Abstract
The red skin color desired by most apple consumers is not easy to achieve in warm climates, as the expression of MYB10, which regulates red pigmentation in apple, is influenced negatively by high temperatures. We describe the development and validation of a genetic marker for red skin coloration that effectively predicts color in a warm summer environment in Spain, as well as more temperate climates in New Zealand and Italy. Following the determination of a major-effect quantitative trait locus (QTL) controlling red skin coloration on linkage group (LG)9, using four segregating populations grown in New Zealand, and screened using the IRSC apple 8-K single-nucleotide polymorphism (SNP) array, the most significant SNP marker (ss475879531) was transformed into a marker suitable for use in a real-time PCR assay. This marker was validated using five apple seedling populations growing in a warm summer environment in Spain, demonstrating that the marker system efficiently predicts red skin coloration and can be used for marker assisted selection, even under conditions considered adverse for skin color development.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The red fruit coloration of apple (Malus × domestica) determined by anthocyanin concentration is known to be controlled both genetically and by environmental conditions, with the MYB1/MYB10 transcription factor identified as the key regulator for red pigmentation in the skin (MYB1) (Takos et al. 2006) and (MYB10) flesh (Espley et al. 2007). The homologs MYB1 and MYB10 have been shown to be probably allelic (Lin-Wang et al. 2011). MYB10 activates the biosynthetic genes from the anthocyanin pathway, acting as part of a protein complex with bHLH and WD40 (Baudry et al. 2004). The expression of MYB10 not only correlates with anthocyanin accumulation in apple fruit skin but is also modulated by temperature (Lin-Wang et al. 2011), indicating that MYB10 is central to the negative effect of warm summer conditions on red color development. Candidate gene-based genetic mapping has located MYB10 and its associated control of red flesh coloration at the bottom of linkage group (LG)9 (Chagne et al. 2007). A genome-wide association study has recently demonstrated that this locus is the principal control for fruit skin coloration in wild and primitive apple collections (Kumar et al. 2014), as was earlier indicated in a breeding population (Lozano et al. 2014). Although a cleaved amplified polymorphic sequence (CAPS) marker based on a single-nucleotide polymorphism (SNP) in the promoter of MYB1 has been developed for the presence of red skin coloration (Takos et al. 2006), this marker was not completely transferable across populations when evaluated for marker-assisted selection (MAS) in a broader genetic background (Zhu et al. 2010). While CAPS markers can be applied by most molecular laboratories, they have a low genotyping throughput. More modern technologies such as capillary electrophoresis, real-time PCR, and SNP arrays offer greater opportunities for multiplexing markers and for reducing genotyping costs and time for screening large breeding populations.
Our study had the goal of developing a new robust genetic marker for red fruit skin color that can be applied by breeders for selection of seedlings that will express high-red color, even when grown under challenging conditions where summer temperatures repress coloration of commonly grown red-skinned cultivars, such as “Gala” (Iglesias et al. 2008). There has been no previous evaluation of the efficiency of MAS for expression of red skin color in climates with hot summer temperatures. To achieve this, we first mapped the locus associated with red skin color, using a large set of 994 apple seedlings from four breeding populations grown in a temperate climate in Hawke’s Bay, New Zealand. We then developed a robust allele-specific co-dominant PCR-based marker for the locus and validated it in a separate set of cultivars from San Michele all’Adige, Italy, and in five breeding populations grown under warm summer conditions at Gimenells, Spain.
Material and methods
Plant material
Four apple (Malus × domestica Borkh.) breeding families (Table 1) of 944 seedlings in total, segregating for fruit skin coloration and grown in Hawke’s Bay, New Zealand, were used for quantitative trait locus (QTL) detection. Three of the four segregating populations are described in Kumar et al. (2012), and the “Royal Gala” × “Granny Smith” population is described in Souleyre et al. (2014). Fruit were harvested in 2010 and 2011. Five biparental families comprising a total of 393 seedlings (Table 2) planted at an Institut de Recerca i Tecnologia Agroalimentàries (IRTA) orchard in Gimenells, Spain, in February 2008 were employed to validate a genetic marker for MAS. The sources of red coloration in these families are indicated in Supplemental Table 1. The fruit picking season spanned from 15 July to 10 September in both years (2011 and 2012) and coincided with high summer temperature periods, which would potentially impair skin reddening. Average maximum temperature was 31.0 and 31.7 °C, and average minimum temperature was 16.1 and 15.9 °C for 2011 and 2012, respectively. Thirty-four apple cultivars grown at the experimental station of the Research and Innovation Centre of Fondazione Edmund Mach, San Michele all’Adige, Italy, were used to evaluate the genetic range for application of the marker for red skin coloration. In all locations (New Zealand, Spain, and Italy), “Malling 9” was used as a rootstock and trees were conducted in a central axis system. Trees were managed according to the standard commercial practices in the regions where they were grown.
Phenotypic evaluation of red skin coloration
Six representative fruits were harvested when they were deemed to be mature based on the development of a green-yellow to pale yellow background color. Fruit skin red color was assessed immediately (Italy) or following 10 weeks of storage at 0.5 °C (Hawke’s Bay, Gimenells) before evaluation. Four variables were used to quantify the degree of red coloration of the apple skin. These phenotyping methods for each of the families and years of assessment are presented in Table 1. In three of the QTL detection families and in all validation populations, the percentage of overcolor was recorded over 2 years by two trained assessors, using a scale where 0 corresponds to absence of red coloration and 9 represents complete coverage of the fruit with coloration. Colorimetric measurements (L*, a*, b*) using a chromameter (Minolta Chroma Meter CR-400) were performed in the Royal Gala × Granny Smith population in 1 year and in the validation populations over 2 years (one spot measurement on the most exposed side of each fruit where red color was visually at the highest intensity). In the validation populations, a red skin intensity score was recorded by three trained assessors, using a scale where 0 is the absence of red color and 9 is the high-intensity red coloration. Anthocyanin concentration in the fruit skin was measured in 2012 in the five validation populations as described in Lozano et al. (2014). Individual fruit data were averaged for each seedling.
Genotyping and QTL mapping
Genomic DNA was extracted as described in Chagne et al. (2012). The four segregating populations were screened using the apple 8-K Infinium® II SNP array (Chagne et al. 2012) (Illumina Inc., San Diego, CA, USA). The SNP array data were analyzed as described in Chagne et al. (2012) using the Genotyping Module of the GenomeStudio Data Analysis software (Illumina Inc., San Diego, CA, USA). QTL analysis for skin coloration was performed with MapQTL® version 5 (www.kyazma.nl) using the non-parametric Kruskal-Wallis test. A QTL was declared if more than five adjacent markers had a significant Kruskal-Wallis K* value at p < 0.01.
Development of allele-specific SNP marker suitable for MAS
The SNP ss475879531 from the apple 8-K Infinium® II SNP array was transformed to a probe-based real-time PCR assay using the following primers and allele-specific probes: forward TGTGATGCCAACTCCGAAC, reverse GTGGTTGTCCTGGTGGATGATG, probe1 TGAAACATACCCTATCAGT, and probe2 TGAAACATACCTTATCAGTT. The probes were labeled with 5′ FAM or Orange-560, respectively, and with 3′ BHQ-1 plus (Biosearch Technologies Inc., Petaluma, CA, USA). The PCR mix contained 1× AccuStart Genotyping ToughMix (Quanta Biosciences, Gaithersburg, MD, USA), with 750-nM forward and reverse primers and 250 nM for each allele probe. PCR conditions were as follows: initial denaturation of 95 °C for 4 min, followed by 45 cycles at 95 °C for 5 s and 60 °C for 45 s (with single fluorescence acquisition), and a final cooling step at 4 °C. PCR was performed in a LightCycler480 (Roche Life Sciences, Basel, Switzerland), and the data were analyzed using the EndPoint Genotyping module with default parameters. The ss475879531 real-time PCR assay was evaluated over DNA from the five validation families from Gimenells, Spain, and 34 cultivars grown in San Michele all’Adige, Italy. For the Spanish families, leaf material was collected from young expanding leaves, freeze-dried, and genomic DNA was extracted using the automated protocol employed for apple MAS by Slipstream Automation Ltd., Palmerston North, New Zealand. DNA from the apple cultivars grown in Italy was extracted using the CTAB method (Doyle 1991). The association between the ss475879531 marker and the phenotype for red skin coloration in the five validation families was tested by one-way analysis of variance using the marker as a factor using GenStat v17.0.
Results
Phenotypic variation for red skin coloration in apple seedling populations
Four populations totaling 944 seedlings segregated for red skin coloration (Fig. 1). The “PremA153” × NZSelectionT51 and Royal Gala × Granny Smith populations were both derived from crosses that had one non-red or low red and one red parent. The PremA153 (low red) × NZSelectionT51 (high red) had a bimodal phenotypic distribution in both 2010 and 2011 for red skin phenotyped using percentage overcolor. The a* colorimetric measurements, where higher values indicate greater skin redness, also showed a binomial distribution for the Royal Gala (high red) × Granny Smith (non-red) population. The two other populations (“Sciros” × NZSelectionT51 and “Fuji” × NZSelectionT51) were families obtained from crossing two red-skinned parents, and their segregation for percentage overcolor in 2010 and 2011 was skewed toward high red, with only a small proportion of low-red seedlings. The Sciros × NZSelectionT51 population contained a larger proportion of seedlings with high-red fruit coverage than the Fuji × NZSelectionT51 population, which had more intermediate individuals with percentage overcolor ranging between 30 and 60 %.
Phenotypic distribution for red coloration in four apple mapping populations. Red coloration of the fruit skin was recorded by trained assessors using a 0 (no red coloration) to 9 (high-red intensity) scale for three segregating populations in 2010 and 2011. In the “Royal Gala” × “Granny Smith” population, colorimetric measurement was performed using a Minolta Chroma Meter. The a* axis of colorimetry ranges from green (low values) to red (high values)
QTL mapping and haplotype segregation at target locus
The number of polymorphic markers from the apple IRSC 8-K SNP array screen that were employed for QTL analysis is presented in Table 3 for each QTL detection family. A single significant QTL located on LG9 was detected for percentage overcolor for each family. The ss475879367 marker that was significant in the Sciros × NZSelectionT51 is likely to be misplaced in the reference apple genome of “Golden Delicious.” Marker ss475879531 (RosBREEDSNP_SNP_TC_31964859_Lg9_00452_MAF40_92351_exon1) was the most significant one for the red parent of the families obtained from non-red × red crosses (Royal Gala × Granny Smith; PremA153 × NZSelectionT51); however, the most significant SNP markers varied among populations and parents. The effect of these most significant markers on the phenotype was estimated in each detection family using analysis of variance (Table 4). The ss475879531 marker was explained between 45.3 and 75.6 % of the phenotypic variation in percentage overcolor (Table 4). To enable the determination of fully informative haplotypes in the PremA153 × NZSelectionT51, Sciros × NZSelectionT51, and Fuji × NZSelectionT51 populations, a second marker located close to ss475879531 (GDsnp00659) was selected based on its linkage phase calculated during the mapping analysis with JoinMap. GDsnp00659 explained less phenotypic variation than ss475879531, except for in the PremA153 × NZSelectionT51 population, where the markers co-segregated and explained the same amount of variance.
A schematic for the haplotype segregation in three families is shown in Fig. 2, illustrating the two, three, and four haplotypes observed for the PremA153 × NZSelectionT51, Sciros × NZSelectionT51, and Fuji × NZSelectionT51 populations, respectively. SNP marker ss475879531 is located at position 31,964,859 bp on LG9, while GDsnp00659 is at position 32,332,810 bp, suggesting that the haplotype developed from these two SNPs spans 368 kb. The MYB10 gene starts and ends at positions 32,851,966 and 32,847,982, respectively, indicating that the haplotype fragment is located 515 kb downstream of MYB10. The haplotype carrying the A allele for both markers (yellow haplotype in Fig. 2) is associated with low-red skin coloration, as illustrated by an average percentage overcolor ranging from 20 to 25 % in both 2010 and 2011, and in both PremA153 × NZSelectionT51 and Sciros × NZSelectionT51 populations. Conversely, three haplotypes carrying at least one G allele from either of the two markers (green, red, and blue haplotypes in Fig. 2) had an average percentage overcolor greater than 59 % across years and populations. In the Fuji × NZSelectionT51 population, a combination of the yellow and green haplotypes was observed and had a lower average percentage overcolor than the other haplotype combination, which indicates that the green haplotype may be less efficient at producing red coloration than the other two. When the haplotypes derived from both ss475879531 and GDsnp00659 markers were subjected to an analysis of variance, between 49.4 and 77.9 % of the phenotypic variation was explained (Table 4). The difference in explained phenotypic variation between a single marker and the haplotyped markers was smaller than the difference between populations. The percentage of explained variation was consistent between years and within populations for all two markers.
Schematic showing distribution of haplotypes influencing red coloration observed for 2 years in three apple mapping populations. Haplotypes were determined using SNP markers ss475879531 and GDsnp00659. The total number of seedlings in each progeny (N) that carry each combination of haplotypes are presented, with the average percentage overcolor (Ave%O) and the standard deviation (SD) of the seedlings carrying that haplotype
Validation of new allele-specific marker for red skin coloration
The ss475879531 marker was transformed into a real-time qPCR assay and screened over 34 cultivars grown in San Michele all’Adige. An example of the real-time qPCR assay output for the allele-specific ss475879531 SNP marker is given in Fig. 3, illustrating the clarity of the haplotype scoring for the two homozygotes and heterozygote. The 34 cultivars grown in San Michele all’Adige had good concordance between the marker genotype and red skin phenotype (Table 5 and Fig. 3).
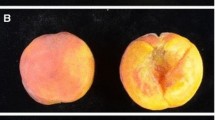
Example of a real-time PCR assay output for apple SNP marker ss475879531. Fluorescence intensity at 523–568 and 483–533 nm was acquired as an endpoint measurement after the PCR amplification using fluorescently labeled probes for alleles A and G, respectively. Homozygous AA and GG genotypes fluoresce only at 523–568 and 483–533 nm, respectively. Heterozygous AG genotype fluoresces at both ranges of wavelength. Ungrouped genotypes with intensity values outside genotypic clusters are scored as “unknown,” while samples with no fluorescence intensity for both probes indicate failed reactions (negative). Examples of apple phenotypes from the IASMA germplasm collection are shown for each genotype, a “Ingrid Marie,” b “Red Free,” c “Sansa,” d Coop-9, e “Renoire,” f “McIntosh,” g “Pinova,” h “Cox’s Orange Pippin,” i “Newton Pippin,” j “Granny Smith,” k “Prime Gold,” and l “Ananas Reinette”
Among the 393 seedlings of the five biparental validation populations grown in Spain and genotyped with the new marker, there were only seven unknown genotypes and three failed reactions. The marker was polymorphic in four of the five validation families (Fig. 4), and one family was monomorphic (PM92). Of the five validation populations, total of 336 and 241 seedlings had both genotype and phenotype information in 2011 and 2012, respectively. With only five seedlings, population PM95 was too small for differentiation between the AG and GG genotype effects; however, all five individuals carried at least one red G allele and exhibited high-red coloration (Supplemental Figure 1). Population PM92 was monomorphic for the red allele (GG), except for three seedlings which may be pollen contaminants, and all individuals exhibited high-red coloration in both years. In the PM91 and PM94 populations, the GG genotype had higher average percentage overcolor than the AG genotype (p < 0.001 in both 2011 and 2012). In the PM96 population, the AA genotype had lower average percentage overcolor than the AG genotype (p < 0.001 in both 2011 and 2012). In the PM94 population, only two and three individuals in 2011 and 2012, respectively, had the AA genotype and were probable pollen contaminants, with these individuals exhibiting average percentage overcolor coverage less than 40 %. The concordance between the marker and the red phenotype measured using anthocyanin concentration (in 2012) and red intensity (in 2011 and 2012) was consistent (Supplemental Figure 1), where overall, the individuals with the AA genotype had a lower concentration of skin anthocyanin (p < 0.001 in 2012) and lower intensity (p < 0.001 in both 2011 and 2012) than both the AG and GG genotypes.
Performance of the red skin marker in four apple breeding populations grown under warm summer temperatures. The red skin phenotype is represented by the percentage of overcolor (% overcolor) measured in 2011 and 2012. The three genotypes of marker ss475879531 (AA, AG, and GG) are presented. NA no amplification (negative), NC no call (unknown genotype). The PM95 validation population is not presented as it had only five seedlings
Discussion
This study has taken advantage of biparental populations from breeding programs with orchards located in very different climates in New Zealand and Spain to develop new information concerning the genetic control of red skin coloration. Furthermore, we have demonstrated that a novel allele-specific genetic marker developed in cool New Zealand summer climates using biparental QTL mapping is also predictive of skin color in a warm summer climate in Spain, where color development is repressed in cultivars that exhibit a high degree of coloration in summer-cool environments.
Toward understanding the genetic control of red coloration in apple
The MYB1 locus controlling red skin color in apple was previously assumed to be the same as the MYB10 red flesh locus on LG9 based on circumstantial evidence and association mapping studies. MYB10 regulates the activity of the genes from the anthocyanin pathway, and an allele of MYB1/MYB10 was demonstrated to modulate red skin coloration and anthocyanin concentration (Takos et al. 2006; Espley et al. 2007). MYB1 and MYB10 were earlier demonstrated as highly likely to be allelic (Lin-Wang et al. 2011) and will from here on be referred to as MYB10 for simplicity. In addition, the association of the MYB10 locus with red skin color has been indicated by association mapping (Kumar et al. 2014; Lozano et al. 2014). We have used QTL mapping to locate a large-effect QTL for apple red skin color on LG9, using four biparental segregating populations involving five breeding parents that exhibit red skin color. This QTL controlling skin color co-locates with the MYB10 candidate gene, as does the red flesh trait (Chagne et al. 2007). Further studies will be required to elucidate the controls determining tissue specificity of red coloration.
Development of red skin color is repressed in some cultivars when fruit ripen under warm conditions, and this effect has been demonstrated to be also under the control of MYB10 (Lin-Wang et al. 2011). The marker we developed from the SNP marker most significantly associated with red skin color, and MYB10 in populations grown under cooler summer temperatures was also effective in predicting skin coloration in a warm climate. In red-fleshed apples, it is likely that other transcription factors may activate MYB10 in response to environmental cues, and the same situation may also apply to temperature-sensitive control of red skin color. Locating the DNA variations in regulatory motifs upstream of MYB10 that might modify its activity and consequently the downstream activity of the anthocyanin biosynthesis genes is one of the next challenges for apple geneticists. Some evidence of cytosine methylation in the 2.5-kb promoter region upstream of MYB10 has been demonstrated in striped “Honeycrisp” apples (Telias et al. 2011) and may indicate that the causative mutation is in the MYB10 regulatory region. The functional alleles of MYB10 might be identified, for example, by evaluating the efficiency of the different haplotypes for anthocyanin gene activation, activation of MYB10 itself, and as a target of transcription factors regulating MYB10. Furthermore, DNA polymorphisms in the proximity of MYB10 should be determined in a wide range of apple germplasm exhibiting a range of skin color responses to warm temperature.
Prospects for using the ss475879531 real-time assay for MAS in apple
The development of a robust allele-specific marker validated both across biparental populations and a genetically diverse cultivar collection for linkage to a QTL explaining most of the phenotypic variation for apple red skin coloration has opened the prospect of using a marker derived from ss475879531 to enhance the efficiency of apple breeding, by employing MAS to identify the seedlings with genetic potential to exhibit red skin.
Our results indicate that although a single marker would not explain all the phenotypic variability that a haplotype-based marker would, a single-marker system would not be greatly outperformed by a system using haplotypes. Use of a single marker for MAS is more practical than a suite of markers on the basis of cost. After testing the hypothesis that marker ss475879531 alone would be efficient for selection, we transformed it into a real-time allele-specific qPCR assay, which is an attractively inexpensive and rapid method for genotyping. The use of a real-time PCR system with labeled probes present in the reaction mix enables the assay to be run in a closed tube, without the requirement to separate the PCR products using electrophoresis, hence greatly speeding up the screening process and reducing the potential human errors during gel loading or data analysis. Most modern research laboratories have access to a real-time PCR machine capable of fluorescence detection.
We used a population of five segregating families with fruit maturing during the warm summer period in northern Spain to validate the ss475879531 real-time PCR assay. The temperatures during the last stages of fruit development were sufficiently high to inhibit reddening of the fruit skin. We demonstrated that our marker efficiently predicts red skin coloration, even under challenging summer conditions. The individuals from the segregating populations that were homozygous for the red allele (GG) using the ss475879531 real-time PCR assay consistently bore fruit with high-red intensity, high-percentage overcolor, and high-anthocyanin concentration. We recommend the use of the ss475879531 real-time marker assay to breeders who have the goal of developing new apple cultivars with red skin that is attractive and develops consistently over a range of summer temperatures.
References
Baudry A, Heim MA, Dubreucq B, Caboche M, Weisshaar B, Lepiniec L (2004) TT2, TT8, and TTG1 synergistically specify the expression of BANYULS and proanthocyanidin biosynthesis in Arabidopsis thaliana. Plant J 39:366–380
Chagne D, Carlisle CM, Blond C, Volz RK, Whitworth CJ, Oraguzie NC, Crowhurst RN, Allan AC, Espley RV, Hellens RP, Gardiner SE (2007) Mapping a candidate gene (MdMYB10) for red flesh and foliage colour in apple. BMC Genomics 8:11. doi:10.1186/1471-2164-8-212
Chagne D, Crowhurst RN, Troggio M, Davey MW, Gilmore B, Lawley C, Vanderzande S, Hellens RP, Kumar S, Cestaro A, Velasco R, Main D, Rees JD, Iezzoni A, Mockler T, Wilhelm L, Van de Weg E, Gardiner SE, Bassil N, Peace C (2012) Genome-wide SNP detection, validation, and development of an 8K SNP array for apple. PLoS ONE 7:12. doi:10.1371/journal.pone.0031745
Doyle JJ (1991) DNA protocols for plants. Springer Verlag, Berlin
Espley RV, Hellens RP, Putterill J, Stevenson DE, Kutty-Amma S, Allan AC (2007) Red colouration in apple fruit is due to the activity of the MYB transcription factor, MdMYB10. Plant J 49:414–427. doi:10.1111/j.1365-313X.2006.02964.x
Iglesias I, Echeverria G, Soria Y (2008) Differences in fruit colour development, anthocyanin content, fruit quality and consumer acceptability of eight “Gala” apple strains. Sci Hortic 119:32–40. doi:10.1016/j.scienta.2008.07.004
Kumar S, Chagne D, Bink MCAM, Volz RK, Whitworth C, Carlisle C (2012) Genomic selection for fruit quality traits in apple (Malus × domestica Borkh.). PLoS ONE 7. doi:10.1371/journal.pone.0036674
Kumar S, Raulier P, Chagné D, Whitworth C (2014) Molecular-level and trait-level differentiation between the cultivated apple (Malus × domestica Borkh.) and its main progenitor Malus sieversii. Plant Genet Resour
Lin-Wang K, Micheletti D, Palmer J, Volz R, Lozano L, Espley R, Hellens RP, Chagne D, Rowan DD, Troggio M, Iglesias I, Allan AC (2011) High temperature reduces apple fruit colour via modulation of the anthocyanin regulatory complex. Plant Cell Environ 34:1176–1190. doi:10.1111/j.1365-3040.2011.02316.x
Lozano L, Iglesias I, Micheletti D, Troggio M, Kumar S, Volz RK, Allan AC, Chagne D, Gardiner SE (2014) Feasibility of genome-wide association analysis using a small single nucleotide polymorphism panel in an apple breeding population segregating for fruit skin color. J Am Soc Hortic Sci 139:619–626
Souleyre EJF, Chagné D, Chen XY, Tomes S, Turner RM, Wang MY, Maddumage R, Hunt MB, Winz RA, Wiedow C, Hamiaux C, Gardiner SE, Rowan DD, Atkinson RG (2014) The AAT1 locus is critical for the biosynthesis of esters contributing to “ripe apple” flavour in “Royal Gala” and “Granny Smith” apples. Plant J 78:903–915. doi:10.1111/tpj.12518
Takos AM, Jaffe FW, Jacob SR, Bogs J, Robinson SP, Walker AR (2006) Light-induced expression of a MYB gene regulates anthocyanin biosynthesis in red apples. Plant Physiol 142:1216–1232. doi:10.1104/pp.106.088104
Telias A, Kui LW, Stevenson DE, Cooney JM, Hellens RP, Allan AC, Hoover EE, Bradeen JM (2011) Apple skin patterning is associated with differential expression of MYB10. Bmc Plant Biol 11. doi:10.1186/1471-2229-11-93
Zhu Y, Evans KM, Peace C (2010) Utility testing of an apple skin color MdMYB1 marker in two progenies. Mol Breed. doi:10.1007/s11032-010-9449-6
Acknowledgments
This work was funded by the New Zealand Ministry for Business Innovation and Employment (MBIE; Contract No. C06X0812) and PREVAR Ltd research consortium (Contract No. 26015), by the European Union funded project in the 7th Framework Programme: Marie Curie Actions, People International Research Staff Exchange Scheme (IRSES) proposals nº 230857 (REDHOTGEN) and nº 295146 (REDHOTGEN-2) and by Spain’s Instituto Nacional de Investigación y Tecnología Agraria y Alimentaria (INIA; project RTA 2009-00069-00-00). We thank Slipstream Automation Ltd. for providing the DNA extraction service.
Data archiving statement
The SNP accession numbers from the apple Infinium 8-K array were deposited into GenBank dbSNP. Marker validation data is shown in the supplementary material.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by E. Dirlewanger
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplemental Figure 1
Marker validation for red skin coloration in five apple families. The red coloration is expressed as anthocyanin concentration and intensity score. The parentage of the five populations is indicated in Supplemental Table 1 (PPTX 306 kb)
Supplemental Table 1
Parentage of the five families used for marker validation for red skin in apple (DOCX 12 kb)
Rights and permissions
About this article
Cite this article
Chagné, D., Kirk, C., How, N. et al. A functional genetic marker for apple red skin coloration across different environments. Tree Genetics & Genomes 12, 67 (2016). https://doi.org/10.1007/s11295-016-1025-8
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11295-016-1025-8