Abstract
Mangrove forests in meso-tidal areas are completely drained during low tides, forming only temporary habitats for fish. We hypothesised that in such temporary habitats, where stranding risks are high, distance from tidal creeks that provided access to inundated areas during receding tides would be the primary determinant of fish distribution. Factors such as depth, root density and shade were hypothesised to have secondary effects. We tested these hypotheses in a tidally drained mangrove patch in the Andaman Islands, India. Using stake nets, we measured fish abundance and species richness relative to distance from creeks, root density/m2, shade, water depth and size (total length) of fish. We also predicted that larger fish (including potential predators) would be closer to creeks, as they faced a greater chance of mortality if stranded. Thus we conducted tethering trials to examine if predation would be greater close to the creeks. Generalised linear mixed effects models showed that fish abundance was negatively influenced by increasing creek distance interacting with fish size and positively influenced by depth. Quantile regression analysis showed that species richness was limited by increasing creek distance. Proportion of predation was greatest close to the creeks (0–25 m) and declined with increasing distance. Abundance was also low very close to the creeks, suggesting that close to the creeks predation pressure may be an important determinant of fish abundance. The overall pattern however indicates that access to permanently inundated areas, may be an important determinant of fish distribution in tidally drained mangrove forests.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Shallow water ecosystems such as mangrove forests and sea grass meadows are widely considered to be nurseries for juvenile fish, several of whose adults inhabit different habitats (Beck et al. 2001; Kathiresan and Bingham 2001; Nagelkerken et al. 2008). Studies propose two hypotheses, abundant food resources and availability of shelter from predators, to explain the value of these ecosystems to juvenile fish (Adams et al. 2006; Nagelkerken 2009a). While the two are not mutually exclusive (Manson et al. 2005; Nagelkerken 2009b) there has traditionally been greater focus on the shelter value of these habitats. Shelter in mangrove forests is thought to come mainly from the structural complexity of the system (Manson et al. 2005). The extensive root systems of mangrove plants such as prop roots, stilt roots and pneumatophores as well as fallen debris are reported to offer refuge for juveniles and hinder movement of large adult piscivores (Laegdsgaard and Johnson 2001; Verweij et al. 2006; Nagelkerken and Faunce 2008). Shade and shallow depth typical of mangrove forests are also considered to provide increased cover and deter larger predatory fishes from entering these habitats, leading to increased juvenile fish abundance (Cocheret de la Morinière et al. 2004; Ellis and Bell 2004; Verweij et al. 2006).
However, most studies that have found a relationship between fish abundance and species richness, with structural complexity, shade and depth come from mangrove forests in micro-tidal environments (tidal range <1 m), such as the Caribbean (Verweij et al. 2006; Nagelkerken 2007; Nagelkerken and Faunce 2008; Kimirei et al. 2011). For instance, in the Curacao Island, which has a mean tidal range of 0.3 m, presence of root structure was found to have a positive relationship with fish abundance (Verweij et al. 2006; Nagelkerken and Faunce 2008). Similarly shade relative to depth was found to positively correlate with fish abundance in Tampa Bay, Florida, which has a mean tidal range of 0.7 m (Ellis and Bell 2004). In such systems, there is little fluctuation in water levels through a tide period, resulting in mangrove forests that can serve as permanently inundated habitats.
However in several regions of the world, such as the Indo-Pacific. mangrove forests also occur in meso-tidal and macro tidal environments and experience tidal variations between two and nine metres (Sheaves 2005; Nagelkerken 2007). In many such areas mangrove forests are often inundated during high tides and completely drained during low tides. Such mangroves form only temporary habitats for fish which have to return to permanently inundated areas, such as main channels or reefs during low tides (Vance et al. 1996; Sheaves 2005). Sampling efforts are often concentrated in those parts of the system that are inundated throughout the tide period, such as the estuary channels (Robertson and Duke 1987; Chong et al. 1990; Blaber et al. 1995). Studies focussing on vegetated parts of tidally drained mangrove systems, have found less evidence of root structure, shade or depth positively correlating with fish abundance or species richness (Mullin 1995; Wang et al. 2009). In fact, Wang et al. compared three habitat types—creeks, mudflats and vegetated areas—within the Dongzhaigang Bay mangrove estuary in China, and found lower fish abundances in vegetated areas (Wang et al. 2009).
Factors such as structure and shade may become important determinants of fish distribution due to biotic pressures such as predation (Laegdsgaard and Johnson 2001; Nagelkerken and Faunce 2007; Nagelkerken and Faunce 2008). However the risk of predation is limited to a few species of fish, i.e., potential prey species. On the other hand, all fish entering temporary habitats such as tidally drained mangrove forests face the risk of being stranded during low tides, unless they have special morphological adaptations that help them survive (e.g., mudskippers).
Thus, we hypothesised that, within tidally drained mangrove forests, ease of access to permanently inundated areas would determine fish distribution (i.e., abundance and species richness). We tested this hypothesis in a tidally drained mangrove patch in the Andaman and Nicobar Islands of India, for day time hours during the dry season. As tidal creeks within the mangrove patch were the first to be inundated during the high tides and last to be drained during the low tides, we assumed that the creeks were the main access routes for fish to travel in and out of the mangrove patch. Tidal creeks were similarly found to be the main entry and exit points for fish entering the (Meynecke et al. 2008) Burram River estuary, Australia, a mangrove forest with a similar tidal range (1–3 m). We thus chose distance from two creeks within the mangrove patch (that connected this habitat to the adjacent sea), as a proxy for access to permanently inundated areas.
We further investigated the effect of three other factors (considered important in micro-tidal mangrove systems), (1) complexity of root structures (measured as density of roots/m2), (2) shade from tree canopy and (3) water depth on fish abundance and species richness in this tidally drained mangrove patch. Following published hypotheses (Laegdsgaard and Johnson 2001; Manson et al. 2005; Nagelkerken et al. 2008), we expected increase in root density and shade to positively correlate with abundance and species richness. We expected increase in water depth to negatively correlate with abundance and richness as past studies suggest that increasing water levels lead to greater predation (Ellis and Bell 2004). Of note here is that water depth in the study site was not correlated with increasing distance to the creeks, because of the topography of the landscape. For instance some areas close to the creeks were shallow because of the increased height of the ground (due to sediment deposit).
Although our predictions were built on the premise that access to permanently inundated areas would be the most important factor for fish entering tidally drained mangroves, predation pressure could not be ignored. Predation in such shallow water ecosystems may arise from both adults as well as piscivorous juveniles (Baker and Sheaves 2005). Piscivorous juveniles could also be a major determinant of fish distribution in mangrove forests (Baker and Sheaves 2005; Baker and Sheaves 2009; Dorenbosch et al. 2009). It is thus important to understand how predation may interact with need for access in shaping fish distribution across a tidally drained mangrove forest.
Our preliminary field observations indicated that with an increase in size (total length), fish faced a greater chance of mortality if stranded during low tides. Smaller fish could survive in tide pools until the next high tide. Thus we expected proportion of predation to be higher close to the creeks (where larger predators may be found) and decline with increasing distance from the creeks.
Materials and methods
Study area
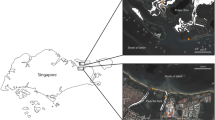
This study was conducted during the dry season, from February 2012 to April 2012, in the Andaman and Nicobar archipelago, situated in the Bay of Bengal (6°–14°N and 91°–94°E). The study was conducted in a riverine mangrove forest of approximately 36 ha in the Lohabarak Crocodile Sanctuary, South Andaman Island. Littoral forests adjoin the mangroves on the landward side, while coral reefs form adjacent habitats on the seaward side (Fig. 1).
Map of the study site showing, the study site, the Lohabarak Crocodile Sanctuary in reference to Andaman Islands and India. Study site is circled in the South Andaman Island. On the right the study site in detail with a permanently inundated areas or the sea, b and c show mangrove forest area of the Lohabarak sanctuary, with c showing sampled areas and d indicating the tidal creeks. Littoral forests and human habitation form the white portions of the Lohabarak map. Map was digitized from a Google Earth layer using graphic design software Adobe Photoshop
Water movement within the patch is a consequence of semi diurnal tides, ranging from 2 to 3 m. Water enters the forest patch through two creeks (400–500 m length), which are inundated during the high tide and are completely dry during low tide.
Salinity in the study site ranged from 30 to 35 ppt during the study period. Plant composition in the mangrove patch is dominated by Rhizophora apiculata, along with patchy distribution of Avicennia officinalis, Bruguiera gymnorrhiza and Lumnitzera racemosa. Sampling was restricted to daylight hours, between 0530 and 1730 h. Using the geo-referencing software Google Earth we divided the study area into 500 circular plots measuring 7 m in diameter. Sampling was done in a subset of these 500 plots (N = 110), covering 22 % of the study area. As we had no prior information about the range of depths, root density or canopy cover occurring within the study area, the plots were chosen randomly using the statistical software R ver. 3.0.1. Plots lying at the creek mouths were eliminated, due to reported presence of saltwater crocodiles in the area, and plots (very close to the landward edge) which were only inundated during spring tides.
Within each plot we measured the distance from the centre of the plot to the nearest creek using Google Earth, water depth at high tide using a calibrated rod, and percentage canopy cover (as a proxy for shade) using a canopy densiometer. To estimate root density per plot, we photographed seven randomly placed non-overlapping 1 m quadrats within each plot from a fixed height of 1.5 m and counted the total number of individual roots in each of these photographs. This was averaged across the total plot to arrive at density of roots per m2.
Fish sampling
We sampled fish communities in each plot, with passive stake nets (of 1 mm mesh size) modified from Mullin (1995) and calculated abundance, species richness and recorded composition of fish later. The nets were set during low tide covering a circular plot of 38.5 m2 area. Two opposing sides, 2 m in width, were left open facing the direction of the incoming tide, such that fish could enter and exit the area during the rising tide. During the slack period (just before the beginning of the ebb tide), when water levels were highest and the tide ceased to rise, nets were closed to trap the fish within the plot. We chose the slack period to trap fish based on pilot studies–visual observation of fish movement near creeks and previous literature. For instance Meynecke et al. (2008), using PIT tags and underwater videos found that while different species of fish entered the mangrove patch at different times during the rising tide, most left at the beginning of the ebb tide. Thus by capturing fish at the slack period we may have a better reflection of where fish occur before they exit the mangroves.
To prevent the nets from rising up with water, the bottom was held down with fishing weights and fixed to the ground with iron pegs. During the subsequent low tide, we counted the stranded fish, measured their total lengths and photographed them for identification before releasing them. Although we did not sample at high tides in the night, fish stranded were always counted in the subsequent low tide, regardless of whether it was day or night. This was done in order to avoid underestimating abundance due to scavengers like hermit crabs.
Fish identification was done using published literature and the online database FishBase and photographs taken at the time of sampling. We identified adults and juveniles by comparing the total length of all individuals trapped with length at maturity and maximum length reported on FishBase. Fish catch was classified into small (<=2 cm) and large (>2 cm) post hoc based on total length and distinct distribution patterns (mentioned in the results section) of these size classes.
Predation
We estimated the proportion of predation at different distances from the creeks, by tethering a cardinal fish species Apogon amboinenesis. The fish were tethered using 1 m long transparent monofilament line inserted through the bone above the nostrils; the other end of the string was attached to a lead fishing weight. All tethered fish measured 3–4 cm and were caught from creeks outside the protected area.
Each assay was conducted in five transects, set at six at distance classes of 0, 25, 50, 75, 100 and 150 m from the creek. The distance classes were chosen after examining initial scatterplots of fish abundance which showed that most fish seemed to occur within 150 m of the creeks. As fish abundance beyond this distance class appeared to be minimal we did not set up any transects beyond this distance class. At each transect, five tethered fish were placed 25 m apart. Each transect was repeated five times. In order to isolate the effects of creek distance we controlled for other factors, by choosing areas with constant levels of root density (35–40 roots/sq m), depth (50–60 cm) and canopy cover (90–100 %). Each assay (comprising one transect) lasted 30 min. We conducted 30 such assays and tethered 150 fish. After each assay, lines with no tethered fish were considered predation events. Fish that had survived were released at the point of catch.
Before the actual trials, we conducted pilot studies to identify if the tethered fish were susceptible to opportunistic predators such as mud crabs (Scyla serrata). No such predation event was observed during the pilot studies. This was potentially because tethered fish were observed to be highly mobile and even feeding during the trials. Scavengers such as hermit crabs fed on only dead fish that were placed on the ground, with feeding times consistently longer than 30 min and only part of the fish being eaten. No birds were observed feeding on tethered fish, potentially because all transects were conducted within dense canopy areas. We observed that all predation attempts by fish such as mangrove snappers resulted in the entire tethered fish being taken. Thus it can be safely assumed that predation events during the final experiment were by piscivorous fish and no other fauna.
All field work was done with permits provided by the Andaman and Nicobar Forest Department No. CWLW/WL/134/378.
Analyses
Fish abundance
To identify the factors influencing fish abundance, we used generalised linear mixed effects models (GLMM) with a Poisson distribution using the R package lme4. Distance from the creeks, root density, shade, depth and size (total length) were considered fixed effects for fish abundance. Random effects were individual plots (sampled on different days leading to variation in tidal fluctuation) and species identity (as biology of each species may cause some variation). We also examined the possibility of each plot having a random effect nested within each species. Several candidate models were removed by comparing Akaike Information Criterion (AIC) values (difference of 3 points considered significant) and significance values (α = 0.05) of estimated parameters. The best model was chosen from the top models by running Likelihood Ratio Tests, and comparing the p values. In order to ascertain which of the predictor variables had the greatest effect on abundance, we standardised the effect sizes. We did this by calculating the mean, median and maximum values of each predictor. We also plotted histograms of the random effects and the residuals of the best model, in order to see if the model met the assumption of normality of residuals imposed by GLMM. We then compared the influence of the predictor variables of the best model, by standardising the effect sizes.
Species richness
Models for species richness did not adhere to the assumption of normality. Preliminary scatterplots however indicated that species richness might be limited by some factors. We therefore used regression quantiles to look at the limits imposed by the distance from the creeks, depth, root density and canopy cover on species richness (Cade and Noon 2003).
Predation
We used a non-linear least squares (NLS) model to examine the relationship between distance from creek and predation. All analyses were done with the statistical software R 3.0.1.
Results
Fish abundance and species composition
We recorded 1108 individuals and 29 species of fish from 110 plots. Nine were reef or reef associated species, while the rest were mangrove or brackish water species. Juveniles constituted 61 % of the total fish sampled. All reef species and 14 of 20 mangrove species were represented by immature fish. Small fish (<= 2 cm) were most abundant comprising 97 % of total abundance. The Malabar rice fish Oryzias setnai and an unidentified juvenile Apogonidae species were the most abundant species (Table 1). Large fish (>2 cm) formed approximately 3 % of the total abundance. Distance from the sampled plots to the creeks, varied from 2 m to 362 m (mean = 90.4 m; SD ± 82), depth varied from 6 cm to110 cm (mean = 46.4 cm; SD ± 21.9) and root density varied from 0 to 82 per m2 (mean = 28.3 roots/m2; SD ± 18). Percentage canopy cover varied from 0 to 100 %, (mean = 81.5 %; SD ± 25.9).
Factors affecting fish abundance
The final model chosen based on AIC values, significant p values (≤0.05) of estimated parameters and Likelihood Ratio Tests, showed that (1) distance from the creeks interacting with log size (of the fish) and (2) depth affected abundance. Plot identity nested within species identity (as a random effect) explained much of the variation in the intercept (Table 2). While distance from the creeks and log fish size had a negative effect on abundance, depth showed a positive effect (Table 2). Standardised effect sizes showed that log (size) appeared to have the greatest effect on fish abundance followed by distance from the creeks and depth had the least effect (Table 3). Models which also included root density and canopy cover did not appear to accurately estimate slopes of all the fixed effects. Likelihood ratio tests also showed that these models were insignificant.
Species richness
Species richness only showed a relationship with distance from the creek. Quantile regression analysis showed that maximum species richness (tau = 0.95) per plot was limited strongly by increasing distance from the creeks (Intercept p = 0.000; slope p = 0.001) (Fig. 2).
Predation across a gradient of increasing distance from the creeks
There was strong inverse relationship between predation and distance from the creeks (Fig. 3). Proportion of predation was greatest within 25 m of the creek and decreased as distance from the creek increased. The model using a logistic equation was the best fit, with predation tending to 0 as distance from the creek increased (Table 3). Scatterplots of fish abundance within 150 m of the creeks showed that fish abundance was low between 0 and 25 m and increased beyond this distance class (Fig. 3).
Proportion of predation was greatest right next to the creek, and dropped with decreasing slope as distance from the creek increased beyond 25 m. The model was fitted by using a nonlinear least squares (NLS) method. The points were best fitted by the logistic equation \(y = {{e^{ - 0.018x} } \mathord{\left/ {\vphantom {{e^{ - 0.018x} } {\left( {1 + 0.52e^{ - 0.66x} } \right)}}} \right. \kern-0pt} {\left( {1 + 0.52e^{ - 0.66x} } \right)}}\) which tends to zero when distances from the creek become very large. Error bars are 95 % binomial confidence intervals. The figure has been superimposed with raw fish abundance data (solid black dots) for the same creek distances. This shows that proportion of abundance is very low within 25 m of the creeks and rise between 25 and 50 m and declines beyond 50 m. The y-axis is proportion of predation and fish abundance
Discussion
Importance of size of the fish
Our results show that fish abundance was influenced by distance from creeks interacting with log size, and depth. The final model explaining abundance shows that a small increase in fish size leads to a sharp decline in fish abundance. This is confirmed by our fish catch data, which shows that small fish (<=2 cm) formed 98 % of total abundance, while large fish (>2 cm) comprised only 2 %. Thus a small increase in fish size would lead to a steep drop in fish abundance. Although fish size was not considered in our original hypothesis these results show that size of the fish could play an important role in shaping fish population structure in mangrove forests. This is particularly interesting as most of the fish catch comprised small fish (<2 cm). Comparing these results to studies in other areas (Blaber and Milton 1990; Chong et al. 1990; Meynecke et al. 2008)indicates that mangrove forests in different regions may be dominated by fish communities of different sizes and habitat associations. The relationship between abundance and size also indicates that temporary mangrove habitats may not be suitable for fish beyond a certain size. Such an inference however is made with caution and can only be confirmed after studying more sites and understanding why fish use temporary mangrove habitats. A rigorous analysis with size as a response variable was however not possible with this dataset, due to the heavy bias towards numbers of small sized fish.
Access to permanently inundated areas and predation
According to the final model, increase in distance from the creeks also resulted in a decrease in fish abundance. Creeks within the study site formed the main routes for water to enter and the last areas from where water receded. Easy access to these creeks would therefore be very important for fish while entering and exiting mangroves. Seen in combination with the log size, the model indicates that there is a greater decline in the abundance of larger fish with a marginal increase in distance from the creeks. Similar size-based distribution patterns were reported by Vance et al. (1996), where the authors found fish with the largest mean lengths in sites closest to the creeks. The mean length of fish caught, declined in sites further away from the creeks.
As large fish were likely to include piscivores, we argued that predation pressure may be greater close to the creeks and may decline with increasing distance from the creeks. Our predation assays confirmed this. At a constant depth of 50–60 cm and constant root density, predation was highest close to the creeks (0–25 m) and declined sharply with increasing distance from the creek. Thus the risk of predation may be greatest close to the creeks leading to smaller fish being more abundant further away from creeks. Similar results were reported by Hammerschlag et al. (2010) in a seagrass-mangrove eco-tone in Biscayne Bay, Florida. The authors found using tethering trials that proportion of predation was highest closest to the mangrove-seagrass eco-tone, the transition zone from mangroves to seagrass meadows (Hammerschlag et al. 2010). Creeks within our study site, which showed the greatest proportion of predation, may be considered similar transition zones between mangrove forests and other habitats such as coral reefs.
The predation model showed a slight increase in proportion of predation between the 0 and 25 m transects. This may further indicate that areas close to the creeks, but within the actual mangrove forest could serve as active hunting zones for predators. It is possible then that large predators may only be using this small area close to the transition zone as hunting grounds. This leads to the question of how effects of predation on abundance may be separated from that of increasing distance from the creeks. A look at Fig. 3 indicates that predation pressure was greatest between 0 and 25 m from the creek while the corresponding graph for fish abundance indicates that overall abundance was very low between 0 and 25 m, and tending to zero at the distance corresponding to the peak in predation predicted by the NLS model. However, abundance rose beyond 25 m and peaks close to 50 m from the creek declining again at 150 m from the creek where we found predation to be the least. This pattern indicates that potential prey species have to make an effective trade-off between predation and stranding and thereby may be avoiding areas very close to the creeks where predation pressure might be highest, without venturing too far out into the mangrove forests where stranding risks are much higher. The decline in proportion of predation at 50, 75, 100 and 150 m indicates that these areas could serve as relatively ‘safe zones’ for smaller potential prey species. Interestingly however transects with the least amount of predation (100 and 150 m) also had very low abundance. This indicates that although predation may be an important biotic pressure shaping fish distribution close to the creek, the overall pattern of fish abundance was determined by access to permanently inundated areas. It might be important to further investigate the dynamics between predation risk and stranding risk in the future.
Comparison of effects of depth, fish size and distance from the creeks
Contrary to our hypothesis increase in depth led to an increase in abundance. Shallow depths are considered to be important for small sized fish to escape larger predators (Ellis and Bell 2004; Meynecke et al. 2008; Nagelkerken and Faunce 2008). In the absence of predation (in much of the mangrove patch), most fish regardless of their size may prefer relatively deeper areas in the mangrove patch. However, a comparison of the effects of all three factors (Table 2) shows that rise in depth had the least effect, resulting in a slight increase in fish abundance. While maximum increase in log size resulted in the greatest reduction in fish abundance, a minimum increase in size did not affect fish abundance. This may have been due to the dominance of small fish (<2 cm) in the mangrove patch. However even a minimum increase in distance from the creeks affected abundance (negatively) pointing to the larger overall impact of this factor on fish abundance.
Finally, species richness was only affected by distance from the creeks. Increasing distance from the creeks limited the maximum number of species that occurred in each plot lending further credence to our hypothesis that access to permanently inundated areas may be the primary determinant of fish distribution. Quantile regression analysis indicates that species richness while limited by distance from the creeks may be affected by other unmeasured factors. For most species, staying close to the creeks may allow fish to minimise time spent travelling during receding tides and maximise on time spent in activities such as feeding (Sheaves 2005). Moving further away from the creeks could increase the risk of mortality for most species. There may be considerable variation between different species and functional groups with regard to the effects of creek distance (Table 1). However the low abundance of most species caught, precludes any formal statistical analysis on the effects of distance from the creeks or other factors on individual species or functional groups, which have provided valuable information in the past (Johnston and Sheaves 2008).
Role of root structure and shade
Root density and canopy cover did not affect fish abundance and species richness. Structure in mangrove forests (such as prop roots or stilt roots) are reported to be important in several studies in micro-tidal areas (Verweij et al. 2006; Nagelkerken et al. 2008) particularly as predation refuges (Laegdsgaard and Johnson 2001; Johnson 2006). In contrast, our results were similar to studies in mangrove forests in meso-tidal regimes, where no effect of root structure was reported (Mullin 1995; Wang et al. 2009), except in Meynecke et al. (2008). These results indicate that the importance of structure and shade as predation refuge may be a significant determinant of fish distribution only in permanently inundated mangrove systems.
Conclusion
In conclusion, access to permanently inundated areas may be an important determinant of fish distribution in temporary habitats such as tidally drained mangrove forests. Other factors such as size of the fish and predation risk (for certain species) also affect the distribution of fish in the study site however, such effects seem to be secondary. The findings of the study are limited by the fact that it is restricted to a single site and does not factor in the effects of climate, diel cycling and within site tidal variation. Thus these results need to be treated with caution while generalising; although they provide valuable insight into the various factors that could affect fish distribution in tidally drained mangrove ecosystems. Studies from more sites across the world which incorporate more spatial and temporal variation, will lead to a more complete understanding of fish distribution in tidally drained mangrove forests and add to the nursery paradigm.
References
Adams AJ, Dahlgren CP, Kellison TG et al (2006) Nursery function of tropical back-reef systems. Mar Ecol Prog Ser 318:287–301. doi:10.3354/meps318287
Antony G, George JP, Mathew A et al (2005) Ichthyofauna of the mangrove ecosystem. Mangrove ecosystems: a manual for the assessment of biodiversity. CMFRI, Chennai, pp 83–116
Baker R, Sheaves M (2005) Redefining the piscivore assemblage of shallow estuarine nursery habitats. Mar Ecol Prog Ser 291:197–213
Baker R, Sheaves M (2009) Overlooked small and juvenile piscivores dominate shallow-water estuarine “refuges” in tropical Australia. Estuar Coast Shelf Sci 85:618–626. doi:10.1016/j.ecss.2009.10.006
Beck MW, Heck KL, Able KW et al (2001) The identification, conservation, and management of estuarine and marine nurseries for fish and invertebrates. Bioscience 51:633–641. doi:10.1641/0006-3568(2001)051[0633:TICAMO]2.0.CO;2
Blaber SJM, Milton DA (1990) Species composition, community structure and zoogeography of fishes of mangrove estuaries in the Solomon Islands. Mar Biol 105:259–267. doi:10.1007/bf01344295
Blaber SJM, Brewer DT, Salini JP (1995) Fish communities and the nursery role of the shallow inshore waters of a tropical bay in the gulf of Carpentaria, Australia. Estuar Coast Shelf Sci 40:177–193. doi:10.1016/s0272-7714(05)80004-6
Cade BS, Noon BR (2003) A gentle introduction to quantile regression for ecologists. Front Ecol Environ 1:412–420. doi:10.1890/1540-9295(2003)001[0412:AGITQR]2.0.CO;2
Chong VC, Sasekumar A, Leh MUC, D’Cruz R (1990) The fish and prawn communities of a Malaysian coastal mangrove system, with comparisons to adjacent mud flats and inshore waters. Estuar Coast Shelf Sci 31:703–722. doi:10.1016/0272-7714(90)90021-i
Cocheret de la Morinière E, Nagelkerken I, Meij H, Velde G (2004) What attracts juvenile coral reef fish to mangroves: habitat complexity or shade? Mar Biol 144:139–145. doi:10.1007/s00227-003-1167-8
Dorenbosch M, Grol MGG, de Groene A et al (2009) Piscivore assemblages and predation pressure affect relative safety of some back-reef habitats forjuvenile fish in a Caribbean bay. Mar Ecol Prog Ser 379:181–196
Ellis WL, Bell SS (2004) conditional use of mangrove habitats by fishes: depth as a cue to avoid predators. Estuaries 27:966–976
Froese R, Pauly D (eds) (2014) FishBase. World Wide Web electronic publication. http://www.fishbase.org (2011)
Hammerschlag N, Morgan AB, Serafy JE (2010) Relative predation risk for fishes along a subtropical mangrove–seagrass ecotone. Mar Ecol Prog Ser 401:259–267
Johnson DW (2006) Predation, habitat complexity and variation in density-dependent mortality of temperate reef fishes. Ecology 87:1179–1188. doi:10.1890/0012-9658(2006)87[1179:PHCAVI]2.0.CO;2
Johnston R, Sheaves M (2008) Cross-channel distribution of small fish in tropical and subtropical coastal wetlands is trophic-, taxonomic-, and wetland depth-dependent. Mar Ecol Prog Ser 357:255–270
Kathiresan K, Bingham BL (2001) Biology of mangroves and mangrove Ecosystems. Adv Mar Biol 40:81–251
Kimirei IA, Nagelkerken I, Griffioen B et al (2011) Ontogenetic habitat use by mangrove/seagrass-associated coral reef fishes shows flexibility in time and space. Estuar Coast Shelf Sci 92:47–58. doi:10.1016/j.ecss.2010.12.016
Laegdsgaard P, Johnson C (2001) Why do juvenile fish utilise mangrove habitats? J Exp Mar Bio Ecol 257:229–253. doi:10.1016/s0022-0981(00)00331-2
Manson FJ, Loneragan NR, Skilleter GA, Phinn SR (2005) An evaluation of the evidence for linkages between mangroves and fisheries : a synthesis of the literature and identification of research directions. In: Gibson RN, Atkinson RJ, Gordon JDM (eds) Oceanography and marine biology: an annual review. University College London Press, London, pp 485–515
Meynecke JO, Poole GC, Werry J, Lee SY (2008) Use of PIT tag and underwater video recording in assessing estuarine fish movement in a high intertidal mangrove and salt marsh creek. Estuar Coast Shelf Sci 79:168–178. doi:10.1016/j.ecss.2008.03.019
Mullin S (1995) Estuarine fish populations among red mangrove prop roots of small overwash Islands. Wetlands 15:324–329. doi:10.1007/bf03160886
Nagelkerken I (2007) Are non-estuarine mangroves connected to coral reefs through fish migration? Bull Mar Sci 80:595–607
Nagelkerken I (2009a) Evaluation of nursery function of mangroves and seagrass beds for tropical decapods and reef fishes: patterns and underlying mechanisms. In: Nagelkerken N (ed) Ecological connectivity among tropical coastal ecosystems. Springer, Dordrecht, pp 357–400
Nagelkerken I (2009b) Ecological connectivity among tropical coastal ecosystems. Springer, Berlin
Nagelkerken I, Faunce CH (2007) Colonisation of artificial mangroves by reef fishes in a marine seascape. Estuar Coast Shelf Sci 75:417–422. doi:10.1016/j.ecss.2007.05.030
Nagelkerken I, Faunce CH (2008) What makes mangroves attractive to fish? Use of artificial units to test the influence of water depth, cross-shelf location, and presence of root structure. Estuar Coast Shelf Sci 79:559–565. doi:10.1016/j.ecss.2008.04.011
Nagelkerken I, Blaber SJM, Bouillon S et al (2008) The habitat function of mangroves for terrestrial and marine fauna: a review. Aquat Bot 89:155–185. doi:10.1016/j.aquabot.2007.12.007
Robertson A, Duke N (1987) Mangroves as nursery sites: comparisons of the abundance and species composition of fish and crustaceans in mangroves and other nearshore habitats in tropical Australia. Mar Biol 96:193–205. doi:10.1007/bf00427019
Sheaves M (2005) Nature and consequences of biological connectivity in mangroves systems. Mar Ecol Prog Ser 302:293–305
Vance DJ, Haywood MDE, Heales DS et al (1996) How far do prawns and fish move into mangroves? Distribution of juvenile banana prawns Penaeus merguiensis and fish in a tropical mangrove forest in northern Australia. Mar Ecol Prog Ser 131:115–124
Verweij MC, Nagelkerken I, De Graaf D et al (2006) Structure, food and shade attract juvenile coral reef fish to mangrove and seagrass habitats : a field experiment. Mar Ecol Prog Ser 306:257–268
Wang M, Huang Z, Shi F, Wang W (2009) Are vegetated areas of mangroves attractive to juvenile and small fish? The case of Dongzhaigang Bay, Hainan Island, China. Estuar Coast Shelf Sci 85:208–216. doi:10.1016/j.ecss.2009.08.022
Acknowledgments
The authors are grateful to the Department of Science and Technology for funding this study and the Forest Department, Andaman and Nicobar Islands for research permits and cooperation. We thank the Andaman and Nicobar Environmental Team, particularly Saw John, Tasneem Khan, Saw Stanley, Saw Isaac, Suresh and Rohan Chakravarthy. We also thank Tara Lawrence for help with fish identification and Suhel Quader and Ashwin Viswanathan for feedback on statistical analyses during this study. Ajith Kumar and Arjun Sivasundar for useful comments on the manuscript and Arjun Srivathsa for help with illustrations. We are grateful to the late K. S. Krishnan and Ajith Kumar for logistical and financial support.
Funding
This study was funded by the Department of Science and Technology, Government of India. Additional support was provided by the Post Graduate Programme in Wildlife Biology and Conservation run jointly by the National Centre for Biological Sciences and Wildlife Conservation Society-India Program. The second author was also supported by the Centre for Ecological Sciences, Indian Institute of Science.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sridharan, B., Namboothri, N. Factors affecting distribution of fish within a tidally drained mangrove forest in the Andaman and Nicobar Islands, India. Wetlands Ecol Manage 23, 909–920 (2015). https://doi.org/10.1007/s11273-015-9428-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11273-015-9428-0