Abstract
Allometric scaling of leaf nitrogen (N) and phosphorus (P) provides information on species adaptation to resource availability and nutrient limitation of primary production. Studies examining how plant functional traits vary spatially have historically used global datasets, which may blur variation at local scale and thus some of the key drivers. Global studies have observed leaf N–P scaling exponents varied across different functional groups, latitudinal zones, ecoregions, and sites. However, whether these trends are consistent in a local habitat is still unclear. Here, we capitalized on a soil P availability gradient in subtropical China to examine scaling exponents of leaf N–P. We investigated 21 woody species at P-rich and P-poor sites for two consecutive years and estimated scaling exponents of leaf N–P across P-type sites, leaf ages, plant functional types, and phylogeny. The exponents varied across the abovementioned levels from 0.356 to 0.699. Averaged across all plant species, the exponent of 0.612 was a departure from the global average of 2/3 (0.66) and 3/4 (0.75). Two types of P-sites did not affect N–P exponents of deciduous-broadleaved trees and evergreen conifers. Our findings indicated that leaf N–P scaling exponents significantly differed within plant functional types and highlighted the resource availability in shaping nutrient cycling and evolutionary processes at local scales.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Nitrogen (N) and phosphorus (P) are key macronutrients in plants supporting growth through both structural and energy-producing parts. Nitrogen is an important component of amino acids (the building blocks of proteins), chlorophyll, and the molecule responsible for photosynthesis. Phosphorus is a critical part of ATP, the energy-carrying molecule used to drive photosynthesis and plant growth (Elser et al. 1996; Güsewell 2004). These two elements are tightly linked and are important parameters in stoichiometric plant growth models (Agren 2004; Niklas et al. 2005). Plants invest disproportional N vs. P due to the difference in physiological growth strategies and varying soil environments, as observed at both local and global scales (Lambers et al. 2008; Townsend et al. 2007). Hence, there is a scaling relationship between leaf N and P concentration which is quantified via slope (α, scaling exponent) and intercept (β, coefficient) on a log–log scale, based on the function N = βPα (Kerkhoff et al. 2005; Reich et al. 2010). The “Growth Rate Hypothesis” (Sterner & Elser 2002) posited that higher proportional P in rRNA concentration in plants correlates to higher growth rates. Two earlier studies claimed that the scaling relationship of N vs. P concentration in plants follows a simple rule at global scales in that the scaling exponent converges around the value 0.66 (Wright et al. 2004) or 0.75 (Niklas et al. 2005). However, the underlying mechanisms behind these numeric laws are still not clear.
Evaluating the scaling relationship between N and P at the global scale may overwrite important variations at the local scale that may arise due to variations in nutrient availability or community composition (Kerkhoff et al. 2006; Tian et al. 2017). Moreover, it is difficult to ascribe a mechanism behind the scaling relationship at the global scale because there are too many unconstrained variables. Global analyses also ignore the role of time and the plasticity of species. Global datasets and short-term nutrient addition experiments are not able to resolve whether the evolutionary history of a plant community is the main driver of scaling relationships under various resource limitations. Therefore, case studies across various habitat and soil nutrient availability gradients are needed to explain the mechanisms behind the general exponent laws on a global scale.
In the following years after the general rules (0.66 and 0.75) were described, several studies developed and completed with larger datasets and quantified scaling factors as a narrower range (from 0.678 to 0.73). (Niklas 2006; Reich et al. 2010; Tian et al. 2017), but little effort has been directed toward determining the underlying mechanisms driving these observations. The acquisition and allocation of N and P in plants vary across geographical locations, soil nutrient status, major functional types, species, and family, and it is expected that scaling exponents of N and P will vary in tandem (Niklas and Cobb 2006; Zhang et al. 2012). For example, deciduous species have higher N and P concentrations than evergreen species to meet fast growth; conifers usually have lower N concentrations than broadleaved species. We expect that N and P scaling exponents will change between conifers and broadleaved species (evergreen and deciduous trees). Actually, a global analysis found conifers have the lowest N and N–P exponents, while no significant differences were found between deciduous and evergreen-broadleaved trees. However, one plant functional type may show a large difference in N and P concentrations in different ecosystems (e.g., temperate vs. tropical ecosystems). In this case, we expect to see a different role of plant functional types in shaping N–P exponents in our study area compared to a global study. Additionally, soil nutrient imbalance, particularly N and P, can also influence leaf N and P concentrations and thus N–P exponents. For instance, in a controlled nutrient addition experiment, Yan et al. (2018) found that the N–P scaling exponent decreased with the increasing levels of N addition, but increased with the increasing levels of P addition. Another community-level leaf N–P scaling exponent’s study showed that the scaling exponents declined with increasing growth rate, which highlighted the role of soil P availability. Here, we hypothesize that along with natural P gradient sites, leaf N–P scaling exponents may increase with soil P concentration and scaling exponents may vary significantly within plant functional types.
Other factors such as leaf age in evergreen trees and phylogeny may also play a role (Aerts 1995; Doust 1980). Generally, both N and P can be resorbed during leaf senescence and with similar magnitude (Vergutz et al. 2012). However, a few case studies found that P concentration decreases disproportionately compared to N across the foliage life span (Achat et al. 2018). Based on the previous work, we hypothesized the scaling exponent of N vs. P in the current-year leaves would be smaller than that of 2-year-old leaves. Different from plant functional groups, phylogeny (species and family) indicates growth strategies in another way. For example, Proteaceae in P-poor tropical ecosystems usually have higher phosphorus use efficiency than other families (Lambers et al. 2012). In this study, as a widely distributed dominant family, whether or not Fagaceae have a significantly different N–P exponent than other families due to local adaptation is intriguing. At the species level, N-P exponents may have large ranges due to large differences in N and P concentrations and growth strategies. Pooled data have advantages to illustrate how species influence N–P exponents, excluding environmental factors; here, we would like to examine within a limited dataset, how N–P exponents vary in species and how sampling numbers may influence the results.
Subtropical soils are generally depleted in “rock-derived” elements such as P, calcium (Ca), and magnesium (Mg) due to strong weathering and leaching as well as efficient retention of P in biota (Vitousek et al. 2010; Walker & Syers 1976). However, soils developed on phosphate rock are rich in P, N, and other important nutrients (Wen et al. 2018). Hence, plants in subtropical areas may present different stoichiometric traits based on adaptations to various nutrient habitats (Townsend et al. 2007). However, the importance of plant functional types and phylogeny vs. environmental factors in shaping exponents might reflect the influence of local habitats. The central hypotheses in this study are that (1) in natural P gradient sites in subtropics, leaf N–P scaling exponents may increase with soil P concentration and scaling exponents may vary significantly within plant functional types. (2) Because P concentration decreases disproportionately compared to N across the foliage life span, the scaling exponent of N vs. P in current-year leaves would be smaller than that of 2-year-old leaves.
Materials and methods
Site description
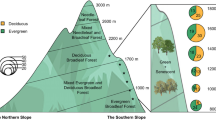
The study region located at Yunnan–Guizhou Plateau (about 2000 m a.s.l.) in Yunnan Province, southwestern China. In this region, the phosphate rock developed randomly within non-phosphate rocks (Fig. 1). The concentration of P2O5 in rocks at the P-rich site is about 0.89% at the P-rich site and 0.04% at the P-deficient site (Table S1). Ferralsols and acrisols are primary soil types in this area, according to World Reference Base for Soil Resources (Group 2014). The climates are similar at the contrasting P-sites (Fig. 1). The zonal vegetation consists of semi-humid evergreen-broadleaved forests and warm temperate conifer forests, including evergreen-broadleaved trees (EB), evergreen conifers (EC), and deciduous-broadleaved trees (DB).
Locations of sampling sites in Yunnan Province, southwestern China. Squares and dots represent phosphorus (P)-rich sites and P-deficient sites, respectively. Site name underlined denotes sampling in 2016 and 2017 while not underlined denotes sampling in 2016. The inserted picture showed monthly mean air temperature and monthly precipitation from 1981 to 2010 at P-rich sites (including Kunming city and Anning county) and P-deficient sites (including Chuxiong city, Mouding, and Lufeng county; China Meteorological Administration). The complete name list of each site is given in Supplementary Table S2. The map is drawn using QGIS version 2.18
Nineteen sites were selected within the study area, including nine on P-rich sites (Kunming) and ten on P-deficient sites (Chuxiong county) (Fig. 1). At each site, three 20 × 20-m plots were set up (Table S2). In this region, the Fagaceae family dominated evergreen and deciduous broadleaf, including Quercus variabilis, Q. acutissima, and Cyclobalanopsis glaucoides. Pinus yunnanensis and Keteleeria evelyniana are primary species in conifers.
Sampling
The leaf and soil samples were collected in August 2016 and 2017. Leaf samples were collected in the upper middle part of the south-facing crown of trees. Samples of full-expanded and medium-sized leaves were collected. For each species at each site, three to five samples were randomly collected (about 200 g per individual) from different healthy individuals and then mixed into one sample. For evergreen trees (EB and EC), we selected current-year leaves and 2-yr-old leaves as two separate samples. In total, we collected 495 leaf samples in two years, involving 21 species representing 8 families (Table S3). Four species were DB, 12 species were EB, and 5 were EC (Table S3). Additionally, nine topsoil samples (0–10 cm) and rock samples were collected per site (rock samples collected in nine sites) and mixed into one sample.
Chemical analyses
We stored and transported samples to the laboratory in coolers immediately after collection. Fresh leaves were dried at 105 °C for 30 min and 60 °C for 48 h (Markert 1994). We put soil and rock samples in a window open room to air dry for one month to constant weight. After that, we ground and sieved samples through a 0.25-mm screen for total element determination and by hand through a 0.2-mm screen for other chemical analysis.
The soil and leaf samples were digested with trace metal-grade nitric acid and diluted with deionized water (Dahlquist and Knoll 1978). Total N concentrations were determined with an elemental analysis-stable isotope ratio mass spectrometer (Vario ELIII; Elementar, Germany). Total and available P concentrations were determined with a plasma optical emission spectrometer (ICP-OES) (Iris Advantage 1000; Thermo Jarrell Ash, Franklin, MA). The contents of NH4+-N and NO3−-N were determined by Automated Discrete Analyzers (SmartChem 200; Alliance, France). Chemical compositions of rocks were determined with Sequential X-ray Fluorescence Spectrometer (XRF) (Ramsey et al., 1995). The concentrations of all elements were expressed as milligrams per gram of dry weight.
Data analyses
The scaling relationships between leaf N and P concentrations in this study were described by a mathematical equation of the type N = βPα, linearized under the form log (N) = log (β) + α log (P) (Niklas 1994). Here, we defined X and Y as leaf P and N concentrations, respectively. The term α—the slope of the scaling relationship, is the allometric coefficient or rate of divergence, while the term β is the intercept or elevation of the relationship. The value of β indicates the differences independent of leaf P concentration when two regression lines share the common slope.
We conducted T tests to examine the differences in nutrient concentrations of soils and rocks between P-rich and P- poor sites. In this paper, we log10 transformed the N and P concentration for all observations and then used reduced major axis (RMA) regression to determine the N vs. P scaling relationship (Warton et al. 2006). Confidence intervals for individual regression slopes followed (Pitman 1939). To answer whether the scaling component in subtropical China approaches 0.66 or 0.75 and whether regional differences are related to parent geology, plant functional types, phylogeny (species and family), and leafage, we used likelihood ratio tests to evaluate the heterogeneity of RMA regression exponents (Warton et al. 2006). Each exponent (α) was tested against the hypothesized value (general scaling exponent of leaf N vs. P concentration: 0.66 or 0.75 using the one-sample test. The calculation of RMA was conducted using “smatr” package (Warton et al. 2012). Additionally, a GLM was used to determine the relationship between soil, leaf traits, and the value of the scaling exponent. To determine whether leaf N and P concentrations and N:P ratios were affected by plant evolutionary history, we determined phylogenetic signals using Pagel’s λ (Pagel 1999). All the data analysis and drawing were conducted by R version 3.4 (R Development Core Team, 2018) and Datagraph 4.3 (Visual Data Tools, Inc, 2018, USA).
Results
Leaf N and P concentrations and N:P ratios at different levels
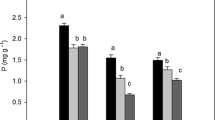
Concentrations of N and P in soils were higher at the P-rich sites than those at the P-poor soils (Table S4). Averaged across all plant species, leaf N and P concentrations were 14.08 and 1.12 mg g−1, respectively. However, at each level, significant differences were found in leaf N and P concentrations and N:P ratios (Table 1). For example, at soil P level, N and P concentrations are higher, while N:P mass ratio is lower at P-rich sites, compared to P-deficient sites. At Plant functional types level, DB had the highest N and P concentrations, while EC and EB had the lowest N and P concentration, respectively. When considering the effects of both plant functional types and soil types, EC or DB did not show significantly different N and P concentrations at P-rich and P-poor sites, while EB at P-poor sites had higher N and P concentrations than at P-poor sites (Table 1).
Scaling exponents of leaf N relative to P at different levels
Averaged across all the species, the scaling exponent was 0.612 (95% CI = 0.516, 0.661) and a statistically significant departure from 0.66 to 0.75 (p < 0.001, Table 2). The scaling exponents differed significantly among the three functional types. EB showed the highest exponent (0.599), while the DB showed the lowest (0.356). The scaling exponent of DB and EC significantly differed from 0.66 to 0.75 (p < 0.001, Table 2). There were no significant differences between Ericaceae (0.471), Pinaceae (0.471), and Fagaceae (0.511) (Table 2, Fig. 1), or between Current-yr. leaves (0.551) and 2-yr-old leaves (0.575) (Table 2), or between P-rich (0.699) and P-poor (0.607) sites. EC or DB did not show significantly different exponents at P-rich and P-poor sites, while EB at P-rich sites (0.281) had lower exponents than at P-poor sites (0.627) (Table 2).
Relationships between scaling exponents of leaf N relative to P and environmental factors
Soil nutrients, pH, and CEC did not significantly affect N–P scaling exponents in this study (Fig. 2). However, seemingly the trends of exponent variation along with soil factors contrast at DB and EC-dominated sites and other sites (Fig. 2). Leaf N and P concentrations affected exponent variations, particularly at functional types and species levels (Fig. 3). Wide leaf P and narrow leaf N concentration range led to lower N–P exponents of DB than the other two functional types, which may indicate different N or P accumulation of plants (Fig. 3). N–P scaling exponents significantly increased with the number of observations in this study indicating that the sampling number may greatly influence the estimation of leaf N–P scaling exponents (Fig. 3f). When the sample size was larger than 150, the N–P scaling exponents located between empirical values 0.66 and 0.75.
Relationships between N–P exponents and leaf N and P concentrations at a site level, b plant functional group level, c leaf age level, d family level, e species level, and f the relationship between N–P exponents and number of observations. Species were used to calculate exponents at species level when samples were more than 10. We only used current-year leaf samples to estimate N–P exponents for figures a, b, d, and e due to limited data for the 2-yr-old leaves. Scaling exponents (α) were calculated from the RMA regression between leaf N and leaf P concentrations and the lower and upper 95% confidence limits were presented in square brackets. All RMA regressions were significant (likelihood ratio tests, p < 0.05)
Discussion
Resource availability and composition effects at P-rich and P-poor sites
Our study examined the scaling exponents of leaf N concentration relative to P in subtropical China. We capitalized on a natural gradient in soil phosphorus concentrations to determine whether nutrient availability, phylogeny, and plant functional traits, influenced nutrient scaling relationships in plants. Our data partly rejected hypothesis 1 that leaf N–P scaling exponents may increase with soil P concentration, although we did see trends that the exponents increased with soil total and available P concentrations (Fig. 2). Although we hypothesized that leaf age influences N–P scaling exponents, we did not find significant differences between current-yr. leaves and 2-yr. leaves. This is consistent with other research between young-leaf and mature-leaf stages in Arabidopsis thaliana (Yan et al. 2018). We also did not find a statistically significant difference in exponents between P-rich and P-deficient sites or between phylogenetic clades.
The scaling exponent was 0.612 for all of the data combined and thus a statistical departure from the previous estimated values of 0.66 and 0.75 (Reich et al. 2010; Tian et al. 2017). The exponent for all the data pooled in our study area was also lower than the exponent in tropical (0.651) biomes (Reich et al. 2010). However, at the leaf age, family, and plant functional type levels, we found distinct differences in the exponents, with a significant departure from general values 0.66 or 0.75 (Table 2). This result highlights how differing plant compositions developed from different resources and nutrient use strategies. A higher exponent at P-rich sites may indicate that N has a higher uptake rate in plants as compared to P-deficient sites, suggesting that higher N and P concentrations at P-rich sites maintain nutrient-rich ecosystems to provide plants with more resources (Table S4). The insignificant relationship at P-sites probably resulted from the local adaptation of species in an unfavorable environment, which amplifies or dampens resource heterogeneity (Urban et al. 2020), thus altering the original nutrient cycling pattern.
Previous studies have shown that geologically derived P variation in soils influenced soil pH, which manifested as nutrient-rich and -poor habitats (Wen et al. 2018). We did not find significant relationships between N–P scaling exponents and soil P, which is different from the studies conducted on a global scale (Tian et al. 2017). Our results underscore that drivers may vary at local scales. Our previous studies have shown that variation in soil P concentration leads to variation in N content, mineral nutrient, and other functional traits (Ji et al. 2019, 2018; Wen et al. 2018). In our study, P-rich sites were developed from phosphate-rich bedrock and thus plants at this location have been exposed to higher P concentrations through their recent evolutionary history. In contrast, nitrogen concentration differences between sites are likely more recent due to elevated nitrogen deposition over the last ~ 150 years (Galloway et al. 2008). Long-term selection is able to develop morphological, physiological, or metabolic adaptations to the local environment, generating ecotypic differentiation in important functional traits (Savolainen et al. 2007). Phosphate-rich bedrock supported a local “favorable island” in P-poor subtropics and thus island plants adopted P-efficient strategies under local adaptation (Ji et al. 2019, 2018). This could lead to less sensitive variations in plant response to P compared to N availability. Additionally, the lack of a significant effect between the two sites reflected plant composition variance based on timing and spatial distribution of resource acquisition. Community and ecosystem stability can potentially change when species have different sensitivities to environmental factors (Tilman & Downing 1994). We also found that the number of observations significantly influenced exponents (Fig. 3f). We have to admit that the limited dataset in the current study may not be sufficient to provide strong support in N–P exponent’s research; more case studies with large sampling are needed in future. Overall, our results suggest that local nutrient-rich habitats, compared to surrounding P-poor sites, improved soil conditions and plant performance, altered plant composition, and ecosystem process.
Plant functional types matter most at a local habitat scale
In this local scale study, plant functional types was more important than phylogeny in influencing leaf N-P scaling exponents. Inconsistent exponents among the three plant functional types suggest diversified growth strategies (Ågren & Weih 2012; Ji et al. 2018; Wen et al. 2018). DB showed the lowest exponent (0.356) in our study, and this exponent was significantly less than the previous value reported for DB (0.712) (Tian et al. 2017). Previous studies reported that DB had lower exponents than EB (Tian et al. 2017) and conifers (McGroddy et al. 2004). This is potentially due to the rapid growth rates of DB, which results in a greater P demand (Elser et al. 2003). The wide leaf P concentration range also supported this suspicion (Fig. 2) and indicated that plant species with faster P accumulation or potentially susceptible to P limitation may have low leaf N–P scaling exponents (Guo et al., 2020).
Admittedly, plant functional types are not completely free of phylogenetic influences (Kerkhoff et al. 2006). Scaling exponents at the phylogeny level are more linked to the inheritance from previous ancestors than present-day environments (Duckworth et al. 2000). Varying scaling exponents at the phylogeny level may indicate evolutionary effects for plant nutrient acquisition and accumulation strategies (Kerkhoff et al. 2006; Sardans et al. 2015). However, our study found that the three families shared similar scaling exponents but had distant phylogenetic clades. This can be explained by there were no phylogenetic signals in leaf N and P concentrations (Table S5); that is to say, phylogeny doesn’t influence leaf N and P concentrations in the current study. The three families are dominant in this area and this suggests that within a local subtropical habitat, even across differing geologies, the dominant families adopted similar N and P acquisition strategies. In contrast, the plant functional type is a classification based on characteristics, such as life form (Raunkiaer 1934), function (deciduous or evergreen) (Dansereau 1951), leaf size, and shape (Dansereau 1951; Küchler and Zonneveld 2012). Evergreen trees are considered suitable in unfavorable conditions due to long leaf lifespans and low nutrient loss, while deciduous trees are reported to avoid environmental stresses through varied stratigies, such as greater nutrient resorption efficiencies, higher nitrogen contents, and rates of photosynthesis, but lower water usage (Aerts 1995; Sobrado 1991; Wen et al. 2018). Plant functional types are believed to represent how plants adaptated to varying environments and thus link plant physiology and ecosystem functions (Dı́az and Cabido 2001).
Implications for tropical ecosystems
Generally, the richest and largest phosphorite deposits form in tropical and subtropical areas in the world where clay minerals and hydroxides are the end products of weathering and aluminum phosphates are formed (Cook & McElhinny 1979; Cook & Shergold 1984; Sheldon 1964). Soils developed on phosphorite bedrock are P-rich due to geochemical and biochemical processes (Cross & Schlesinger 1995; Walker & Syers 1976). Previous experiments in subtropical areas revealed that P and P-related traits were altered at P-rich sites (Wen et al. 2018; Yan et al. 2015). In this study, the N–P scaling exponents were different from both a global dataset (Tian et al. 2017) and controlled nutrient addition experiments (Zhang et al. 2018). This suggests that phosphorite bedrock in subtropics/tropics create a unique habitat where potentially ecological functions are varying, including nutrient uptake and use strategies in species (Ji et al. 2018), plant metabolic regulation (Ji et al. 2019), and even faster biogeochemical cycling (unpublished data).
Supporting information
Additional Supporting Information may be found in the online version of this article.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Achat DL, Pousse N, Nicolas M, Augusto L (2018) Nutrient remobilization in tree foliage as affected by soil nutrients and leaf life span. Ecol Monogr 88(3):408–428
Aerts R (1995) The advantages of being evergreen. Trends Ecol Evol 10:402
Agren GI (2004) The C: N: P stoichiometry of autotrophs—theory and observations. Ecol Lett 7:185–191
Ågren GI, Weih M (2012) Plant stoichiometry at different scales: element concentration patterns reflect environment more than genotype. New Phytol 194:944–952
Chen D, Zhang X, Kang H, Sun X, Yin S, Du H, Yamanaka N, Gapare W, Wu HX, Liu C (2012) Phylogeography of Quercus variabilis based on chloroplast DNA sequence in East Asia: multiple glacial refugia and mainland-migrated island populations. PLoS One 7:e47268
Cook PJ, McElhinny M (1979) A reevaluation of the spatial and temporal distribution of sedimentary phosphate deposits in the light of plate tectonics. Econ Geol 74:315–330
Cook PJ, Shergold JH (1984) Phosphorus, phosphorites and skeletal evolution at the Precambrian—Cambrian boundary. Nature 308:231
Cross AF, Schlesinger WH (1995) A literature review and evaluation of the. Hedley fractionation: applications to the biogeochemical cycle of soil phosphorus in natural ecosystems. Geoderma 64:197–214
Dahlquist RL, Knoll JW (1978) Inductively coupled plasma-atomic emission spectrometry: analysis of biological materials and soils for major, trace, and ultra-trace elements. Appl Spectrosc 32:1–30
Dansereau P (1951) Description and recording of vegetation upon a structural basis. Ecology 32:172–229
Dı́az S, Cabido M (2001) Vive la différence: plant functional diversity matters to ecosystem processes. Trends in Ecology & Evolution 16:646–655
Doust JL (1980) A comparative study of life history and resource allocation in selected Umbelliferae. Biol J Lin Soc 13:139–154
Duckworth JC, Kent M, Ramsay PM (2000) Plant functional types: an alternative to taxonomic plant community description in biogeography? Prog Phys Geogr 24:515–542
Elser JJ, Dobberfuhl DR, Mackay NA, Schampel JH (1996) Organism size, life history, and N: P stoichiometry. Bioscience 46:674–684
Elser J, Acharya K, Kyle M, Cotner J, Makino W, Markow T, Watts T, Hobbie S, Fagan W, Schade J (2003) Growth rate–stoichiometry couplings in diverse biota. Ecol Lett 6:936–943
Galloway JN, Townsend AR, Erisman JW, Bekunda M, Cai Z, Freney JR, Martinelli LA, Seitzinger SP, Sutton MA (2008) Transformation of the nitrogen cycle: recent trends, questions, and potential solutions. Science 320:889–892
Group IW (2014) World reference base for soil resources 2014 international soil classification system for naming soils and creating legends for soil maps. FAO, Rome
Guo Y, Yan Z, Gheyret G, Zhou G, Xie Z, Tang Z (2020) The community-level scaling relationship between leaf nitrogen and phosphorus changes with plant growth, climate and nutrient limitation. J Ecol 108(4):1276–1286
Güsewell S (2004) N: P ratios in terrestrial plants: variation and functional significance. New Phytol, 243–266.
Ji H, Wen J, Du B, Sun N, Berg B, Liu C (2018) Comparison of the nutrient resorption stoichiometry of Quercus variabilis Blume growing in two sites contrasting in soil phosphorus content. Ann for Sci 75:59
Ji H, Ossipov V, Du B, Wen J, Liu C (2019) Differences in the relationship between metabolomic and ionomic traits of Quercus variabilis growing at contrasting geologic-phosphorus sites in subtropics. Plant Soil 439:1–17
Kerkhoff AJ, Enquist BJ, Elser JJ, Fagan WF (2005) Plant allometry, stoichiometry and the temperature-dependence of primary productivity. Glob Ecol Biogeogr 14:585–598
Kerkhoff AJ, Fagan WF, Elser JJ, Enquist BJ (2006) Phylogenetic and growth form variation in the scaling of nitrogen and phosphorus in the seed plants. Am Naturalist 168:E103–E122
Küchler AW, Zonneveld IS (2012) Vegetation mapping. Springer Science & Business Media
Lambers H, Raven JA, Shaver GR, Smith SE (2008) Plant nutrient-acquisition strategies change with soil age. Trends Ecol Evol 23:95–103
Lambers H, Cawthray GR, Giavalisco P, Kuo J, Laliberté E, Pearse SJ et al (2012) Proteaceae from severely phosphorus-impoverished soils extensively replace phospholipids with galactolipids and sulfolipids during leaf development to achieve a high photosynthetic phosphorus-use-efficiency. New Phytol 196(4):1098–1108
Markert B (1994) Environmental sampling for trace analysis. Wiley
McGroddy ME, Daufresne T, Hedin LO (2004) Scaling of C: N: P stoichiometry in forests worldwide: Implications of terrestrial redfield-type ratios. Ecology 85:2390–2401
Niklas KJ (1994) Plant allometry: the scaling of form and process. University of Chicago Press
Niklas KJ (2006) Plant allometry, leaf nitrogen and phosphorus stoichiometry, and interspecific trends in annual growth rates. Ann Bot 97:155–163
Niklas KJ, Cobb ED (2006) Biomass partitioning and leaf N, P—stoichiometry: comparisons between tree and herbaceous current-year shoots. Plant, Cell Environ 29:2030–2042
Niklas KJ, Owens T, Reich PB, Cobb ED (2005) Nitrogen/phosphorus leaf stoichiometry and the scaling of plant growth. Ecol Lett 8:636–642
Pagel M (1999) Inferring the historical patterns of biological evolution. Nature 401:877–884
Pitman E (1939) A note on normal correlation. Biometrika 31:9–12
Ramsey MH, Potts PJ, Webb PC, Watkins P, Watson JS, Coles BJ (1995) An objective assessment of analytical method precision: comparison of ICP-AES and XRF for the analysis of silicate rocks. Chem Geol 124(1–2):1–19
Raunkiaer C (1934) The life forms of plants and statistical plant geography; being the collected papers of C. Raunkiaer.
Reich PB, Oleksyn J, Wright IJ, Niklas KJ, Hedin L, Elser JJ (2010) Evidence of a general 2/3-power law of scaling leaf nitrogen to phosphorus among major plant groups and biomes. Proc Biol Sci 277:877–883
Sardans J, Janssens IA, Alonso R, Veresoglou SD, Rillig MC, Sanders TGM, Carnicer J, Filella I, Farrearmengol G, Penuelas J (2015) Foliar elemental composition of European forest tree species associated with evolutionary traits and present environmental and competitive conditions. Glob Ecol Biogeogr 24:240–255
Savolainen O, Pyhäjärvi T, Knürr T (2007) Gene flow and local adaptation in trees. Annu Rev Ecol Evol Syst 38:595–619
Sheldon RP (1964) Paleolatitudinal and paleogeographic distribution of phosphorite. US Geol Surv Prof Pap 501:C106–C113
Sobrado M (1991) Cost-benefit relationships in deciduous and evergreen leaves of tropical dry forest species. Funct Ecol 5:608–616
Sterner RW, Elser JJ (2002) Ecological stoichiometry: the biology of elements from molecules to the biosphere. Princeton University Press
Tian D, Yan Z, Niklas KJ, Han W, Kattge J, Reich PB, Luo Y, Chen Y, Tang Z, Hu H (2017) Global leaf nitrogen and phosphorus stoichiometry and their scaling exponent. National Sci Rev 5:728–739
Tilman D, Downing JA (1994) Biodiversity and stability in grasslands. Nature 367:363–365
Townsend AR, Cleveland CC, Asner GP, Bustamante MMC (2007) Controls over foliar N:P ratios in tropical rain forests. Ecology 88:107–118
Urban MC, Strauss SY, Pelletier F, Palkovacs EP, Leibold MA, Hendry AP, De Meester L, Carlson SM, Angert AL, Giery ST (2020) Evolutionary origins for ecological patterns in space. Proc Natl Acad Sci 117:17482–17490
Vergutz L, Manzoni S, Porporato A, Novais RF, Jackson RB (2012) Global resorption efficiencies and concentrations of carbon and nutrients in leaves of terrestrial plants. Ecol Monogr 82(2):205–220
Vitousek PM, Porder S, Houlton BZ, Chadwick OA (2010) Terrestrial phosphorus limitation: mechanisms, implications, and nitrogen–phosphorus interactions. Ecol App 20:5
Walker TW, Syers JK (1976) The fate of phosphorus during pedogenesis. Geoderma 15:1–19
Warton DI, Wright IJ, Falster DS, Westoby M (2006) Bivariate line-fitting methods for allometry. Biol Rev 81:259–291
Warton DI, Duursma RA, Falster DS, Taskinen S (2012) SMATR 3—an R package for estimation and inference about allometric lines. Methods Ecol Evol 3:257–259
Wen J, Ji H, Sun N, Tao H, Du B, Hui D, Liu C (2018) Imbalanced plant stoichiometry at contrasting geologic-derived phosphorus sites in subtropics: the role of microelements and plant functional group. Plant and Soil.
Team RC (2018) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna. https://www.R-project.org
Wright IJ, Reich PB, Westoby M, Ackerly DD, Baruch Z, Bongers F, Cavenderbares J, Chapin T, Cornelissen JH, Diemer M (2004) The worldwide leaf economics spectrum. Nature 428:821
Yan K, Duan C, Fu D, Li J, Wong MHG, Qian L, Tian Y (2015) Leaf nitrogen and phosphorus stoichiometry of plant communities in geochemically phosphorus-enriched soils in a subtropical mountainous region, SW China. Environ Earth Sci 74:3867–3876
Yan Z, Li X, Tian D, Han W, Hou X, Shen H, Guo Y, Fang J (2018) Nutrient addition affects scaling relationship of leaf nitrogen to phosphorus in Arabidopsis thaliana. Funct Ecol 32:2689–2698
Zhang SB, Zhang JL, Slik JWF, Cao KF (2012) Leaf element concentrations of terrestrial plants across China are influenced by taxonomy and the environment. Glob Ecol Biogeogr 21:809–818
Zhang J, Yan X, Su F, Li Z, Wang Y, Wei Y, Ji Y, Yang Y, Zhou X, Guo H (2018) Long-term N and P additions alter the scaling of plant nitrogen to phosphorus in a Tibetan alpine meadow. Sci Total Environ 625:440–448
Acknowledgements
This work was financially supported by the National Key R&D Program of China (2017YFC0505501). We are grateful for the support by the Instrumental Analysis Center of Shanghai Jiao Tong University. We thank Dr. Wuyuan Yin for his invaluable help in identifying tree species. We thank three anonymous reviewers for their careful reading of our manuscript and their many insightful comments and suggestions.
Author information
Authors and Affiliations
Contributions
CL and JW conceived the presented idea. JW, NS, HJ, BD, and MU conducted the field sampling. JW carried out the lab experiment and statistical analyses. JZ verified the analytical methods. JW wrote the manuscript draft. YL, JB, LM, HK, GL, and CL discussed the results and contributed to the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent to Publish
Not applicable.
Additional information
Communicated by Dafeng Hui.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wen, J., Brahney, J., Lin, Y. et al. The scaling of leaf nitrogen and phosphorus along a phosphorus availability gradient in a subtropical forest. Plant Ecol 223, 995–1006 (2022). https://doi.org/10.1007/s11258-022-01252-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11258-022-01252-7