Abstract
To assess the status of severity of fluoride contamination in lateritic Bankura and Purulia districts of West Bengal, concentrations of fluoride in different water sources and agricultural field soils were investigated. The fluoride content (mg/l) was observed to differ with aquifer depths: 0.19–0.47 in dug wells, 0.01–0.17 in shallow tube wells, and 0.07–1.6 in deep tube wells. Fluoride within the World Health Organization (WHO) prescribed range (1.0–1.5 mg/l) was estimated only in ~17 % of the total collected water samples while ~67 % showed <0.7 mg/l fluoride and thus may impede in the production and maintenance of healthy teeth and bones of the residents, especially children. Fluoride in water was found to be significantly correlated (r = 0.63) with pH. The exposure dose of fluoride (mg/kg/day) from drinking water in infants, children, and adults was estimated in the ranges 0.02–0.53, 0.01–0.24, and 0.01–0.14, respectively against the standard value of 0.05. A clear risk of dental fluorosis is apparent in infants and children of the study area. The fluoride in soil (55–399 mg/kg) was detected to be significantly correlated with the fluoride content in deep tube wells and soil pH (r = 0.56 and 0.71, respectively). The relationships of soil fluoride with total hardness and that with phosphate were not significant. There is a high possibility of bioaccumulation of fluoride from contaminated soil and water of the study area to cultivated crops. This will enhance the quantity of fluoride intake into human food chain in addition to drinking water pathway.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Alarming concentration of fluoride in groundwater associated with weathering of igneous and metamorphic rocks such as granites and gneisses has been globally reported to be a burning environmental issue from Algeria, Brazil, Canada, China, Ethiopia, Ghana, India, Iran, Italy, Japan, Jordan, Kenya, Korea, Malawi, Mexico, Norway, Pakistan, Sri Lanka, Thailand, Turkey, and the USA (WHO 2006; Brindha and Elango 2011). An estimated 200 million people from 29 countries are under health-related risks due to the consumption of high fluoride-contaminated groundwater (Ayoob and Gupta 2006; Bhattacharya and Chakrabarti 2011). In India, fluorosis has been pronounced endemic in 20 out of total 29 states including 65 % of rural habitations by the United Nations International Children’s Emergency Fund (UNICEF) (UNICEF 1999; Kundu and Mandal 2009). Presence of moderate to high concentration of fluoride in groundwater has been reported from different states of India (Salve et al. 2008; Sankararamakrishnan et al. 2008; Suthar et al. 2008; Mishra et al. 2009; Brindha et al. 2011; Dar et al. 2011) including West Bengal (Chatterjee et al. 2008; Kundu and Mandal 2009; Bhattacharya and Chakrabarti 2011; Chakrabarti and Bhattacharya 2013). More than 65 million Indians including 6 million children are at risk due to the presence of excessive concentration of fluoride beyond the desirable limits in groundwater (Andezhath et al. 1999; UNICEF 1999). The natural dissolution of fluorite, apatite, and topaz from local bedrocks leads to high fluoride concentration in groundwater (Suthar et al. 2008). Along with it, different man-made activities such as usage of phosphate fertilizers and pesticides, discharge of untreated or partially treated sewage and sludge in groundwater, overexploitation of groundwater for irrigation leading to depletion of water table, etc. have been argued to cause an increase in fluoride concentration in groundwater (EPA 1997; Ramanaiah et al. 2006).
Fluoride may enter into the biological systems via water, food, industrial exposure, drug, and cosmetic products although drinking water has been established to be the major contributor (Susheela 1999). The intake of fluoride within permissible limits of 0.5–1.0 mg/l is beneficial for human health in production and maintenance of healthy teeth and bones (Wood 1974), while excessive intake causes chronic dental and skeletal fluorosis, manifested by mottling teeth in mild cases, softening of bones and neurological damage in severe cases (Steinberg et al. 1955). The World Health Organization (WHO) has made 1.5 mg/l as the permissible limit for fluoride (WHO 2006) while the Bureau of Indian Standards (BIS) and Indian Council of Medical Research (ICMR) have made 0.6–1.2 and 1.5 mg/l as the desirable range and permissible limit for fluoride, respectively (ICMR 1975; BIS 2003). Fluoride concentration in excess of 2.0 mg/l causes conditions for dense and brittle bone, and also dental problems (Chatterjee et al. 2008). The United States Public Health Service (USPHS) has recommended a range of allowable concentrations for fluoride in drinking water for a region depending on its climatic conditions. This is because the amount of water consumed and consequently the amount of fluoride ingested being influenced primarily by the air temperature of that region (USPHS 1962).
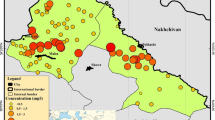
In West Bengal, fluoride contamination has been reported from 60 blocks spread over eight districts (Chatterjee et al. 2008). The present study was carried out in lateritic Bankura and Purulia districts of West Bengal, where groundwater is the main source of drinking as well as irrigation water. But, there is no centralized drinking water treatment facility in the area. The Public Health Engineering Department (PHED), Government of West Bengal by conducting a preliminary survey in 2007 had previously reported the presence of fluoride in some of the blocks of these two districts (PHED Report 2007). On the basis of the report, the PHED had sealed numerous unsafe deep tube wells of this region and painted them with red color. Keeping an account of the importance of public health, the main objectives of this survey were to investigate the present status of quality and safety of drinking water (with respect to fluoride contents) for the villagers dependent on groundwater from the currently used deep tube wells in Bankura and Purulia districts, and to determine the fluoride content of agricultural field soil for understanding the possibility of bioconcentration of fluoride from soil and water to cultivated crops. An attempt has also been made to assess the exposure dose of fluoride in drinking water from the deep tube wells of the study area.
Materials and methods
Study area
The Bankura district is geographically located between latitude 22°38′–23°38′ N and longitude 86°36′–87°46′ E, covering an area of 6882 km2 with a population of 3,596,674, and out of this, nearly 91.7 % live in rural areas whereas the Purulia district is located between latitude 22°42′–23°42′ N and longitude 85°49′–86°54′ E, having an area of 6259 km2 with 2,930,115 populations, and out of this nearly 87.3 % live in rural areas (Census 2011). During summer, the climate in the study area is dry and tremendously hot (the maximum temperature rises to ~52 °C) with low to moderate monsoons (average rainfall in Bankura district is 1345 mm and that in Purulia district is 1329 mm), while in winter, it is very cool with the minimum temperature dropping down to ~4 °C. The average elevation of Bankura and Purulia districts are 78 and 228 m, respectively. The overall drainage pattern of the area is parallel to sub-parallel and is mainly controlled by geological structural elements. The average slope of land in the districts varies from 0.4 to 10 %. Some parts of the two districts were reported to be affected with fluoride (PHED Report 2007). The mostly affected ten blocks of Bankura district (Chhatna, Shaltora, Gangajal Ghati, Bankura II, Indpur, Hirbundh, Khatra, Sarenga, Raipur, and Simlapal) and six blocks of Purulia district (Hura, Kashipur, Raghunathpur I, Santuri, Puncha, and Manbazar I) have been chosen for the present study (Fig. 1). The major occupation of the residents of these two districts is farming, and irrigation is mainly carried out through shallow and deep tube wells. Rice is the main crop of both the districts. Besides rice, the major cultivated crops are potato, wheat, vegetables, mustard, til, and maize.
The Bankura district is the connecting link between the plains of Bengal on the east and Chotanagpur plateau on the west. The areas to the east and north-east are low-lying recent alluvial plains containing seams of coal belonging to the Raniganj system. The surface gradually rises to the west, giving way to undulating lateritic terrain, interspersed with hillocks of metamorphic or gneissose rocks (O’Malley 1995). Thus, geomorphologically, the Bankura district can be divided into three zones. The western part is dominated by hilly hard rock (comprising 8 blocks out of the total 22 blocks) while the eastern part by sedimentary alluvial plains (comprising 6 blocks). The central part is a mixed formation of both rocky and alluvial segments (comprising 8 blocks) (Chakrabarti and Bhattacharya 2013). Strong massive runs of hornblendic varieties stretch across the region in tolerably continuous lines, the general strike being nearly east and west. Due to undulated topography of the Purulia district (comprising total 20 blocks) nearly 50 % of the rainfall flows away as surface runoff. The district is mostly covered with residual soil formed by weathering of bed rocks. Lateritic soil prevails in the uplands while in the plains, reddish clay loam or white to reddish clay are common. The entire area is reported to be underlain by Pre-Cambrian metamorphic rocks like (Chotanagpur) granite gneiss, biotite granite gneiss, calc-granulites, ultra-basic and meta-basic rocks, meta-sedimentaries, including crystalline limestone, hornblende schist, biotite gneiss, pegmatite, and quartz-vein containing fluoride bearing minerals like apatite, fluorite, and mica (Bhattacharya and Chakrabarti 2011).
Sample collection
A total of 124 water samples were randomly collected from different sources such as deep tube well, shallow tube well, dug well, and pond spread over the two districts during the month of December, 2013, following standard sampling techniques. A total of 79 agricultural field soil samples were also collected from a depth of 0–45 cm by composite sampling technique and stored in plastic zipper bags with proper labeling for further analysis. The global positioning system (GPS) was used to map the sampling sites. The sampling was done purposefully in December with the premise that it will help to record the highest possible value of fluoride in groundwater over the other months since the heavy rains received during monsoonal months (July–October) would leach out whatever amount of fluoride present in soil and also applied through nitrogen and phosphate fertilizers to groundwater aquifers (Kundu and Mandal 2009). The sampling was done at a spatial distance of around ~2 km away from each other points. Before collection, the water was left to run from the sources (shallow and deep tube wells) for about 10 min to get representative samples with stable electrical conductivity (Khaiwal and Garg 2007). All the water samples were collected in pre-cleaned polyethylene bottles of 500-ml capacity. The sampling bottles were soaked in 1:1 diluted HCl solution for 24 h, washed with distilled water, and were washed again prior to each sampling with the filtrates of the sample.
Sample treatment
The water samples were filtered through 0.45-μ Millipore filter paper, and 50-ml filtered samples were kept in polyethylene bottles at 4 °C for analysis. The soil samples were immediately sun-dried after collection and later dried in the hot air oven at 60 °C for 72 h. The dried soil samples were then grinded by cautiously disaggregating in a mortar and screened through 2.0-mm-pore-sized sieve to get homogenized representative powder sample. Finally, the samples were stored in airtight polyethylene bags at room temperature. Proper care was taken at each step to minimize any contamination.
Sample analysis
The fluoride concentration in water samples was determined electrochemically using the Thermo Scientific Orion 4star pH/Ion Selective Electrode bench top meter by an approved American Society for Testing and Materials (ASTM) standard test method for fluoride ion in water (ASTM D 1179). This method is applicable to the measurement of fluoride in water in the concentration range of 0–19,990 ppm. The electrode used was an Orion ISE 9609BNWP and 960900 fluoride combination electrode. Standard fluoride solutions (0.1–10 mg/l) were prepared from a stock solution (100 mg/l) of Na2F. As per experimental requirement, 1 ml of total ionic strength adjusting buffer grade III (TISAB III, Orion) was added in 10 ml of sample. The ion meter was calibrated for a slope of −59.2 ± 2.0. Total fluoride content in soil samples was estimated through NaOH fusion method (McQuaker and Gurney 1977). This method involves fusion of soil samples with 16 N NaOH in Ni crucibles placed in muffle furnace and slowly raising the temperature to 600 °C for 30 min, followed by dissolving the residue by heating with water on a hot plate. After the treatment samples were removed and allowed to cool, then 10 ml of distilled water was added to the samples with stirring to adjust the pH to 8–9. Then, the samples were filtered, transferred to 100-ml volumetric flasks, and finally diluted with double-distilled water to make 100 ml. To 10 ml of the above extract, 1 ml of TISAB III solution was added and mixed well, and the fluoride content was estimated using the fluoride selective electrode in the same way as discussed in case of water sample analysis. The detection limit of this method is 0.005 mg/l. All the experiments were carried out in triplicate and the results were found reproducible with ±2 % error.
The pH values of the water and soil samples were measured in situ immediately after collection with the help of a portable pH meter (Eutech multi-parameter tester: PCSTestr 35). Total hardness of water and soil samples (by titrimetric method using standard EDTA solutions) and total phosphate content of soil samples (by molybdenum blue method using a Systronics 166 VIS spectrophotometer) were analyzed following standard method (APHA 2005).
Assessment of exposure dose of fluoride
The exposure doses for infants (6-kg body weight), children (20 kg), and adults (70 kg) were calculated by following the generic equation (Jha et al. 2009),
where
- ED:
-
Exposure dose of fluoride (mg/kg/day)
- C :
-
Concentration of fluoride in water (mg/l)
- BW:
-
Body weight (kg)
- WI:
-
Water intake (l/day)
The evaluated minimum, maximum, and average fluoride concentrations of deep tube wells of the study area were used for the calculation of fluoride intake. Sixty-eight families comprising of total 242 individuals residing in the 16 blocks of the study area were interrogated during our investigation to evaluate the daily average water intake values for infants, children, and adults. On average, infants, children, and adults living in the study area drink 1, 3, and 6 l of water per day, respectively. The exposure doses of fluoride were estimated on the above said water intake values.
Statistical analysis
Means of three replicates, standard deviation, and Pearson’s correlation coefficient (r) on the experimental data were calculated using SPSS, version 15.0 for windows (SPSS Inc., Chicago, USA). The sampling location map and contour map were prepared using Golden surfer, version 8.0 (Golden Software Inc., Colorado, USA).
Results and discussion
Fluoride contamination in water
The collected water samples irrespective of their sources were found to be free from color, odor, and turbidity. The fluoride contents in water samples of the study area varied greatly in different blocks as depicted in Table 1, but the concentration of fluoride in most of the samples was within the WHO- and BIS-recommended maximum permissible limit of 1.5 mg/l. The intake of fluoride in the range of 0.7–1.2 mg/l is beneficial for the production and maintenance of healthy teeth and bones in human beings (USEPA 2010). But, the results revealed that ~67 % water samples had fluoride contents <0.7 mg/l and thus cannot be utilized as sources of potable water. Figure 2 presents the district wise distribution of fluoride in water samples in different concentration ranges (<0.50, 0.50–1.0, 1.0–1.5, and >1.5 mg/l). In the Bankura district, none of the collected water sample was detected to exceed the permissible 1.5 mg/l fluoride content but in the Purulia district, four deep tube well water samples from Kashipur block were found to cross that limit. Figure 2 clearly indicates that the maximum habitations (more than 49 % in the Bankura district and 63 % in the Purulia district) have fluoride concentrations <0.50 mg/l. The maximum and minimum concentrations of fluoride in sampled water was 1.6 mg/l (deep tube well water, Kashipur block, Purulia) and 0.01 mg/l (shallow tube well water, Raipur block, Bankura), respectively, with the average value of 0.55 ± 0.24 mg/l. After evaluating the data, it is suggested that water from deep tube wells in the Kashipur block of Purulia district is unsuitable for drinking purpose as prolonged use of this fluoride-contaminated water (1.3 ± 0.32 mg/l) can lead to dental and skeletal fluorosis in residents. In surface water, the concentration of fluoride generally has a range of 0.01–0.30 mg/l (USEPA 2010). The pond water samples collected from the Chhatna block of Bankura district thus had presence of fluoride (0.08 ± 0.02 mg/l) within the permissible range. The fluoride content in water of the study area was observed to differ with depth of aquifers. For example, it ranged 0.19–0.47 mg/l in dug wells, 0.01–0.17 mg/l in shallow tube wells, and 0.07–1.6 mg/l in deep tube wells (Table 1). The spatial variation of fluoride concentration in water resources of the study area is shown in Fig. 3. After publication of the PHED report (2007), different mitigation steps like sealing of unsafe deep tube wells (fluoride concentration >1.5 mg/l), mass awareness campaigns, rain water harvesting initiatives, etc. had been taken by the Government of West Bengal. The present results show that those have enlightened the status of groundwater fluoride pollution of the study area. But, the presence of fluoride in ~67 % water samples below the desirable range (0.7–1.2 mg/l) has to be taken care of immediately. In drinking water treatment, fluoride may be applied at low levels to aid in dental and skeletal health of the residents, especially children of the study area.
The presence of high pH and low calcium and magnesium contents in water had been reported to be the two major factors responsible for rapid leaching of fluoride and consequently resulting into elevated concentration of it in water (Rao et al. 1993). In the study area, the pH in the sampled water was found to vary from 6.5 ± 0.2 to 8.0 ± 0.3 (Table 1) and was significantly correlated (r = 0.63) with the fluoride contents (Fig. 4a). This finding is in agreement with earlier reports (Jha et al. 2009; Rao 2009; Brindha et al. 2011). According to Salve et al. (2008), fluoride is desorbed in clay in alkaline form and thus, alkaline pH is a more favorable condition for fluoride dissolution activity than acidic pH condition. In lateritic regions of West Bengal, alkaline groundwater had been previously reported to be associated with higher leaching of fluoride (Chatterjee et al. 2008; Chakrabarti and Bhattacharya 2013). Moreover, hardness, arising mainly due to the presence of calcium and magnesium ions in water, was observed to have a negative correlation with fluoride concentration (r = −0.32) of the study area (Fig. 4b). The hardness of the studied water samples (90–766 mg/l) was found to gradually decrease with increase in fluoride concentration (0.01–1.6 mg/l). This is also concurrent with the previous findings by Jha et al. (2009) and Brindha et al. (2011). The inverse relationship of fluoride with calcium and magnesium is possibly due to ion-exchange processes occurring in water systems (Salve et al. 2008).
Assessment of exposure dose of fluoride from drinking water
Residents of the study area in the absence of centralized drinking water treatment and distribution facilities only use the deep tube wells installed by the PHED for consuming drinking water. The exposure doses of fluoride from those deep tube wells of the study area were evaluated for infants, children, and adults (Table 2). For the calculation of fluoride intake, the minimum, maximum, and average fluoride levels of deep tube wells (0.07, 1.6, and 0.55 mg/l, respectively) as observed by us were employed. Due to prevailing scorching hot and dry tropical climate of the study area, on average, infants, children, and adults were found to consume 1, 3, and 6 l of water per day, respectively. The exposure doses were estimated on the above said water intake values. For infants, the exposure doses were found between 0.02 and 0.53 mg/kg/day against the standard value of 0.05 mg/kg/day, which is the minimum risk level (MRL) value recommended by the Agency for Toxic Substances and Disease Registry (ATSDR) (Ortiz et al. 1998). In children and adults, the exposure doses were evaluated in the ranges 0.01–0.24 and 0.01–0.14 mg/kg/day, respectively. Thus, the maximum exposure doses of fluoride for infants, children, and adults of the study area were estimated to be around 11, 5, and 3 times higher than the ATSDR’s MRL value. On average, infants, children, and adults were found to consuming 0.18 ± 0.11, 0.08 ± 0.04, and 0.05 ± 0.03 mg/kg/day fluoride, respectively. A clear endangerment of dental fluorosis is therefore apparent in infants and children of the study area. The results show that the average exposure dose of fluoride to adults is within the ATSDR’s MRL value from drinking water but, when intakes of fluoride from other sources (such as food, tea, vegetables, fruits, milk, meat, etc.) are taken into consideration, then the total exposure dose of fluoride to adults is expected to be much higher (Jha et al. 2009).
Fluoride contamination in soil
Soil receives fluoride from weathering of various minerals like apatite, fluorite, and mica which are the major natural sources of fluoride in the environment. Different anthropogenic activities including agriculture are also responsible for disposal of fluoride in soil. Irrigation increases the sodicity of the soil while fertilizers applied for cultivation lead to high fluoride content in soil which ultimately leaches to the aquifers (Brindha et al. 2011). The fluoride content in agricultural field soils of the study area is presented in Table 3. The maximum and minimum fluoride in sampled soil was 399 mg/kg (Raghunathpur I block, Purulia) and 51 mg/kg (Raipur block, Bankura), respectively. The observed range is higher than those that are reported for various tropical areas of the world (Hall and Cain 1972). Moreover, a significant correlation (r = 0.56) was detected between the fluoride content in deep tube wells and that in soils, collected from nearby cropping fields (Fig. 5a). Thus, it can be concluded that the soil of the study area contains considerable labile fluorine which is not tightly bound by silicate and phosphate minerals (Jha et al. 2009; Saini et al. 2013). Soil pH, formation of stable aluminum and calcium complexes, rate of deposition, and climatic conditions are established to be the major factors that influence the mobility of fluoride in soil (Pickering 1985). In this study, a significant positive correlation (r = 0.71) was observed between soil fluoride content and soil pH (Fig. 5b) whereas relationship between soil fluoride content and total hardness of soil (r = 0.30) and that between soil fluoride content and soil phosphate (r = 0.04) were not significant (Fig. 5c, d, respectively). The results thus show that the enrichment of fluoride of the study area is associated with the slightly alkaline nature of soil while use of phosphate fertilizers in agriculture has negligible impact. This is concurrent with the previous findings by Farooqi et al. (2009), and Kundu and Mandal (2009).
If the present trend of using large amount of fluoride-contaminated groundwater for irrigation continues, the significant positive correlation between water fluoride and soil fluoride contents of the study area indicates that there is a high possibility of increase of fluoride concentration in water and ultimately its bioaccumulation into irrigated crops in near future. Plants by using their roots absorb fluoride from soil and then transport it to different organs, mainly the leaves via xylematic flow (Davison and Weinstein 1998). Different researchers had shown bioconcentration of fluoride from soil and water to plants at various degrees (Fornasiero 2001; Kalinic et al. 2005; Kozyrenko et al. 2007; Saini et al. 2013). Thus, consumption of food, vegetables, and fruits is becoming another potent route for fluoride entry into human food chain along with the present drinking water pathway. Ingestion of plants with a high fluorine concentration has been reported to cause chronic toxicity in grazing animals and humans and leads to bone damage as well as tooth wear (Clark and Stewart 1983).
Conclusions
The present status of fluoride contamination in groundwater and agricultural field soil of lateritic Bankura and Purulia districts (West Bengal) was investigated in this study. The range of fluoride in different water sources was estimated to be 0.01–1.6 mg/l with average value of 0.55 ± 0.24 mg/l. In most of the samples, fluoride was present within the WHO-recommended maximum permissible limit (1.5 mg/l). Use of deep tube wells for drinking purpose in the Kashipur block (fluoride content >1.5 mg/l) has to be restricted at an earliest as it may cause health hazards to the local population. The fluoride in water was observed to be significantly positively correlated with pH and negatively correlated with hardness. The sealing of unsafe deep tube wells has largely sorted out the groundwater fluoride contamination in the study area where fluorosis was previously prominent. The maximum exposure doses of fluoride from drinking water for infants, children, and adults were estimated to be around 11, 5, and 3 times higher than the MRL value (0.05 mg/kg/day). If intakes of fluoride from other sources are taken into consideration, then the total exposure dose of fluoride is anticipated to give much higher values. The fluoride in soils (55–399 mg/kg) was found to be related with pH of the soil while agricultural activities had almost no impact. Transfer of fluoride from contaminated soil and water to plants would enhance the severity of fluoride toxicity in residents. Extensive monitoring has to be thus conducted to assess the degree of bioaccumulation of fluoride by locally cultivated crops and vegetables. A centralized drinking water treatment and distribution facility, defluoridation of the groundwater, and recharging groundwater by rain water harvesting are recommended to improve the fluoride contamination problem in this area on long-term basis.
References
Andezhath SK, Susheela AK, Ghosh G (1999) Fluorosis management in India: the impact due to networking between health and rural drinking water supply agencies. Interdiscip Perspect Drink Water Risk Assess Manag 260:159–165
APHA (2005) Standard methods for the examination of water and wastewater, 21st edn. American Public Health Association, Washington
Ayoob S, Gupta AK (2006) Fluoride in drinking water: a review on the status and stress effects. Crit Rev Environ Sci Technol 36:433–487
Bhattacharya HN, Chakrabarti S (2011) Incidence of fluoride in the groundwater of Purulia district, West Bengal: a geo-environmental appraisal. Curr Sci 101(2):152–155
BIS (2003) Indian standard specifications for drinking water. IS:10500, Bureau of Indian Standards, New Delhi. http://wbpcb.gov.in. Accessed 2 Aug 2014
Brindha K and Elango L (2011) Fluoride in groundwater: causes, implications and mitigation measures. In: Monroy SD (ed) Fluoride properties, applications and environmental management. Nova Science Publishing, pp 111–136
Brindha K, Rajesh R, Murugan R, Elango L (2011) Fluoride contamination in groundwater in parts of Nalgonda District, Andhra Pradesh, India. Environ Monit Assess 172:481–492
Census (2011) Bankura district: Census 2011 data, Government of India. http://www.census2011.co.in/census/district/13-bankura.html. Accessed 13 Aug 2014
Chakrabarti S, Bhattacharya HN (2013) Inferring the hydro-geochemistry of fluoride contamination in Bankura district, West Bengal: a case study. J Geol Soc India 82(4):379–391
Chatterjee A, Roy RK, Ghosh UC, Pramanik T, Kabi SP, Biswas K (2008) Fluoride in water in parts of Raniganj Coalfield, West Bengal. Curr Sci 94(3):309–311
Clark RG, Stewart DJ (1983) Fluorine. In: Grace NE (ed) The mineral requirements of grazing ruminants. New Zealand Society of Animal Production, Occasional Publication 9:129–134
Dar MA, Sankar K, Dar IA (2011) Fluorine contamination in groundwater: a major challenge. Environ Monit Assess 173:955–968
Davison A, Weinstein LW (1998) The effects of fluorides on plants. Earth Island 13:257–264
EPA (1997) Public health global for fluoride in drinking water. Pesticide and Environmental Toxicology, Section Office of Environmental Health Hazard Assessment, Environmental Protection Agency, California
Farooqi A, Masuda H, Siddiqui R, Naseem M (2009) Sources of arsenic and fluoride in highly contaminated soils causing groundwater contamination in Punjab, Pakistan. Arch Environ Contam Toxicol 56:693–706
Fornasiero RB (2001) Phytotoxic effects of fluoride. Plant Sci 161:979–985
Grimaldo M, Borja V, Ramirez AL, Poncce M, Rosas M, Diaz-Barriga F (1995) Endemic fluorosis in San Luis Potosi, Mexico: identification of risk factor associated with human exposure to fluoride. Environ Res 68:25–30
Hall RJ, Cain RG (1972) Organic fluorine in tropical soils. New Phytol 71:839–853
ICMR (1975) Manual of standards of quality for drinking water supplies, special report series number 44, 2nd edn. Indian Council of Medical Research, New Delhi
Jha SK, Nayak AK, Sharma YK (2009) Fluoride occurrence and assessment of exposure dose of fluoride in shallow aquifers of Makur, Unnao district, Uttar Pradesh, India. Environ Monit Assess 156:561–566
Kalinic N, Hrsak J, Vadjic V, Lambasa-Belak Z, Mihelcic V, Perkovic B (2005) Fluoride content in soil and vegetation. Bull Environ Contam Toxicol 75:157–162
Khaiwal R, Garg VK (2007) Hydro-chemical survey of groundwater of Hisar city and assessment of defluoridation methods used in India. Environ Monit Assess 132:33–43
Kozyrenko MM, Artyukovo EV, Shmakov VN, Konstantinov YM (2007) Effect of fluoride pollution on genetic variability of Larix gmelinii (Poaceae) in East Siberia. J For Res 12:388–392
Kundu MC, Mandal B (2009) Agricultural activities influence nitrate and fluoride contamination in drinking groundwater of an intensively cultivated district in India. Water Air Soil Pollut 198:243–252
McQuaker RN, Gurney M (1977) Determination of total fluoride in soil and vegetation using an alkali fusion selective ion electrode technique. Anal Chem 49:53–56
Mishra PC, Meher K, Bhosagar D, Pradhan K (2009) Fluoride distribution in different environmental segment at Hirakud Orissa (India). Afr J Environ Sci Technol 3(9):260–264
O’Malley (1995) Bankura. Bengal District Gazetteers, Government of West Bengal, pp 1–20
Ortiz D, Castro L, Turrubiartes F, Milan J, DiazBarriga F (1998) Assessment of the exposure to fluoride from drinking water in Durango, Mexico, using a geographic information system. Fluoride 31(4):183–187
PHED Report (2007) Activities & achievements in rural drinking water supply and other areas. http://www.wbphed.gov.in/applications/im/uploads/000643.pdf. Accessed 18 Aug 2014
Pickering WF (1985) The mobility of soluble fluoride in soils. Environ Pollut B9:281–308
Ramanaiah SV, Venkatamohan S, Rajkumar B, Sarma PN (2006) Monitoring of fluoride concentration in groundwater of Prakasham district in India: correlation with physico-chemical parameters. J Environ Sci Eng 48:129–134
Rao NS (2009) Fluoride in groundwater, Varaha River Basin, Visakhapatnam District, Andhra Pradesh, India. Environ Monit Assess 152:47–60
Rao NVR, Rao KS, Schuiling RD (1993) Fluoride distribution in waters of Nalgonda district, Andhra Pradesh, India. Environ Geol 21:84–89
Saini P, Khan S, Baunthiyal M, Sharma V (2013) Mapping of fluoride endemic area assessment of F−1 accumulation in soil and vegetation. Environ Monit Assess 185(2):2001–2008
Salve PR, Maurya A, Kumbhare PS, Ramteke DS, Wate SR (2008) Assessment of groundwater quality with respect to fluoride. Bull Environ Contam Toxicol 81:289–293
Sankararamakrishnan N, Sharma AK, Iyengar L (2008) Contamination of nitrate and fluoride in groundwater along the Ganges Alluvial Plain of Kanpur district, Uttar Pradesh, India. Environ Monit Assess 146:375–382
Steinberg CL, Gardner DE, Smith FA, Hofge HC (1955) Comparison of rheumatoid (ankylosing) spondilitis and crippling fluorosis. Ann Rheum Dis 14:378–384
Susheela AK (1999) Fluorosis management programme in India. Curr Sci 77:1250–1256
Suthar S, Garg VK, Jangir S, Kaur S, Goswami N, Singh S (2008) Fluoride contamination in drinking water in rural habitations of Northern Rajasthan, India. Environ Monit Assess 145:1–6
UNICEF (1999) States of the art report on the extent of fluoride in drinking water and the resulting endemicity in India. Report by fluorosis and rural development foundation for. UNICEF, New Delhi
USEPA (2010) Drinking water treatability database: fluoride. http://iaspub.epa.gov/tdb/pages/contaminant/contaminantOverview.do?contaminantId=10700. Accessed 23 Oct 2014
USPHS (1962) Drinking water standards. USPHS publications, USGPO, Washington DC, p 956
WHO (2006) Fluoride in drinking water. IWA publishing, London, p 144
Wood JM (1974) Biological cycles for toxic elements in the environment. Science 183:1049–1052
Acknowledgments
The authors are thankful to the Department of Environmental Science, University of Kalyani, West Bengal, India, for providing the laboratory facilities. This study would not have been possible without the help of many. Among them, the greatest help in preparing the samples prior to analysis came from Amrita Chaudhuri and Soume Pyne. Piyal Bhattacharya is thankful to Mousumi Bhattacharya for her cooperation during writing this article. The authors are also thankful to the critical comments of the anonymous reviewers that helped to improve the manuscript considerably.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Philippe Garrigues
Alok C. Samal and Piyal Bhattacharya contributed equally to the work
Rights and permissions
About this article
Cite this article
Samal, A.C., Bhattacharya, P., Mallick, A. et al. A study to investigate fluoride contamination and fluoride exposure dose assessment in lateritic zones of West Bengal, India. Environ Sci Pollut Res 22, 6220–6229 (2015). https://doi.org/10.1007/s11356-014-3817-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-014-3817-4