Abstract
Five differently adapted natural populations of the native salt desert grass Lasiurus scindicus Henrard from Lesser Cholistan Desert (Pakistan) in South Punjab of east central Pakistan, were evaluated to examine their mechanism of adaptation to saline stress based on some key morpho–anatomical and physiological characteristics. Five ecotypes were collected from one saline site, two moderately saline sites, and two highly saline sites. Anatomical adaptations in each ecotype critically supported the physiological, but the adaptations were of specific nature depending on the type of each site’s normal habitat conditions. Higher salinities resulted in increased Na+, Cl−, Mg2+, Ca2+ and K+ content in root and shoot. At root level, some specific structural modifications like increased sclerification in cortical and pith regions, endodermal thickness, and number and size of xylem vessels are vital for water conservation under osmotic stress. Several characteristics were promising for increasing the plants ability to deal with osmotic stress, including at the stem level, increased sclerification, stem area, cortical region thickness and vascular bundle area, and at the leaf level, significant structural modifications such as leaf thickness, epidermal thickness, sclerenchymatous area, cortical area, metaxylem area and bulliform cell area were promising. All these may contribute towards water conservation, which ultimately account for ecotype survival under saline–induced physiological droughts.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
World agriculture is severely affected by abiotic stresses, the most important are salinity, drought, heat and cold, may reduce yield by more than 50 % or more for many crop plants (Wang et al. 2003; Öztürk et al. 2006; Ashraf et al. 2009, 2012; Mittler and Blumwald 2010; Hakeem et al.2013). Salinization of water and soil is among the environmental hazards that severely reduce the agricultural production all over the world (Maqsood et al. 2013). High soil salt content affects plant growth by imposing physiological drought, ion toxicity and oxidative stress (Chen et al. 2007; Cuin and Shabala 2007) which ultimately modify plant morphology, anatomy and physiology. The plant’s response to external stress is complex involving not only morpho-anatomical changes but also internal metabolic adjustments that enable the plant to survive under adverse conditions (Demmig-Adams et al. 2008). The increase in population pressure along with severe climatic changes sternly affects the crop productivity and as a result, the development of tolerant varieties is the first priority of scientist (Newton et al. 2011; Lobell et al. 2011).
Highly complex mechanisms are involved in plant responses to multiple stresses. These mechanisms enable the plant to detect and respond to hazardous environmental constraints in order to minimize their harmful effects (Cramer et al. 2011; Atkinson and Urwin 2012). Determental environmental hazards like water and soil salinization severely reduce the agricultural production all over the world (Jouyban 2012).
The plants inhabiting desert environments subjected to vagaries of nature like water, heat and salt stress. These plants have developed specific features in their morphology and anatomy which help them to thrive under adverse conditions (Naz et al. 2009; Hameed et al. 2011). Lasiurus scindicus is an inhabitant of saline patches in the Cholistan Desert, and a highly nutritive and palatable grass. It dominates the vegetation of sandy habitat that receives low rainfall, however, height and cover may decrease significantly along the increasing salinity gradient (Naz et al. 2010a, 2010b). As this species is native to xero-halophytic conditions by growing there for a long period of time, so must exhibit the tremendous tolerance to abiotic stresses, therefore, the present study was focused on examination of structural and functional aspects of adaptation of this grass to habitats differing in salinity stress.
Materials and methods
Study area
The Cholistan Desert in Pakistan spreads over about 26,000 km2 in the South Punjab. Huge landmass of the Cholistan desert comprises three distinct ecological zones, inter-dune flats, sand dunes and salt-affected flats. Hard and compact clayey flats (dahars) inter-spread among loose sand dunes. Soil of dahars varies in soil phyco-chemical characteristics, in particular, texture and salinity. Soils of the Cholistan desert are very poor, lacking organic matter <1.0 %. A single dunes may reach to average height of over 100 m or so. The annual rainfall varies from 100 to 200 mm, mean temperature 6.6 °C (December–January) 46 °C (June), with absolute maximum up to 52 °C. The relative humidity varies from 35 to 65 %. Rain water is stored in temporary ponds that evaporates very quickly. Underground water is generally brackish, and relatively deeper about 40 m or so below the soil surface (Arshad et al. 2008).
Plant material
Lasiurus scindicus is a rhizomatous, tussock-forming grass that can reach to 1.5 m in height (Fig. 1). It is native to drier regions of Asia and Africa (Cope 1982), and shows better growth under low rainfall as in the Cholistan Desert (El-Keblawy et al. 2009). This grass is relished by domestic livestock and local wildlife, such as nilgai, chinkara and blackbuck (Assaeed 1997). This species of grass, a major component of the vegetation of the flat plains in the desert, is more palatable than any other native species of the Cholistan Desert, and also stabilizes loose sand dunes (Khan et al. 1999).
Habitat ecology of Lasiurus scindicus in the Cholistan desert. a Lasiurus scindicus a highly palatable, nutritious, dominant, drought tolerant grass of the area. It is found almost everywhere, particularly, on sand dunes and interdunal sandy plain areas. b Sand dune vegetation dominated by L. scindicus along with Ochthochloa compressa. c Pure lush green community of L. scindicus after plenty of monsoon rainfall. In some areas it has pure communities spread over kilometers. d Pure community of dry L. scindicus in the Cholistan Desert
Data collection and analysis
Five saline habitats were selected from the Cholistan desert for the present study. The selection was made on the basis of specific halophytic plants inhabiting at that site as well as on specific features of the soil including soil type, soil colour, and moisture content (Table 1). Five ecotypes were collected as follows from: a low saline site, named Derawar Fort (DF); two moderately saline sites, Trawaywala Toba (TT) and Bailahwala Dahar (BD); and two highly saline sites, Ladam Sir (LS) and Pati Sir (PS).
Soil analysis
Soil sampling was conducted at 15 and 24 cm depth in the rhizosphere because these depths have the maximum root mass. 200 g of dried soil was used to measure the saturation percentage, pH and electrical conductivity (ECe). ECe and pH was recorded on pH/EC meter (WTW series InoLab pH/Cond 720, California, USA). Other soil analysis were carried out in lines of the procedure adopted by USDA Laboratory Staff (1954). Soil Na+, K+, and Ca2+ were estimated with a flame photometer (PFP–7, Jenway, Essex, UK). Serial grades of 10–100 mg L−1 standard solutions were prepared for each cation. These standard solutions were used to draw a standard curve for each element. The optical densities of cations were estimated on flame photometer and total concentration of each cation in each soil sample was calculated. Cl− content was recorded on a digital chloride meter (Model 926, Sherwood Scientific Ltd., Cambridge, UK).
Morpho-anatomical studies
Data for morphological characteristics like root and shoot length, leaf area, leaf hairiness and plant fresh and dry weights were recorded from fully mature plants. For the anatomical studies, a 2 cm piece was taken from fully expanded largest leaf for leaf anatomy, from the 3rd internode of the largest tiller for stem anatomy, and the thickest adventitious root near the root/shoot junction for root anatomy. All adventitious root are almost of equal thickness, so we selected the thickest among them to have a consistency. Formalin–acetic–alcohol solution was used as a fixative that includes v/v 35 % distilled water, 50 % ethanol, 10 % acetic acid and 5 % formalin. The material was then transferred to acetic alcohol preservative solution (v/v 25 % acetic acid and 75 % ethanol). Free-hand sectioning technique was used for the preparation of permanent slides. A serial grades of ethanol were used for dehydration of the material. For staining, safranin was used for lignified tissue and fast green for parenchymatous cells. Anatomical data were recorded on a light microscope (Nikon SE, Anti–Mould, Tokyo, Japan) and ocular and stage micrometers were used for calibration of ocular micrometer. Micrographs of the transverse sections were taken with a digital camera (Nikon FDX-35) on a stereo–microscope (Nikon 104, Japan).
Physiological parameters
Analysis of inorganic elements
Concentrated sulphuric acid (H2SO4) was added to dried ground material (0.5 g) in digestion tubes (size 10 mL), incubated overnight at room temperature. 35 % hydrogen peroxide (H2O2) was then slowly added to digestion tube, and the tubes were ported in a digestion block system (KJELDATHERM basic, C. Gerhardt GmbH & Co. KG, Bonn, Germany) and heated at 350 °C. The digestion tubes were removed from the block after 30 min and then allowed to cool at room temperature. Hydrogen peroxide was then again added the tubes were placed back into the block. The procedure was repeated till the solution turned colourless. The extract was filtered and used for determining K+, Ca2+, Na+ Cl− and Mg2+ concentrations. Na+ and K+ with recorded a flame photometer (Model 410, Sherwood Scientific Ltd., Cambridge, UK), Ca2+ and Mg2+ on an atomic absorption spectrophotometer (Model Analyst 3000; Perkin Elmer, Norwalk, CT), and Cl− with a chloride meter (Model 926; Sherwood Scientific Ltd., Cambridge, UK).
Ion washing
The same leaf (that was used for ionic concentration) was used for the determination of excreted ions, which are deposited on leaf surface via salt hairs and glands. The leaves were immediately washed in 20 ml of deionised distilled water. The excreted ions were recorded from the wash.
Statistical analysis
The experiment was designed in a completely randomized design with ten replications. The data were subjected to analysis of variance, and the mean values were compared by the least significance difference test. Standard error was calculated for each mean value.
Results
Soil analysis
Soil textural classes, saturation percentage, moisture contents, organic matter, soil pH, ECe, and soil Na+, K+, Ca2+, and Cl− contents were determined (Table 1). The degree of salinity of the sites could be ranked as: least (DF) <moderately (TT < BD) <highly salt–affected (LS < PS).
Morpho–physiological parameters
Shoot growth (measured by growth in leagth) of L. scindicus was the highest at moderately saline habitats, Trawaywala Toba (TT) and Bailahwala Dahar (BD). Shoot length significantly decreased at highly saline sites Ladam Sir (LS) and Pati Sir (PS); however, the plants inhabiting the least saline Derawar Fort (DF) were smaller in height as compared to those from moderately saline sites. Root length, in contrast, gradually and consistently decreased with increase in salinity level of the habitat, but the difference between the two moderately saline sites TT and BD was not significant (Table 2).
Flag leaf area and total leaf area per plant were considerably high in plants inhabiting moderately saline TT, which were about two-fold greater than those recorded in plants from the other habitats (Table 2). Plants from the highest saline PS showed the minimum value of these two characteristics. Leaf area of the plants from moderately saline BD was relatively higher than that from the least saline DF.
Shoot and root Na+ content gradually increased in plants as salinity increased to moderately saline BD, but the differences were non-significant (Table 2). However, higher salinities at LS and PS showed a significant increase in this characteristics. The maximum of this parameter was recorded in the plants from highly saline PS. There was a gradual and significant increase in K+ content of shoot and root with increase in salinity level. Shoot Ca2+ was the maximum in plants from moderately saline TT, but further increase in salinity level of the habitats showed a gradual decrease in this parameter. In contrast, root Ca2+ showed a consistent increase with increase in salinity level, the plants from highly saline PS showed a significantly increased accumulation of Ca2+ in roots than plants from all other habitats.
Shoot and root Mg2+ gradually increased with increasing salinity levels of the habitat, but decreased at the highest level of PS (Table 2). However, the difference among the different habitats with respect to shoot and root Mg2+ was not significant. A significant decrease was noticed in shoot with increase in salinity levels, whereas, root was maximum at moderate saline TT, and thereafter significantly decreased with increasing salinity levels of the habitats.
A consistent and significant increase in excreted Na+ and Cl− was recorded in L. sindicus ecotypes with increasing salinity levels of these habitats (Table 2). Excreted Ca2+ also showed a significant increase, but up to high salinity of LS. This parameter decreased significantly at the highest saline site PS. An increase in excreted Mg2+ was also recorded, but the differences among different ecotypes were not significant. Excreted K+ was the maximum at moderately saline TT, but higher salinity level of the habitats resulted in a gradual but non-significant decrease.
Anatomical parameters
Root anatomy
Root cross-section area was significantly higher at moderately saline TT, but a decrease was recorded along with salinity levels. Tissues outside the endodermis were disintegrated in the ecotype from moderately saline BD and highly saline LS and PS. Length of root hairs was significantly higher at moderately saline TT as compared to that at the least saline DF (Table 3). A similar trend was seen in the case of cortical cell area and thickness of epidermal and sclerenchymatous region, but the difference was not significant in the case of cortical region thickness. All these parameters were not possible to record in the ecotypes from BD, LS, and PS due to disintegration of tissues.
A gradual and significant increase in endodermal cell area was recorded with increasing levels of salinity in the habitats (Table 3). Pericyle thickness, however, increased up to moderately saline BD. A further increase in salinity level of the habitat depicted a significant decrease.
Metaxylem vessel number in ecotypes increased significantly along with salinity level of the habitats, but up to highly saline LS (Table 3). Plants inhabiting the highest saline PS showed a significant decrease in this parameter. Area of metaxylem gradually and significantly increased with increasing salinity levels. Phloem area showed a significant and consistent increase with increase in salinity level of the habitats, but up to high saline LS. A significant decrease in this attribute was noted in plants inhabiting the highest saline PS.
A gradual increase was recorded in pith area as the salinity level of the habitat increased, but the differences among the moderately saline BD and highly saline LS and PS were not significant. Pith cell area increased with increasing salinity level of the habitat up to moderately saline BD. Plants inhabiting the highest saline PS site showed significantly reduced cells of pith parenchyma (Table 3).
Stem anatomy
A significant and consistent increase was recorded in the stem cross-section area as the salinity of various habitats increased (Table 3). The ecotypes inhabiting highly saline sites (LS and PS) showed considerably thicker stem than those from the other sites. Cuticle thickness did not alter at lower saline DF and moderately saline TT and BD, but it increased significantly at higher salinities.
Epidermal thickness was the maximum in plants inhabiting moderately saline TT, but the differences in this parameter among plants from various habitats were not significant (Table 3). Sclerencyma was not observed in the plants from lower salinities (DF and TT). However, thickness of the sclerenchymatous tissue was significantly higher in the plants from higher salinities (LS and PS) than that recorded in those inhabiting moderately saline BD habitat.
A significant increase in cortical region thickness was observed as the salinity level of the habitat increased (Table 3). However, quite an opposite trend was recorded in the cellular area of cortical cells, wherein a significant decrease along the increasing salinity gradient was recorded.
Vascular bundle area increased significantly, but the phloem area decreased as the salinity level of the habitats increased. Metaxylem vessel area, however, increased up to moderately saline BD, but thereafter a significant decrease was recorded with a further increase in salinity level (Table 3).
Leaf anatomy
An increase in leaf (midrib and lamina) thickness was recorded with increasing salinity levels of the habitats, but the differences among the different ecotypes were not significant, particularly in those inhabiting highly saline habitats (Table 3). A significant increase in epidermal thickness at both leaf surfaces was recorded with increase in salinity gradient of different habitats. A significant increase in sclerenchymatous area and cortical region thickness was also recorded as the salt level increased. A similar trend was recorded in the case of cellular area of cortical parenchyma, but the plants inhabiting the highest saline PS showed a slight but non–significant decrease.
A consistent decrease in bundle sheath thickness and mesophyll thickness was observed with increase in salinity level, however, the differences among different ecotypes were not significant with respect to these parameters (Table 3). A significant and sequential increase was recorded in vascular bundle area, metaxylem area, and phloem area with increase in salinity levels, but plants inhabiting the highest saline PS showed a significant decrease in these characteristics. Bulliform cell number significantly increased with increase in salinity gradient of the habitats.
Number of stomata on abaxial leaf surface significantly decreased with increase in salinity level of the habitats, but the differences between the ecotypes from the highly saline LS and PS were not significant (Table 3). In contrast, stomatal number on adaxial leaf surface increased up to moderately saline BD, but higher levels resulted in a significant decrease in this attribute. Stomatal area on the abaxial leaf surface, however, was the maximum at moderately saline BD, but it decreased significantly at higher salinities (LS and PS). Stomatal area on adaxial surface showed a consistent decrease along salinity gradient, but the differences among different ecotypes did not decrease significantly. However, the number of stomata increased as salinity level of the habitats increased. Plants inhabiting the highest saline PS had extremely reduced trichome size, but exceedingly high in density per mm2 (Table 3).
Discussion
Differentially adapted populations of L. scindicus responded very specifically under habitat with varying salt levels in relation to structural and functional features. Since this species is moderately tolerant to salt stress (Naz et al. 2010a, 2010b), reduced growth, and particularly under higher salinities is beneficial for its survival (Table 2), because successful survival is more important than their vigorous growth (Hameed and Ashraf 2008).
Increasing salinity gradient of the habitats imparted specific morphological and anatomical changes, which indicates adaptability potential of this grass to cope with environmental hazards in the Cholistan desert (Hameed et al. 2013). Increasing salinity of the habitats hampered root growth in this species (Table 2), which is a characteristic feature of most plant species that have relatively less tolerance against salinity stress. A number of similar observations have earlier been reported by many researchers (Monteverdi et al. 2008; Munns and Tester 2008). Moderate salinities at Trawaywala Toba (TT) were more suitable for leaf development, as both flag leaf area and total leaf area per plant were the maximum in plants from this site. However, higher salinities had a negative impact on leaf area, as well as overall growth in L. scindicus. Alem et al. (2002) reported a similar decrease in leaf area in durum wheat in response to increased salinity.
As it is expected, concentration of toxic ions like Na+ and Cl− in roots and shoots increased as the salinity level of the habitat increased (Table 2), but at the same time, increased concentration of beneficial ions, in particular, that of K+ and Ca2+, may certainly contribute to high degree of salt tolerance in this species. Ashraf (2004) also reported high K+:Na+ and Ca2+:Na+ ration in salt tolerant grasses. At the same time, there was no visible effect of increasing salinity gradient on uptake of Mg2+, but the concentration significantly decreased. Qian et al. (2001) in Kentucky bluegrass and Grieve et al. (2004) in wheatgrass also reported similar findings.
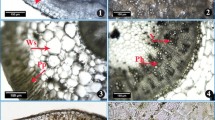
At the root level, L. sindicus showed some specific structural modifications like sclerification in cortical and pith region, which may enhance mechanical strength of root, critical for preventing root damage under harsh climates (Fig. 2). Root of L. scondicus are exposed to extreme unfavourable environmental conditions in the Cholistan Desert, where the soils is compact saline soil or saline-sodic. Parenchymatous tissues, in particular cortex and epidermis disintegrated as a result in three ecotypes from higher salinities. Increased endodermal thickness may control radial movement of water in roots (Table 3), and increased number and size of xylem vessels is vital for efficient conduction and translocation of water and other nutrients, and it may again contribute to its survival under harsh environments (Rashid and Ahmed 2011). The major impact of salts on roots was the disintegration of epidermis and outer cortical region, particularly at higher salinities. The survival of this species in such circumstances may depend on increased sclerification and endodermal thickness, which is critical for minimizing water loss through roots (Ahmad 1996).
Anatomical studies of Lasiurus scindicus ecotypeas from the Cholistan Desert. Root transverse sections. a Derawar Fort, b Trawaywala Toba, c Bailahwala Dahar, d Ladam Sir, and e Pati Sir, stem, f Derawar Fort, g Trawaywala Toba, h Bailahwala Dahar, i Ladam Sir, and j Pati Sir, and leaf, k Derawar Fort, l Trawaywala Toba, m Bailahwala Dahar, n Ladam Sir, and o Pati Sir (c cortex, en endodermis, ep epidermis, l leaf lamina, m leaf midrib, mv metaxylem vessels, p pith, sc sclerenchyma, vb vascular bundle)
At stem level, increased sclerification was one of the most prominent features in L. scindicus ecotypes (Fig. 2). Moreover, a significant increase in stem area, cortical region thickness and vascular bundle area along a salinity gradient may increase its potential for water conservation in terms of water loss from plant surface and increased storage ability (Table 3), both vital for the survival under limited moisture availability caused by osmotic stress. Increased sclerification in stem also been observed by several authors in the stem of different plant species, such as Aeluropus lagopoides (Hameed et al. 2013) and Prosopis strombulifera (Reinoso et al. 2004).
At leaf level, significant structural modifications in L. sindicus ecotypes included increased leaf thickness, epidermal thickness, sclerenchymatous area, cortical area, metaxylem area and bulliform cell area (Fig. 2). All these may contribute to water conservation, which ultimately accounts for species survival under physiological droughts like salinity (Balsamo et al. 2006). Another distinctive feature is the reduction in stomatal density and area (Table 3), which can significantly control transpirational water loss from leaf surface (Fig. 3). This was further supported by increased trichome density on leaf margins (Abdel and Al-Rawi 2011).
Lasiurus scindicus is a salt excretory grass, which is capable of excreting excess of toxic ions. It can also accumulate toxic ions such as Na+ and Cl− in the shoot tissue to some extent (Gulzar et al. 2003; Arndt et al. 2004). At higher salinities, mechanism of salinity tolerance seemed to be shifted towards excretion, rather than dumping off the toxic ions in cell organelles and tissues, as also reported by Naz et al. (2009). The predominant ions secreted by plants are generally Na+ and Cl− with K+, Ca2+ and Mg2+ in minor quantities (Munns 2002), as was also recprded in L. scindicus ecotypes. However, for Mg2+ and K+ ions, excretion patterns at the studied sites were inconsistent. A decrease in Cl− content under higher levels of salinity indicates the involvement of some other mechanisms to prevent the plant from excreting K+ that hampers its growth under higher levels of salinity. A sharp increase in root and shoot K+ and root Ca2+ seems to be involved in its survival under saline conditions.
The Na+ is used in the metabolic processes in a number of halophytic species (Munns and Tester 2008). Lasiurus scindicus not only excreted excessive ions, but also accumulated them in the leaves independent of their habitats (Table 3). Retention of the physiologically advantageous ions like Ca2+ and K+ in shoot is certainly useful for osmotic balance regulation, and this is critically important for the successful survival (Ashraf 2004).
Anatomical adaptations investigated in this grass critically support the physiological adaptations confirming its successful survival in extreme desert saline conditions. The highly saline LS population, showed increased sclerification in the vascular region, inside endodermis and in outer cortical region. Moreover, intensive sclerification was recorded in stem and midrib region of the leaf and leaf sheath. Highly developed bulliform cells were prominent in the leaf lamina. These modifications may give rigidity to root tissues and also significantly reduce water loss through plant surfaces (Alvarez et al. 2008). Salt accumulation is the prominent feature of this species to cope with high salinities.
References
Abdel CG, Al-Rawi IMT (2011) Anatomical alteration in response to irrigation and water stress in some legume crops. Am J Exp Agric 1:231–264
Ahmad T (1996) Eucalyptus in Pakistan. FAO Corporate Document Repository. (http://www.fao.org/)
Alem C, Labhilili M, Brahmi K, Jlibene M, Nasrallah N, Maltouf AF (2002) Hydrous and photosynthetic adaptations of common and durum wheat to saline stress. Comp Ren Biol 325:1097–1109
Alvarez JM, Rocha JF, Machado SR (2008) Bulliform cells in Loudetiopsis chrysothrix (Nees) Conert and Tristachya leiostachya Nees (Poaceae): structure in relation to function. Braz Arch Biol Technol 51:113–119
Arndt SK, Arampatsis C, Foetzki A, Li X, Zeng F, Zhang X (2004) Contrasting patterns of leaf solute accumulation and salt adaptation in four phreatophytic desert plants in a hyperarid desert with saline groundwater. J Arid Environ 59:259–270
Arshad M, Hussan AU, Ashraf MY, Noureen S, Moazzam M (2008) Edaphic factors and distribution of vegetation in the Cholistan Desert, Pakistan. Pak J Bot 40:1923–1931
Ashraf M (2004) Some important physiological selection criteria for salt tolerance in plants. Flora 199:361–376
Ashraf M, Ozturk M, Athar HR (2009) Salinity and water stress: improving crop efficiency, series: tasks for vegetation science, vol 44. Springer Verlag, New York, p 245
Ashraf M, Ozturk M, Ahmad MSA, Aksoy A (2012) Crop production for agricultural improvement. Springer Verlag, New York, p 796
Assaeed AM (1997) Estimation of biomass and utilization of three perennial range grasses in Saudi Arabia. J Arid Environ 36:103–111
Atkinson NJ, Urwin PE (2012) The interaction of plant biotic and abiotic stresses: from genes to the field. J Exp Bot 63:3523–3543
Balsamo RA, Willigen CV, Bauer AM, Farrant J (2006) Drought tolerance of selected Eragrostis species correlates with leaf tensile properties. Ann Bot 97:985–991
Chen Z, Pottosin II, Cuin TA, Fuglsang AT, Tester M, Jha D, Zepeda-Jazo I, Zhou M, Palmgren MG, Newman IA, Shabala S (2007) Root plasma membrane transporters controlling K+/Na+ homeostasis in salt-stressed barley. Plant Physiol 145:1714–1725
Cope TA (1982) Poaceae. In: Nasir E, Ali SI (eds) Flora of West Pakistan, 143rd edn. University of Karachi, Karachi, pp 1–678
Cramer GR, Urano K, Delrot S, Pezzotti M, Shinozaki K (2011) Effects of abiotic stress on plants: a systems biology perspective. BMC Plant Biol 11:163
Cuin TA, Shabala S (2007) Compatible solutes reduce ROS-induced potassium efflux in Arabidopsis roots. Plant Cell Environ 30:875–885
Demmig-Adams B, Dumlao MR, Herzenach MK, Adams WW (2008) Acclimation. Elsevier, Colorado
El-Keblawy A, Ksiksi T, El Alqamy H (2009) Camel grazing affects species diversity and community structure in the deserts of the UAE. J Arid Environ 73:347–354
Grieve CM, Poss JA, Grattan SR, Suarez DL, Benes SE, Robinson PH (2004) Evaluation of salt-tolerant forages for sequential water reuse systems, II: plant-ion relations. Agric Water Manag 70:121–135
Gulzar S, Khan MA, Ungar IA (2003) Effects of salinity on growth, ionic content, and plant-water status of Aeluropus lagopoides. Commun Soil Sci Plant Anal 34:1657–1668
Hakeem KR, Parvaiz A, Ozturk M (2013) Crop improvement-new approaches and modern techniques, vol XXVII. Springer Verlag, New York, p 494
Hameed M, Ashraf M (2008) Physiological and biochemical adaptations of Cynodon dactylon (L.) Pers. from the Salt range (Pakistan) to salinity stress. Flora 203:683–694
Hameed M, Ashraf M, Al–Quriany F, Nawaz T, Ahmad MSA, Younis A, Naz N (2011) Medicinal flora of the Cholistan desert: a review. Pak J Bot 43:39–50
Hameed M, Naz N, Nawaz T, Batool R, Ashraf M, Ahmad F, Ruby T (2013) Structural adaptations in a desert halophyte Aeluropus lagopoides (Linn.) Trin. ex Thw. under high salinities. J Biol Res-Thessalon 19:150–164
Jouyban Z (2012) Ethylene biosynthesis. Tech J Eng Appl Sci 1:100–107
Khan MF, Anderson DM, Nutkani MI, Butt NM (1999) Preliminary results from reseeding degraded Dera Ghazi Khan rangeland to improve small ruminant production in Pakistan. Small Rumin Res 32:43–49
Lobell DB, Schlenker WS, Osta-Roberts J (2011) Climate trends and global crop production since 1980. Sci 333:616–620
Maqsood MA, Hussain S, Aziz T, Ashraf M (2013) Sustainable agriculture through integrated soil fertility management on degraded lands. Development in soil salinity assessment and reclamation. Springer, New York, pp 759–768
Mittler R, Blumwald E (2010) Genetic engineering for modern agriculture: challenges and perspectives. Ann Rev Plant Biol 61:443–462
Monteverdi CM, Lauteri M, Valentini R (2008) Biodiversity of plant species and adaptation to drought and salt conditions. Selection of species for sustainable reforestation activity to combat desertification. In: Abdelly C, Öztürk M, Ashraf M, Grignon C (eds) Biosaline agriculture and high salinity tolerance. Birkhaüser Verlag, Switzerland, pp 197–206
Munns R (2002) Comparative physiology of salt and water stress. Plant Cell Environ 25:239–250
Munns R, Tester M (2008) Mechanisms of salinity tolerance. Ann Rev Plant Biol 59:651–681
Naz N, Hameed M, Wahid A, Arshad M, Ahmad MSA (2009) Patterns of ion excretion and survival in two stoloniferous arid zone grasses. Physiol Plant 135:185–195
Naz N, Hameed M, Ashraf M (2010a) Eco-morphic response to salt stress in two halophytic grasses from the Cholistan desert, Pakistan. Pak J Bot 42:1343–1351
Naz N, Hameed M, Ashraf M, Al-qurainy F, Arshad M (2010b) Relationships between gas-exchange characteristics and stomatal structural modifications in some desert grasses under high salinity. Photosynthetica 3:446–456
Newton RJ, Jones SE, Eiler A, McMahon KD, Bertilsson S (2011) A guide to the natural history of freshwater lake bacteria. Microbiol Mol Biol Rev 75:14–49
Öztürk M, Waisel Y, Khan MA, Gork G (2006) Biosaline agriculture and salinity tolerance in plants. Birkhauser Verlag, Basel, p 205
Qian YL, Wilhelm SJ, Marcum KB (2001) Comparative responses of two Kentucky bluegrass cultivars to salinity stress. Crop Sci 41:1895–1900
Rashid P, Ahmed A (2011) Anatomical adaptations of Myriostachya wightiana Hook. F. to salt stress. J Biol Sci 20:205–208
Reinoso H, Sosa L, Ramirez L (2004) Salt-induced changes in the vegetative anatomy of Prosopis strombulifera (Leguminosae). Can J Bot 82:618–628
Wang H, Miyazaki S, Kawai K, Deyholos M, Galbraith DW, Bohnert HJ (2003) Temporal progression of gene expression responses to salt shock in maize roots. Plant Mol Biol 52:873–891
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Naz, N., Batool, R., Fatima, S. et al. Adaptive components of tolerance to salinity in a saline desert grass Lasiurus scindicus Henrard. Ecol Res 30, 429–438 (2015). https://doi.org/10.1007/s11284-014-1236-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11284-014-1236-0