Abstract
This manuscript reports a 3-year study on occurrence of pharmaceuticals, hormones, and triclosan in surface waters of a central urban region of São Paulo State of Southeast Brazil (the Monjolinho River in São Carlos). Water samples collected once at every 2 months were pre-concentrated by solid-phase extraction (SPE) and analyzed by liquid chromatography coupled with tandem mass spectrometry (LC-MS/MS). The most frequently detected compounds in higher concentrations were caffeine, paracetamol, and atenolol (maximum concentrations 129,585, 30,421, and 8199 ng L−1, respectively), while hormones estrone and 17-β-estradiol were the least detected, in levels up to 14.8 ng L−1. There was an increasing trend in concentrations of most of the compounds along the river course, especially downstream of the river where there is discharge of both wastewater treatment plant effluent and raw sewage from a particular region of São Carlos city. Concentrations of contaminants were higher during dry periods as a result of decline in the water levels. Decrease in concentrations near the river mouth occurred to different extents for each compound. It was high for caffeine and atenolol, but was very low for carbamazepine and diclofenac. The present study reports the first data about the occurrence of some major emerging contaminants in the Monjolinho River. Besides its regional significance, this work may assist in composing a dataset for water contamination diagnosis focusing on emerging contaminants, both in the Brazilian as well as in the Global studies related to aquatic ecosystems. Such datasets can be helpful for making future public policies on water quality, since these compounds are not yet legally regulated.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Over the years, the anthropic activities have caused degradation of aquatic systems by introducing several kinds of contaminants, thus endangering aquatic life (Geist 2011; Schwarzenbach et al. 2006). Therefore, in the last 20 years, the scientific community has turned its attention to investigate occurrence of emerging compounds in the environment (Fatta-Kassinos et al. 2011). These compounds are not yet regulated by the environmental laws, and most of them are still unknown for their adverse effects to the aquatic life. Several different classes of compounds have been considered as emerging contaminants such as hormones, pharmaceuticals, and personal care products (PPCP). Pharmaceuticals and hormones reach the sewers as a result of their ingestion and posterior excretion, while personal care products originate from daily hygiene activities, such as bathing, skin washing, and tooth brushing (Brausch and Rand 2011; Jjemba 2006). However, incomplete removal of such biologically active emerging contaminants in the wastewater treatment plants (WWTPs) allows them to reach the water bodies (Collado et al. 2014), where they may cause undesirable adverse effects to non-target organisms.
Among the large range of contaminants that are introduced in aquatic systems, pharmaceuticals and hormones have caught significant attention of the main international environmental agencies. The development of new drugs and raise in the world life expectancy with growth of the elderly population has caused high drug consumption (European Community 2013). A list of relevant pharmaceuticals to the water cycle suggested by the Global Water Research Coalition included atenolol, diclofenac, ibuprofen, naproxen, and carbamazepine as pharmaceuticals of high priority for the occurrence studies and risk assessment (Global Water Research Coalition 2008). Although the concentrations of pharmaceuticals found in natural waters apparently do not offer risks to the human health, their continuous exposure to aquatic organisms together with other contaminants is of seriously growing concern due to the known and unknown adverse effects (Deblonde and Hartemann 2013; Madureira et al. 2012; Petersen et al. 2014). On the other hand, it is known that hormones act as endocrine disruptor, causing fish feminization and reproductive problems, which may lead to the ecological imbalance in the aquatic environment (Aris et al. 2014; Orlando and Ellestad; Yan et al. 2012).
The occurrence of PPCP and hormones in aquatic ecosystems has been widely studied in many countries (Bendz et al. 2005; Camacho-Muñoz et al. 2010; Esteban et al. 2014; Moldovan 2006; Petrović et al. 2014; Ternes 1998; Verlicchi et al. 2014; Wang et al. 2012). However, these studies are still very limited in Brazil (Caldas et al. 2013; de Sousa et al. 2014; Jardim et al. 2012; Kuster et al. 2009; Montagner and Jardim 2011; Sodré et al. 2010). Previously reported studies regarding rivers of the metropolitan region of Campinas from São Paulo State of Brazil pointed out that the levels of emerging compounds is a result of the poor sewage treatment facilities (Jardim et al. 2012; Montagner and Jardim 2011).
The Monjolinho River is an important part of the Tietê-Jacaré rivers’ hydrographical basin and is located in São Carlos city, central region of São Paulo State. This city houses about 220 thousand inhabitants (IBGE Brazilian Institute of Geography and Statistics 2013) and has an extensive industrial park, especially in the sectors of automotives, refrigeration, paper and cardboard, school supplies, cosmetics, and textiles. Almost 99.6 % of the sewage produced by São Carlos city is collected; however, 85 % of this amount is treated (CETESB - São Paulo State Environmental Agency 2012). In this context, the Monjolinho River is the receiving water body of both treated and untreated effluents. Eventually, this water body suffers due to the urban runoff from São Carlos city and, therefore, urbanization is causing the degradation of the Monjolinho River as reported by some previous studies (Bianchi et al. 2011; Campagna et al. 2008; Chiba et al. 2011; Printes et al. 2011).
Although the water from the Monjolinho River is not used for public supply, there is a general concern regarding water resource conservation and protection of aquatic life. Furthermore, there is insufficient data regarding occurrence of emerging contaminants in the Monjolinho River, what drives studies on their occurrence in this water body as initial tools for diagnosing its environmental quality in addition to the priority contaminants. Since these contaminants are not yet regulated by environmental laws, studies related to their occurrence and concentration levels can assist in composing a dataset that can help in making future public policies on water quality in Brazil.
The aim of this study was to investigate the occurrence and spatiotemporal distribution of some important pharmaceuticals, hormones, and triclosan in surface water of the Monjolinho River. Caffeine was also included in this study as it has been suggested by several authors to be a good tracer of contamination by domestic sewage (Buerge et al. 2003; Daneshvar et al. 2012).
Material and methods
Sampling sites and sample collection
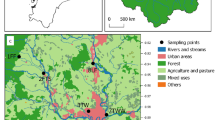
Water samples were collected from six different sampling sites located in the Monjolinho River except site 4 that was located in one of the river tributaries (Água Quente stream). The sites were selected on the basis of different external factors that have a significant impact on the Monjolinho River (Fig. 1).
Site 1 (reference site, 21°59′56.63″S; 47°50′26.55″W) is the source of the Monjolinho River and is situated in an agricultural area, subjected to minor impacts. Site 2 (21°59′12.40″S; 47°52′27.40″W) is located in the beginning of the urban area, representing the influence of agricultural zone, while site 3 (21°01′19.88″S; 47°54′51.63″W) is situated in a locality where the river has received the urban runoff and eventual clandestine discharge of sewage in the pluvial drainage system. Site 4 refers to the Água Quente stream, which is the main receptor of non-treated sewage from a specific neighborhood of São Carlos city. The Água Quente stream flows into the Monjolinho River next to the release of WWTP effluent. Site 5 is situated at the downstream of the WWTP (22°02′07.19″S; 47°57′28.13″W), which represents impacts resulting from discharge of both treated (from WWTP) and untreated sewage (Água Quente stream; 22°1′48.58″S; 47°55′55.44″W). Finally, site 6 (22°01′50.60″S; 48°01′58.94″W) is located just before it flows into the Jacaré-Guaçu River (tributary of the Tietê River). This last site represents the water quality after all the discharges of pollutants and removal by natural physicochemical processes. A total of 21 sampling campaigns in field were covered from the period of January/2011 to November/2013.
Water samples were collected at 30-cm depth in 500-mL amber PET bottles, previously rinsed thoroughly with ultrapure water. After collection, samples were refrigerated at 4 °C during their transport to the laboratory and processed within 48 h.
Standards and reagents
All the analytical standards were of high purity grade (>90 %). Caffeine (CAF), paracetamol (PAR), ibuprofen (IBU), naproxen (NPX), diclofenac (DCF), carbamazepine (CMZ), atenolol (ATN), propranolol (PRO), triclosan (TCS), estrone (E1), 17-β-estradiol (E2), and 17-α-ethinylestradiol (EE2) were purchased from Dr. Ehrenstorfer (Augsburg, Germany). The isotopically labeled compounds, used as surrogates, were caffeine-d3, naproxen-d3, atenolol-d7, carbamazepine-d10, ibuprofen-d3, diclofenac-d4, triclosan-d3, propranolol-d7, estrone-d4, 17-β-estradiol-d5, and 17-α-ethinylestradiol-d4; all of them were purchased from CDN Isotopes (Quebec, Canada).
HPLC-grade methanol and acetone were purchased from J.T. Baker (Ecapetec, Mexico) and Mallinkrodt (Phillipsburg, USA), respectively. Formic acid (98 % purity) and HPLC-grade ammonium hydroxide were bought from Sigma-Aldrich (Schweiz, Switzerland). Ultrapure water was obtained from a Milli-Q device (Merck Millipore, São Paulo, Brazil). Cartridges used in solid-phase extraction (SPE) were Oasis® HLB (200 mg, 6 mL) purchased from Waters Corporation (Milford, Massachusetts, USA).
Individual standard solutions and surrogate solutions were prepared in methanol at a concentration of 100 mg L−1 and stored at −18 °C. Working standard solutions were prepared before each analytical determination through appropriate dilution of individual stock solutions in methanol-water (25:75, v v −1).
Determination of physicochemical parameters
Physicochemical parameters such pH, dissolved oxygen (DO), electrical conductivity (EC), and turbidity (Turb) were determined in field directly in the water body, using a multi-parameter probe YSI 6820 V2 (Yellow Springs, Ohio, USA). Total dissolved carbon (DOC) was analyzed by a Shimadzu TOC-L CPN analyzer (Tokyo, Japan).
Analytical method for determination of emerging contaminants
Procedures for sample preparation and analysis are described in details in de Sousa et al. (2014). After collection, samples were vacuum-filtered and target-compound were extracted from each 500-mL water sample by SPE. Previously to each extraction, a mix of isotopically labeled compounds (surrogates) was added to samples to get a final concentration of 100 ng L−1. Target-compounds were eluted with 2 × 3 mL of methanol followed by 3 mL of methanol-acetone (50:50, v v−1). The extracts were evaporated to dryness under nitrogen stream and reconstituted in methanol-water (25:75, v v−1).
Target compounds were analyzed by ultra-performance liquid chromatography coupled to a triple quadrupole mass spectrometer, using a Waters ACQUITY UPLC system. UPLC was equipped with an ACQUITY UPLC binary solvent manager and an AQUITY UPLC manager (USA). Chromatographic separation was achieved with an ACQUITY UPLC BEH C18 (50 mm × 2.1 mm, 1.7 μm) preceded by a guard column of the same material, both supplied by Waters (USA). The mobile phases used were 0.05 % aqueous solution of ammonium hydroxide (v v−1) (eluent A) and methanol (eluent B), with a flow rate of 0.45 mL min−1. The column and sample temperatures were 40 and 10 °C, respectively. The gradient elution was started with 5 % of B for 0.5 min, increasing to 45 % of B over 1.5 min, raising to 95 % of B in 4 min, held for 0.5 min, and back to initial conditions within 1.5 min. Total run time was 6 min and injection volume was set to 10 μL.
Mass spectrometer (TQD, Waters, UK) was equipped with an electrospray ion (ESI) source. Analyses were performed both in negative (NI) and positive ionization (PI) modes, as the physicochemical properties of the target-compounds were very different. MS parameters were set up as follows: desolvation temperature was 500 °C; ESI source block temperature was 150 °C; capillary voltage for PI and NI were 3 kV and 2.5 kV, respectively; cone nitrogen gas flow, 20 L h−1; desolvation nitrogen gas flow, 750 L h−1; and argon collision gas, 3 × 10−3 mbar. Quantitative analyses were performed in a multiple reaction monitoring (MRM) mode, in which two transitions between the precursor ion and the most abundant fragment ion were monitored for each analyte. MRM 1 was used for quantification while MRM 2 was used for confirmation purposes.
Method validation
Recovery studies were performed to evaluate the efficiency of SPE procedure. Water samples from site 1 (reference site) were spiked before SPE with a mix of all target-compounds and surrogates in two concentration levels: 10 and 100 ng L−1. Recovery was obtained from comparison between peak areas of the analytes in samples spiked with the same standard concentrations only after SPE proceedings. Method detection limits (MDL) and method quantification limits (MQL) were obtained from spiked water samples before SPE and determined as the analyte concentration that gave a signal-to-noise (S/N) ratio of 3 and 10, respectively. Linearity was evaluated from calibration curves with at least six points, and intra-batch precision was determined from relative standard deviation (RSD) of six replicates of fortified samples at the abovementioned two concentrations.
Statistical treatment
Principal component analysis (PCA) was used with the aim of reducing the number of variables and to observe relationships among the studied variables and the one that determines data variability of a sample set. Physicochemical data and concentrations of emerging contaminants (except PAR, because its data is available only for a half of the monitoring period) were organized in a data matrix containing 108 samples. As pre-processing step, an auto-scaling of the data matrices was performed to give the same weight to all variables. PCA was performed using the Pirouette 4.0 rev. 2 software (Infometrix, Bothell, E.U.A.).
Results and discussion
Method performance
Results of all validation parameters of the method are available in Online Supplementary Resource 1. Recoveries of the SPE proceedings ranged from 31.6 (for PAR) to 101.2 % (for ATN and E1). The lowest recoveries were obtained for PAR and TCS (i.e., 68 %). However, precision of extractions was acceptable (RSD <15 %), which makes the SPE method satisfactory for the target-compounds (Cassiano et al. 2009). Linear correlations obtained for calibration curves were always higher than 0.99. Additionally, the chromatographic method was able to separate most of the selected compounds, as evidenced by the retention times (Online Supplementary Resource 1). For compounds whose retention times were similar, selectivity was guaranteed by mass spectrometry detector with triple quadrupole analyzer.
Method precision (RSD) was lower than 15 %, which was satisfactory for all selected compounds according to method validation guidelines (ANVISA - Brazilian Health Surveillance Agency 2003; US-FDA - U.S. Food and Drug Administration 2001) and European Directive (EC 2002). MDL and MQL ranged from 0.04 to 3.0 ng L−1 and from 0.1 to 5.0 ng L−1, respectively, which was appropriate to detect the analytes in water samples at concentrations of the same order of magnitude already reported in previous studies (Baker and Kasprzyk-Hordern 2013; Collado et al. 2014; Vazquez-Roig et al. 2012; Wang et al. 2012).
Physicochemical parameters of water samples
Table 1 presents the average values of physicochemical parameters of the Monjolinho River during the monitoring period. Average pH ranged from 6.4 to 7.0 between the six sampling sites. The values of EC, Turb, and DOC increased toward downstream of the WWTP, indicating the harmful influence of urbanization on the surface water quality, dominantly by the release of raw and treated sewage. Coherently, there was a decrease of DO concentrations at site 5 (downstream of the WWTP) to an average of 4.12 mg L−1. This variation is attributed to the introduction of labile organic matter, especially from non-treated sewage, as evidenced by the high DOC concentrations at site 4 (Água Quente stream). In accordance with that, water samples from site 4 showed the lowest average DO concentration (3.15 mg L−1) and the highest DOC value (24.4 mg L−1).
Occurrence of emerging contaminants
Figure 2 presents the percentage of positive samples and average concentrations of the selected emerging contaminants found in the Monjolinho River during the monitoring period. Of all selected contaminants, only the synthetic hormone EE2 was not detected in any sample. Furthermore, hormones were found to be in low percentage of positive samples at the study area. On the other hand, CAF, ATN, and PAR were frequently detected in high concentrations. TCS and PRO were also frequently detected but in lower levels.
Table 2 shows the minimum, maximum, and average concentrations of the target-compounds at five sampling sites along the Monjolinho River and the Água Quente stream. CAF was detected in 93 % of the samples, and it was present in higher concentrations compared to the other contaminants (average of 14,955 ng L−1; Fig. 2), reaching a maximum of 129,585 ng L−1. This compound is ubiquitous and being human-related; therefore, several authors have proposed caffeine as a tracer of domestic sanitary contamination in urban water bodies (Buerge et al. 2003; Froehner et al. 2011) although other authors have demonstrated that there are better markers, such as CMZ and DCF (Daneshvar et al. 2012; de Sousa et al. 2014), also included in the present study. CAF is present in high concentrations not only in pharmaceutical formulations but also in beverages like refrigerants, coffee, tea, and stimulants in general. Therefore, high concentrations are expected to be found in urban aquatic bodies due to its wide consumption in daily routines.
High average concentrations of CAF were also found in other Brazilian water bodies, such as the Atibaia River (São Paulo State; 10,152 ng L−1) (Montagner and Jardim 2011), Iguaçu River (Paraná State; 51,360 ng L−1) (Froehner et al. 2011), and Jundiaí River (São Paulo State; 6550 ng L−1) (de Sousa et al. 2014). These concentrations are quite similar to that found in the Monjolinho River (Fig. 2, Table 2). However, they are higher than those found in aquatic bodies of the USA (average concentration 28.8 ng L−1) and Europe (average concentrations from 137 to 455 ng L−1) (Bendz et al. 2005; Camacho-Muñoz et al. 2010; Conley et al. 2008; Loos et al. 2010). It might be associated with the higher sewage treatment index in developed countries in relation to Brazil (World Health Organization/UNICEF - 2013), since previous studies pointed out that removal rates of CAF are higher than 90 % in WWTPs with biological treatments (Kosma et al. 2014; Martínez Bueno et al. 2011; Sui et al. 2010).
Another frequently detected compound in relatively high concentrations in the Monjolinho River was the beta-blocker ATN (78 % of frequency). The average concentration was 1182 ng L−1 (Fig. 2), while the maximum detected was 8199 ng L−1. On the other hand, PRO that belongs to the same therapeutic group of ATN presented lower concentrations in relation to this compound (average of 15.2 ng L−1; maximum of 77.3 ng L−1; Table 2). This behavior was also observed by several authors in other water bodies (Bendz et al. 2005; de Sousa et al. 2014; Kasprzyk-Hordern et al. 2008; Osorio et al. 2012; Valcárcel et al. 2011). About 90 % of ATN is excreted in the non-metabolized form, while less than 1 % of PRO is excreted in the original form (Ternes 1998; Zuccato et al. 2005), what can explain the higher concentrations of ATN compared to PRO. Furthermore, photodegradation experiments in surface waters reported that PRO had a half-life time ranging from 6.0 to 8.3 h (Yamamoto et al. 2009), which was lower than other pharmaceuticals like ATN, IBU, PAR, and CMZ. Thus, it indicates that photodegradation may be an important attenuation factor for PRO in surface waters.
Among the anti-inflammatories/analgesic selected, PAR was the most frequently detected (77 % of positive samples), generally found in high concentrations (average 3702 ng L−1; maximum: 30,421 ng L−1; Fig. 2, Table 2). NPX, IBU, and DCF were detected in about 60 % of the samples and presented low concentrations, up to 655.2, 743.9, and 385.7 ng L−1, respectively. The higher concentrations of PAR relative to the other anti-inflammatories might be associated to its nonprescription sale and widespread consumption in Brazil as analgesic for fever and pains of several origins. Similar levels of PAR were found in the Atibaia River (Campinas, São Paulo State; maximum of 13,440 ng L−1) (Montagner and Jardim 2011). However, these concentrations are higher compared to other aquatic bodies in Europe. In the Ebro River watershed, Spain, concentrations of PAR reached 872 ng L−1 (Silva et al. 2011). Unlike, the range of concentrations of IBU and DCF were near to that found in some rivers of Spain (the Llobregat and Ebro Rivers; IBU 2.7–868 ng L−1; DCF <0.4–796 ng L−1) (Osorio et al. 2012; Silva et al. 2011) and Sweden (the Höje River; IBU 80–780 ng L−1; DCF 10–180 ng L−1) (Bendz et al. 2005).
Differences in concentrations of pharmaceuticals can be attributed to different consumption patterns between populations, removal in WWTP or hydrological factors like river flow, which determines the dilution of treated or untreated sewage, or even sorption/sedimentation and photodegradation that are dependent of the compound and peculiarity of the study area (Lindström et al. 2002).
The psychiatric drug CMZ was detected in 74 % of samples in the Monjolinho River, and the concentrations reached 171.3 ng L−1 (Table 2). The concentration range of this compound is comparable to that found in the Llobregat River, Spain (1.2–267 ng L−1) (Osorio et al. 2012). However, they are higher than that reported in surface waters of the USA (2.9–23 ng L−1) (Conley et al. 2008), Spain (<LD 53.8 ng L−1) (Collado et al. 2014; Silva et al. 2011), and Serbia (<LQ 35.5 ng L−1) (Petrović et al. 2014).
The natural hormones E1 and E2 were hardly detected and the measurable concentrations were low, presenting averages of 6.90 and 1.85 ng L−1, respectively (Fig. 2). These concentrations are lower compared to that found for pharmaceuticals. However, the results are in agreement with most of the data reported in the literature (Camacho-Muñoz et al. 2010; de Sousa et al. 2014; Esteban et al. 2014; Loos et al. 2010; López-Roldán et al. 2010; Wang et al. 2012; Zhao et al. 2009). The concentration range of E1 in the Monjolinho River (<0.1–14.7 ng L−1) is similar to that found in the Jundiaí River (São Paulo State, Brazil; 4.75–8.01 ng L−1) (de Sousa et al. 2014) and in some aquatic bodies in Spain, like the Jarama and Manzanares Rivers (<0.05–17 ng L−1) and Llobregat River (0.82–5.81 ng L−1) (Esteban et al. 2014; López-Roldán et al. 2010). For E2, the average concentration in the present study (1.85 ng L−1) was consistent with maximum values obtained in rivers in China (7.7 ng L−1) and the USA (1.91 ng L−1) (Peng et al. 2008; Wang et al. 2012; Zhao et al. 2009). These values are lower than those found for E1. However, numerous studies have noticed the same behavior (Esteban et al. 2014; Peng et al. 2008; Wang et al. 2012), and some of them not even detected E2 and EE2 (Loos et al. 2010; López-Roldán et al. 2010; Moreira et al. 2009; Yu and Chu 2009). This fact can be attributed to the conversion of E2 and EE2 into E1. According to Ribeiro et al. (2010), E1 is a metabolite produced by degradation of E2, while EE2 has been found to be transformed in conjugated estrogens during biodegradation.
TCS is an antiseptic agent added in several personal care products of daily use, such as toothpaste, hand soaps, deodorants, and skin creams. The main concern regarding the presence of TCS in aquatic systems is its toxicity to some species of algae and bacteria in biofilms, due to its direct mode of action as bactericide (Ricart et al. 2010). TCS was frequently detected in the Monjolinho River (80 % of frequency), with average concentration of 35.2 ng L−1 (Fig. 2) and maximum of 281.1 ng L−1. These concentrations are in agreement with the results obtained from the Jundiaí River (Brazil, 4.95–324.0 ng L−1) and in aquatic bodies from the UK (<5–95 ng L−1), Spain (46–184 ng L−1), and Sweden (20–190 ng L−1) (Bendz et al. 2005; de Sousa et al. 2014; Esteban et al. 2014; Kasprzyk-Hordern et al. 2008).
Spatiotemporal distribution
The spatial distribution pattern of emerging contaminants demonstrates an increase of concentrations from the source of the river (site 1) to the downstream of WWTP (site 5) (Table 2). PCA graph illustrates this trend, as shown in Fig. 3, where scores and loadings are represented together. Two principal components (PC) were extracted, which together corresponded to 54.5 % of the total data variance (43.6 % for PC1 and 10.9 % for PC2). It is possible to observe that samples were distributed along the PC1, and there was a separation from the left to the right along this axis, according to the urbanization grade and impacts received, as the following: sites 1, 2, 3, 6, 4 and finally the site 5 on the inferior right corner.
Considering the variable DO as a good water quality parameter and other variables (EC, DOC, and emerging contaminants) as representatives of a poor water quality, it can be confirmed from the graph (Fig. 3) that there is a subsequent increase of anthropic impacts from left to right. It demonstrates that the river is subject to a considerable raise in contamination from upstream to downstream, where the urbanization has a negative influence, since it also increases along the river course.
Sampling sites with less urban influence (sites 1 to 3; filled symbols inside the dotted square of Fig. 3) were discriminated by the parameter DO, presenting high DO concentrations and low concentrations of emerging contaminants and other physicochemical parameters. On the other hand, sites 4 and 5, with more pronounced urban influence, were discriminated by pharmaceuticals, TCS, CAF, EC, and DOC. Both sites exhibited high concentrations of emerging contaminants, EC, and DOC in all samples, but low DO concentrations.
As shown in Fig. 3, site 4 (Água Quente stream) is separated on the inferior right corner. This site presented higher concentrations of contaminants than site 5, especially CAF, NPX, and ATN. Site 4 is receptor of non-treated sewage from an area of São Carlos which houses 50,000 inhabitants, while site 5 is downstream of the WWTP, whose samples are representative of discharge of effluent coming from WWTP and non-treated sewage coming from the site 4, which flows into the river together with the effluent. Therefore, the higher concentrations at site 4 relative to site 5 may be associated to the dilution of raw sewage (from site 4) by the river flow. Our results show that the release of non-treated sewage by Água Quente stream and the WWTP effluent are the main sources of emerging contaminants in the Monjolinho River.
Despite of the fact that site 6 is situated at the river mouth, its samples were next to the intersection of PC1 and PC2 axis, between samples from sites 3 and 4. Concentrations of emerging contaminants at this sampling point were lower than downstream to WWTP. It reveals that a variety of natural processes simultaneously work to remove contaminants from water flowing in the river stream, such as adsorption on particulate matter, biodegradation, and photodegradation (Andreozzi et al. 2003; Benotti and Brownawell 2009; Buerge et al. 2003; Lin and Reinhard 2005; Yamamoto et al. 2009). Furthermore, dilution is another factor that may occur, since there are several other tributaries that flow into the water body between site 5 and the river mouth, which may not contribute significantly with the target compounds since they are in a rural area.
The decrease in concentrations between site 5 and 6 was different for each compound, since their persistence and physicochemical characteristics are different, and also depends on the characteristics of the water body. The grouping of variables in the PCA graph (Fig. 3) reveals different behaviors of contaminants in the aquatic body. The group of CAF, ATN, and NPX at the negative quadrant of PC2, opposite to the position of CMZ (at the positive quadrant), shows different trends for these two groups. From site 5 to site 6, there was a considerable decrease in concentrations of CAF, ATN, and NPX, which was also observed for PAR (although it was not included on PCA) (Table 2). On the other hand, CMZ presented similar concentrations in both sampling sites. This observation reveals that CMZ was persistent, and little difference in its concentration can be attributed to the dilution in the river mouth, while CAF, ATN, NPX, and PAR could be gone through biodegradation and/or photodegradation and/or adsorption processes (Benotti and Brownawell 2009; Buerge et al. 2003; Lin and Reinhard 2005; Yamamoto et al. 2009). CAF and PAR are considered biodegradable (t b1/2 of 5.2–13 days for CAF and t b1/2 of 1.2–13 days for PAR) while NPX is stable toward biodegradation but is photolabile (photodegradation t 1/2 of 0.7–1.9 h) (Benotti and Brownawell 2009; Lin et al. 2006; Lin and Reinhard 2005; Yamamoto et al. 2009). Besides this, sorption onto suspended particulate matter or sediments can occur. High distribution coefficients (k d) were observed in other studies for CAF (17.9–250 L kg−1), ATN (8.1 L kg−1), and NPX (1.9 L kg−1), although for PAR sorption is reported to be negligible (k d 0.5–2.8 L kg−1) (Lin et al. 2010; Martínez-Hernández et al. 2014; Yamamoto et al. 2009).
It can be also observed in Fig. 3 that CMZ is geometrically next to EC. A correlation graph of these two variables (Fig. 4) reveals a positive correlation between them (r = 0.59). This reinforces the persistent behavior of CMZ, as EC is considered a conservative parameter, predominantly resulting from chloride and sodium ions that remain in the water phase (Andreozzi et al. 2003; de Sousa et al. 2014; Kasprzyk-Hordern et al. 2009; Vieno et al. 2006). CMZ is resistant to photodegradation (half-life from 75 to 525 days) (Andreozzi et al. 2003) and biotransformation (Benotti and Brownawell 2009), besides having low sorption potential (k ow of 2.45). Furthermore, a 93 % of excretion in the form of glucuronides implies in its conversion into the original compound afterward, acting like a reservoir of this later (Bendz et al. 2005).
Other groups formed in the PCA graph were as follows: DOC, PRO, IBU, and DCF. The spatial variation of these emerging compounds indicates intermediary attenuation in relation to that observed for CMZ, CAF, ATN, and NPX. The decrease in concentrations between sites 5 and 6 ranged from about 50 to 60 % for PRO and IBU. Nevertheless, DCF which was in the positive quadrant of PC2 presented lower attenuation (about 20 %) similar to CMZ. As already mentioned, PRO can be photodegraded, having a short half-life (Andreozzi et al. 2003), while IBU presents satisfactory removal in WWTPs, indicating good biodegradability (Martínez Bueno et al. 2011). Unlike, DCF can be considered more persistent like CMZ. Previous studies report high persistence for both compounds in WWTPs (Zhang et al. 2008) that can explain the same behavior in surface waters. Due to their stability and persistence, CMZ and DCF have been suggested as a good tracer of wastewaters in rivers, even better than caffeine (de Sousa et al. 2014; Gasser et al. 2011), and several studies have been employed CMZ as tracer of wastewater contamination in river waters and groundwater (Fenech et al. 2013; Guérineau et al. 2014; Harwood 2014; Madoux-Humery et al. 2013). In the present study, simultaneous detection of CMZ and CAF at the site 3 clearly shows some release of sewage even before the WWTP.
In general, the seasonal variation showed an increase in the concentrations of pharmaceuticals and TCS in the dry periods, especially downstream of the WWTP (site 5) and in the river mouth (site 6). Contaminants like IBU, DCF, and CMZ, together with DOC, presented a more pronounced and well-defined seasonal variation compared to other compounds in the site 5 and are presented in Fig. 5, together with monthly precipitation in the São Carlos region. Higher concentrations in the dry period are related to the constant discharge of sewage/effluent associated to the low river flow in this period that decreases the dilution capacity of water body (Osorio et al. 2014). Another factor that can also contribute to the slight increase in the concentrations of IBU and DCF in the dry period is their higher consumption, since this period coincides with the winter season in the study area, due to which high incidence of pain, cold, and flu are reported.
It is worth noting that in September/2013, the overall monthly precipitation was quite low but during the sampling event, heavy rain was observed, due to which the concentrations of emerging contaminants were lower than the other campaigns. This caused the eventual dilution of contaminants that resulted in a different behavior from that expected for the seasonal profile obtained during the monitoring period as a whole.
Conclusion
This is the first study on the occurrence of emerging contaminants in the Monjolinho River, the main aquatic body of São Carlos, a prominent city of São Paulo state, Brazil. During 3 years of study, 12 compounds were investigated. Of all those target-compounds, only 17-α-ethinylestradiol hormone was not detected. The most frequently detected compounds and in higher concentrations were caffeine, paracetamol, and atenolol, while the most hardly detected were estrone and 17-β-estradiol, both in low concentrations. Along the river, the contamination increased from the source toward downstream of the WWTP, evidenced by raising concentrations of emerging contaminants and physicochemical parameters. From this, we conclude that the main source of emerging contaminants in the Monjolinho River is the discharge of effluent from WWTP and the non-treated sewage from the tributary Água Quente stream. Natural attenuation was very expressive for caffeine, atenolol, and naproxen, while carbamazepine and diclofenac were more persistent. However, these processes can result in the generation of by-products that might be more toxic than the original compounds. In this paper, we have reported nonexistent data about the occurrence and concentrations of some major emerging contaminants in the Monjolinho River. Considering that these compounds are not yet legally regulated by environmental laws, the reported results may assist in composing a dataset for water contamination with emerging contaminants in Brazil, which is extremely necessary to generate discussions that can result in future public policies on water quality related to these compounds.
References
Andreozzi R, Raffaele M, Nicklas P (2003) Pharmaceuticals in STP effluents and their solar photodegradation in aquatic environment. Chemosphere 50:1319–1330. doi:10.1016/S0045-6535(02)00769-5
ANVISA - Brazilian Health Surveillance Agency (2003) Guia para validação de métodos analíticos e bioanalíticos, Resolução n. 899 de 29/05/2003 vol 2014
Aris AZ, Shamsuddin AS, Praveena SM (2014) Occurrence of 17α-ethynylestradiol (EE2) in the environment and effect on exposed biota: a review. Envinron Int 69:104–119. doi:10.1016/j.envint.2014.04.011
Baker DR, Kasprzyk-Hordern B (2013) Spatial and temporal occurrence of pharmaceuticals and illicit drugs in the aqueous environment and during wastewater treatment: new developments. Sci Total Environ 454–455:442–456. doi:10.1016/j.scitotenv.2013.03.043
Bendz D, Paxéus NA, Ginn TR, Loge FJ (2005) Occurrence and fate of pharmaceutically active compounds in the environment, a case study: Höje River in Sweden. J Hazard Mater 122:195–204. doi:10.1016/j.jhazmat.2005.03.012
Benotti MJ, Brownawell BJ (2009) Microbial degradation of pharmaceuticals in estuarine and coastal seawater. Environ Pollut 157:994–1002. doi:10.1016/j.envpol.2008.10.009
Bianchi J, Espindola ELG, Marin-Morales MA (2011) Genotoxicity and mutagenicity of water samples from the Monjolinho River (Brazil) after receiving untreated effluents. Ecotoxicol Environ Saf 74:826–833. doi:10.1016/j.ecoenv.2010.11.006
Brausch JM, Rand GM (2011) A review of personal care products in the aquatic environment: environmental concentrations and toxicity. Chemosphere 82:1518–1532. doi:10.1016/j.chemosphere.2010.11.018
Buerge IJ, Poiger T, Müller MD, Buser H-R (2003) Caffeine, an anthropogenic marker for wastewater contamination of surface waters. Environ Sci Technol 37:691–700. doi:10.1021/es020125z
Caldas S, Bolzan C, Guilherme J, Silveira M, Escarrone A, Primel E (2013) Determination of pharmaceuticals, personal care products, and pesticides in surface and treated waters: method development and survey. Environ Sci Pollut Res 20:5855–5863. doi:10.1007/s11356-013-1650-9
Camacho-Muñoz D, Martín J, Santos JL, Aparicio I, Alonso E (2010) Occurrence, temporal evolution and risk assessment of pharmaceutically active compounds in Donana Park (Spain). J Hazard Mater 183:602–608
Campagna AF, Fracácio R, Rodrigues BK, Eler MN, Verani NF, Espíndola ELG (2008) Analyses of the sediment toxicity of Monjolinho River, São Carlos, São Paulo State, Brazil, using survey, growth and gill morphology of two fish species (Danio rerio and Poecilia reticulata). Braz Arch Biol Technol 51:193–201
Cassiano NM, Barreiro JC, Martins LRR, Oliveira RV, Cass QB (2009) Chromatographic methods validation for analysis of small molecules in biological matrices. Quim Nova 32:1021–1030
Chiba W, Passerini M, Baio J, Torres J, Tundisi J (2011) Seasonal study of contamination by metal in water and sediment in a sub-basin in the southeast of Brazil. Braz J Biol 71:833–843
Collado N (2014) Pharmaceuticals occurrence in a WWTP with significant industrial contribution and its input into the river system. Environ Pollut 185:202–212. doi:10.1016/j.envpol.2013.10.040
Conley JM, Symes SJ, Schorr MS, Richards SM (2008) Spatial and temporal analysis of pharmaceutical concentrations in the upper Tennessee River basin. Chemosphere 73:1178–1187. doi:10.1016/j.chemosphere.2008.07.062
CETESB - São Paulo State Environmental Agency (2012) Qualidade das Águas Superficiais no Estado de São Paulo. http://www.cetesb.sp.gov.br/userfiles/file/agua/aguas-superficiais/relatorio-aguas-superficiais-2012-substituido-em-060513.zip. Accessed May 2014
Daneshvar A (2012) Evaluating pharmaceuticals and caffeine as indicators of fecal contamination in drinking water sources of the Greater Montreal region. Chemosphere 88:131–139. doi:10.1016/j.chemosphere.2012.03.016
de Sousa DNR, Mozeto AA, Carneiro RL, Fadini PS (2014) Electrical conductivity and emerging contaminant as markers of surface freshwater contamination by wastewater. Sci Total Environ 484:19–26. doi:10.1016/j.scitotenv.2014.02.135
Deblonde T, Hartemann P (2013) Environmental impact of medical prescriptions: assessing the risks and hazards of persistence, bioaccumulation and toxicity of pharmaceuticals. Public Health 127:312–317. doi:10.1016/j.puhe.2013.01.026
Esteban S, Gorga M, Petrovic M, González-Alonso S, Barceló D, Valcárcel Y (2014) Analysis and occurrence of endocrine-disrupting compounds and estrogenic activity in the surface waters of Central Spain. Sci Total Environ 466–467:939–951. doi:10.1016/j.scitotenv.2013.07.101
EC - European Community (2002) Comission Decision 2002/657/EC vol 2002/657/EC Official Journal of the European Communities
EC - European Community (2013) Directive 2013/39/EU of the European Parliament and of the Council of 12 August 2013
Fatta-Kassinos D, Meric S, Nikolaou A (2011) Pharmaceutical residues in environmental waters and wastewater: current state of knowledge and future research. Anal Bioanal Chem 399:251–275. doi:10.1007/s00216-010-4300-9
Fenech C, Nolan K, Rock L, Morrissey A (2013) An SPE LC–MS/MS method for the analysis of human and veterinary chemical markers within surface waters: an environmental forensics application. Environ Pollut 181:250–256. doi:10.1016/j.envpol.2013.06.012
Froehner S, Machado K, Falcão F, Monnich C, Bessa M (2011) Inputs of domestic and industrial sewage in Upper Iguassu, Brazil identified by emerging compounds. Water Air Soil Pollut 215:251–259. doi:10.1007/s11270-010-0475-0
Gasser G (2011) Evaluation of micropollutant tracers. II. Carbamazepine tracer for wastewater contamination from a nearby water recharge system and from non-specific sources. Desalination 273:398–404. doi:10.1016/j.desal.2011.01.058
Geist J (2011) Integrative freshwater ecology and biodiversity conservation. Ecol Indic 11:1507–1516. doi:10.1016/j.ecolind.2011.04.002
GWRC - Global Water Research Coalition (2008) Development of an international priority list of parmaceuticals relevant for fhe water cycle. http://edepot.wur.nl/138086. Accessed may 2014
Guérineau H (2014) Source tracking of leaky sewers: a novel approach combining fecal indicators in water and sediments. Water Res 58:50–61. doi:10.1016/j.watres.2014.03.057
Harwood JJ (2014) Molecular markers for identifying municipal, domestic and agricultural sources of organic matter in natural waters. Chemosphere 95:3–8. doi:10.1016/j.chemosphere.2013.09.104
IBGE Brazilian Institute of Geography and Statistics (2013) Cities - São Carlos
Jardim WF, Montagner CC, Pescara IC, Umbuzeiro GA, Di Dea Bergamasco AM, Eldridge ML, Sodré FF (2012) An integrated approach to evaluate emerging contaminants in drinking water. Sep Purif Technol 84:3–8. doi:10.1016/j.seppur.2011.06.020
Jjemba PK (2006) Excretion and ecotoxicity of pharmaceutical and personal care products in the environment. Ecotoxicol Environ Saf 63:113–130. doi:10.1016/j.ecoenv.2004.11.011
Kasprzyk-Hordern B, Dinsdale RM, Guwy AJ (2008) The occurrence of pharmaceuticals, personal care products, endocrine disruptors and illicit drugs in surface water in South Wales, UK. Water Res 42:3498–3518. doi:10.1016/j.watres.2008.04.026
Kasprzyk-Hordern B, Dinsdale RM, Guwy AJ (2009) The removal of pharmaceuticals, personal care products, endocrine disruptors and illicit drugs during wastewater treatment and its impact on the quality of receiving waters. Water Res 43:363–380. doi:10.1016/j.watres.2008.10.047
Kosma CI, Lambropoulou DA, Albanis TA (2014) Investigation of PPCPs in wastewater treatment plants in Greece: occurrence, removal and environmental risk assessment. Sci Total Environ 466–467:421–438. doi:10.1016/j.scitotenv.2013.07.044
Kuster M, Azevedo DA, de Alda MJ L, Aquino Neto FR, Barceló D (2009) Analysis of phytoestrogens, progestogens and estrogens in environmental waters from Rio de Janeiro (Brazil). Envinron Int 35:997–1003. doi:10.1016/j.envint.2009.04.006
Lin AY, Reinhard M (2005) Photodegradation of common environmental pharmaceuticals and estrogens in river water. Environ Toxicol Chem 24:1303–1309
Lin AY-C, Plumlee MH, Reinhard M (2006) Natural attenuation of pharmaceuticals and alkylphenol polyethoxylate metabolites during river transport: photochemical and biological transformation. Environ Toxicol Chem 25:1458–1464. doi:10.1897/05-412R.1
Lin AY-C, Lin C-A, Tung H-H, Chary NS (2010) Potential for biodegradation and sorption of acetaminophen, caffeine, propranolol and acebutolol in lab-scale aqueous environments. J Hazard Mater 183:242–250. doi:10.1016/j.jhazmat.2010.07.017
Lindström A, Buerge IJ, Poiger T, Bergqvist P-A, Müller MD, Buser H-R (2002) Occurrence and environmental behavior of the bactericide triclosan and its methyl derivative in surface waters and in wastewater. Environ Sci Technol 36:2322–2329. doi:10.1021/es0114254
Loos R, Locoro G, Contini S (2010) Occurrence of polar organic contaminants in the dissolved water phase of the Danube River and its major tributaries using SPE-LC-MS2 analysis. Water Res 44:2325–2335. doi:10.1016/j.watres.2009.12.035
López-Roldán R, de Alda ML, Gros M, Petrovic M, Martín-Alonso J, Barceló D (2010) Advanced monitoring of pharmaceuticals and estrogens in the Llobregat River basin (Spain) by liquid chromatography–triple quadrupole-tandem mass spectrometry in combination with ultra performance liquid chromatography–time of flight-mass spectrometry. Chemosphere 80:1337–1344. doi:10.1016/j.chemosphere.2010.06.042
Madoux-Humery A-S, Dorner S, Sauvé S, Aboulfadl K, Galarneau M, Servais P, Prévost M (2013) Temporal variability of combined sewer overflow contaminants: evaluation of wastewater micropollutants as tracers of fecal contamination. Water Res 47:4370–4382. doi:10.1016/j.watres.2013.04.030
Madureira TV, Rocha MJ, Cruzeiro C, Rodrigues I, Monteiro RAF, Rocha E (2012) The toxicity potential of pharmaceuticals found in the Douro River estuary (Portugal): evaluation of impacts on fish liver, by histopathology, stereology, vitellogenin and CYP1A immunohistochemistry, after sub-acute exposures of the zebrafish model. Environ Toxicol Pharmacol 34:34–45. doi:10.1016/j.etap.2012.02.007
Martínez Bueno MJ, Uclés S, Hernando MD, Dávoli E, Fernández-Alba AR (2011) Evaluation of selected ubiquitous contaminants in the aquatic environment and their transformation products. a pilot study of their removal from a sewage treatment plant. Water Res 45:2331–2341. doi:10.1016/j.watres.2011.01.011
Martínez-Hernández V, Meffe R, Herrera S, Arranz E, de Bustamante I (2014) Sorption/desorption of non-hydrophobic and ionisable pharmaceutical and personal care products from reclaimed water onto/from a natural sediment. Sci Total Environ 472:273–281. doi:10.1016/j.scitotenv.2013.11.036
Moldovan Z (2006) Occurrences of pharmaceutical and personal care products as micropollutants in rivers from Romania. Chemosphere 64:1808–1817. doi:10.1016/j.chemosphere.2006.02.003
Montagner CC, Jardim WF (2011) Spatial and seasonal variations of pharmaceuticals and endocrine disruptors in the Atibaia River, São Paulo State (Brazil). J Braz Chem Soc 22:1452–1462
Moreira DS, Aquino SF, Afonso RJ, Santos EP, de Padua VL (2009) Occurrence of endocrine disrupting compounds in water sources of Belo Horizonte Metropolitan Area, Brazil. Environ Technol 30:1041–1049. doi:10.1080/09593330903052830
Orlando EF, Ellestad LE Sources, concentrations, and exposure effects of environmental gestagens on fish and other aquatic wildlife, with an emphasis on reproduction. Gen Comp Endocrinol. doi:http://dx.doi.org/10.1016/j.ygcen.2014.03.038
Osorio V (2014) Hydrological variation modulates pharmaceutical levels and biofilm responses in a Mediterranean river. Sci Total Environ 472:1052–1061. doi:10.1016/j.scitotenv.2013.11.069
Osorio V, Marcé R, Pérez S, Ginebreda A, Cortina JL, Barceló D (2012) Occurrence and modeling of pharmaceuticals on a sewage-impacted Mediterranean river and their dynamics under different hydrological conditions. Sci Total Environ 440:3–13. doi:10.1016/j.scitotenv.2012.08.040
Peng X, Yu Y, Tang C, Tan J, Huang Q, Wang Z (2008) Occurrence of steroid estrogens, endocrine-disrupting phenols, and acid pharmaceutical residues in urban riverine water of the Pearl River Delta, South China. Sci Total Environ 397:158–166. doi:10.1016/j.scitotenv.2008.02.059
Petersen K, Heiaas HH, Tollefsen KE (2014) Combined effects of pharmaceuticals, personal care products, biocides and organic contaminants on the growth of Skeletonema pseudocostatum. Aquat Toxicol 150:45–54. doi:10.1016/j.aquatox.2014.02.013
Petrović M, Skrbić B, Zivančev J, Ferrando-Climent L, Barcelo D (2014) Determination of 81 pharmaceutical drugs by high performance liquid chromatography coupled to mass spectrometry with hybrid triple quadrupole–linear ion trap in different types of water in Serbia. Sci Total Environ 468–469:415–428. doi:10.1016/j.scitotenv.2013.08.079
Printes LB, Fernandes MN, Espíndola ELG (2011) Laboratory measurements of biomarkers and individual performances in Chironomus xanthus to evaluate pesticide contamination of sediments in a river of southeastern Brazil. Ecotoxicol Environ Saf 74:424–430. doi:10.1016/j.ecoenv.2010.10.033
Ribeiro AR, Carvalho MF, Afonso CMM, Tiritan ME, Castro PML (2010) Microbial degradation of 17β-estradiol and 17α-ethinylestradiol followed by a validated HPLC-DAD method. J Environ Sci Health B 45:265–273. doi:10.1080/03601231003704523
Ricart M (2010) Triclosan persistence through wastewater treatment plants and its potential toxic effects on river biofilms. Aquat Toxicol 100:346–353. doi:10.1016/j.aquatox.2010.08.010
Schwarzenbach RP, Escher BI, Fenner K, Hofstetter TB, Johnson CA, von Gunten U, Wehrli B (2006) The challenge of micropollutants in aquatic systems. Science 313:1072–1077
Silva BF, Jelic A, López-Serna R, Mozeto AA, Petrovic M, Barceló D (2011) Occurrence and distribution of pharmaceuticals in surface water, suspended solids and sediments of the Ebro river basin, Spain. Chemosphere 85:1331–1339. doi:10.1016/j.chemosphere.2011.07.051
Sodré FF, Pescara IC, Montagner CC, Jardim WF (2010) Assessing selected estrogens and xenoestrogens in Brazilian surface waters by liquid chromatography–tandem mass spectrometry. Microchem J 96:92–98. doi:10.1016/j.microc.2010.02.012
Sui Q, Huang J, Deng S, Yu G, Fan Q (2010) Occurrence and removal of pharmaceuticals, caffeine and DEET in wastewater treatment plants of Beijing, China. Water Res 44:417–426. doi:10.1016/j.watres.2009.07.010
Ternes TA (1998) Occurrence of drugs in German sewage treatment plants and rivers. Water Res 32:3245–3260. doi:10.1016/S0043-1354(98)00099-2
US-FDA - U.S. Food and Drug Administration (2001) Guidance for industry, bioanalytical method validation
Valcárcel Y, Alonso SG, Rodríguez-Gil JL, Maroto RR, Gil A, Catalá M (2011) Analysis of the presence of cardiovascular and analgesic/anti-inflammatory/antipyretic pharmaceuticals in river- and drinking-water of the Madrid Region in Spain. Chemosphere 82:1062–1071. doi:10.1016/j.chemosphere.2010.10.041
Vazquez-Roig P, Andreu V, Blasco C, Picó Y (2012) Risk assessment on the presence of pharmaceuticals in sediments, soils and waters of the Pego–Oliva Marshlands (Valencia, eastern Spain). Sci Total Environ 440:24–32. doi:10.1016/j.scitotenv.2012.08.036
Verlicchi P, Al Aukidy M, Jelic A, Petrović M, Barceló D (2014) Comparison of measured and predicted concentrations of selected pharmaceuticals in wastewater and surface water: a case study of a catchment area in the Po Valley (Italy). Sci Total Environ 470–471:844–854. doi:10.1016/j.scitotenv.2013.10.026
Vieno NM, Tuhkanen T, Kronberg L (2006) Analysis of neutral and basic pharmaceuticals in sewage treatment plants and in recipient rivers using solid phase extraction and liquid chromatography–tandem mass spectrometry detection. J Chromatogr A 1134:101–111. doi:10.1016/j.chroma.2006.08.077
Wang G et al (2012) Endocrine disrupting chemicals in New Orleans surface waters and Mississippi Sound sediments. J Environ Monit 14:1353–1364. doi:10.1039/c2em30095h
World Health Organization/UNICEF - (2013) Progress on sanitation and drinking water – update 2013. http://www.wssinfo.org/fileadmin/user_upload/resources/JMPreport2013.pdf. Accessed February 2014
Yamamoto H (2009) Persistence and partitioning of eight selected pharmaceuticals in the aquatic environment: laboratory photolysis, biodegradation, and sorption experiments. Water Res 43:351–362. doi:10.1016/j.watres.2008.10.039
Yan Z, Lu G, Liu J, Jin S (2012) An integrated assessment of estrogenic contamination and feminization risk in fish in Taihu Lake, China. Ecotoxicol Environ Saf 84:334–340. doi:10.1016/j.ecoenv.2012.08.010
Yu C-P, Chu K-H (2009) Occurrence of pharmaceuticals and personal care products along the West Prong Little Pigeon River in east Tennessee, USA. Chemosphere 75:1281–1286. doi:10.1016/j.chemosphere.2009.03.043
Zhang Y, Geißen S-U, Gal C (2008) Carbamazepine and diclofenac: removal in wastewater treatment plants and occurrence in water bodies. Chemosphere 73:1151–1161. doi:10.1016/j.chemosphere.2008.07.086
Zhao J-L, Ying G-G, Wang L, Yang J-F, Yang X-B, Yang L-H, Li X (2009) Determination of phenolic endocrine disrupting chemicals and acidic pharmaceuticals in surface water of the Pearl Rivers in South China by gas chromatography–negative chemical ionization–mass spectrometry. Sci Total Environ 407:962–974. doi:10.1016/j.scitotenv.2008.09.048
Zuccato E, Castiglioni S, Fanelli R (2005) Identification of the pharmaceuticals for human use contaminating the Italian aquatic environment. J Hazard Mater 122:205–209. doi:10.1016/j.jhazmat.2005.03.001
Acknowledgments
The authors would like to thank the Coordination of Improvement of Higher Education Personal (CAPES, research project n° 3039/2010), the National Council of Scientific and Tecnological Development (CNPq, grants #140771/2012-3 and 403580/2013-7), the São Paulo Research Foundation (FAPESP, processes 2012/14403-7 and 10/01731-0), and ANP-Petrobrás (project 0050.0043180.08.4; agreement 4600295977) for the financial support of this project.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Hongwen Sun
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 16 kb)
Rights and permissions
About this article
Cite this article
Campanha, M.B., Awan, A.T., de Sousa, D.N.R. et al. A 3-year study on occurrence of emerging contaminants in an urban stream of São Paulo State of Southeast Brazil. Environ Sci Pollut Res 22, 7936–7947 (2015). https://doi.org/10.1007/s11356-014-3929-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-014-3929-x