Abstract
In addition to physicochemical methods, benthic foraminifera have become an essential tool for the assessment of polluted environments. The main objectives of the present work were to study the distribution of benthic foraminifera along the coastline of Skhira and Gabes (southern coast of Gulf of Gabes) and to predict the impact of pollution on these organisms. Thirty-one samples were studied and a polluted area was delimited by chemical analysis, where heavy metal, fluoride, phosphorus, nitrogen, and COT contents are very high. Thirty-four species of benthic foraminifera were identified and their response to pollution is very remarkable, in which their distribution shows barren area, corresponding to the highly polluted area. Away from the contaminated area, the density and the diversity of these organisms increase. Statistical analyses (principal component analysis (PCA)/FA and matrix correlation) show a possible control of these pollutants on biotic indices (with negative correlation), in addition to the presence of tolerant and sensitive species to pollution. A variety of test malformations were noticed especially in Ammonia beccarii, Peneroplis planatus, Sorites variabilis, and Adelosina pulchella. Unpolluted stations were dominated by species characteristic of shallow water environments with sandy sediment such Ammonia parkinsoniana, Triloculina trigonula, Quinqueloculina agglutinans, and P. planatus.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Foraminifera are unicellular Protista that construct shells of one and many chambers. They are the most abundant protozoa in most marine and brackish water habitat with more than 40,000 cited species (Loeblich and Tappan 1988). During the last few decades, many studies dealing with benthic foraminifera as bioindicators of marine pollution have been carried out. Several factors such as (i) hard exoskeleton which records fundamental environmental changes, (ii) small size and higher abundance in small size sample, (iii) wide distribution over all marine environments, (iv) high taxonomic diversity, and (v) very short reproductive cycles can justify the use of those Protista as biomarkers (Coccioni 2000). Studies of pollution effects on benthic foraminifera were initiated by Resig (1960) and Watkins (1961) (Armynot du Châtelet et al. 2004), although effects on foraminifera had been investigated in all kinds of areas exposed to different types of pollution such as industrial, domestic, and agricultural waste, oil, or heavy metal contamination (Debenay et al. 2001; Armynot du Châtelet et al. 2004; Bergin et al. 2006; Ferraro et al. 2006; Yalçýn et al. 2008; Aloulou et al. 2012; Donnici et al. 2012; Meriç et al. 2012). The reaction of benthic foraminifera to pollutant may be expressed by (i) morphological abnormalities of their test structure (Yanko et al. 1994; Geslin et al. 1998), (ii) by a change in the density and the diversity of the assemblage (Bergin et al. 2006; Ferraro et al. 2006; Valenti et al. 2008; Vilela et al. 2014), or (iii) by colored shells of specimens (Yalçýn et al. 2008; Meriç et al. 2012).
Although recent benthic foraminifera have been widely studied in the Mediterranean Sea, few studies have been carried out on the Gulf of Gabes. They were reported, the first time, by Glaçon (1963), then by Blanc-Vernet in 1974. During Mer Pélagienne campaign, Blanc-Vernet (1974) studied both benthic and pelagic foraminifera in the Gulf of Gabes sensu lato, but within a description or the systematic of those species. However, studies on the utilities of benthic foraminifera as bioindicators in the Gulf of Gabes, Tunisia, have been neglected, and the only published work in this issue was edited by Aloulou et al. (2012). Through this study, the author showed a clear relation between pollution mainly by phosphogypsum sewage and fishing activities and the density and the diversity of the benthic foraminiferal assemblages in the coastline of Sfax—Skhira (northern part of the Gulf of Gabes).
The main objectives of this work were (i) to examine the distribution of heavy metal, fluoride, total phosphorus, nitrogen, and total organic carbon (TOC) concentrations in the surface sediment of the coast of Ghannouch—Gabes (southern part of the Gulf of Gabes, Tunisia), in order to delimit polluted area, (ii) to determine the assemblage of benthic foraminifera, and (iii) to identify a possible relation between pollutant concentrations and benthic foraminiferal distribution. This study is the first one on this area, and no similar study was carried out before.
Study area
Geographical setting
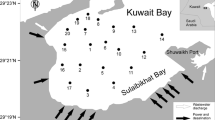
The Gulf of Gabes is an eastward-facing embayment area located in the Tunisia coast (Fig. 1). It is 100 km long and 100 km wide (Sanchez-Arcilla et al. 2010). The gulf basin is very shallow, reaching only 50-m depths and 110 km offshore (Hattab et al. 2013). It is boarded by a subdued topography formed mainly by low plateaus (20–70 m) and plains (1–5 m) (Oueslati 1992). The climate of this region is characterized by temperate, humid, and hot Mediterranean air coming from the east part, beside the subtropical, dry, and hot with Saharan dust coming from the southwest part (Essersi et al. 2010). The average rainfall ranges between 250 mm/year in the north and 100–150 mm/year in the south, although the rainfall rate is highly irregular (Sanchez-Arcilla et al. 2010). The tide in this region is semidiurnal; the tidal range is greatest in the southern Gulf and decreases at the edges and its amplitude is the highest in the Mediterranean Sea, which gets to 1.7 m (Sammari et al. 2006). The gulf supports high fishery productivity (which represents 65 % of the national fish production in Tunisia) and serves as a nursery, feeding, and breeding ground for numerous populations of fish and crustaceans (Hattab et al. 2013).
Pollution condition
In the last years, the Gulf of Gabes has suffered high anthropic pressures due to the huge industrial activity, in addition to urban development. In Gabes city, quantities of phosphogypsum, from the phosphoric acid and chemical product industry, are released into the marine environment (Rabaoui et al. 2014). According to Drira (2009), production of dry phosphogypsum is estimated to be 5 tonnes per 1 tonne of P2O5. In addition, other potential heavy metal sources such as lead, ceramic, textile, building material factories, and harbor’s activities exist in this area.
The coastline of Skhira City hosts also a phosphate fertilizer factory. While phosphogypsum waste is stored near the factory, effluents from treatment unit are released into the sea and present risks of coast pollution (Aloulou Ben Ayed 2010).
Materials and methods
In July 2010, superficial sediments samples were collected from 31 stations (Fig. 1); five from Skhira coastal shelf (stations S) and 26 stations from Ghannouch—Gabes area (stations L and R). Sampling stations “S” and “L” were selected along the coast (about 10 m from the coastline), which the depth of seawater not exceeding 0.5 m. However, sampling station “R” was taken around the commercial port of Ghannouch and the fishing port of Gabes, using a Peterson grab sampler where seawater depth is ranged between 5 and 12 m (Table 1). Only the top 2 cm of the surface sediment was sampled, sealed in clean polyethylene bags, and stored at 4 °C until analysis in laboratory.
Grain-size analysis
The samples for the grain-size analysis were treated with an H2O2 solution, and then dried at 40 °C and sieved using AFNOR mesh-type sieves varied from 2 mm to 63 microns. Shepard (1954) grain size classification was followed. The results of this analysis (gravels, sand, and mud percentages) are reported in Table 1.
Geochemical analysis
For the geochemical characterization, sediments samples were dried at 50 °C and pass through a 63-μm sieve. For heavy metal and total phosphorus concentrations, samples were digested with HNO3 (65 %)/HCl (37 %) solution. Pb, Zn, Cd, Cu, Cr, and Al concentrations were measured using absorption atomic spectrometer, and the detection limits (mg/l) were 0.005 for Cd, 0.05 for Cr, 0.022 for Cu, 0.05 for Pb, 0.008 for Zn, and 0.013 for Al. Total phoshorus content was carried out using a Hitachi U-2000 spectrometer, with a wavelength of 880 nm following the NFT 90.023. For fluoride determination, sediment samples (0.5–1 g) were dissolved in 3 ml of HCl solution (6 M) and diluted to 100 ml with deionized water and an aliquot of the extract was used for the determination of fluoride by a specific ion electrode (Abu Hilal 1985). The total organic carbon (TOC) content in superficial sediment was determined by measurements of the amount of dichromate which did not interact with the sample using titration process (CEAEQ 2006). The content of total nitrogen was determined by mineralization of sediment sample by sulfuric acid, followed by a titration with HCl solution (0.01 M), following NFT 90.110.
In order to establish further accuracy for the contamination of the sediment, enrichment factor (EF) is used to evaluate the degree of heavy metal contamination sediments of the Gulf of Gabes. EF presents state of natural or anthropogenic enrichments of sediment by heavy metals, which is calculated as follows (Choi et al. 2008; Garcia et al. 2008):
where X/Al is the ratio of the concentration of element X to Al in both contaminated and uncontaminated sediment. The EF value of 1 indicates predominant natural origin of the element in the sediment. The EF above 1.5 indicates enrichment of natural processes (e.g., biota contribution) or anthropogenic influences.
Foraminiferal analysis
For benthic foraminifera analysis, a constant volume of 50 cm3 of sediment was stored in 2 g/l ethanol-rose bengal solution for 48 h to distinguish between living and dead foraminifera (Walton 1952). Then, samples were washed through a set of sieves (63, 125, 250, and 500 μm) and oven-dried at 50 °C. At each station, three hundred specimens from all fraction together were hand-picked and identified following the generic classification of Loeblich and Tappan (1988), Cimerman and Langer (1991), Hottinger et al. (1993), and Sgarrella and Moncharmont-Zei (1993). According to Fatela and Taborda (2002), this number is considered sufficient for statistical analysis.
Due to low number of living individuals (1–10 in the 300 picked specimens), the total assemblage (dead and living) showed to be the best to use for statistical purposes. The dead foraminifera provide a large database, and the total populations are often statistically identical and only diverge when living/total becomes large (Samir and El-Din 2001). Therefore, Armynot du Châtelet et al. (2004) suggested the use of total assemblage due to possible seasonal cycles affecting living foraminifera.
Foraminiferal parameters were estimated using (a) foraminiferal density defined as the number of specimens per 50 cm3, (b) species diversity (S) as the number of species in samples (Murray 1991, 2006), and (c) the Fisher α index which considers both the number of species and specimens in an assemblage (Fisher et al. 1943).
The species diversity and the Fisher α index were calculated using the Palaeontological Statistics (PAST) data analysis package (version 1.68).
Statistical analysis
For statistical analysis, species occurring in more than 5 % of the total assemblage, at least one sample was taken into consideration in order to reduce background noise (Armynot du Châtelet et al. 2004). Infrequent occurring taxa (<5 % relative abundance) were omitted as they have insignificant effects on the formation of the major groups (Frezza and Carboni 2007; Romano et al. 2008). Since the probability distribution of the raw data, both biotic and abiotic, fails the Kolmogorov-Smirnov test for normality, a logarithmic transformation Log (1 + X) was performed (Frontalini and Coccioni 2008). This transformation was essential to remove the effects of orders of magnitude difference between variables, to avoid negative numbers, to normalize the data, and to increase the importance of smaller values (Burone et al. 2006; Frontalini and Coccioni 2008).
Relationship among transformed variables (assemblage’s parameters, heavy metals, fluoride, total phosphorus, N, and TOC) was tested using Spearman’s rho correlation with statistical significance set at p < 0.05.
Multivariate statistical analyses (Q-mode hierarchical cluster analysis (HCA) and principal component analysis (PCA)) were performed using SPSS statistical software (version 16.0 for Windows).
The HCA, using Ward’s method as the amalgamation rule and the squared Euclidean distance as similarity measurement (Romano et al. 2008), was applied for the classification of stations (Q-mode) into associations (clusters). Each cluster is characterized by a similar spatial distribution pattern (Samir et al. 2003). PCA was applied to experimental data in order to examine the relationship among them. This statistical analysis attempts to identify underlying factors that explain the pattern of correlation within a set of observed variables. It is used to reduce data, in a way to identify a small number of factors that explain most of the variance observed in a much larger number of manifested variables (Frezza and Carboni 2007).
Results
Grain-size distribution patterns
Grain size analysis showed that superficial sediment of both areas is composed mainly of sand (between 2 mm and 63 μm), which widely ranges between 10.46 and 99 %. The mud fraction (<63 μm) is dominant in stations in front of commercial harbor of Ghannouch (Table 1). The coarser fraction was mad up, primarily, of gravels and/or fragments of mollusks and gastropods. This sandy facies can be the result of the hydrodynamic proprieties of the intertidal area, where the combination of the wave and tide action produces high energy preventing the deposition of fine fraction (Aloulou et al. 2012).
Chemical analysis
The results of the chemical analysis are given in Table 2. The concentration in each studied element varied highly among the sampling sites in both areas. Ranges of heavy metals in sediments are 0–11 ppm for Pb, 6.22–10,175 ppm for Zn, 1.76–120 ppm for Cr, 1.5–58 ppm for Cu, and 0–1225 ppm for Cd. Fluoride concentrations varied from 0.003 to 7.2 %, while total phosphorus content ranges between 0.17 and 25,100 ppm. The highest concentrations of Cd, Cr, Cu, Zn, F, and P were found between the commercial harbor of Ghannouch and the fishing harbor of Gabes, both in offshore and coastline stations. In addition to this area, lead showed maximum concentrations at stations L9 and L10. Total organic carbon (TOC) concentration of the superficial sediment varied between 0.1 and 4 %, with a general tendency for higher values in fine- to silt-sized fraction. The spatial distribution of nitrogen showed high contents in stations of the southern sector of Ghannouch—Gabes area, compared to other sampled sites.
Benthic foraminifera
Total foraminiferal assemblages were made up almost of benthic species usually living in shallow water as the study area. On the whole, 34 species (24 porcelaneous, 7 hyaline, and 3 agglutinated) have been identified, belonging to 15 genera and 11 families (Plate 1 and 2). Benthic foraminifera were found in 24 of the total 31 samples, where foraminifer’s density varies from 87 to 37,720 specimens per 50 cm3. Species diversity (S), Fisher’s alpha (α) index, Simpson index, and evenness index were determined for each site (Fig. 2). Species richness (S) and Fisher alpha index show higher values in the extremities of the study area. Simpson index and equitability index vary respectively from 0.823 to 0.932 and from 0.765 to 0.90, while evenness ranges between 0.49 and 0.733. Those indexes feature the same distribution patterns, in which the lowest values are observed in stations L2, R3, and R8.
Scale bar 100 μm. 1: Ammonia beccarii Linné, 1758. 2: Ammonia parkinsoniana d’Orbigny, 1839. 3: Elphidium crispum Linné, 1758. 4: Elphidium aculeatum d’Orbigny, 1846. 5: Elphidium advenum, Cushman, 1922. 6: Peneroplis planatus Fichtel and Moll, 1798. 7: Peneroplis pertusus Froskal, 1775. 8: Coscinospira hemprichii Ehrenberg, 1839. 9: Sorites variabilis Lacroix, 1941. 10: Rosalina bradyi Cushman, 1915. 11: Massilina secans d’Orbigny, 1826. 12: Lachlanella variolata d’Orbigny, 1826. 13: Quinqueloculina lata Terquem, 1876. 14: Quinqueloculina agglutinans d’Orbigny, 1839a. 15: Quinqueloculina variolata d’Orbigny, 1826. 16: Quinqueloculina berthelotiana d’Orbigny, 1839. 17: Quinqueloculina berthelotiana d’Orbigny var. wiesneri, Le Calvez et Le Calvez, 1958
Scale bar 100 μm. 1: Quinqueloculina rugosa d’Orbigny, 1839. 2: Quinqueloculina vulgaris d’Orbigny, 1826. 3: Quinqueloculina costata d’Orbigny, 1826. 4: Quinqueloculina limbata d’Orbigny, 1826. 5: Triloculina trigonula Lamarck, 1804. 6: Clavulina angularis d’Orbigny, 1826. 7: Adelosina longirostra d’Orbigny, 1826. 8: Adelosina mediterranensis Le Calvez, J. & Y., 1958. 9: Adelosina italica Terquem, 1878. 10: Adelosina pulchella d’Orbigny, 1846. 11: Spiroloculina excavata d’Orbigny, 1846. 12: Spiroloculina antillarum d’Orbigny, 1839. 13: Spiroloculina depressa d’Orbigny, 1826. 14: Spiroloculina cf. S. angulata Cushman. 15: Sigmoilinata costata Schlumberger, 1893. 16: Buccella granulata Di Napoli Alliata, 1952. 17: Textularia agglutinans d’Orbigny, 1839
Only 19 species show relative abundances exceeding 5 % in at least one sample. Among them, six species are more abundant: Ammonia parkinsoniana, Ammonia beccarii, Adelosina longirostra, Peneroplis planatus, Quinqueloculina agglutinans, and Triloculina trigonula.
The abundance of abnormal specimens in the total assemblage does not exceed 5 % in all samples (4.60 % at station R9), and only four species showed morphological deformation: A. beccarii, P. planatus, Sorites variabilis, and Adelosina pulchella. According to Sharifi et al. (1991), Yanko et al. (1994), and Frontalini and Coccioni (2008), four modes of abnormalities have been identified: (i) Siamese twins, (ii) reduced chambers size, (iii) aberrant chambers chaps, and (iv) distorted chamber arrangement or change in coiling (Plate 3). In addition to morphological deformation, porcelain worn test were distinguished in the southern part of Ghannouch—Gabes area.
Statistics
Spearman’s rho correlation was calculated in order to establish relationships among geochemical parameters, foraminiferal taxa, and biotic parameters (Table 3). This matrix shows high coefficient of correlation (up to 0.9) among geochemical parameters such as Zn, Cd, Cu, C, Pb, P, F, and N, suggesting an association possibility between them and a similar source input. However, biotic parameters (density, specific richness, and Fisher α index) are negatively correlated with chemical elements except for P and pH of the sediment, indicating the toxicity of those elements. Few species show strong correlation (both positive and negative) with one or several chemical parameters. Total organic carbon presents negative correlation with A. beccarii (r = −0.600), Ad. longirostra (r = −0.424), and Quinqueloculina rugosa (r = −0.503), none for the opportunistic aspect of those species in the marine environment. However, Rosalina bradyi is positively correlated with TOC (r = 0.411), indicating an opportunistic behavior by taking advantage of nutrient availability under adverse conditions due to pollution (Romano et al. 2009). Peneroplis planatus, Elphidium crispum, and S. variabilis show negative relationships with all elements, except Pb and TOC. The pH of the sediment has a negative correlation coefficient with Buccella granulata (r = −0.527), while Q. agglutinans is negatively correlated only with fluoride (r = −0.443). Spiroloculina antillarum has negative coefficients with all elements studied, except the pH of the sediment. Thus, these species could be considered as sensitive species to pollution. In fact, the presence of pollutants in marine environment could play an important and negative role in the distribution, in the fulfillment, and even in the biological functions of these species. However, R. bradyi, Quinqueloculina berthelotiana var. weinsri, and Sigmoilinata costata have positive correlations with most of the chemical elements. Thus, Pb has a high correlation with R. bradyi (r = 0.568) and Q. berthelotiana var. weinsri (r = 0.560). Sigmoilinata costata is positively correlated with the Zn (r = 0.542), Cr (r = 0.510), Cu (r = 0.579), NTK (r = 0.627), P (r = 0.515), Cd (r = 0.500), and F (r = 0.443). The positive correlation of R. bradyi, Q. beth. var. weinsri, and S. costata with Pb and S. costata with Cd shows the resistance aspect of these species towards those elements, which are considered non-essential. Nevertheless, positive affinity of essential elements (Cu, Cr, Zn), F, P, and N with these species suggests their important roles in the biological functions of the metabolism of these species (Donnici et al. 2012).
Despite their relative abundances in the samples, A. parkinsoniana, Ad. longirostra, and T. trigonula show no significant correlation with contaminants. Therefore, these species cannot be regarded as appropriate indicators for pollution in our study area.
Principal component analysis (PCA) is an effective method to evaluate biotic and abiotic parameters obtained from sediment samples and to identify the factor influencing each one. Two principal components have been identified, explaining together 54.83 % of the data variance, especially the first and the second PC account for 38.11 and 16.71 % of the total variance (Fig. 3). The dominant elements in the first PC were the pollutants (heavy metals, F, TOC, N, P), along with some foraminiferal species such as R. bradyi, S. costata, and Quinqueloculina berthelotiana var. weinsri. On the opposite, the highest values of component 2 were recorded by biotic parameters (density, richness, and Fisher α index), in addition to P. planatus, S. variabilis, A. beccarii, Ad. longirostra, Adelosina mediterranensis, and Q. rugosa. Ammonia parkinsoniana, E. crispum, Q. berthelotiana, and pH sediment are negatively correlated with both components 1 and 2.
PCA ordination diagram of samples based on biotic and abiotic selected parameters. Abec: Ammonia beccarii; Apark: Ammonia parkinsoniana; Along: Adelosina longirostra; Amedit: Adelosina mediterranensis; Apul: Adelosina pulchella; Bgran: Buccella granulata; Ecris: Elphidium crispum; Eacul: Elphidium aculeatum; Ppla: Peneroplis planatus; Qagg: Quinqueloculina agglutinans; Qrug: Quinqueloculina rugosa; Qberth: Quinqueloculina berthelotiana; Qberthvarwie: Quinqueloculina berthelotiana var. wiesneri; Rbrad: Rosalina bradyii; Scost: Sigmoilinata costata; Svar: Sorites variabilis; Sant: Spiroloculina antillarum; ScfSang: Spiroloculina cf. S. angulata; Ttrig: Triloculina trigonula
The Q mode HCA (Ward method) has identified two major clusters A and B, based on chemical parameters and benthic foraminifera distribution and abundance (Fig. 4). The cluster A is associated with stations R9, R10, and R11, located next to the Ghannouch commercial port. This cluster is mainly known by the high concentrations of pollutants and the significant percentage of the fine fraction of the sediment (68 % on average). The most abundant species between the three stations are Ad. longirostra and T. trigonula, with a mean of 14.41 and 10.81 %, respectively.
The cluster B, containing all other stations, is divided into two clusters of lower hierarchical order B1 and B2: The subgroup B1, associated with coastal stations, is also divided into two subgroups; B1a and B1b. B1a consists of the coastal sites in Skhira (S1, S2, S3, S4, S5), in addition to L1 and L2 points. These points are characterized by low fluoride concentration relative to other stations. Abundant foraminifera species are A. parkinsoniana (14 % on average), followed by Ad. longirostra, A. beccarii, and P. planatus (13, 11.95, and 9.66 %, respectively). B1b is formed by L6, L9, and L10, whose abundant species are A. parkinsoniana (18.33 %), P. planatus (13.11 %), Ad. longirostra (10.11 %), and T. trigonula (7.67 %). Subgroup B2 is also divided into two sub groups B2a and B2b. B2a, associated with R12, R14, R17, and R18 stations, is characterized by the abundance of Q. agglutinans (11.83 % on average) and S. variabilis (6.33 %), in addition to Ad. longirostra (13.08 % on average). Finally, B2b subgroup consists of stations R2, R3, R7, R8, R21, R22, and R23. The dominant species in those stations are P. planatus, A. beccarii, and Ad. longirostra with average 12.76, 10.71, and 10.47 %, respectively.
Discussion
The obtained results in this study can be used for the characterization of the state of Skhira and Ghannouch—Gabes coastline, in terms of chemical properties, and their impacts on benthic foraminifera.
The study areas have undergone environmental stress for several years. The discharge of different types of effluents has influenced directly or indirectly on marine ecosystem. Chemical analysis showed important accumulation of pollutants between commercial harbor of Ghannouch and fishing harbor of Gabes. This accumulation can be due to the presence of drainage channel of the chemical group in this zone. Both harbor have acted as barriers and transformed the area into a confined space, so the dispersion of pollutants was limited, and their concentrations tended to decrease with increasing distance away from the source of pollution. Similar pattern was observed in Skhira coastline, where high values of heavy metals, fluoride, and phosphorus were detected in stations S5 and S4, located near the chemical plane of Skhira.
To evaluate metallic contamination in sediment samples, enrichment factor determination was carried out (Table 4). EF values were higher than 1.5, suggesting anthropogenic impacts on the metal levels in the whole area. Heavy metal concentrations were compared to the effect range-low (ER-L) and effect range-median (ER-M) values (Table 2) reported for the sediment guideline of USEPA (Ferraro et al. 2006; Frontalini and Coccioni 2008). Pb, Cr, and Cu show median concentration values lower than ER-L. However, only two stations (R9 and R10) exceeded the ER-L value for Cu and Cr, but not the ER-M. Median concentration values of Zn and Cd are widely higher than the ER-M level. The highest level of heavy metals is found, where mud fraction constitutes the important fraction, which can be explained by the adsorptive proprieties of clay minerals and the large specific surface area of finer particles (Coccioni et al. 2009).
Several factors control the levels of organic matter (or TOC) in sediments such as the rich contributions of terrestrial organic matter (OM), low oxygen concentrations, and particle size (Aloulou Ben Ayed 2010). In fact, over the grain size is finer, their specific surfaces is small, increasing their retention capacity of the OM. However, remarkable concentrations were observed in stations with both sandy and silty facies. To note, the absence of vegetation in those samples, in addition to the harbor’s activities and maritime traffic, could be the cause of this organic pollution. The highest concentrations of TOC correspond to the highest values of heavy metals. According to Donnici et al. (2012), organic matter can be identified as a geochemical carrier of metals due to its capacity of absorbing metals of both terrestrial and marine origins.
The statistical analysis (BC and PCA) has identified significant correlations among studied elements, especially Cd, Cu, Cr, Zn, TOC, F, and P, suggesting possible association between them and a similarity of the input source. Despite the presence of multitudes of industries in the study area and different types of effluents, the positive association between heavy metals, phosphorus, and fluoride showed that pollution generated by the chemical group Ghannouch is the most dominant compared to other existed sources.
Since 1959, benthic foraminifer’s reaction towards pollution was the subject of several studies, covered different types of environments. This relationship differs depending on the contaminants nature, the type of marine environment, and the benthic foraminifera assemblage. According to Debenay et al. (1996), a total disappearance of benthic foraminifera was observed only in areas under a direct influence of effluent discharge, while at the periphery of the highly polluted and virtually barren zone, both density and diversity increase gradually. This phenomenon has been observed by other authors (Ferraro et al. 2006; Bergamin et al. 2009), where they suggested that high concentrations of heavy metals can strongly influence the mortality of species in the Gulf of Naples. The change that occurred in density and diversity of benthic foraminifera towards pollution has not been limited to the study of surface sediments in the contaminated areas. Vilela et al. (2014) have shown, through the study realized in cores at Guanabara Bay, that Ammonia tepida and Buliminella elangantissima are inversely proportional. Indeed, A. tepida is dominant in upper intervals of cores, while B. elangantissima dominates at deeper intervals. Thus, the change is explained by the degradation of the ecosystem quality during the late Holocene and the influence of human occupation.
In the present work, lowering in density and diversity of benthic foraminifera was clearly observed. It has been noted that as higher concentrations of heavy metals and other contaminants are detected, the number of species and specimens decreased, leading to a barren zone located between the commercial harbor of Ghannouch and the fishing harbor of Gabes. The influence of pollution on benthic foraminifera distribution has been proved by BC, where biotic parameters have negative correlation with most of contaminants. Besides density and diversity variations, statistical correlation matrix showed the presence of both tolerant and sensitive species. Rosalina bradyi, Q. berthelotiana var. weinsri, and S. costata were considered significant at stations R9, R10, and R11, and they showed very low abundance in “unpolluted sites.” Thus, those species have positive correlations with most of the chemical elements. Rosalina bradyi correlates positively with heavy metals, indicating tolerant behavior towards pollution, despite its low abundance in sediment samples. Conversely, R. bradyi was considered as sensitive species, according to Bergamin et al. (2009) and Romano et al. (2009), and showed significant abundance in the Gulf of Naples, Italy. The Quinqueloculina population expresses both tolerant (Q. berthelotiana var. weinsri) and sensitive (Q. berthelotiana) behavior towards pollution. Nevertheless, Quinqueloculina lata, Quinqueloculina stelligera, and Quinqueloculina parvula showed only a tolerant aspect towards pollution in the coastal area of Bagnoli (Italy). In addition, they exhibit an opportunistic behavior by taking advantages of nutrient availability under adverse condition due to the pollution (Romano et al. 2009).
The genus Ammonia, present in the studied areas, was composed mainly of A. parkinsoniana and A. beccarii. A. parkinsoniana, known as typical for shallow water environment, was found at depth of 10–45 m in the Gulf of Naples and can reach 107-m water depth in the Rhône delta (Rasmussen 2005). Frontalini and Coccioni (2008) reported that A. parkinsoniana was strongly affected by heavy metals. According to their results, this species is very sensitive and un-tolerant to heavy metal pollution and prefer a clean to slightly polluted environment. The same result was observed in Rodrigo de Freitas lagoon (Brazil), where A. parkinsoniana density decreased as heavy metal concentrations increase (Vilela et al. 2011). The multivariate analysis (HCA) showed the dominance of A. parkinsoniana in sediment samples of cluster B. This cluster consists of coastal stations from Skhira and Ghannouch—Gabes areas, characterized by low concentrations of heavy metals, fluoride, and phosphorus. However, BC revealed the absence of correlation (negative or positive) of this species with toxic elements.
In the Guanabara Bay (Brazil), A. beccarii possesses an opportunistic aspect. In fact, this species has a high potential to survive with high inputs of nutrients and trace metal concentrations, especially chromium (Donnici et al. 2012). According to Thomas et al. (2004), the relative abundance of A. beccarii may reflect an increase in nitrates. Though, in the present study, A. beccarii showed a sensitive behavior towards chromium and fluoride and a negative correlation with TOC.
In the literature, test deformation of benthic foraminifera could be caused either by natural or anthropogenic effects. According to Yanko et al. (1998), this phenomenon could occur in cold or hot water, regardless of latitudes, taxonomic affinity, nutriment strategy, or test morphology. A detailed analysis of test abnormalities and their possible causes was given by Boltovskoy et al. (1991) and Alve (1995a, b). According to those authors, test deformation can be the result of multiple effects and it would be difficult to isolate a single specific cause (Burone et al. 2006). In experimental analysis, under normal conditions, 1 % of deformed Ammonia is observed (Stouff et al. 1999), while Morovan et al. (2004) found 1.75 % of abnormal test in a single specific culture of A. tepida, living in unpolluted environment (Frontalini et al. 2009). Nevertheless, Samir and El-Din (2001) and Burone et al. (2006) revealed a strong correlation between number of deformed tests and heavy metal concentrations, in El Mex Bay (Egypt) and Montevideo coastal area (Uruguay).
In the present work, besides the negative assemblage response to the presence of important concentrations of different chemical parameters, abnormal test were observed in the surveyed areas, where the percentage of deformed specimens varies between 1 and 4 %. Important deformed test numbers were observed at stations with high concentrations of pollutants, suggesting that pollution is the main cause of those deformities.
Many studies have focused on the relationship between sediment grain size and benthic foraminifera distribution (Samir and El-Din 2001; Armynot du Châtelet et al. 2009). Armynot du Châtelet et al. (2009) noted that both density and richness of the specimens are clearly related to the size of the sediment, considered as the main factor limiting the distribution of benthic foraminifera. Nevertheless, few authors have confronted this issue from a quantitative viewpoint. Alve and Murray (1999) found a significant correlation between relative abundance of species and the percentage of fine sediments (<63 μm). Mendes et al. (2004) reported the abundance of some species into the granulometric ternary diagrams of Folk (1954), indicating the preference of some species to a selected type of sediment (Celia Magno et al. 2012). Gabes and Skhira areas were characterized, generally, by sandy sediment, coarse to very fine. Observations of benthic foraminifera density and diversity do not show any preference for the particle size of the sediment. According to Armynot du Châtelet et al. (2009), density and species richness decrease when the size of sediment increases due to the oligotrophic nature of coarse sediments. In our case, the minima of density and diversity were observed at stations R9, R10, and R11, where fine fraction (<63 microns) varied between 52.39 and 86.33 %, suggesting the importance of pollution control on benthic foraminifera distribution compared to the particle size of sediment. Sigmoilinita costata showed remarkable relative abundance in sites R10 and R11 with respectively 11 and 14 %. These two stations were characterized by high percentage of mud fraction (R10 (65 %) and R11 (86 %)). Celia Magno et al. (2012) also found a positive correlation between S. costata and clay fraction in the area of Augusta (Sicily, Italy). Taking into account the tolerant behavior of S. costata, we have come to conclude that obtained results are matching those of Bergamin et al. (2003) and Romano et al. (2008, 2009). These authors found that resistant species to pollution are well correlated with muddy fraction.
Conclusion
In this paper, we aimed to present the contribution of benthic foraminifera in the evaluation of coastal environment state. Geochemical analysis show different levels of contamination and helped to restraint the most polluted area which is located between both harbors of Ghannouch and Gabes. Important concentrations of chemical elements can be due to the presence of multiple sources of pollution in this area. A comparison between the distribution of pollution pattern and biotic indices results is then preceded. We have come to conclude a possible relationship between those two factors. This link is demonstrated by low density and diversity towards increasing pollutant concentrations. On the basis of statistical analysis, we showed the presence of both tolerant and sensitive species to pollution. Besides, unpolluted to moderately polluted stations were dominated by species resulted from shallow water environments with sandy sediment. High percentages of morphological abnormalities were found at stations with high concentrations of pollutants, indicating the role of pollution in those deformities.
This study showed that benthic foraminifera may be used as bioindicators for monitoring coastal pollution of the sediment of Skhira and Gabes (Tunisia). Our results suggest more analysis on deformed benthic foraminifera to identify the principal cause of abnormalities in tests of benthic foraminifera.
References
Abu Hilal A (1985) Phosphate pollution in the Jordan Gulf of Aqaba. Mar Pollut Bull 16:281–285
Aloulou Ben Ayed F (2010) Contamination des sédiments marins par les hydrocarbures et les métaux lourds et utilisation des foraminifères comme bio-indicateurs de pollution. University of Sfax (Tunisia), Dissertation
Aloulou F, EllEuch B, Kallel M (2012) Benthic foraminiferal assemblages as pollution proxies in the northern coast of Gabes Gulf, Tunisia. Environ Monit Assess 84:777–795
Alve E (1995a) Benthic foraminiferal responses to estuarine pollution; a review. J Foraminifer Res 25:190–203
Alve E (1995b) Benthic foraminiferal distribution and recolonization of formerly anoxic environments in Drammensfjord, southern Norway. Mar Micropaleontol 25:169–186
Alve E, Murray JW (1999) Marginal marine environments of the Skagerrak and Kattegat: a baseline study of living (stained) benthic foraminiferal ecology. Palaeogeogr Palaeoclimatol Palaeoecol 146:171–193
Armynot du Châtelet EA, Debenay JP, Soulard R (2004) Foraminiferal proxies for pollution monitoring in moderately polluted harbors. Environ Pollut 127:27–40
Armynot du Châtelet ÉA, Bout-Roumazeilles V, Riboulleau A, Trentesaux A (2009) Sediment (grain size and clay mineralogy) and organic matter quality control on living benthic foraminifera. Rev Micropaleontol 52:75–84
Bergamin L, Romano E, Gabellini M, Ausili A, Carboni MG (2003) Chemical–physical and ecological characterisation in the environmental project of a polluted coastal area: the Bagnoli case study. Mediterr Mar Sci 4:5–20
Bergamin L, Romano E, Finoia MG, Venti F, Bianchi J, Colasanti A, Ausili A (2009) Benthic foraminifera from the coastal zone of Baia (Naples, Italy): assemblage distribution and modification as tools for environmental characterization. Mar Pollut Bull 59:234–244
Bergin F, Kucuksezgin F, Uluturhan E, Barut IF, Meric E, Avsar N, Nazik A (2006) The response of benthic foraminifera and Ostracoda to heavy metal pollution in Gulf of Izmir. Estuar Coast Shelf Sci 66:368–386
Blanc-Vernet L (1974) Microfauna of dredging and coring conducted in the coast of Tunisia (Gulf of Gabes) and Libya (Tripolitania). Géol Mediterr 1:9–26 (in Frensh)
Boltovskoy E, Scott DB, Medioli FS (1991) Morphological variations of benthic foraminiferal tests in response to changes in ecological parameters: a review. J Paleontol 65:175–185
Burone L, Venturini N, Sprechmann P, Valente P, Muniz P (2006) Foraminiferal responses to polluted sediments in the Montevideo coastal zone, Uruguay. Mar Pollut Bull 52:61–73
Celia Magno M, Bergamin L, Finoia MG, Pierfranceschi G, Venti F, Romano E (2012) Correlation between textural characteristics of marine sediments and benthic foraminifera in highly anthropogenically-altered coastal areas. Mar Geol 315–318:143–161
Centre d’Expertise en Analyse Environnementale du Québec (CEAEQ) (2006) Détermination du carbone organique total dans les solides : dosage par titrage, MA.405–C1.0. Ministère du développement durable, de l’Environnement et des Parcs du Québec
Choi KY, Kim SH, Chon HT (2008) Distributions and accumulations of heavy metals in the sediments of harbors and coastal areas in Korea. Proceedings of the International Symposia on Geoscience Resources and Environments of Asian Terranes, 4th IGCP 516,and 5th APSEG, Thailand
Cimerman F, Langer M (1991) Mediterranean Foraminifera. Academia Scientiarum et Artium Slovenica Classis IV: Historia Naturalis, 30 : 1–119
Coccioni R (2000) Benthic foraminifera as bioindicators of heavy metal pollution. In: Martin RE (ed) Environmental micropaleontology: the application of microfossils to environmental geology. Kluwe Academic/Plenum Publishers, New York, pp 71–103
Coccioni R, Frontalini F, Marsili A, Mana D (2009) Benthic foraminifera and trace element distribution: a case-study from the heavily polluted lagoon of Venice (Italy). Mar Pollut Bull 59:257–267
Debenay JP, Pawlowski J, Decrouez D (1996) Les foraminifères actuels. Masson, Paris
Debenay JP, Tsakiridis E, Soulard R, Grossel H (2001) Factors determining the distribution of foraminiferal assemblages in Port Joinville Harbor (Ile d’Yeu, France): the influence of pollution. Mar Micropaleontol 43:75–118
Donnici S, Serandrei-Barbero R, Bonardi M, Sperle M (2012) Benthic foraminifera as proxies of pollution: the case of Guanabara Bay (Brazil). Mar Pollut Bull 64(10):2015–2028
Drira Z (2009) Contribution à la compréhension du fonctionnement du golfe de Gabès : étude des caractéristiques dynamiques et structurales des communautés phyto-zooplanctonique en relation avec la variabilité environnementale et les caractéristiques hydrographiques des zones côtières et océaniques. University of Sfax (Tunisia), Dissertation
Essersi R, Nechad B, Ruddick K, Zargouni F (2010) Optical remote sensing of the Gulf of Gabès—relation between turbidity, Secchi depth and total suspended matter. Ocean Sci Discuss 7:1767–1783
Fatela F, Taborda R (2002) Confidence limits of species proportions in microfossil assemblages. Mar Micropaleontol 45:169–174
Ferraro L, Sprovieri M, Alberico I, Lirer F, Prevedello L, Marsella E (2006) Benthic foraminifera and heavy metals distribution: a case study from the Naples Harbour (Tyrrhenian Sea, Southern Italy). Environ Pollut 142:274–287
Fisher RA, Corbet AS, Williams CB (1943) The relationship between the number of species and the number of individuals in a random sample of an animal population. J Anim Ecol 12:42–58
Folk RL (1954) The distinction between grain-size and mineral composition in sedimentary-rock nomenclature. J Geol 62:344–359
Frezza V, Carboni MG (2007) Distribution of recent foraminiferal assemblages near the Ombrone River mouth (northern Tyrrhenian Sea, Italy). Rev Micropaleontol 52:43–66
Frontalini F, Coccioni R (2008) Benthic foraminifera for heavy metal pollution monitoring: a case study from the central Adriatic Sea coast of Italy. Estuar Coast Shelf Sci 76:404–417
Frontalini F, Buosi C, Da Pelo S, Coccioni R, Cherchi A, Bucci C (2009) Benthic foraminifera as bio-indicators of trace element pollution in the heavily contaminated Santa Gilla lagoon (Cagliari, Italy). Mar Pollut Bull 58:858–877
Garcia EM, Cruz-Motta JJ, Farina O, Bastidas C (2008) Anthropogenic influences on heavy metals across marine habitats in the western coast of Venezuela. Cont Shelf Res 28:2757–2766
Geslin E, Debenay JP, Lesourd M (1998) Abnormal wall textures and test deformation in Ammonia (hyaline foraminifer). J Foraminifer Res 28:148–156
Glaçon G (1963) Foraminifères des dépôts actuels des côtes de Tunisie Sud Orientale. University of Montpellier, Dissertation
Hattab T, Ben Rais Lasram F, Albouy C, Romdhane MS, Jarboui O, Halouani G, Cury P, Le Loc'h F (2013) An ecosystem model of an exploited southern Mediterranean shelf region (Gulf of Gabes, Tunisia) and a comparison with other Mediterranean ecosystem model properties. J Mar Syst 128:159–174
Hottinger L, Halicz E, Reiss Z (1993) Recent foraminiferida from the Gulf of Aqaba, Red Sea. Slovenska Akademija Znanosti in Umenosti, Ljubljana
Loeblich AR, Tappan H (1988) Foraminiferal genera and their classification. Van Nostrant Reinhold, New York
Mendes I, Gonzalez R, Dias JMA, Lobo F, Martins V (2004) Factors influencing recent benthic foraminifera distribution on the Guadiana shelf (Southwestern Iberia). Mar Micropaleontol 51:171–192
Meriç E, Avşar N, Nazik A, Yokeş B, Barut IF, Eryilmaz M, Kom E, Taşkin H, Başsari A, Dinçer F, Bircan C, Kaygun A (2012) Benthic forminifer-ostracod assemblages and Pacific Ocean–Red Sea originated foraminifera and abnormal individuals. Bull Mineral Res Explor 145:62–103
Morovan J, Le Cadre V, Jorissen F, Debenay JP (2004) Foraminifera as potential bioindicators of the "Erika" oil spill in the Bay of Bourgneuf: field and experimental studies. Aquat Living Resour 17:317–322
Murray JW (1991) Ecology and paleoecology of benthic foraminifera. Longman Scientific and Technical Edition, New York
Murray JW (2006) Ecology and applications of benthic foraminifera. Cambridge University Press, London
Oueslati A (1992) Salt marshes in the Gulf of Gabes (Southeastern Tunisia): their morphology and recent dynamics. J Coast Res 8:727–733
Rabaoui L, Balti R, EL Zrelli R, Tlig-Zouari S (2014) Assessment of heavy metal pollution in the Gulf of Gabes (Tunisia) using four mollusc species. Medit Mar Sci 15:45–58
Rasmussen TL (2005) Systematic paleontology and ecology of benthic foraminifera from the Plio-Pleistocene Kallithea Bay section. Cushman Foundation Special Publication, Rhodes (Greece)
Resig JM (1960) Foraminiferal ecology around ocean outfalls off southern California. In: Pearson PA (ed) Waste disposal in the marine environment. Pergamon press, London, pp 104–121
Romano E, Bergamin L, Finoia MG, Carboni MG, Ausili A, Gabellini M (2008) Industrial pollution at Bagnoli (Naples, Italy): benthic foraminifera as a tool in integrated programs of environmental characterization. Mar Pollut Bull 53:439–457
Romano E, Bergamin L, Ausili A, Pierfranceschi G, Maggi C, Sesta G, Gabellini M (2009) The impact of the Bagnoli industrial site (Naples, Italy) on sea-bottom environment. Chemical and textural features of sediments and the related response of benthic foraminifera. Mar Pollut Bull 59:245–256
Samir AM, El-Din AB (2001) Benthic foraminiferal assemblages and morphological abnormalities as pollution proxies in two Egyptian bays. Mar Micropaleontol 41:193–227
Samir AM, Abdou HF, Zazou SM, El-Menhawey WH (2003) Cluster analysis of recent benthic foraminifera from the northwestern Mediterranean coast of Egypt. Rev Micropaleontol 46:111–130
Sammari C, Koutitonsky VG, Moussa M (2006) Sea level variability and tidal resonance in the Gulf of Gabes. Tunisia. Cont Shelf Res 26:338–350
Sanchez-Arcilla A, Mosso C, Sierra JP, Mestres M, Harzallah A, Senouci M, El Raey M (2010) Climatic drivers of potential hazards in Mediterranean coasts. Reg Environ Chang. doi:10.1007/s10113-010-0193-6
Sgarrella F, Moncharmont-Zei M (1993) Benthic foraminifera of the Gulf of Naples (Italy): systematic and autoecology. Boll Soc Paleontol Ital 32:145–264
Sharifi AR, Croudace IW, Austin RL (1991) Benthic foraminiferids as pollution indicators in Southampton Water, southern England, UK. J Micropalaeontol 10:109–113
Shepard FP (1954) Nomenclature based on sand, silt, clay ratios. J Sediment Petrol 24:151–158
Stouff V, Geslin E, Debenay JP, Lesourd M (1999) Origin of morphological abnormalities in Ammonia (foraminifera): studies in laboratory and natural environments. J Foraminifer Res 29:152–170
Thomas E, Abramson I, Varekamp JC, Buchholtz ten Brink MR (2004) Eutrophication of Long Island Sound as traced by benthic foraminifera. Proceedings of the 6th Biennual Long Island Sound Research Conference: 87–91
Valenti D, Tranchina L, Brai M, Caruso A, Cosentino C, Spagnolo B (2008) Environmental metal pollution considered as noise: effects on the spatial distribution of benthic foraminifera in two coastal marine areas of Sicily (Southern Italy). Ecol Model 213(3–4):449–462
Vilela CG, Batista DS, Baptista-Neto JA, Ghiselli RO Jr (2011) Benthic foraminifera distribution in a tourist lagoon in Rio de Janeiro, Brazil: a response to anthropogenic impacts. Mar Pollut Bull 62:2055–2074
Vilela CG, Fgueira BO, Macedo MC, Baptista Neto JA (2014) Late Holocene evolution and increasing pollution in Guanabara Bay, Rio de Janeiro, SE Brazil. Mar Pollut Bull 79:175–187
Walton WR (1952) Techniques of recognition of living foraminifera. Contribution from the Cushman Foundation 3: 56–60
Watkins JG (1961) Foraminiferal ecology around the Orange Country, California, ocean sewer outfall. Micropaleontology 7:199–206
Yalçýn H, Meriç E, Avsar N, Tetiker S, Barut YF, Yýlmaz P, Dinçer F (2008) Mineralogical and geochemical features of colored benthic foraminifers from Aegean and southwestern coasts of Turkey. Micropaleontology 54(3):351–370
Yanko V, Kronfeld J, Flexer A (1994) Response of benthic foraminifera to various pollution sources: implications for pollution monitoring. J Foraminifer Res 24:1–17
Yanko V, Ahmad M, Kaminski M (1998) Morphological deformities of benthic foraminiferal tests in response to pollution by heavy metals: implications for pollution monitoring. J Foraminifer Res 28:177–200
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Thomas Hein
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOC 199 kb)
Rights and permissions
About this article
Cite this article
Ayadi, N., Zghal, I., Aloulou, F. et al. Impacts of several pollutants on the distribution of recent benthic foraminifera: the southern coast of Gulf of Gabes, Tunisia. Environ Sci Pollut Res 23, 6414–6429 (2016). https://doi.org/10.1007/s11356-015-5872-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-015-5872-x