Abstract
Background
Powerlifting exercises are commonly performed by athletes and recreational trainers for increasing muscle strength. Increased performance for these exercises may promote beneficial outcomes, especially in terms of bone health. The aim of this study was to examine whether powerlifting exercise performance and muscle mass indices are associated with bone mineral density.
Methods
Fifty-one males (median age 24.0 years) with resistance training experience (median 5.0 years) performed one-repetition maximum tests for the bench press, squat and deadlift. Muscle mass indices and bone mineral density were assessed via a whole-body dual energy X-ray absorptiometry (DEXA) scan. Stronger and weaker participants were directly compared for fat-free mass, lean mass and bone mineral density. Spearman rank-order correlation coefficient analyses were used to determine whether powerlifting exercise performance is related with muscle mass indices and bone mineral density.
Results
Stronger participants had greater fat-free mass, appendicular lean mass, and bone density (p < 0.01). For all muscle mass indices (e.g., fat-free mass and appendicular lean mass) there were small to strong positive relationships found with bone mineral density (rs = 0.28–0.65; p < 0.05). Small to strong relationships were found between powerlifting exercise performance and bone mineral density for the majority of regions (rs = 0.29–0.65, p < 0.05). Powerlifting exercise performance was not related with leg bone mineral density.
Conclusion
Focusing on improving powerlifting exercise performance and muscle mass appears to be beneficial for improving bone mineral density in young men. However, since this was a cross-sectional study causality cannot be established.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Progressive resistance training is an effective intervention for increasing muscle mass [1] as well as improving bone health [2]. The bench press, squat and deadlift are arguably the most commonly prescribed resistance exercises by advanced trainers [3]. These three exercises are also performed in the sport of powerlifting where the objective is for competitors to lift a maximal external load for a single repetition for each exercise [4]. Powerlifting exercises place significant mechanical stress on various musculoskeletal regions including the hip and spine, especially during the squat and deadlift [5]. Since mechanical loading is a fundamental factor for bone mass growth [6] and muscle hypertrophy [7], it is coherent that stronger compared to weaker competitive powerlifters have greater muscle mass and bone mass [8, 9]. However, there appears to be a scarcity of research that have investigated comprehensively the muscle mass and regional bone mineral density of stronger and weaker trainers based on powerlifting performances.
In young healthy males, Guimarães et al. [10] showed that muscle strength and lean body mass were associated with bone mineral content and bone mineral density. Therefore, suggesting that even in younger adults these factors influence bone health and might be important to monitor in younger adults with a family history of osteoporosis. [11]. It should be noted that the hip and spine are considered the most important regions to target when performing resistance training in older adults because these are the predominant sites where fractures occur [12]. However, Guimarães et al. [10] did not include the squat and deadlift in their study, opting for mostly machine weights with the exception of the bench press. Currently resistance training recommendations for increasing bone mineral density focuses on the progression towards using heavy loads (e.g., 70–90% one-repetition maximum—1RM), using exercises that target muscles surrounding the hips and spine, and performed at least three times per week [13]. Anecdotally, recreational trainers that regularly perform the bench press, squat, and deadlift will mainly focus on increasing the maximal loads lifted, like competitive powerlifters. Therefore, it would be expected that heavy loads would generally be used in training to enhance maximal loads lifted. It is of importance to identify factors that may influence bone mineral density of younger recreational resistance trainers to assist with reducing bone loss due to aging and potentially avoid the development of osteopenia and osteoporosis. Greater insight into the relationships between muscle strength for the powerlifting exercises, indices of muscle mass and bone mineral density will assist with understanding how to manipulate resistance training variables to optimise bone health in younger adults.
The aim of this study was to investigate the muscle mass indices and bone mineral density of stronger versus weaker resistance trainers, based on their Wilks score [14], with similar experience. Additionally, we also sought to examine whether strength (1RM) absolute and normalised to body mass allometrically scaled and Wilks score and muscle mass indices were related to bone mineral density in young men. We hypothesised that the stronger resistance trainers (i.e., greater Wilks scores) would have superior muscle mass and bone mineral density compared to weaker resistance trainers. It was also hypothesised that numerous positive correlations would be observed between absolute and normalised strength, fat-free mass, appendicular lean mass, and bone mineral density. The findings from this study may assist exercise professionals with resistance training prescription for young men, especially for the improvement of bone health.
Methods
Study design
A cross-sectional study designed was employed to address the study aims. Males aged 18–45 years were recruited and needed to have at least 6 months resistance training experience with ≥ 2 sessions per week currently being performed. It was also a study requirement that participants were able perform the bench press, squat and deadlift. Participants were required to visit the laboratory on 1–2 occasions to complete the assessments. Muscle strength was assessed using the one-repetition maximum (1RM) test for the bench press, squat and deadlift. Fat-free mass, lean mass, and bone mineral density was assessed via a whole-body dual energy X-ray absorptiometry (DEXA) scan. The DEXA scan could be completed prior to the 1RM tests (i.e., total of 1 visit) or during an additional visit (i.e., total of 2 visits), with an average of 2 days between visits.
Participants
Fifty-one males (median age 24.1 [interquartile range (IQR) 20.9–28.1] years; height 178.0 [172.6–180.9] cm; body mass 81.7 [76.6–88.7] kg; resistance training experience 5.0 [3.0–9.0] years) participated in this study. Participants reported performing resistance training 4.0 (3.0–5.0) days per week and performing the bench press, squat and deadlift in 1.0 (1.0–2.0) session per week. Nine participants were known to have experience competing in either powerlifting or Olympic weightlifting competitions; however, none of these participants were currently preparing for a competition. The remaining participants performed resistance training on a recreational level. Twenty-eight of the 51 participants also regularly performed aerobic exercise as part of their training with the median weekly volume being 120.0 (60.0–180.0) minutes. Following completion of the testing sessions, participants were divided into two groups based on their powerlifting performances. The Wilks score is a validated method to compare the performance of powerlifters between varying body weight classes [15]. Briefly, the Wilks formula is based on a 5th order polynomial reflecting the best fit relationship between body mass and an estimation of lifting performance of world class powerlifters. For this study Wilks scores > 280 were considered to be strong and < 280 as weak. Individual lifts were also expressed relative to body weight, which is a method commonly used to evaluate strength between lifters [16], as well as relative to body mass allometrically scaled [17]. The demographic and powerlifting exercise performance characteristics of the strong and weak participants are presented in Tables 1 and 2, respectively. Participants provided verbal and written consent prior to study commencement. This study was approved by the University of Sydney Human Research Ethics Committee, project number 2015/706.
Procedures
All testing was conducted at the Exercise Physiology Laboratories at The University of Sydney. Participants were instructed to refrain from any strenuous physical activity for at least 24 h before the testing sessions and if they reported any fatigue or soreness from previous exercise the testing was postponed to another day for these symptoms to subside. Participants were also asked to refrain from using caffeine or pre-workout supplements prior to the testing session. In preparation for the DEXA scan, participants were instructed to be in fasted state (10–12 h) with no more than 200 ml of water permitted during this period.
Muscle strength
Muscular strength was assessed via completing a 1RM, which is defined as the heaviest load a participant could successfully lift. Participants could choose to complete the 1RM tests for the bench press or squat first, with the deadlift being the last exercise completed. The 1RM test involved a protocol that has previously been used by the researchers [18, 19]. For each exercise, a thorough warm-up was conducted which involved performing repetitions at light loads and then progressing to heavier loads. However, participants were instructed not to exceed approximately 80% of maximal effort during the warm-up sets to prevent fatigue from affecting their 1RM performance. During 1RM attempts, if the lift was successful the barbell load was increased and there was 3–5 min recovery between attempts. The technique for the bench press involved lying flat on a bench, lowering the barbell to no greater than approximately 2.5 cm from the chest and then pressing the barbell upwards until arms were fully extended. A research assistant helped to lift the barbell from the rack in readiness to begin attempts. The back squat was performed using a squat rack with the positioning of spotter arms just below the point where the thighs were parallel to the floor. A successful squat attempt involved descending by flexing the knees and hips until they were in a horizontal plane (i.e., thighs parallel to the floor) and then ascending to an upright position. The deadlift began with a loaded barbell placed on the floor in front of the participant, having them bend over to grab the bar and then lift the barbell until standing erect in a fully upright position with knees extended. For the squat and deadlift 1RM, participants were asked not to use weight belts, but for the deadlift wrist straps and chalk could be used to assist with holding the bar.
Body composition
A DEXA scan was conducted to assess fat-free mass, lean mass and bone mineral density using the GE Lunar Prodigy (Medical Systems, Madison, WI). Participants had one whole body scan which was performed under standardised conditions. As mentioned above, participants were required to be in a fasted state for at least 10 h prior to the scan. Bladder/bowel was to be voided, any jewellery removed, clothes were removed down to the underwear and a hospital gown was worn. Participants laid flat on the scanning bed and were positioned in the centre, aligned with the long axis of the scanner, with arms and legs evenly separated to allow accurate analysis of body regions. The DEXA scanner was calibrated within 24 h prior to every scan. Following the scan, in-built analysis software (version 13.60.033; enCORE 2011, GE Healthcare, Madison, WI) allowed the calculation of total and regional (predefined by the software) lean mass, fat-free mass (FFM), bone mineral content, and bone mineral density. One researcher checked all analyses to ensure that the region of interest sites were consistent. Since body height can influence body composition [20], FFM were divided by height2 (kg/m2) to calculate FFM index (FFMI). Additionally, total appendicular lean mass (ALM) relative to height squared was calculated to provide ALM index (ALMI).
Statistical analyses
All statistical analyses were performed using SPSS version 24.0 for Windows (IBM Corp. Armonk, NY USA). Following the Kolmogorov–Smirnov test, there appeared to be inconsistent normal data distribution and given the relatively small sample size it was decided that non-parametric tests would be used for all analyses. Therefore, data were presented as median with interquartile range (IQR). Differences between stronger and weaker participants for all variables of interest was analysed using the Mann–Whitney U test. Relationships between powerlifting exercise performance, muscle mass indices and bone mineral density were assessed using Spearman rank-order correlation coefficient analyses expressed as rho (rs). Strength of correlations were qualitatively assessed as: trivial (r < 0.1), small (r > 0.1–0.3), moderate (rs > 0.3–0.5), strong (rs > 0.5–0.7), very strong (rs > 0.7–0.9), nearly perfect (rs > 0.9), and perfect (rs = 1.0) [21]. The alpha level for significance for tests was set at p < 0.05 and trends declared at p = 0.05–0.10.
Results
The muscle mass indices and bone mineral density of the stronger and weaker participants are presented in Table 3. Overall, the stronger compared to the weaker participants were more muscular as reflected by greater FFM (p = 0.006) and ALM (p = 0.005). When expressed relative to height these differences between groups remained, favouring the stronger participants (FFMI, p < 0.00; ALMI, p < 0.001). Stronger compared to weaker participants had greater bone mineral density for the arms, pelvis, trunk and spine (p < 0.05), but no significant difference between groups was found for the legs. Total bone mineral density was greater for stronger participants (p = 0.02) but no significant differences between groups were found for bone mineral content.
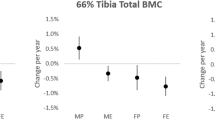
The relationships between bone mineral density, powerlifting exercise performance and muscle mass indices are presented in Table 4. Very strong positive relationships were found between total bone mineral content and FFM (rs = 0.79, p < 0.001) and ALM (rs = 0.77, p < 0.001). Relationships were slightly weaker for bone mineral content and FFMI (rs = 0.41, p = 0.003) and ALMI (rs = 0.47, p = 0.001). There was moderate to strong positive relationships between FFM and total and regional bone mineral density (rs = 0.43–0.65, p < 0.05) (Fig. 1). For all remaining muscle mass indices (FFMI, ALM, and ALMI) there were small to strong positive relationships found with bone mineral density (rs = 0.28–0.65, p < 0.05). Moderate to strong positive relationships were found between total bone mineral content and all powerlifting exercises (1RM) (rs = 0.32–0.56, p < 0.05). Small to strong positive relationships were found between bone mineral density for the majority of regions and all powerlifting exercises (1RM and 1RM/BM0.67) (rs = 0.29–0.65, p < 0.05), Wilks score (rs = 0.36–0.45, p < 0.05), and total strength (rs = 0.42–0.52, p < 0.01). Leg bone mineral density was not related with powerlifting exercise (1RM and 1RM/BM0.67), Wilks score, and total strength.
Discussion
The first aim of this study were to investigate the muscle mass indices and bone mineral density of stronger versus weaker resistance trainers with similar experience. The second aim was to examine the relationship between powerlifting performance, muscle mass indices and bone mineral density. In agreement with the original hypothesis stronger participants were more muscular (i.e., greater FFM and ALM), generally had greater bone mineral density, and appeared to have lower body fat percentage. Bone mineral density of all body regions was positively related with muscle mass. However, bone mineral density of all regions except for the legs was positively related with powerlifting exercise performance. Therefore, it seems that better bone mineral density is present within resistance trainers that have greater muscle mass and strength as assessed by loads lifted for the powerlifts. Focusing on improving powerlifting exercise performance may be beneficial for improving bone mineral density in young men. However, since this was a cross-sectional study causality cannot be established and due to the small sample size caution is warranted when interpreting the findings.
Fat-free mass (FFM) consists of skeletal muscle, internal organs, bone, water, and connective tissue; however, skeletal muscle represents the largest proportion [1]. Therefore, following a resistance training intervention changes in FFM are mainly due to increased skeletal muscle mass [1]. Powerlifters are known to have greater FFM compared to the general population and it has also been proposed that FFM is the greatest determinant of powerlifting performance [22,23,24]. Additionally, more successful competitive powerlifters possess higher degrees of muscle mass compared to less successful peers [8, 9]. This is consistent with the findings from the present study when comparing the stronger and weaker participants. Since there was no difference between the demographic characteristics between groups (e.g., resistance training experience, frequency, lifts trained), it appears that differences in powerlifting performance may be related to factors such as exercise prescription [25], diet [26, 27] and genetics [28]. Based on the reference values for FFMI in Caucasians [20], the stronger and weaker participants in the present study were better than 95% and 90%, respectively, of males aged 18–34 years.
The bench press, squat and deadlift involve large muscle groups and when sufficient loads are used (> 60% 1RM), increased mechanical stress on the activated muscle groups will stimulate muscle hypertrophy [29]. During these powerlifting exercises musculoskeletal regions emphasised include the lumbar and hip region which are the predominant sites where fractures occur in aging [12]. The increased mechanical stress placed on the involved musculoskeletal regions during the powerlifting exercises would likely provide an ideal stimulus for bone mass growth [6]. These thoughts are supported by previous research showing greater bone mineral density of the arms, lumbar spine, and whole body in young male powerlifters (n = 10) compared to healthy age-matched controls [30]. However, in this previous study there was no difference between groups for bone mineral density of the proximal femur. Interestingly, in the present study the only region that did not differ between groups in terms of bone density was the legs. Potentially bone mineral density may be more difficult to change in the legs through resistance training in younger males. The finding may have also been influenced by the weight-bearing status of the lower body and other frequent high-intensity lower body dominant activities generally performed by young adults. Therefore, leading to this region of the body being less sensitive for detecting changes in bone mineral density based on resistance exercise performance.
Total bone mineral content did not differ between groups which is difficult to explain since we found that it was very strongly related with FFM. Chumlea et al. [31] showed that the size of the skeletal frame is an important factor that influences bone mineral content with greater absolutes of bone observed with larger frames. This previous study found increases in bone mineral density in men with broader shoulders and wrists, which were sites related with large amounts of muscle. However, even though broad knees (as well as shoulders and wrists) were associated with high fat-free mass, there was no association with bone mineral content or density [31]. These previous findings support the hypothesis developed from the present study concerning the difficultly in differentiating bone mineral density of the lower body between young adults based on classification of stronger and weaker participants (from the Wilks score). However, it appears that compared to machine weights and isolated resistance exercises, powerlifting exercises may provide an ideal stimulus for improving overall bone health in populations that these exercises are not contraindicated. Although, to maximise the benefits there should be an emphasis on increasing the maximal loads lifted.
The positive relationships between the muscle mass variables (FFM, FFMI, ALM, and ALMI) and bone mineral content and density found in the present study are in agreement with the findings from Guimarães et al. [10] where moderate to very strong relationships were found in a group of young male undergraduate students. The relationship between lean mass and bone mineral content and density among older aged men is well established [32, 33]. Even though females have greater losses in bone content and density with aging (30% compared to 16% for men) there is a similar degree of loss in lean mass [34]. Since lean mass plays a major role in health through the regulation of bodily processes and maintenance of physical function [35], it is no surprise that it is a predictor of longevity in older adults [36].
The findings from this study indicated that maximal loads lifted for the powerlifting exercises are generally related to bone mineral density and this was also consistent with the findings from Guimarães et al. [10]. The novelty of the present study was that muscle strength was expressed in both absolute (load lifted) and relative (load lifted divided by body mass allometrically scaled) terms. Through differentiating strength in this manner it was possible to make more sensitive comparisons between individuals [17]. Additionally, relative strength is commonly used to provide greater insight into physical function and athletic performance [37]. Since powerlifting performance expressed in both absolute and relative terms was similarly related with bone mineral content and density, it provides strong confirmation of the influence of muscle strength on bone health. However, leg bone mineral density was not significantly related with muscle strength which supports the finding of no difference in bone density for this region between the stronger and weaker participants (as discussed above). The only variables that were related with leg bone density were all of the muscle mass variables (FFM, FFMI, ALM, and ALMI). Future research is required to investigate the effects of resistance training interventions targeting muscle strength versus muscle hypertrophy on bone mineral density.
The present results may be applicable to middle-aged and older adults who are at risk of developing sarcopenia, osteoporosis or the newly defined geriatric condition of osteosarcopenia [38]. Resistance training is one of several preventive and therapeutic interventions for osteosarcopenia and based on the results of the present study the inclusion of the powerlifting exercises should be considered. The resistance exercise prescription should also target maximising muscle strength of the squat and deadlift due the associations with bone mineral density of the arm, spine, pelvic and trunk found in the present study. The results of the present cross-sectional study are consistent with evidence from a randomised control trial conducted by Harding et al. [39] that involved an 8-month high-intensity resistance training program featuring the squat, deadlift and shoulder press in males with low bone mineral density. The participants in this previous study showed improvement in bone strength, physical function, and muscle strength compared with an isometric axial exercise group and control group, as well as being well accepted and tolerated. Enhancement of fat-free mass should also be emphasised in people with osteosarcopenia based on the present study findings and previous research. An increase in fat-free mass can be achieved through aforementioned resistance training recommendations in conjunction with implementation of specific dietary practices, e.g., protein intake of 2 g/kg/day) [26, 27, 40].
There are certain limitations that should be acknowledged when interpreting the findings from the present study. This study did not assess bone mineral density at the common specific sites including the hip and lumbar spine. Therefore, it was not possible to make comparisons with other relevant studies and this will be a requirement for future research into the effect of powerlifting exercises on bone health, since there is scarcity of research on this topic. However, our study could be compared to the findings from Guimarães et al. [10] with the novelty being the inclusion of powerlifting exercises and the extensive exploration of muscle strength and its relationship with bone mineral density. Further information gathered from participants would have been helpful to explain some of the findings of the study. In particular, details about individual exercise prescription (e.g., sets, repetitions, loads, perceived effort etc.) and diet such as protein intake and total daily calories, which can affect fat-free mass [26, 27]. It was unknown whether participants used any performance enhancing drugs over the previous 12 months which could have influenced the results. The sample size of the study was relatively small although it was still larger than the study by Guimarães et al. [10] which included 36 young males. Nevertheless, there is a risk of Type 2 errors (false negative) especially since data normality was inconsistent. However, the findings from this study do provide novel information concerning the potential use of powerlifting exercises for prevention of significant bone loss during the aging process. Although there have been many studies exploring the effects of resistance training on body composition and bone mineral density [1, 2], powerlifting lifting interventions are lacking especially in non-athletic populations. Information from this study may be helpful towards the development of adequately powered and well-designed studies on this topic in the future.
Conclusion
Greater powerlifting exercise performance and increased muscle mass appears to be related to greater bone mineral density in young males. Muscle mass appears to have a larger influence on leg bone mineral density compared to lower body strength. For healthy adults, implementation of powerlifting exercises following resistance training recommendations for maximising strength may assist with increasing muscle mass and maintaining healthy bones [41]. However, for people that have certain musculoskeletal or cardiovascular conditions where powerlifting exercises may be contraindicated, alternative resistance training practices should be considered in consultation with appropriately qualified exercise specialists.
References
Benito PJ, Cupeiro R, Ramos-Campo DJ, Alcaraz PE, Rubio-Arias JÁ (2020) A systematic review with meta-analysis of the effect of resistance training on whole-body muscle growth in healthy adult males. Int J Environ Res 17(4):1285
Souza D, Barbalho M, Ramirez-Campillo R, Martins W, Gentil P (2020) High and low-load resistance training produce similar effects on bone mineral density of middle-aged and older people: a systematic review with meta-analysis of randomized clinical trials. Exp Gerontol 138:110973. https://doi.org/10.1016/j.exger.2020.110973
Kompf J, Arandjelović O (2017) The sticking point in the bench press, the squat, and the deadlift: Similarities and differences, and their significance for research and practice. Sports Med 47(4):631–640. https://doi.org/10.1007/s40279-016-0615-9
Ferland PM, Comtois AS (2019) Classic powerlifting performance: a systematic review. J Strength Cond Res 33(Suppl 1):S194-s201. https://doi.org/10.1519/jsc.0000000000003099
Bengtsson V, Berglund L, Aasa U (2018) Narrative review of injuries in powerlifting with special reference to their association to the squat, bench press and deadlift. BMJ Open Sport Exerc Med 4(1):e000382–e000382. https://doi.org/10.1136/bmjsem-2018-000382
Hong AR, Kim SW (2018) Effects of resistance exercise on bone health. Endocrinol Metab (Seoul) 33(4):435–444. https://doi.org/10.3803/EnM.2018.33.4.435
Aguilar-Agon KW, Capel AJ, Martin NRW, Player DJ, Lewis MP (2019) Mechanical loading stimulates hypertrophy in tissue-engineered skeletal muscle: Molecular and phenotypic responses. J Cell Physiol 234(12):23547–23558. https://doi.org/10.1002/jcp.28923
Keogh JWL, Hume PA, Pearson SN, Mellow PJ (2009) Can absolute and proportional anthropometric characteristics distinguish stronger and weaker powerlifters? J Strength Cond Res 23(8):2256–2265. https://doi.org/10.1519/JSC.0b013e3181b8d67a
Lovera M, Keogh J (2015) Anthropometric profile of powerlifters: differences as a function of bodyweight class and competitive success. J Sports Med Phys Fitness. 55(5):478–487
Guimarães BR, Pimenta LD, Massini DA et al (2018) Muscle strength and regional lean body mass influence on mineral bone health in young male adults. PLoS ONE 13(1):e0191769–e0191769. https://doi.org/10.1371/journal.pone.0191769
Henderson LB, Adams JS, Goldstein DR, Braunstein GD, Rotter JI, Scheuner MT (2000) A familial risk profile for osteoporosis. Genet Med 2(4):222–225. https://doi.org/10.1097/00125817-200007000-00004
Holroyd C, Cooper C, Dennison E (2008) Epidemiology of osteoporosis. Best Pract Res Clin Endocrinol Metab 22(5):671–685. https://doi.org/10.1016/j.beem.2008.06.001
Zehnacker CH, Bemis-Dougherty A (2007) Effect of weighted exercises on bone mineral density in post menopausal women. A systematic review. J Geriatr Phys Ther. 30(2):79–88. https://doi.org/10.1519/00139143-200708000-00007
Vanderburgh PM, Dooman C (2000) Considering body mass differences, who are the world’s strongest women? Med Sci Sports Exerc 32(1):197–201. https://doi.org/10.1097/00005768-200001000-00029
Vanderburgh PM, Batterham AM (1999) Validation of the Wilks powerlifting formula. Med Sci Sports Exerc 31(12):1869–1875. https://doi.org/10.1097/00005768-199912000-00027
Jaric S (2002) Muscle strength testing: use of normalisation for body size. Sports Med 32(10):615–631. https://doi.org/10.2165/00007256-200232100-00002
Jacobson B, Thompson BJ, Conchola E, Glass R (2013) A comparison of absolute, ratio and allometric scaling methods for normalizing strength in elite American football players. J Athl Enhanc. 2:2. https://doi.org/10.4172/2324-9080.1000110
Amirthalingam T, Mavros Y, Wilson GC, Clarke JL, Mitchell L, Hackett DA (2017) Effects of a modified german volume training program on muscular hypertrophy and strength. J Strength Cond Res 31(11):3109–3119. https://doi.org/10.1519/jsc.0000000000001747
Hackett DA, Amirthalingam T, Mitchell L, Mavros Y, Wilson GC, Halaki M (2018) Effects of a 12-week modified German volume training program on muscle strength and hypertrophy-a pilot study. Sports. 6:1. https://doi.org/10.3390/sports6010007
Schutz Y, Kyle UUG, Pichard C (2002) Fat-free mass index and fat mass index percentiles in Caucasians aged 18–98 y. IJO 26(7):953–960. https://doi.org/10.1038/sj.ijo.0802037
Hopkins WG, Marshall SW, Batterham AM, Hanin J (2009) Progressive statistics for studies in sports medicine and exercise science. Med Sci Sports Exerc 41(1):3–13. https://doi.org/10.1249/MSS.0b013e31818cb278
Brechue WF, Abe T (2002) The role of FFM accumulation and skeletal muscle architecture in powerlifting performance. Eur J Appl Physiol 86(4):327–336. https://doi.org/10.1007/s00421-001-0543-7
Keogh JW, Hume PA, Pearson SN, Mellow P (2007) Anthropometric dimensions of male powerlifters of varying body mass. J Sports Sci 25(12):1365–1376. https://doi.org/10.1080/02640410601059630
Mayhew JL, McCormick TP, Piper FC, Kurth AL, Arnold MD (1993) Relationships of body dimensions to strength performance in novice adolescent male powerlifters. Pediatr Exerc Sci 5(4):347–356
Tan B (1999) Manipulating resistance training program variables to optimize maximum strength in men: a review. J Strength Cond Res 13(3):289–304. https://journals.lww.com/nsca-jscr/Fulltext/1999/08000/Manipulating_Resistance_Training_Program_Variables.19.aspx
Hayes A, Cribb PJ (2008) Effect of whey protein isolate on strength, body composition and muscle hypertrophy during resistance training. Curr Opin Clin Nutr Metab Care 11(1):40–44
Slater GJ, Dieter BP, Marsh DJ, Helms ER, Shaw G, Iraki J (2019) Is an energy surplus required to maximize skeletal muscle hypertrophy associated with resistance training. Front Nutr 6:131. https://doi.org/10.3389/fnut.2019.00131
MacArthur DG, North KN (2007) ACTN3: a genetic influence on muscle function and athletic performance. Exerc Sport Sci Rev 35(1):30–34. https://doi.org/10.1097/JES.0b013e31802d8874
Wackerhage H, Schoenfeld BJ, Hamilton DL, Lehti M, Hulmi JJ (2019) Stimuli and sensors that initiate skeletal muscle hypertrophy following resistance exercise. J Appl Physiol 126(1):30–43
Tsuzuku S, Ikegami Y, Yabe K (1998) Effects of high-intensity resistance training on bone mineral density in young male powerlifters. Calcif Tissue Int 63(4):283–286. https://doi.org/10.1007/s002239900527
Chumlea WC, Wisemandle W, Guo SS, Siervogel RM (2002) Relations between frame size and body composition and bone mineral status. Am J Clin Nutr 75(6):1012–1016. https://doi.org/10.1093/ajcn/75.6.1012
Gómez-Cabello A, Ara I, González-Agüero A, Casajús JA, Vicente-Rodríguez G (2012) Effects of training on bone mass in older adults: a systematic review. Sports Med 42(4):301–325. https://doi.org/10.2165/11597670-000000000-00000
Makovey J, Naganathan V, Sambrook P (2005) Gender differences in relationships between body composition components, their distribution and bone mineral density: a cross-sectional opposite sex twin study. Osteoporos Int 16(12):1495–1505. https://doi.org/10.1007/s00198-005-1841-4
Proctor DN, Melton LJ, Khosla S, Crowson CS, O’Connor MK, Riggs BL (2000) Relative influence of physical activity, muscle mass and strength on bone density. Osteoporos Int 11(11):944–952. https://doi.org/10.1007/s001980070033
Wolfe RR (2006) The underappreciated role of muscle in health and disease. Am J Clin Nutr 84(3):475–482. https://doi.org/10.1093/ajcn/84.3.475
Srikanthan P, Karlamangla AS (2014) Muscle mass index as a predictor of longevity in older adults. Am J Med 127(6):547–553. https://doi.org/10.1016/j.amjmed.2014.02.007
de Lira C, Vargas V, Silva W, Bachi A, Vancini R, Andrade M (2019) Relative strength, but not absolute muscle strength, is higher in exercising compared to non-exercising older women. Sports (Basel) 7(1):19. https://doi.org/10.3390/sports7010019
Hassan E, Duque G (2017) Osteosarcopenia: a new geriatric syndrome. Aust Fam Phys 46:849–853. http://www.racgp.org.au/afp/2017/november/osteosarcopenia-a-new-geriatric-syndrome/
Harding AT, Weeks BK, Lambert C, Watson SL, Weis LJ, Beck BR (2020) A comparison of bone-targeted exercise strategies to reduce fracture risk in middle-aged and older men with osteopenia and osteoporosis: LIFTMOR-M semi-randomized controlled trial. J Bone Miner Res 35(8):1404–1414. https://doi.org/10.1002/jbmr.4008
Phu S, Boersma D, Duque G (2015) Exercise and Sarcopenia. J Clin Densitom 18(4):488–492. https://doi.org/10.1016/j.jocd.2015.04.011
Ratamess N, Alvar B, Evetoch T et al (2009) American College of Sports Medicine position stand. Progression models in resistance training for healthy adults. Med Sci Sports Exerc. 41(3):687–708
Funding
No funding was received to conduct this project.
Author information
Authors and Affiliations
Contributions
Conceptualization, DAH; methodology, DAH; formal analysis, DAH; investigation, DAH; AS; writing—original draft preparation, DAH; writing—review and editing, DAD; AS; supervision, DAH; project administration, DAH.
Corresponding author
Ethics declarations
Conflicts of interest
No potential conflicts of interest were reported by the authors.
Ethical approval
This study was approved by the University of Sydney Human Research Ethics Committee, project number 2015/706.
Informed consent
Participants provided verbal and written consent prior to study commencement.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Hackett, D.A., Sabag, A. Powerlifting exercise performance and muscle mass indices and their relationship with bone mineral density. Sport Sci Health 17, 735–743 (2021). https://doi.org/10.1007/s11332-021-00740-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11332-021-00740-z