:875–881
Choi KC, Chung WT, Kwon JK, Yu JY, Jang YS, Park SM, Lee SY, Lee JC (2010) Inhibitory effects of quercetin on aflatoxin B1-induced hepatic damage in mice. Food Chem Toxicol 48:2747–2753
Choudhary A, Verma RJ (2005) Ameliorative effects of black tea extract on aflatoxin-induced lipid peroxidation in the liver of mice. Food Chem Toxicol 43:99–104
Corcuera LA, Amézqueta S, Arbillaga L, Vettorazzi A, Touriño S, Torres JL, López de Cerain A (2012) A polyphenol-enriched cocoa extract reduces free radicals produced by mycotoxins. Food Chem Toxicol 50:989–895
Csala M, Kardon T, Legeza B, Lizák B, Mandl J, Margittai É, Puskás F, Száraz P, Szelényi P, Bánhegyi G (2015) On the role of 4-hydroxynonenal in health and disease. Biochim Biophys Acta 1852:826–838
Demet Ö, Oğuz H, Çelik I, Nizamlıoğlu F (1995) Pirinçte aflatoksin üretilmesi. Vet Bil Derg 11:19–23
Dohnal V, Wu Q, Kuča K (2014) Metabolism of aflatoxins: key enzymes and interindividual as well as interspecies differences. Arch Toxicol 88:1635–1644
El-Agamy DS (2010) Comparative effects of curcumin and resveratrol on aflatoxin B (1)-induced liver injury in rats. Arch Toxicol 84:389–396
El-Nekeety AA, Mohamed SR, Hathout AS, Hassan NS, Aly SE, Abdel-Wahhab MA (2011) Antioxidant properties of Thymus vulgaris oil against aflatoxin-induce oxidative stress in male rats. Toxicon 57:984–991
Eraslan G, Kanbur M, Aslan Ö, Karabacak M (2013) The antioxidant effects of pumpkin seed oil on subacute aflatoxin poisoning in mice. Environ Toxicol 28:681–688
Eraslan G, Kanbur M, Karabacak M, Sarica Soyer Z (2015) Ameliorative effect of colostrum on aflatoxin-induced biochemical changes in mice. Fresenius Environ Bull 24:546–551
Fairbanks VF, Klee GG (1987) Biochemical aspect of hematology. In: Tietz NW (ed) Fundamentals of clinical chemistry, 3rd edn. WB Saunders, Philadelphia, pp 803–806
Ferrari R, Ceconi C, Curello S, Cargnoni A, Alfieri O, Pardini A, Marzollo P, Visioli O (1991) Oxygen free radicals and myocardial damage: protective role of thiol-containing agents. Am J Med 91:95–105
Germoush MO (2016) Diosmin protects against cyclophosphamide-induced liver injury through attenuation of oxidative stress, inflammation and apoptosis. Int J Pharmacol 12:644–654
Guengerich FP, Raney KD, Kim DH, Shimada T, Meyer DJ, Ketterer B, Harris TM (1992) Oxidation and conjugation of aflatoxins by humans and experimental animals. Prog Clin Biol Res 374:157–165
Guéraud F, Atalay M, Bresgen N, Cipak A, Eckl PM, Huc L, Jouanin I, Siems W, Uchida K (2010) Chemistry and biochemistry of lipid peroxidation products. Free Radic Res 44:1098–1124
Gupta R, Sharma V (2011) Ameliorative effects of tinospora cordifolia root extract on histopathological and biochemical changes induced by aflatoxin-b(1) in mice kidney. Toxicol Int 18:94–98
Gutteridge JM (1995) Lipid peroxidation and antioxidants as biomarkers of tissue damage. Clin Chem 41:1819–1828
Halliwell B (1999) Antioxidant defense mechanisms: from the beginning to the end (of the beginning). Free Radic Res 31:261–272
Halliwell B, Chirico S (1993) Lipid peroxidation: its mechanism, measurement, and significance. Am J Clin Nutr 57:715–724
Hathout AS, Mohamed SR, El-Nekeety AA, Hassan NS, Aly SE, Abdel-Wahhab MA (2011) Ability of Lactobacillus casei and Lactobacillus reuteri to protect against oxidative stress in rats fed aflatoxins-contaminated diet. Toxicon 58:179–186
Hou YJ, Zhao YY, Xiong B, Cui XS, Kim NH, Xu YX, Sun SC (2013) Mycotoxin-containing diet causes oxidative stress in the mouse. PLoS One 8:e60374
Hussain S, Khan MZ, Khan A, Javed I, Asi MR (2009) Toxico-pathological effects in rats induced by concurrent exposure to aflatoxin and cypermethrin. Toxicon 53:33–41
Ingawale DK, Mandlik SK, Naik SR (2014) Models of hepatotoxicity and the underlying cellular, biochemical and immunological mechanism(s): a critical discussion. Environ Toxicol Pharmacol 37:118–133
Jain D, Bansal MK, Dalvi R, Upganlawar A, Somani R (2014) Protective effect of diosmin against diabetic neuropathy in experimental rats. J Integr Med 12:35–41
Jodynis-Liebert J, Matławska I, Bylka W, Murias M (2006) Protective effect of Aquilegia vulgaris L. on aflatoxin B(1)-induced hepatic damage in rats. Environ Toxicol Pharmacol 22:58–63
Kanaze FI, Termentzi A, Gabrieli C, Niopas I, Georgarakis M, Kokkalou E (2009) The phytochemical analysis and antioxidant activity assessment of orange peel (Citrus sinensis) cultivated in Greece-Crete indicates a new commercial source of hesperidin. Biomed Chromatogr 23:239–249
Kanbur M, Eraslan G, Sarıca Soyer Z, Aslan O (2011) The effects of evening primrose oil on lipid peroxidation induced by subacute aflatoxin exposure in mice. Food Chem Toxicol 49:1960–1964
Karabacak M, Eraslan G, Kanbur M, Sarıca Soyer Z (2015) Effects of Tarantula cubensis D6 on aflatoxin-induced injury in biochemical parameters in rats. Homeopathy 104:205–210
Larsson P, Persson E, Tyden E, Tjalve H (2003) Cell-specific activation of aflatoxin B1 correlates with presence of some cytochrome P450 enzymes in olfactory and respiratory tissues in horse. Res Vet Sci 74:227–233
Li Y, Ma QG, Zhao LH, Guo YQ, Duan GX, Zhang JY, Ji C (2013) Protective efficacy of alpha-lipoic acid against aflatoxinB1-induced oxidative damage in the liver. Asian-Australas J Anim Sci 27:907–915
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Luck HS (1965) Catalase. In: Bergmeyer HU (ed) Methods of enzymatic analysis. Academic Press, London, pp 885–894
Ma Q, Li Y, Fan Y, Zhao L, Wei H, Ji C, Zhang J (2015) Molecular mechanisms of lipoic acid protection against aflatoxin B1-induced liver oxidative damage and inflammatory responses in broilers. Toxins (Basel) 7:5435–5447
Marin S, Ramos AJ, Cano-Sancho G, Sanchis V (2013) Mycotoxins: occurrence, toxicology, and exposure assessment. Food Chem Toxicol 60:218–237
Mary VS, Theumer MG, Arias SL, Rubinstein HR (2012) Reactive oxygen species sources and biomolecular oxidative damage induced by aflatoxin B1 and fumonisin B1 in rat spleen mononuclear cells. Toxicology 302:299–307
McLean M, Dutton MF (1995) Cellular interactions and metabolism of aflatoxin: an update. Pharmacol Ther 65:163–192
Michel F, Bonnefont-Rousselot D, Mas E, Drai J, Thérond P (2008) Biomarkers of lipid peroxidation: analytical aspects. Ann Biol Clin (Paris) 66:605–620
Miller GL (1959) Protein determination for large numbers of samples. Anal Chem 31:964
Nabney J, Nesbitt BF (1965) A spectrophotometric method for determining the aflatoxins. Analyst 90:155–160
Niki E (2009) Lipid peroxidation: physiological levels and dual biological effects. Free Radic Biol Med 47:469–484
Nili-Ahmadabadi A, Tavakoli F, Hasanzadeh G, Rahimi H, Sabzevari O (2011) Protective effect of pretreatment with thymoquinone against Aflatoxin B(1) induced liver toxicity in mice. Daru 19:282–287
O'Brien PJ (1988) Free-radical-mediated chemical carcinogenesis. Ann N Y Acad Sci 534:552–564
Ohkawa H, Ohishi N, Yagi K (1978) Reaction of linoleic acid hydroperoxide with thiobarbituric acid. J Lipid Res 19:1053–1057
Opara EC (2006) Oxidative stress. Dis Mon 52:183–198
Paglia DE, Valentine WN (1967) Studies on the quantitative and qualitative characterization of erythrocyte glutathione peroxidase. J Lab Clin Med 70:158–169
Pimpukdee K, Kubena LF, Bailey CA, Huebner HJ, Afriyie-Gyawu E, Phillips TD (2004) Aflatoxin-induced toxicity and depletion of hepatic vitamin A in young broiler chicks: protection of chicks in the presence of low levels of NovaSil PLUS in the diet. Poult Sci 83:737–744
Pisoschi AM, Pop A (2015) The role of antioxidants in the chemistry of oxidative stress: a review. Eur J Med Chem 97:55–74
Pitt JI (2000) Toxigenic fungi and mycotoxins. Br Med Bull 56:184–192
Preetha SP, Kanniappan M, Selvakumar E, Nagaraj M, Varalakshmi P (2006) Lupeol ameliorates aflatoxin B1-induced peroxidative hepatic damage in rats. Comp Biochem Physiol C Toxicol Pharmacol 143:333–339
Queenthy SS, John B (2013) Diosmin exhibits anti-hyperlipidemic effects in isoproterenol induced myocardial infarcted rats. Eur J Pharmacol 718:213–218
Rawal S, Kim JE, Coulombe R Jr (2010) Aflatoxin B1 in poultry: toxicology, metabolism and prevention. Res Vet Sci 89:325–331
Rehman MU, Tahir M, Khan AQ, Khan R, Lateef A, Hamiza OO, Ali F, Sultana S (2013) Diosmin protects against trichloroethylene-induced renal injury in Wistar rats: plausible role of p53, Bax and caspases. Br J Nutr 110:699–710
Roberts BA, Patterson DS (1975) Detection of twelve mycotoxins in mixed animal feedstuffs, using a novel membrane clean up procedure. J Assoc Off Anal Chem 58:1178–1181
Roze LV, Hong SY, Linz JE (2013) Aflatoxin biosynthesis: current frontiers. Annu Rev Food Sci Technol 4:293–311
Senthamizhselvan O, Manivannan J, Silambarasan T, Raja B (2014) Diosmin pretreatment improves cardiac function and suppresses oxidative stress in rat heart after ischemia/reperfusion. Eur J Pharmacol 736:131–137
Shotwell OL, Hesseltine CW, Stubblefield RD, Sorenson WG (1966) Production of aflatoxin on rice. Appl Microbiol 14:425–428
Sirajudeen M, Gopi K, Tyagi JS, Moudgal RP, Mohan J, Singh R (2011) Protective effects of melatonin in reduction of oxidative damage and immunosuppression induced by aflatoxin B1-contaminated diets in young chicks. Environ Toxicol 26:153–160
Souza MF, Tomé AR, Rao VS (1999) Inhibition by the bioflavonoid ternatin of aflatoxin B1-induced lipid peroxidation in rat liver. J Pharm Pharmacol 51:125–129
Srinivasan S, Pari L (2012) Ameliorative effect of diosmin, a citrus flavonoid against streptozotocin-nicotinamide generated oxidative stress induced diabetic rats. Chem Biol Interact 195:43–51
Sun Y, Oberley LW, Li Y (1988) A simple method for clinical assay of superoxide dismutase. Clin Chem 34:497–500
Tanrikulu Y, Sahin M, Kismet K, Kilicoglu SS, Devrim E, Tanrikulu CS, Erdemli E, Erel S, Bayraktar K, Akkus MA (2013) The protective effect of diosmin on hepatic ischemia reperfusion injury: an experimental study. Bosn J Basic Med Sci 13:218–224
Tracey WR, Tse J, Carter G (1995) Lipopolysaccharide-induced changes in plasma nitrite and nitrate concentrations in rats and mice: pharmacological evaluation of nitric oxide synthase inhibitors. J Pharmacol Exp Ther 272:1011–1015
Villa P, Cova D, De Francesco L, Guaitani A, Palladini G, Perego R (1992) Protective effect of diosmetin on in vitro cell membrane damage and oxidative stress in cultured rat hepatocytes. Toxicology 73:179–189
Vipin AV, Raksha Rao K, Nawneet KK, Anu Appaiah KA, Venkateswaran G (2017) Protective effects of phenolics rich extract of ginger against Aflatoxin B1-induced oxidative stress and hepatotoxicity. Biomed Pharmacother 91:415–424
Vladimirov IuA (1998) Free radicals and antioxidants. Vestn Ross Akad Med Nauk 7:43–51
Vladimirov YA, Proskurnina EV (2009) Free radicals and cell chemiluminescence. Biochemistry (Mosc) 74:1545–1566
Wang JS, Groopman JD (1999) DNA damage by mycotoxins. Mutat Res 424:167–181
Wild CP, Turner PC (2002) The toxicology of aflatoxins as a basis for public health decisions. Mutagenesis 17:471–481
Winterbourn CC, Hawkins RE, Brian M, Carrell RW (1975) The estimation of red cell superoxide dismutase activity. J Lab Clin Med 85:337–341
Wu Q, Jezkova A, Yuan Z, Pavlikova L, Dohnal V, Kuca K (2009) Biological degradation of aflatoxins. Drug Metab Rev 41:1–7
Yang CF, Liu J, Shen HM, Ong CN (2000) Protective effect of ebselen on aflatoxin B1-induced cytotoxicity in primary rat hepatocytes. Pharmacol Toxicol 86:156–161
Yoshioka T, Kawada K, Shimada T, Mori M (1979) Lipid peroxidation in maternal and cord blood and protective mechanism against activated-oxygen toxicity in the blood. Am J Obstet Gynecol 135:372–376
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Philippe Garrigues
Rights and permissions
About this article
Cite this article
Eraslan, G., Sarıca, Z.S., Bayram, L.Ç. et al. The effects of diosmin on aflatoxin-induced liver and kidney damage. Environ Sci Pollut Res 24, 27931–27941 (2017). https://doi.org/10.1007/s11356-017-0232-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-017-0232-7