Abstract
Purpose
Obesity is a multifactorial disease and it is related to the occurrence of metabolic syndrome and nonalcoholic fatty liver disease (NAFDL) in youth. This study aimed to investigate the effects of a 12-week multicomponent intervention program in markers of metabolic syndrome and NAFLD in Brazilian overweight/obese low-income school-aged children.
Methods
This quasi-experimental study comprised overweight/obese school-aged children, aged 7–13 years. The participants were assigned to intervention (n = 17) or control group (n = 18). The multicomponent intervention was developed during 12 weeks, consisting of exercise sessions (twice/week; 1 h), nutritional education sessions (once/month), and parental support (twice/week). The following variables were evaluated: anthropometric measures (height, body weight, waist circumference, percentage of body fat); biochemical assays (total cholesterol (TC), triglycerides-TG, high-density lipoprotein-HDL, glucose, aspartate aminotransferase-AST, alanine aminotransferase-ALT), cardiorespiratory fitness, and maturational stage. A cardiovascular disease (CVD) composite z-scores (percentage of body fat, glucose, AST, ALT, TG, and TC/HDL ratio) was also calculated. General linear models were used for data analysis.
Results
Compared to the control group, intervention group participants decreased percentage of body fat (Δ − 0.97; p < 0.001), glucose levels (Δ − 0.15; p = 0.005), ALT (Δ − 2.84; p = 0.021), TC/HDL ratio (Δ − 0.93; p < 0.001), CVD composite score (Δ − 0.97; p < 0.001), and total food intake (Δ − 131.44; p = 0.03), while there was no differences between groups on waist circumference, AST, triglycerides, and CRF.
Conclusion
A 12-week multicomponent intervention was effective on decreasing some metabolic syndrome parameters in overweight/obese school-aged children.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Detrimental behavior to health, such as physical inactivity, sedentary habits, and unhealthy eating, is increasingly recurrent in children and adolescent's routine [1]. Apart from the genetic disposition, this lifestyle factors are the main responsible for the rise in the prevalence of overweight and obesity, as well as the physical fitness decrease observed in pediatric population [2, 3].
Obesity is a multifactorial disease and is the central risk factor for metabolic syndrome (MetS). Mets is characterized by a group of disorders that include, in addition to obesity, elevated levels of triglycerides (TG), low levels of high-density lipoprotein cholesterol (HDL-C), insulin resistance, and arterial hypertension [4]. Despite the absence of global consensus regarding MetS definition in the pediatric population, studies have indicated that there is a high occurrence of cardiovascular disease (CVD) risk factors clustering even in early ages [5]. Moreover, Nonalcoholic Fatty Liver Disease (NAFLD) is another obesity-health problem [6] and identified as the leading cause of liver disease in pediatric populations worldwide [7]. It is characterized by pathological fat accumulation in the liver, which may lead to liver damage in the form of inflammation and fibrosis [8] placing NAFLD as the hepatic representation of MetS. Previous studies found that increases levels of serum aspartate aminotransferase (AST), alanine aminotransferase (ALT), uric acid, fasting glucose, and body mass index (BMI) are predictors of NAFLD among obese children and adolescents [9, 10]. Although the MetS and the NAFLD prevalence are higher in adolescents, young children also exhibit MetS and NAFLD as obesity-related complications [11].
It is important to highlight that low-income children are the most affected by unhealthy behavior opportunities as well as having less access to health care, and, therefore, being more prone to develop comorbidities associated with obesity [12]. Moreover, because of the multifactorial characteristic of obesity´s etiology, an effective behavioral change intervention should be considered along with a sustained commitment of children´s families [13].
In this context, a multicomponent approach [14] involving physical activity, nutritional education, and family support as well as working on individual and social aspects was recommended [15]. Indeed, interventions including physical exercise and nutritional education were effective to decrease obesity and related metabolic diseases in Latino youth [16, 17]. However, these studies were developed with adolescents and did not considered NAFLD markers in the analyses. Furthermore, the interventions were based on physical activity and/or nutrition, disregarding the essential role of parental support on those strategies. Thus, there is a lack of studies using a multicomponent intervention to improve cardiometabolic profile, especially regarding NAFLD parameters, in low-income Brazilian children. This strategy seems to be worthy to consider, because low cost preventive strategies should be adopted for the target population. Based on these aspects, we hypothesized that a 12-week multicomponent intervention program focused on overweight/obese low-income school-aged children is effective in improving markers of metabolic syndrome, and NAFLD.
Therefore, this study aimed to investigate the effects of a 12-week multicomponent intervention program in markers of metabolic syndrome and NAFLD in Brazilian overweight/obese low-income school-aged children.
Methods
Study design and approach
This quasi-experimental study is part of the “ACTION FOR HEALTH” (Adolescents and Children in a Training Intervention for Health) project. This project was developed to target overweight/obese low-income school-aged children of both genders aged 7–13 years. It is a multicomponent intervention program aiming to promote PA after-school classes and included nutritional education as well as parental support sessions. The program used the Behavior Change Ecological Model, which calls for the implementation of multidisciplinary actions [14]. Physical exercise sessions, nutritional education and parental support were offered to systematically target mechanisms of change at each level of influence (intrapersonal, interpersonal and environmental). Detailed information about study design and intervention process was published elsewhere 18].
All the Helsinki Declarations’ ethical aspects were followed [19]. Procedures and evaluation methods were approved by the Scientific Board of the Research Unit that lead the project (excluded information for blind review) and is registered in www.clinicaltrials.gov (Number: NCT02929472).
The program was carried-out by four physical education (PE) teachers, one nutritionist, and one pediatrician. They previously participated in weekly meetings (over 6 months), where methodologies and intervention sessions were prepared and tested. Seven graduate PE students and two graduate nutrition students collaborated in the weekly sessions and were trained and supervised by the project coordinator.
The overall protocol was organized as follow: during the first day blood samples were collected, anthropometric variables were assessed as well as the evaluation of maturational stages. At the second day, body scan was performed in the children, while parents were interviewed about children´s 3-day food intake record. Lastly, during the third and fourth days, physical fitness tests were applied. Physical tests were carried out at the Physical Education Department of the Federal University of Paraiba. The blood analyses were carried-out at a convened specialized laboratory in João Pessoa/Brazil. The same procedures were followed for post-intervention evaluation.
Participants and procedures
The project was publicized in two educational poles (six public schools) from João Pessoa-PB (Brazil), which were located in regions nearby the intervention site. All schools were located in a deprived area, with low socioeconomic status: 62.5% of the mothers or fathers were unemployed and 45% of the mothers and 64% of the fathers had concluded the 9th grade or less.
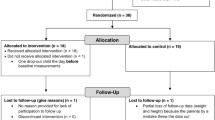
Parents of potential participants (n = 276, according to school teacher´s or parent´s information) were invited to participate in early evening information meetings with the multidisciplinary staff. From those, ninety-six parents attended the meetings, where all the potential participants were screened for inclusion criteria: (a) being overweight or obese, according to Cole et al. criteria [20] and (b) not being involved in any other physical exercise or diet program. From those attending the first assessment, fifty-three were excluded and eight did not complete baseline evaluations. Thus, the final sample comprised 35 participants allocated to control or intervention groups, respectively (Fig. 1). Children and parents of the control group were advised to do not change their lifestyle during the period between the evaluations (12 weeks). Furthermore, they were informed that after the 12-week intervention period their children will have access to the same activities as the intervention group had.
Physical exercise intervention
The exercise classes comprised 60 min sessions, twice a week, for 12 weeks. Physical eduaction lead the exercise sessions. Each session included 4 phases: 10 min of warm-up; 30 min of circuit training; 15 min of pre-sports and recreational games; and 5 min of cool-down activities. Warm-up included aerobic/anaerobic and recreational activities. The circuit-training, comprised six stations and included activities that prioritized physical fitness [21]. Exercises were structured allowing participants to maintain a high intensity throughout the circuit. This protocol has been used and tested in other intervention studies with a similar sample [22, 23].
Training intensity and compliance between individuals were defined to induce heart rate (HR) higher than 65% of HRmax of each participant. To monitor the activity, ten randomly selected participants wore a portable HR monitor (Polar Team2 Pro, Polar, Finland) during sessions. Attendance average for INT group was 85% (ranging between 73.6% and 88.9%).
Nutritional education intervention
A nutritionist designed the nutritional session and consisted in two actions. The first one named dietary counseling. Each month, all participants and their parents were invited for a nutritional appointment in which it was provided information about the food pyramid, weight loss importance, fat-free and low-energy foods, food calories, good nutritional choices on preparing low-coast meals, and decisions on food choices. The three sessions (1/month) were based on the information recorded through the participant’s 24 h-recorded-dietary evaluation, which was done by the nutritional staff at baseline. The attendance to appointments was 100%. The second action consisted on the attempt to accomplishment of three daily dietary goals. These goals were focused on three main points: (a) increase frequency and quantity intake of adequate food (fruit and vegetables); (b) decrease of inadequate food intake (processed food and added sugar); and (c) increase water consumption. Interventions focusing on these goals have been used in similar populations [16]. At the beginning of the intervention, participants received a worksheet to record day by day whether they met the daily goals. No significant inter subject variance was observed for the achievement of the proposed goals in the intervention group (data not shown).
Food intake was carried out by a 24-h recall, which was applied three times (participants, along with their parents, informed food intake during the past 2 weekdays and 1 weekend day). Data from these 3 days were tabulated in the software “Virtual Nutri” to obtain the total energy intake values for each of the 3 days. The mean value of caloric intake among the 3 days was used and data were recorded in kcal.
Parental support intervention
Simultaneous to children’s program, parents were invited to attend to specific exercise sessions to encourage family support. Adults’ participation was voluntary and the parental support intervention focused on improvement of three aspects: (a) encouragement through provision of transportation to PA facilities (100% attendance); (b) participation in PA with the children, when applicable (82% attendance); and (c) watching the children during physical activities (95% attendance). The activities were also carried out by a trained PE teacher at the center of sports of Federal University of Paraíba, João Pessoa (Brazil) at the same time as their respective children.
Anthropometric measures
Height and body weight were assessed using an “Holtain” stadiometer, and percentage of body fat (%BF) was measured by a bioimpedance scale after 4-h-fasting and low water intake (Inbody 720, Biospace Co. Ltd.), [24]. These measures were performed at a specialized laboratory in João Pessoa and followed standardized procedures [24]. To measure waist circumference, NHANES protocol (US Department of Health and Human Services, 1996) [25] was followed.
Cardiorespiratory fitness (CRF)
CRF was measured using the 20 m Shuttle-run Test [26]. Participants completed 20 m shuttle runs keeping in time with an audible ‘bleep’ signal. The frequency of the sound signals was increased every minute by 0.5 km/h, increasing the intensity of the test, and youngsters were encouraged to run to exhaustion [26]. The number of completed laps was recorded for each participant and retained for analysis. This procedure has been used previously in studies with similar samples [27, 28]. Evidence for the acceptable reliability and validity of the 20 m shuttle run test has been proven, with test–retest reliability coefficients ranging from r = 0.78 to r = 0.93 [29].
Biochemical assays
The circulating levels of plasma insulin, plasma glucose, plasma cholesterols (TC and LDL-C), plasma TG, ALT, AST, were measured through peripheral puncture in the cubital vein after a nocturnal 12 h-fasting, by laboratory specialists, using standard techniques at an accredited partner laboratory. The analysis of TC, HDL-C, TG, and glucose was carried out by spectrophotometry (Cobas Integra 400 Plus) with Roche® kits. The LDL-C fraction was indirectly calculated using the Friedewald formula [30]. The AST and ALT levels were determined by enzyme kinetic assay for spectrophotometrically obtained after 10 min’ centrifugation at 3500 rpm. Insulin was determined by Luminex-100 IS (Integrated System: Luminex Corporation, Austin, TX, USA), using the Linco Human Gut Hormone panel kit (Linco Research Inc., MO, USA). All samples ran in duplicate and the mean was calculated. The intra-assay coefficient of variation was less than 20%, according to the manufacturer.
Confounder variables
Maturational stages
The breast stages (B1–B5) in girls and the genital stages (G1–G5) in boys were assessed according to Tanner’s criteria [31] by a pediatrician at the first day of measurements.
Statistical analyses
Stata for windows (version 13) was used for all the analyses. The composite CVD risk factor was calculated from the sum of z-scores of %BF, glucose, AST, ALT, TG, TC/HDL ratio, and CRF, by sex and age category (< 9 years and 9 + years). This approach was already used in similar studies [5].
Means and standard deviations (SD) were calculated to characterize the participants. Baseline differences between groups were calculated using general linear model adjusted for sex. Each of the dependent variables was standardized as follow at the given time point: standardized value = (value − mean)/standard deviation. Change scores were calculated by subtracting post-intervention values from baseline values. General linear models were performed to analyze differences between IG and CG in change scores from baseline to post-intervention adjusted for sex, sexual maturation, age category and CRF baseline level. It was also presented the confidence interval (CI). The effect size (Cohen’s d) of the intervention are also shown. The magnitude of effect size was classified according to Cohen [32] as: ≤ 0.20 (trivial), between 0.21 and 0.50 (small), between 0.51 and 0.80 (moderate), and > 0.80 (large). We accepted 5% of error in our analyses.
The sample size was calculated a posteriori in the software G*power version 3.1, considering that participants were volunteered selected. Thus, for general linear model, a medium effect of the intervention on dependent variables (F2 = 0.15 to 0.35) was considered and the value of the statistical power was between 0.60 and 0.80, as well as the level of statistical significance was established as p < 0.05.
Results
Descriptive characteristics of the participants at baseline are shown in Table 1. We only found statistically significant differences at baseline for CRF between intervention and control group.
Table 2 presents physical characteristics, change scores (mean ± SD), adjusted differences by group (IG and CG) and effect size at baseline and post-intervention. Compared to the control group, participants allocated to intervention group decreased %BF, with an adjusted difference of − 1.747 (CI − 2.529 to − 0.964; p < 0.001) and a trivial effect size (d = 0.10). Similar results were found for glucose levels (adjusted difference = − 5.483; CI 9.163 to − 1.803; p = 0.005; d = 0.03), TC/HDL ratio (adjusted difference = − 1.478; CI = − 1.917 to − 1.039; p ≤ 0.001; d = 1.28), CVD composite score (adjusted difference = − 3.951; CI − 5.684 to − 2.218; p < 0.001; d = 0.87) and total food intake (adjusted difference = − 134.28; CI − 258.53 to − 10.03; p < 0.03; d = 0.52). There was no differences between groups on waist circumference, AST, triglycerides and CRF.
Discussion
We investigated the effects of a 12-week multicomponent intervention program in markers of metabolic syndrome and NAFLD among overweight/obese low-income school-aged children. Despite its importance, it is not well documented how such programs can be implemented. Indeed, only few investigations analyzed the implementation of a multicomponent approach [33,34,35,36], and at the best of our knowledge, none in low-income children. The most important findings lie on the fact that the 12-week intervention had positive effect in several variables such as %BF, glucose levels, ALT, TC/HDL ratio, the CVD composite score, and total food intake.
Previous studies using similar interventions showed the same results [37,38,39,40,41], especially those targeting youth metabolic syndrome [37, 39,40,41]. Likewise, a systematic review and meta-analysis investigating the effectiveness of lifestyle interventions based on nutrition and physical exercise on cardiometabolic risk factors of obese children showed positive effects in reducing body weight as well as improvements in LDL, TG, fasting insulin and blood pressure [13]. In addition, in agreement with our findings, Bianchini et al. [42] indicated that after a multidisciplinary intervention, including nutritional, parental and pediatrician actions, over 16 weeks with obese youngsters, there was a decrease in cardiometabolic risk factors and adiposity.
Regarding %BF, it was not found difference between groups at baseline. The large variability of the baseline results was maintained in post-test, meaning that differences found at the end of the study would be a consequence of the intervention program and not really influenced by the procedure used to evaluate this variable. Thus, the observed decrease in body fat, associated to better scores of lipid profile indicators and glucose level, is an important indicator of the effectiveness of the proposed multicomponent intervention. Indeed, although Nascimento et al. (23), after an 8-month regular physical exercise program found similar results, our study promoted those improvements in a shorter period of time, which seems to hold promise for childhood obesity worldwide [43, 44].
In addition, the observed significant reduction in TC/HDL ratio (from 3.84 to 2.91), which is an important marker of atherosclerotic risk [23] emphasize the multicomponent intervention relevance. In other study, although with untrained men, Nybo et al. [45] reported that the TC/HDL ratio was the only component of the lipid profile that improved (decreasing from 3.41 to 2.92) after an exercise intervention focused on promoting health.
Despite the positive effects in general lipid profile, our study showed a negative and unexpected outcome regarding TG values, which increased from baseline to post-intervention in both groups. However, similar findings were reported after a multicomponent intervention in obese children in both, control and intervention group [46]. Despite literature suggests that programs focused on physical exercise is effective in reducing or even maintaining TG levels [47] as well as those using diet [13], there still are some discrepancies regarding this issue. Moreover, the increasing of TG levels observed in the present study could also be associated with different factors besides diet or physical exercise, such as regional lipid concentration, genetics, or blood test technology [47].
Furthermore, it is worth to highlight that control group showed worse values of %BF, glucose, TC/HDL ratio and ALT than intervention group after only 12 weeks. Therefore, these negative changes indicate that children of the control group could be at higher risk of developing metabolic dysfunction, including type 2 diabetes and NAFLD. Thus, beyond the inherent damages and difficulties ascribed to both conditions, being overweight/obese and low-income do not be engage in some preventive action such as the intervention program might result in further additional threat.
Concerning CRF, intervention and control groups presented difference at baseline. Likewise, both groups showed improvements after 12 weeks. Nonetheless, it is important to highlight that intervention group presented higher difference from baseline to post-intervention, which could indicate a trend of improvement in this group, as seen in other studies applying longer periods of intervention with overweight children and adolescents [42, 48].
While most of studies that implemented interventions targeting NAFLD in youth were based on exercise and nutrition intervention [38], our program besides the exercise and diet focused on parental support (twice a week). ALT is one important marker of NAFLD. For instance, it was shown that 9 out 11 studies pointed out decreases of ALT levels as a result of intervention [38] that agrees with our findings and stressed the importance that multicomponent interventions might have on decreasing NAFLD markers in youth. Noteworthy to mention is the fact that our data focused on overweight/obese individuals belonging to a low socioeconomic level, which could lead to a potential extra value for the development of strategies for health prevention and promotion.
Therefore, a multicomponent intervention as developed in the present study seems to be an effective strategy to reduce %BF and improve several cardiometabolic risk factors among overweight/obese school-aged children. We also emphasize the relevance of implementing this intervention strategy in a low-income area, once children living in vulnerable zones have fewer physical activity facilities, low access to healthy nutrition, and some of them live without their parents decreasing their healthy opportunities compared to higher income families [49].
In summary, this study provides some evidences of a successful multicomponent intervention among low socioeconomic overweight/obese school-aged children. Given the positive effect, our approach could be taken in further studies as well as to be considered by public health policy responsible for implementing large-scale intervention actions mainly when targeting obese children and low socioeconomic populations.
Limitations and strengths
The present study has some limitations. The small sample size of intervention and control groups require caution regarding to generalization of these results to the wider population of Brazilian overweight/obese school-aged children. Nonetheless, the results were positive and added information to previous data. Besides, given the fact that we dealt with overweight/obese population our data would be of great clinical significance. Furthermore, the intervention covered only limited period of time (12 weeks), and it would be interesting to know whether a long-term program would result in continued improvement. Strengths of this study are the recruitment of low-income overweight/obese participants and the multicomponent design of intervention that included besides physical exercise and nutritional education an additional intervention with the children’s parents.
Conclusions
A 12-week multicomponent intervention was effective on decreasing some metabolic parameters in low-income overweight/obese youngsters. Therefore, this kind of intervention could be considered a tool for health promotion in this population.
Abbreviations
- MetS:
-
Metabolic syndrome
- TG:
-
Triglycerides
- HDL-C:
-
High-density lipoprotein cholesterol
- LDL-C:
-
Low-density lipoprotein cholesterol
- CVD:
-
Cardiovascular disease
- NAFLD:
-
Nonalcoholic Fatty Liver Disease
- AST:
-
Aspartate aminotransferase
- ALT:
-
Alanine aminotransferase
- BMI:
-
Body mass index
- PE:
-
Physical education
- HR:
-
Heart rate
- PA:
-
Physical activity
- %BF:
-
Percentage of body fat
- CRF:
-
Cardiorespiratory fitness
- IG:
-
Intervention group
- CG:
-
Control group
References
Tremblay MS, LeBlanc AG, Kho ME, Saunders TJ, Larouche R, Colley RC et al (2011) Systematic review of sedentary behaviour and health indicators in school-aged children and youth. Int J Behav Nutr Phys Act 8:98
Ogden CL, Carrol MD, Kit BK, Flegal KM (2012) Prevalence of obesity and trends in body mass index among US Children and Adolescents, 1999–2010. J Am Med Assoc 307:483
Wijnhoven TM, Van Raaij JM, Spinelli A, Starc G, Hassapidou M, Spiroski I et al (2014) WHO European Childhood Obesity Surveillance Initiative: body mass index and level of overweight among 6–9-year-old children from school year 2007/2008 to school year 2009/2010. BMC Public Health 14:806
Weiss R, Bremer AA, Lustig RH (2013) What is metabolic syndrome, and why are children getting it? Ann N Y Acad Sci 1281:123–140
Andersen LB, Lauersen JB, Brønd JC, Anderssen SA, Sardinha LB, Steene-Johannessen J et al (2015) A new approach to define and diagnose cardiometabolic disorder in children. J Diabetes Res 2015
Papandreou D, Karavetian M, Karabouta Z, Andreou E (2017) Obese children with metabolic syndrome have 3 times higher risk to have nonalcoholic fatty liver disease compared with those without metabolic syndrome. Int J Endocrinol 2017:1–5
Barshop NJ, Sirlin CB, Schwimmer JB, Lavine JE (2008) Review article: epidemiology, pathogenesis and potential treatments of paediatric non-alcoholic fatty liver disease. Aliment Pharmacol Ther 28:13–24
Siegel AB, Zhu AX (2009) Metabolic syndrome and hepatocellular carcinoma: two growing epidemics with a potential link. J Asian Econ 19:389–399
Vos MB, Abrams SH, Barlow SE, Caprio S, Daniels SR, Kohli R et al (2017) NASPGHAN clinical practice guideline for the diagnosis and treatment of nonalcoholic fatty liver disease in children: recommendations from the Expert Committee on NAFLD (ECON) and the North American Society of Pediatric Gastroenterology, Hepatology and Nutrition. J Pediatr Gastroenterol Nutr 64:319–334
Sartorio A, Del Col A, Agosti F, Mazzilli G, Bellentani S, Tiribelli C et al (2007) Predictors of non-alcoholic fatty liver disease in obese children. Eur J Clin Nutr 61:877–883
Kim JY, Cho J, Yang HR (2018) Biochemical predictors of early onset non-alcoholic fatty liver disease in young children with obesity study subjects. J Korean Med Sci 33:1–11
World Health Organization (2016) Draft Final Report of the Commission on Ending Childhood Obesity. World Health Organization, Geneva
Ho M, Garnett SP, Baur L, Burrows T, Stewart L, Neve M et al (2012) Effectiveness of lifestyle interventions in child obesity: systematic review with meta-analysis. Pediatrics 130:2012-1176
Sallis JF, Cervero RB, Ascher W, Henderson KA, Kraft MK, Kerr J (2006) An ecological approach to creating active living communitites. Annu Rev Public Health 27:297–322
Ranucci C, Pippi R, Buratta L, Aiello C, Gianfredi V, Piana N et al (2017) Effects of an intensive lifestyle intervention to treat overweight/obese children and adolescents. Biomed Res Int 2017
Davis JN, Ventura EE, Shaibi GQ, Byrd-williams CE, Alexander KE, Vanni AK et al (2010) Interventions for improving metabolic risk in overweight Latino youth 5:451–455
Davis JN, Kelly LA, Lane CJ, Ventura EE, Byrd- CE, Alexandar KA et al (2010) Randomized control trial to improve adiposity and insulin resistance in overweight Latino adolescents. Obesity 17:1542–1548
Silveira DS, Lemos LFGBF, Tassitano RM, Cattuzzo MT, Feitoza AHP, Aires LMSMC, Mota JP et al (2018) Effect of a pilot multi-component intervention on motor performance and metabolic risks in overweight/obese youth. J Sport Sci 36:2317–2326
World Medical Association (2013) Declaration of Helsinki Ethical Principles for Medical Research Involving Human Subjects. JAMA 310:2191–2194
Cole TJ, Bellizzi MC, Flegal KM, Dietz WH (2000) Establishing a standard definition for child overweight and obesity worldwide: international survey. BMJ 320:1240–1243
Weineck J (2005) Biologia do Esporte. Manole, São Paulo
Aires L, Silva G, Martins C, Marques E, Lagoa MJ, Ribeiro JC et al (2016) Exercise intervention and cardiovascular risk factors in obese children. Comparison between obese youngsters taking part in a physical activity school-based programme with and without individualised diet counselling: the ACORDA project. Ann Hum Biol 43:183–190
Nascimento H, Costa E, Rocha S, Lucena C, Rocha-Pereira P, Rêgo C et al (2014) Adiponectin and markers of metabolic syndrome in obese children and adolescents: impact of 8-mo regular physical exercise program. Pediatr Res 6:1–7
Lohman TG, Roche AF, Martorell R (1988) Anthropometric standardization reference manual. Human Kinetics Books, Champaign
Services UD (1996) NHANES III. Anthropometric procedures. Washington
Leger LA, Mercier D, Gadoury C, Lambert J (1988) The multistage 20 metre shuttle run test for aerobic fitness. J Sports Sci 6:93–101
Noonan RJ, Boddy LM, Knowles ZR, Fairclough SJ (2017) Fitness, fatness and active school commuting among liverpool schoolchildren. Int J Environ Res Public Health 14:995
Rodriguez-Ayllon M, Cadenas-Sanchez C, Esteban-Cornejo I, Migueles JH, Mora-Gonzalez J, Henriksson P et al (2017) Physical fitness and psychological health in overweight/obese children: a cross-sectional study from the ActiveBrains project. J Sci Med Sport 21:179–184
Artero EG, España-Romero V, Castro-Piñero J, Ortega FB, Suni J, Castillo-Garzon MJ et al (2011) Reliability of field-based fitness tests in youth. Int J Sports Med 32:159–169
Friedewald WT, Levy RI, Fredrickson DS (1972) Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin Chem 18:499–502
Tanner JM (1986) Normal growth and techniques of growth assessment. Clin Endocrinol Metab 15:411–451
Cohen J (1988) Statistical power analysis for the behavioral sciences. Erlbaum, Hillsdale
Kelder SH, Mitchell PD, McKenzie TL, Derby C, Strikmiller PK, Luepker RV et al (2003) Long-term implementation of the catch physical education program. Heal Educ Behav 30:463–475
Perry CL, Sellers DE, Johnson C, Pedersen S, Bachman KJ, Parcel GS et al (1997) The Child and Adolescent Trial for Cardiovascular Health (CATCH): intervention, Implementation, and Feasibility for Elementary Schools in the United States. Heal Educ Behav 24:716–735
McKay HA, Macdonald HM, Nettlefold L, Masse LC, Day M, Naylor P-J (2005) Action Schools! BC implementation: from efficacy to effectiveness to scale-up. Br J Sports Med 49:210–218
McKenzie TL, Strikmiller PK, Stone EJ, Woods SE, Ehlinger SS, Romero KA et al (1994) CATCH: physical activity process evaluation in a multicenter trial. Health Educ Q 2:73–89
Bugge A, El-Naaman B, Dencker M, Froberg K, Holme IMK, McMurray RG et al (2012) Effects of a three-year intervention: the Copenhagen School Child Intervention Study. Med Sci Sport Exerc 44:1310–1317
Africa JA, Newton KP, Schwimmer JB (2016) Lifestyle interventions including nutrition, exercise, and supplements for nonalcoholic fatty liver disease in children. Dig Dis Sci 61:1375–1386
Tanrikulu MA, Agirbasli M, Berenson G (2016) Primordial prevention of cardiometabolic risk in childhood. Adv Exp Med Biol 956:489–496
Van Buren DJ, Tibbs TL (2014) Lifestyle interventions to reduce diabetes and cardiovascular disease risk among children. Curr Diab Rep 14:557
García-Hermoso A, Carmona-López MI, Saavedra JM, Escalante Y (2014) Physical exercise, detraining and lipid profile in obese children: a systematic review. Arch Argent Pediatr 112:519–525
Bianchini JAA, da Silva DF, Nardo CCS, Carolino IDR, Hernandes F, Nardo NJ (2013) Multidisciplinary therapy reduces risk factors for metabolic syndrome in obese adolescents. Eur J Pediatr 172:215–221
Elvsaas IKO, Giske L, Fure B, Juvet LK (2017) Multicomponent lifestyle interventions for treating overweight and obesity in children and adolescents: a systematic review and meta-analyses. J Obes 2017:5021902
Bleich SN, Vercammen KA, Zatz LY, Frelier JM, Ebbeling CB, Peeters A (2018) Interventions to prevent global childhood overweight and obesity: a systematic review. Lancet Diabetes Endocrinol 6(4):332–346
Nybo L, Sundstrup E, Jakobsen MD, Mohr M, Hornstrup T, Simonsen L et al (2010) High-intensity training versus traditional exercise interventions for promoting health. Med Sci Sports Exerc 42:1951–1958
Guo H, Zeng X, Zhuang Q, Zheng Y, Chen S (2015) Intervention of childhood and adolescents obesity in Shantou city. Obes Res Clin Pract 9(4):357–364
Wang Y, Xu D (2017) Effects of aerobic exercise on lipids and lipoproteins. Lipids Health Dis 16:1–8
Carrel AL, Clark RR, Peterson SE, Nemeth BA, Sullivan J, Allen DB (2005) Improvement of fitness, body composition, and insulin sensitivity in overweight children in a school-based exercise program: a randomized, controlled study. Arch Pediatr Adolesc Med 159:963–968
Romero AJ (2005) Low-income neighborhood barriers and resources for adolescents’ physical activity. J Adolesc Heal 36:253–259
Acknowledgements
This work was supported by the “National Council for Scientific and Technological Development” (ID: 477893/2013-9), Coordination for the Improvement of Higher Education Personnel (CAPES) and Foundation for science and technology: SFRH/BSAB/142983/2018 and UID/DTP/00617/2019.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee (Ethics and Research Committee of the Health Sciences Center of the Federal University of Paraíba-number 0390/14) and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Brand, C., Lima, R.A., Silva, T.F. et al. Effect of a multicomponent intervention in components of metabolic syndrome: a study with overweight/obese low-income school-aged children. Sport Sci Health 16, 137–145 (2020). https://doi.org/10.1007/s11332-019-00590-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11332-019-00590-w