Abstract
Understanding how individuals respond to habitat discontinuities could provide a first step in the large-scale evaluation of their responses to habitat fragmentation. Our objective was to evaluate habitat fragmentation effects on individual and population parameters of two native species of the Pampas grassland (Senecio pampeanus and Margyricarpus pinnatus). Both species grow in vegetation remnants surrounded by pine afforestation in the coastal dunes of the southern Pampas (Argentina). Plants reproductive aspects and offspring fitness were examined, and the influence of environmental maternal effects was analyzed through a common garden experiment. Individuals of S. pampeanus growing in smaller fragments produced fewer heads and fewer fruits per head than those associated with the continuous grassland and with larger fragments. However, M. pinnatus did not exhibit any difference in flower and fruit production with respect to fragment size. The seeds that came from the smallest fragments took longer to germinate for the two species, and in the case of M. pinnatus they also showed reductions in percent germination. We found evidence for influence of the maternal environment on percent germination and in seedling establishment time of M. pinnatus. Our results provide clues about the effects of fragmentation on plant reproduction, with consequences for the offspring, and also show that species respond differently according to their biological characteristics. Furthermore, the results obtained contribute to the understanding of mechanisms by which the subdivision of natural environments affects the persistence of populations and alters the structure of plant communities.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Habitat fragmentation is considered one of the main forces responsible for the global biodiversity crisis (Saunders et al. 1991; Fahrig 2003; García 2011; Haddad et al. 2015), threatening the persistence and survival of species through the reduction of the available habitat, the isolation of the remaining fragments, the influence from the resulting edges, and the increase in vulnerability to extrinsic disturbances (Lord and Norton 1990). Identifying the causes and consequences of habitat fragmentation is essential to preserve biodiversity and the functioning of ecosystems. Additionally, understanding how individuals respond to habitat discontinuities could provide a first step in the large-scale evaluation of responses to habitat fragmentation (Bowers et al. 1996). Debinski and Holt (2000) highlight an absence of homogeneity in the results of studies on habitat degradation, mainly in those that analyze species richness and abundance and their relationship with fragment size. Many of these different responses to fragmentation could be explained by detecting the factors that make certain species particularly sensitive to habitat degradation and identify possible effects on other organisms of the community, including pollinators and granivores (Ewers and Didham 2006; García 2011).
Fragmentation influences the individual performance of plants, affecting their growth and reproductive capacity by creating barriers to pollination and dispersal (Aizen and Feinsinger 1994), and causes reductions in population sizes by dividing large populations into two or more smaller subpopulations within a restricted area (Andrén 1994; Fahrig 2003). Subpopulations are consequently more prone to inbreeding depression and genetic drift (Ewers and Didham 2006; García 2011). At the individual and population level, the most commonly evaluated aspects of the effects of fragmentation have been on plant fertility and plant-pollinator interactions (Didham et al. 1996; Honnay et al. 2005). Although reproduction is crucial for the persistence of plant populations, it is not the only process; other stages of their life cycle, such as seed dispersal and germination or seedling recruitment, are also important and may affect the viability of future generations (Aguilar et al. 2006; García and Chacoff 2007). However, the effect of fragmentation on these ecological processes has been much less considered (Benitez-Malvido 1998; Henríquez 2004; Ashworth and Marti 2011).
In small and fragmented plant populations, persistence may depend not only on the degree of inbreeding but also on the ability of individual plants to live in increasingly smaller areas and lower ecological quality due to their proximity to an edge (Granados et al. 2014). The environmental conditions experienced by an individual plant during its growth and development may affect not only that particular individual but also its progeny during one or more generations (Wulff et al. 1999). The stressful environmental conditions can cause a maternal plant to inadequately provision its progeny, resulting in poor seedling growth and decreased competitive ability (Riginos et al. 2007). This influence of the maternal ecological environment on the offspring phenotype, known as the environmental maternal effect, occurs after embryo formation but before seed dispersal (Roach and Wulff 1987; Aarssen and Burton 1990; Donohue 2009; Wolf and Wade 2009) and can contribute to the differentiation in the traits of the progeny if the growth conditions of the mother plants are different from those of the offspring (Bischoff and Müller-Schärer 2010).
The natural heterogeneity of landscapes and habitats determines species interactions and community dynamics, where patches of suitable and unsuitable habitats alternate in a heterogeneous mosaic, at scales that are relevant to the acquisition of resources, dispersal, reproduction, and other components of the individual's fitness (Didham 2010). This results in an adaptation of many species to a certain natural degree of fragmentation (Honnay et al. 2005). However, in anthropogenically altered landscapes, humans destroy and modify the habitat at increasing rates, exceeding the capacity of most species to adapt and respond to the decreasing availability and increasing isolation of suitable habitat (Lienert 2004; Didham 2010). In this context, plants that differ in habitat requirements and in their biology are likely to respond differently to habitat fragmentation (Heinken and Weber 2013).
Grasslands are recognized as the terrestrial biome where biodiversity and ecosystem processes are at greatest risk worldwide, due to the large disparity between the rate of habitat loss and the degree of effective protection (Hoekstra et al. 2005; Suttie et al. 2005). The Pampas biome comprises the natural grasslands of central and eastern Argentina, the entire territory of Uruguay and the southern tip of Brazil, and constitutes one of the largest temperate grassland regions in the world (Bilenca and Miñarro 2004). Since the second half of the nineteenth century, the Pampas region has undergone a process of transformation and fragmentation due to the progress of various forms of anthropogenic intervention (Bilenca and Miñarro 2004; Azpiroz et al. 2012), including the replacement of natural grasslands with forest plantations (Guadagnin et al. 2009; Simberloff et al. 2010; Fonseca et al. 2013).
The natural systems of the coastal dunes of the Sothern Pampas, in the southwestern area of the province of Buenos Aires (Argentina), are characterized by their environmental heterogeneity, with dune strips interrupted by interdunal depressions and lagoons (Monserrat 2010). These are high quality environments, encompassing a high biological and environmental richness, and providing important ecosystem services (Kittlein et al. 2004). The restrictions that they impose on traditional agricultural exploitation because of being nutrient-poor soils keep them better preserved than the rest of the Pampas grasslands (Zalba and Villamil 2002; Baldi et al. 2006). However, different anthropogenic activities, including the establishment and expansion of urban centers and the afforestation with exotic species (Bilenca and Miñarro 2004; Guadagnin et al. 2009; Azpiroz et al. 2012), fragment the ecosystem limiting the original vegetation to relictual areas (Dadon 2002). Maritime pine (Pinus pinaster), a species with invasive behavior in Argentina and Uruguay (Picasso and Rodríguez 2008; Brugnoli et al. 2009), is the most commonly planted alien tree in this area. Planted and spontaneously expanded stands of Maritime pine frequently result in a matrix that surrounds grassland remnants, reducing and isolating natural habitats. Previous studies have shown that the fragmentation of the ecosystem by forest plantations results in changes in the composition of the vegetation and increases the number of exotic plants, mainly in the smaller remnants (Yezzi et al. 2018), in addition to altering their resilience and their successional response to fires (Yezzi et al. 2019). Finding general response patterns of plant species to the effects of fragmentation allows to identify the ecological processes most susceptible to disruption of habitat continuity (Aguilar et al. 2006). However, up to this moment, the extent to which the individual native species of the Pampas grassland and their populations are affected by the fragmentation has not been thoroughly evaluated, despite the importance that this type of information has for conservation.
Our objective was to evaluate the effect of fragmentation generated by pine afforestation and expansion on individual and population parameters of two species of plants native to grasslands in the coastal dunes of the province of Buenos Aires (Argentina). We examined reproductive aspects of the individual plants and the offspring fitness of two common grassland species growing in remnants of vegetation surrounded by a P. pinaster plantation. We propose that the reduction in the size of the grassland fragments surrounded by pine trees affects the habitat quality for native plants, modifying their growth and reproduction and cascading towards their offspring through maternal effects. We expect to find (1) lower rates of flowering and fruiting as the area of the fragments decreases, (2) lower percentage of germination and establishment of the seeds collected from smaller fragments compared to those that collected in larger fragments and in continuous grassland, and (3) a decrease in the influence of environmental maternal effects after a generation growing under homogeneous conditions.
Materials and methods
Study area
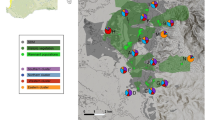
This work was developed in an area of coastal dunes in southwestern Buenos Aires province, Argentina. The coastal strip includes an extensive field of active and fixed dunes, wide beaches, and interspersed temporary and permanent ponds and small lakes, and ranges between two and seven kilometers wide (Celsi et al. 2010). The climate is subhumid temperate, with annual average values of temperature and precipitation of 14.1 °C and 850 mm, respectively (Burgos and Vidal 1951). Psammophilous shrubs such as Hyalis argentea and Baccharis divaricata, and grasses such as Poa lanuginosa, Schizachyrium spicatum and Aristida spegazzinii are dominant species in dunes. Interdunal depressions are covered by Cortaderia selloana, Juncus acutus, Typha sp. and other hygrophilous herbs (Celsi and Giussani 2019). The natural landscape is well-preserved compared to other areas of coastal dunes of the Southern Pampa (Celsi et al. 2010), with grazing by domestic cattle, urbanization and roads, and the spontaneous expansion of exotic trees among the most conspicuous anthropogenic disturbances (Zalba and Villamil 2002). We studied a P. pinaster afforestation, implanted ca. 35 years ago that received no further management and currently covers a total area of approximately 30 ha, surrounding a set of natural grassland remnants in a coastal dune sector (38° 55′ S, 60° 33′ W). While pines grow on an area of ca. 30 ha, the whole set of trees and grassland remnants occupy 1 km2. Grassland remnants immersed in the forested area were identified from a satellite image of the site taken from Google Earth and dated October 11th, 2015, and the area and perimeter of each fragment were measured using Google Earth Pro. Three grassland fragments of up to 0.1 ha, three from 0.1 to 0.5 ha, three from 0.5 to 1 ha and three from 1 to 2.5 ha were selected, and a neighboring continuous grassland of 40 ha was used as a control (Fig. 1).
Location of the ecosystem of coastal dunes of the Southern Pampa, in the southwest of the province of Buenos Aires, Argentina (a). Study area indicating grassland fragments of different sizes (up to 0.1 ha, 0.1 to 0.5 ha, 0.5 to 1 ha and 1 to 2.5 ha) surrounded by Pinus pinaster afforestation and the control of continuous grassland (40 ha; b). Image from Google Earth (2019)
Plant species
Two common native species of the plant communities of the Pampas grasslands that were present in the fragments were selected: Senecio pampeanus Cabrera and Margyricarpus pinnatus (Lam.) Kuntze (nomenclature according to Zuloaga et al. 2019).
Senecio pampeanus (Asteraceae) is a perennial shrub frequent in low and humid places. Dimorphic flowers grow gathered in heads (female in the margin, 11–13; hermaphrodites in the center, numerous). Flowering occurs in spring. The cypselae are cylindrical with white pappus and hairs (Cabrera 1963). Pollination is entomophilous, without evidence of spontaneous self-pollination in the field (Torres and Galetto 2008). The species is dispersed by the wind (Torres and Galleto 2011).
Margyricarpus pinnatus (Rosaceae) is a perennial shrub frequent in sandy and dry sites, with solitary, hermaphrodites chasmogamous and mixed cleistogamous flowers. Flowering occurs in early spring. It has fleshy, drupaceous fruits, 5 to 7 cm in diameter, more or less globose (Cabrera 1967). Pollination is entomophilous, with self-pollination in the case of cleistogamous flowers (Zardini 1973). It has zoochory dispersal (Novara 1993), including birds within their biological vectors (Bernardello et al. 2006).
Due to the different flowering and fruiting times of the studied species, the field work was carried out at different times.
Flowering and fruiting
Five individuals of each species were randomly selected for each size of grassland fragment and ten of each for the continuous grassland. All selected plants were marked and georeferenced.
Senecio pampeanus: in December 2015, the number of heads per plant was recorded, assigning them to five categories: closed heads (immature), floriferous heads (with flowers with the ligule extended), immature fruitful heads (with ripe fruits, ready for dispersal), mature fruitful heads (with ripe already dispersing fruits), and empty heads (with all fruits dispersed). Counts were made in three non-adjacent portions of each plant that were randomly chosen and delimited as angular sectors of 12°, centered on the main stem of the specimen; the average of these values for each plant was calculated and multiplied by thirty to estimate the total number of heads for the whole individual. Three immature fruitful heads were collected per plant and the total number of fruits per head was counted, also registering the percentage of fertile fruits, considering as non-fertile those who looked empty under a stereomicroscope.
Margyricarpus pinnatus: in October 2015 the total number of flowers per plant was counted. For each plant, two counts of the total number of fruits were made (one at the beginning of the fruiting period: end of November 2015, and one at the end of the fruiting period: mid-April 2016). At the beginning of fruiting, 60 fruits were collected from not less than three plants for each fragment size and for the continuous grassland area. At the end of fruiting, the fruits were categorized according to their maturation status into three categories: immature (green), intermediate maturity (white), and ripe (pink).
Germination of seeds collected in the field
Senecio pampeanus: the cypselas were stored in paper bags at room temperature and kept in darkness for 90 days until the beginning of the experiments. Following Funes et al. (2009), they were not subjected to any pre-germination treatment.
Margyricarpus pinnatus: to promote germination, the pulp was completely eliminated from the fruits and they were stored in paper bags in a dry environment and protected from light for 40 days. Cold stratification was applied to break physiological dormancy reported for other species in the genus (Baskin and Baskin 2001), keeping them at a constant temperature of 4–6 °C for 35 days.
Germination tests in the laboratory were performed in a constant temperature chamber at 20 °C (Funes et al. 2009; Nuciari 2012) exposed to 24 h of light. The seeds were placed on filter paper in Petri dishes. Ten seeds were sown per dish, and 6 dishes per treatment, totaling 30 dishes and 300 seeds for each species. Seeds were irrigated with distilled water, keeping the substrate moist. Observations were made every 2 to 3 days and germination was considered to occur when the radicle emerged at least 1 mm from the seed. The average germination time (TMG) was calculated according to Nakagawa (1999).
Establishment of seedlings of Margyricarpus pinnatus from seeds collected in the field
Each emerged seedling was transplanted to trays with individual cavities containing soil, a tray per treatment (F1 generation; Fig. 2). The trays were kept in a greenhouse, watered and checked daily, recording the number of seedlings established and the time of establishment.
Schematic diagram of the experimental design. P: plants of Margyricarpus pinnatus growing in the field, F1: seeds, seedlings, and plants obtained from plants growing in the field, and cultivated under controlled conditions, F2: seeds, seedlings, and plants obtained from cultivated plants, and also grown under controlled conditions
Germination of Margyricarpus pinnatus seeds obtained from cultivated plants and seedlings establishment
Six months after establishment, the surviving plants were transplanted into pots and placed under homogeneous conditions in a common garden. During the months of February and March 2017 the fruits produced by these plants (F2 generation; Fig. 2) were collected. Germination and establishment tests were performed in the same way as those described for the seeds collected in the field.
Data analysis
Contingency tables and Chi-squared tests were used to compare the maturation state of the S. pampeanus heads for plants growing in remnants of different area and in the continuous grassland. The same analysis was applied to the fruits of M. pinnatus.
Number of heads per plant, number of fruits per head and percentage of fertile fruits per head for S. pampeanus, total number of flowers and total number of fruits for M. pinnatus, percentage of germination and average germination time for the seeds of both species collected in the field, and time of establishment and number of established seedlings of M. pinnatus were compared to analyze possible effects of fragment area, by means of ANOVA, when the assumptions of homoscedasticity and normality were met. When necessary, data were transformed (square root for number of heads of S. pampeanus and total number of fruits of M. pinnatus; natural logarithm for time of establishment for seedlings of M. pinnatus) to correct for the lack of normality. When the lack of normality persisted, the Kruskal–Wallis test was applied to the original data (Zar 1996). When an effect of fragment area was found, post hoc tests were applied to determine which of the treatments differed from each other.
When an effect of fragment area on the offspring fitness of M. pinnatus plants growing in the field was found, the influence of maternal environmental effects was assessed by ANOVA (for average germination time, establishment time, and number of established seedlings) and by Kruskal–Wallis test (for germination percentage). The data corresponding to the generation growing from seeds collected in the field (F1) were compared with that from seeds grown in homogeneous conditions (F2). We considered that a significant interaction between the effects of the origin of the seeds (coming from plants growing in the field or from cultivated individuals), and treatment (grassland fragments of different size and continuous grassland) in this analysis would indicate a contribution of maternal environmental effect to the differentiation of treatments. Where such a significant interaction was detected, comparisons were made by post hoc tests to identify the effects of fragment area that were still significant in the F2 plants. To further explore the influence of maternal effects, inter-individual variations for plants of the two generations (F1 and F2) were compared using the Mann–Whitney test (for germination percentage) and Fisher's F test (at the time of establishment), in order to look for a possible reduction in the variation from F1 to F2, which would indicate that the effect of fragment area was transient, and persisted only for a generation.
Data normality were evaluated by residuals analysis and normal probability plots. Levene’s test was used to verify homogeneity of variances. All analyses and graphs were performed using the statistical packages Infostat (Di Rienzo et al. 2008) and XLSTAT (versions 5.7.2 and 2018.2.50198).
Results
Flowering and fruiting
Senecio pampeanus: the maturational state of the heads was influenced by the fragments area in which the plants grew (X2 = 448.414, P < 0.0001). A delay in the maturation of the heads in fragmented grasslands was observed: while in December the plants of the continuous grassland had a high percentage of fruitful heads and also some heads that had already released all their fruits, individuals growing on fragmented grasslands still exhibited a considerable percentage of floriferous heads and a variable number of closed, unripe heads (Fig. 3).
Fragment area also influenced the number of heads per plant (F = 3.60, P = 0.0190) and the number of fruits per head (F = 5.63, P = 0.0026). Plants that grew in grassland fragments of smaller area (0.1 ha) had fewer heads and fewer fruits per head than those plants growing in the continuous grassland and in larger fragments (Fig. 4). No significant differences were detected in the percentage of fertile fruits per head between treatments (F = 0.96, P = 0.4488).
Comparison between the number of heads (a) and the number of fruits per head (b) of Senecio pampeanus plants growing in grassland fragments of different sizes (white bars) and in a continuous grassland (black bar), in the coastal dunes of the Southern Pampa (Argentina). Different letters indicate significant differences (P < 0.05); bars represent the standard error
Margyricarpus pinnatus: ripening of the fruits was influenced by fragment area (X2 = 303.491, P < 0.0001). Plants in the continuous grassland showed fruit earlier ripening compared to those growing in grassland fragments of all sizes (Fig. 5). No significant differences were detected between the fragment areas with respect to the number of flowers (H = 3.22, P = 0.5209) and the number of fruits (F = 0.60, P = 0.6688).
Germination of seeds collected in the field
Fragment area influenced germination in both species (S. pampeanus: H = 9.85, P = 0.0346; M. pinnatus: H = 8.76, P = 0.05). In the case of S. pampeanus, the seeds from plants that grew in the continuous grassland had the highest percentage of germination (75%), statistically higher only from those produced by plants of the 1 ha fragments, that were associated to the lowest germination percentage recorded (52%; Fig. 6a). In the case of M. pinnatus, seeds from plants that grew in the continuous grassland also had the highest germination percentage (72%), significantly higher than those that from plants of the smallest fragments (50%; Fig. 6b).
Comparison between the percentage of germination of seeds of Senecio pampeanus (a) and of Margyricarpus pinnatus (b) produced by plants growing in the field in grassland fragments of different size (white bars) and in a continuous grassland (black bar), in the coastal dunes of the Southern Pampa (Argentina). Different letters indicate significant differences (P < 0.05); bars represent the standard error
Fragment area also influenced the average germination time (S. pampeanus: F = 13.25, P < 0.0001; M. pinnatus: F = 3.04, P = 0.0361). In the case of S. pampeanus, seeds from the continuous grassland and from larger fragments (2.5 ha) had a shorter mean germination time (10 days) compared to those from the other grassland fragments analyzed (15 days; Fig. 7a). The seeds of M. pinnatus that were produced by plants in the smallest fragments (0.1 ha) had a longer mean germination time (28 days) than those from bigger fragments and from the continuous grassland (19 days; Fig. 7b).
Comparison between the average germination time of seeds of Senecio pampeanus (a) and of Margyricarpus pinnatus (b) produced by plants growing in the field in grassland fragments of different size (white bars) and in a continuous grassland (black bar), in the coastal dunes of the Southern Pampa (Argentina). Different letters indicate significant differences (P < 0.05); bars represent the standard error
Establishment of seedlings of Margyricarpus pinnatus from seeds collected in the field
The influence of fragment area where M. pinnatus mother plants grew extended towards the phase of seedling establishment. Seedlings from seeds produced in smallest fragments showed lower establishment and took longer to do so, compared to those of the continuous grassland (number of established seedlings: three and five, F = 2.73, P = 0.05; time of establishment: 24 and 15 days, F = 4.32, P = 0.0027; Fig. 8).
Comparison between the time of establishment (a) and the number of established Margyricarpus pinnatus seedlings (b) originating from seeds produced by plants growing in the field (F1) in grassland fragments of different sizes (white bars) and in a continuous grassland (black bar), in the coastal dunes of the Southern Pampa (Argentina). Different letters indicate significant differences (P < 0.05); bars represent the standard error
Influence of maternal environment on germination and establishment of Margyricarpus pinnatus
The origin of the seeds (coming from plants growing in the field, F1, or from cultivated individuals, F2) and fragment area (grassland fragments of different sizes and continuous grassland) had a statistically significant interaction on the percentage of germination (H = 25.17, P = 0.0024) and on the time of seedling establishment (F = 2.34, P = 0.05). Such an interaction was not detected for mean germination time (F = 2.00, P = 0.1087) and number of established seedlings (F = 1.56, P = 0.1999).
While the F1 seeds from smaller areas (0.1 ha) showed a germination percentage significantly lower than those from the continuous grassland (40 ha; Figs. 6b, 9a), this difference was no longer significant for the F2. In this case, the seeds of 1 ha areas showed lower germination compared to those of the continuous grassland (40 ha) and to those of 2.5 and 0.5 ha (Fig. 9a). No reduction was found in the inter-individual variation between fragments areas from F1 to F2 (U = 257, P = 0.998).
Comparison between the percentage of germination (a) and the time to establishment (b) of individuals originated from seeds produced by plants growing in the field (F1; dark gray) and by plants grown under homogeneous conditions (F2; light gray), coming from grassland fragments of different sizes and from a continuous grassland. Different letters indicate significant differences (P < 0.05); bars represent the standard error
In F1, seedlings of the smallest areas (0.1 ha) took longer to establish with respect to those of the larger areas (1 and 2.5 ha), and those of the continuous grassland (40 ha; Figs. 8a, 9b). This difference extended toward F2, with seedlings of the smallest areas (0.1 ha) also showing longer establishment times compared to those of the 2.5 and 0.5 ha areas (Fig. 9b). In this case, a significant reduction in the inter-individual variation between areas from F1 to F2 was observed (F = 2.82, P < 0.0001).
Discussion
The results obtained in this work provide clues about the effects of fragmentation on different aspects of the reproductive biology of two common plants of the Pampas grassland, which also influence characteristics of their offspring, and show that species respond differently according to their biological characteristics. Furthermore, they contribute to the understanding of mechanisms by which the subdivision of natural environments affects the persistence of populations and alters the structure of plant communities.
Both studied species showed a delay in fruit maturation in the fragmented grassland with respect to the continuous habitat. Different authors maintain that changes in certain environmental processes alter plant phenology, modifying the time of the year in which flowering occurs (Inouye et al. 2003; López et al. 2018). Modification of abiotic conditions has been demonstrated, for instance, in a work carried out in the Brazilian woody savanna, where changes in light conditions at fragment edges led to alterations in the reproductive phenology of a species with a preference for dry environments (Camargo et al. 2011). This effect could alter the timing of plant-animal interactions (Fabina et al. 2010), and also delay fruit and seed production and maturation.
Fragmentation also affected propagule production of S. pampeanus. Individuals growing in smaller fragments produced fewer heads and fewer fruits per head than those associated with the continuous grassland and with the larger fragments. However, M. pinnatus did not exhibit any difference in flower and fruit production when comparing individuals growing in grassland fragments of different size and in continuous grassland. Different studies document a decrease in the richness of pollinating insects and in fruiting of plants when comparing continuous habitats or large fragments with small relicts (Aizen and Feinsinger 1994; Morgan 1999; Goverde et al. 2002; Rosch et al. 2013; Álvarez et al. 2014). The tree matrix surrounding grassland remnants in this study could create barriers for some insects and limit pollination, and consequently could affect plant reproduction, as has been observed in other studies focused on forest-grassland boundaries (Spector and Ayzama 2003; Matter et al. 2004; Cant et al. 2005). In addition, small fragments may not contain ecologically effective populations of pollinators due to the effect of the area itself. The limitation observed in the reproduction of S. pampeanus could be explained by the fact that it is a species that does not produce seeds in the absence of floral visitors (Torres and Galetto 2008), unlike M. pinnatus, which has the ability to produce both flowers that require pollinators (chasmogamous) and flowers that are capable of selfing (cleistogamous; Steibel and Troiani 1999). This feature could have allowed it to compensate for the eventual drop in fruit production associated with limitations in pollination, since self-pollination may be especially important in small populations (Lienert 2004). Therefore, the most marked fragment size effects observed for S. pampeanus in this work support the idea that species with the ability to self-fertilize could be less affected by habitat fragmentation than those that do not reproduce in the absence of floral visitors. This difference in the effects of fragmentation upon the reproductive success of two species with different reproductive traits could reflect one of the proximate causes of changes in the composition of plant communities as a response to fragmentation, strengthening the understanding of the processes that underlie the biological impoverishment associated with this type of habitat degradation. In addition to compromising pollination, the forest matrix could also act as a barrier to dispersal. S. pampeanus produces seeds that are dispersed by wind, while M. pinnatus has fleshy fruits that are dispersed by animals. Both mechanisms could be conditioned by the matrix of trees, in the first case by mechanical interference with wind (Soons and Heil 2002) and in the second by possible changes in the abundance of the populations of animal dispersersand in their behavior (Rey and Alcántara 2014). Considering the latter, it cannot be ruled out that the lack of fragmentation effects on the number of M. pinnatus fruits could be related to a reduction in the removal rate by predators in the smallest areas, a phenomenon that has already been described for grassland fragments surrounded by a forest matrix (Cintra et al. 2013; Rey and Alcántara 2014; Pretelli et al. 2018).
The two potential aforementioned limitations, a reduction in cross-pollination and an interference with seed dispersal, could contribute to the genetic isolation of populations, generating a process of depression by inbreeding, reducing the offspring fitness, and resulting in the production of seeds of lower quality, which could eventually reduce the population’s ability to respond to future environmental change (Soons and Heil 2002; Honnay and Jacquemyn 2007; Nora et al. 2011; Hooftman et al. 2003). The time elapsed since fragmentation is an important feature to assess genetic erosion in plants, and it is considered that in recently fragmented ecosystems (less than 50 years) the loss of genetic variability may be may be less evident compared with fragmented ecosystems more than 50 years (Morgan 1999; Aguilar et al. 2008). However, most of the species studied have been woody species with long lifespans (Aguilar et al. 2019), while the effects of genetic erosion could be more pronounced for herbaceous plants, with shorter generation times (Aguilar et al. 2008). Therefore, and although the studied fragmentation process is relatively recent, we could expect potential genetic erosion effects caused by grassland fragmentation on offspring fitness of the studied species. This possibility is consistent with the results obtained in our germination tests, in which the seeds that came from the smallest fragments took longer to germinate for the two species, and in the case of M. pinnatus they also showed reductions in the percentage of germination. In addition to this, seedlings of M. pinnatus developed from seeds that came from small fragments took longer to establish and did so in fewer numbers than those from the continuous grassland, even though they were grown under identical environmental conditions. These effects could reflect fitness reduction processes associated with genetic drift and would be even stronger under natural conditions, where seedlings are subject to competition with each other and with the surrounding plant community (Henríquez 2004).
Alternatively or in a complementary way, the lower fitness of the offspring could be a consequence of the adverse abiotic conditions that prevail in the smaller fragments (McGarigal and Cushman 2002), which would result in a lower availability of nutrients for the mother plant to invest in seed development (Soons and Heil 2002; Ashworth and Marti 2011). These maternal environmental effects may have a significant impact on the early fitness traits of the offspring (Roach and Wulff 1987), but they are expected to be transient and to influence only direct descendants or to be reduced significantly in the following generations (Roach and Wulff 1987; Agrawal 2002). This transience can help to identify if the cause of the reductions in fitness is effectively linked to the maternal effect or if it includes genetic components of greater persistence. Although in this work we found no evidence of an influence of the maternal environment on the mean germination time of the seeds, nor on the number of established M. pinnatus seedlings in the F2 generation, we did detect an effect on germination percentage and on time for seedling establishment. The interaction between the effects of the origin of the seeds, and fragment area upon both variables would also indicate that there is an environmental maternal effect. In the same way, the reduction in the variation of the time for seedling establishment in F2 is another piece of evidence in favor of the possible influence of the maternal environment. Our results support the idea that the fitness of the offspring is affected by the intensity of the fragmentation of the maternal environment, decreasing the chances of seedling recruitment and the competitive capacity of young individuals. This could compromise population viability, since juvenile stages are the most vulnerable and crucial for the establishment and survival of plant populations (Henríquez 2004; Padilla Ruíz 2008; Aguilar et al. 2019). The latter would be consistent with the results obtained in a community-level study carried out at the same site, where M. pinnatus was abundant in continuous grassland, but was extremely scarce or even absent in the smaller fragments (Yezzi et al. 2018).
According to the results obtained, 2.5 ha could be proposed a priori as a minimum effective size allowing the conservation of the two studied species. However, and as is also clear from this study, species respond to fragmentation according to their reproductive strategies, and therefore what may be a safe size for one species may not be enough for others. Therefore, understanding the responses of species with contrasting life history traits could help reveal what ecological processes are being affected, select species that indicate impact, and take possible mitigation actions. On the other hand, it is also interesting to note that the effects can be transferred to the following generation. Therefore, incorporating the evaluation of the aptitude of the offspring developed in fragmented environments into fragmentation studies, can provide important information on the viability of plant populations over time. This type of information is important for the conservation and management of the Pampas biodiversity in the current scenario in which its loss by human activities has reached levels unprecedented in history.
References
Aarssen LW, Burton SM (1990) Maternal effects at four levels in Senecio vulgaris (Asteraceae) grown on a soil nutrient gradient. Am J Bot 77(9):1231–1240
Agrawal A (2002) Herbivory and maternal effects: mechanisms and consequences of transgenerational induced plant resistance. Ecology 83:3408–3415. https://doi.org/10.2307/3072089
Aguilar R, Ashworth L, Galetto L, Aizen M (2006) Plant reproductive susceptibility to habitat fragmentation: review and synthesis through a meta-analysis. Ecol Lett 9:968–980. https://doi.org/10.1111/j.1461-0248.2006.00927.x
Aguilar R, Quesada M, Ashworth L, Herrerias-Diego Y, Lobo J (2008) Genetic consequences of habitat fragmentation in plant populations: susceptible signals in plant traits and methodological approaches. Mol Ecol 17(24):5177–5188. https://doi.org/10.1111/j.1365-294X.2008.03971.x
Aguilar R, Cristóbal-Pérez EJ, Balvino-Olvera FJ, Aguilar-Aguilar MJ, Aguirre-Acosta N, Ashworth L, Lobo JA, Martén-Rodríguez S, Fuchs EJ, Sanchez-Montoya G, Bernardello G, Quesada M (2019) Habitat fragmentation reduces plant progeny quality: a global synthesis. Ecol Lett 22(7):1163–1173. https://doi.org/10.1111/ele.13272
Aizen M, Feinsinger P (1994) Forest fragmentation, pollination, and plant reproduction in a Chaco dry forest, Argentina. Ecology 75:330–351. https://doi.org/10.2307/1939538
Álvarez A, Pérez MA, Cuevas E (2014) Efecto de la fragmentación del hábitat en el éxito reproductivo de Fuchsia parviflora, una especie dioica. Biológicas 16(1):30–32
Andrén H (1994) Effects of habitat fragmentation on birds and mammals in landscapes with different proportions of suitable habitat: a review. Oikos 71:355–366. https://doi.org/10.2307/3545823
Ashworth L, Marti ML (2011) Forest fragmentation and seed germination of native species from the Chaco Serrano forest. Biotropica 43(4):496–503. https://doi.org/10.1111/j.1744-7429.2010.00721.x
Azpiroz AB, Isacch JP, Dias RA, Di Giacomo AS, Suertegaray Fontana C, Morales Palarea C (2012) Ecology and conservation of grassland birds in southeastern South America: a review. J Field Ornithol 83(3):217–246. https://doi.org/10.1111/j.1557-9263.2012.00372.x
Baldi G, Guerschman JP, Paruelo JM (2006) Characterizing fragmentation in temperate South America grasslands. Agric Ecosyst Environ 116:197–208
Baskin CCA, Baskin JMA (2001) Seeds: ecology, biogeography, and evolution of dormancy and germination. Academic Press, San Diego
Benitez-Malvido J (1998) Impact of forest fragmentation on seedling abundance in a Tropical Rain Forest. Conserv Biol 12(2):380–389. https://doi.org/10.1111/j.1523-1739.1998.96295.x
Bernardello G, Anderson GJ, Stuessy TF, Crawford DJ (2006) The angiosperm flora of the Archipelago Juan Fernandez (Chile): Origin and dispersal. Can J Bot 84:1266–1281
Bilenca D, Miñarro F (2004) Identificación de Áreas Valiosas de Pastizal (AVPs) en las Pampas y Campos de Argentina, Uruguay y sur de Brasil. Fundación Vida Silvestre, Buenos Aires
Bischoff A, Müller-Schärer H (2010) Testing population differentiation in plant species—how important are environmental maternal effects. Oikos 119(3):445–454. https://doi.org/10.1111/j.1600-0706.2009.17776.x
Bowers MA, Matter SF, Dooley JL, Dauten JL, Simkins JA (1996) Controlled experiments of habitat fragmentation: a simple computer simulation and a test using small mammals. Oecologia 108:182–191. https://doi.org/10.1007/BF00333230
Brugnoli E, Masciadri S, Muniz P (2009) Base de datos de especies exóticas e invasoras en Uruguay, un instrumento para la gestión ambiental y costera. Ecoplata, Montevideo
Burgos J, Vidal AL (1951) Los climas de la República Argentina según la nueva clasificación de Thornthwaite. Meteoros 1:1–32
Cabrera AL (1963) Flora de la Provincia de Buenos Aires. Colección Científica del INTA, Buenos Aires
Cabrera AL (1967) Flora de la Provincia de Buenos Aires. Colección Científica del INTA, Buenos Aires
Camargo MGG, Souza RM, Reys P, Morellato LPC (2011) Effects of environmental conditions associated to the cardinal orientation on the reproductive phenology of the cerrado savanna tree Xylopia aromatica (Annonaceae). An Acad Bras Ciênc 83(3):1007–1020. https://doi.org/10.1590/S0001-37652011005000014
Cant ET, Smith AD, Reynolds DR, Osborne JL (2005) Tracking butterfly flight paths across the landscape with harmonic radar. Proc R Soc B 272:785–790. https://doi.org/10.1098/rspb.2004.3002
Celsi CE, Giussani LM (2019) Geographical distribution and habitat characterization of Poa schizantha (Poaceae), a narrow endemic of the coastal sand dunes of the southern Pampas, Argentina. Bot J Linn Soc 192(1):296–313
Celsi C, Mac-Lean D, Yezzi A, Triches M (2010) Dunas costeras de la Pampa Austral. Biodiversidad, ecología y conservación entre el río Quequén Salado y el balneario Pehuen-có. Fundación de Historia Natural Félix de Azara, Buenos Aires
Cintra R, Magnusson WE, Albernaz A (2013) Spatial and temporal changes in bird assemblages in forest fragments in an eastern Amazonian savannah. Ecol Evol 3(10):3249–3262
Dadon JR (2002) El impacto del turismo sobre los recursos naturales costeros en la costa pampeana. In: Dadon JR, Matteucci SD (eds) Zona Costera de la Pampa Argentina. Lugar Editorial, Buenos Aires, pp 101–121
Debinski DM, Holt RD (2000) A survey and overview of habitat fragmentation experiments. Conserv Biol 14(2):342–355. https://doi.org/10.1046/j.1523-1739.2000.98081.x
Didham RK (2010) Ecological consequences of habitat fragmentation. Encyclopedia of Life Sciences (ELS). Wiley, Chichester. https://doi.org/10.1002/9780470015902.a0021904
Didham RK, Ghazoul J, Stork NE, Davis AJ (1996) Insects in fragmented forests: a functional approach. Trends Ecol Evol 11:255–260. https://doi.org/10.1016/0169-5347(96)20047-3
Di Rienzo JA, Casanoves F, Balzarini MG, Gonzalez L, Tablada M, Robledo CW (2008) InfoStat, versión 2008. Universidad Nacional de Córdoba, Córdoba
Donohue K (2009) Completing the cycle: maternal effects as the missing link in plant life histories. Phil Trans R Soc B 364:1059–1074. https://doi.org/10.1098/rstb.2008.0291
Ewers RM, Didham RK (2006) Confounding factors in the detection of species responses to habitat fragmentation. Biol Rev 81:117–142. https://doi.org/10.1017/S1464793105006949
Fabina NS, Abbott KC, Gilman RT (2010) Sensitivity of plant-pollinator-herbivore communities to changes in phenology. Ecol Modell 221(3):453–458. https://doi.org/10.1016/j.ecolmodel.2009.10.020
Fahrig L (2003) Effects of habitat fragmentation on biodiversity. Annu Rev Ecol Evol Syst 34:487–515. https://doi.org/10.1146/annurev.ecolsys.34.011802.132419
Fonseca C, Guadagnin DL, Emer C, Masciadri S, Germain P, Zalba SM (2013) Invasive alien plants in the Pampas grasslands: a tri-national cooperation challenge. Biol Invasions 15:1751–1763. https://doi.org/10.1007/s10530-013-0406-2
Funes G, Díaz S, Venier P (2009) La temperatura como principal determinante de la germinación en especies del Chaco seco de Argentina. Ecol Austral 19:129–138
García D (2011) Efectos biológicos de la fragmentación de hábitats: nuevas aproximaciones para resolver un viejo problema. Ecosistemas 20(2):1–10
García D, Chacoff NP (2007) Scale-dependent effects of habitat fragmentation on hawthorn pollination, frugivory, and seed predation. Conserv Biol 21(2):400–411. https://doi.org/10.1111/j.1523-1739.2006.00593.x
Goverde M, Schweizer K, Baur B, Erhardt A (2002) Small-scale habitat fragmentation effects on pollinator behaviour: experimental evidence from the bumblebee Bombus veteranuson calcareous grasslands. Biol Conserv 104:293–299
Granados C, Serrano Giné D, García-Romero A (2014) Efecto de borde en la composición y en la estructura de los bosques templados. Sierra de Monte-Alto, Centro de México. Caldasia 36(2):269–287. https://doi.org/10.15446/caldasia.v36n2.47486
Guadagnin DL, Zalba SM, Costa Gorriz B, Fonseca CR, Nebbia AJ, Cuevas YA, Emer C, Germain P, Wendland E, Perello LFC, Bastos MCS, Sanhueza C, Masciadri Bálsamo S, de Villalobos AE (2009) Árvores e arbustos exóticos invasores no Bioma Pampa—questões ecológicas, culturais e socioeconômicas de umdesafiocrescente. In: De Patta PV, Müller SC, de Souza ZM, Ávila Jacques AV (eds) Campos Sulinos: conservação e uso sustentable da biodiversidade. Ministerio do Meio Ambiente, Brasilia, pp 300–316
Haddad NM, Brudvig LA, Clobert J, Davies KF, Gonzalez A, Holt RD, Lovejoy TE, Sexton JO, Austin MP, Collins CD, Cook WM, Damschen EI, Ewers RM, Foster BL, Jenkins CN, King AJ, Laurance WF, Levey DJ, Margules CR, Melbourne BA, Nicholls AO, Orrock JL, Song DX, Townshend JR (2015) Habitat fragmentation and its lasting impact on Earth’s ecosystems. Sci Adv. https://doi.org/10.1126/sciadv.1500052
Heinken T, Weber E (2013) Consequences of habitat fragmentation for plant species: do we know enough? Perspect Plant Ecol Evol Syst 15:205–216
Henríquez CA (2004) Efecto de la fragmentación del hábitat sobre la calidad de las semillas en Lapageria rosea. Rev Chil de Hist Nat 77:177–184. https://doi.org/10.4067/S0716-078X2004000100013
Hoekstra JM, Boucher TM, Ricketts TH, Roberts C (2005) Confronting a biome crisis: global disparities of habitat loss and protection. Ecol Lett 8:23–29. https://doi.org/10.1111/j.1461-0248.2004.00686.x
Honnay O, Jacquemyn H (2007) Susceptibility of common and rare plant species to the genetic consequences of habitat fragmentation. Conserv Biol 21(3):823–831. https://doi.org/10.1111/j.1523-1739.2006.00646.x
Honnay O, Jacquemyn H, Bossuyt B, Hermy M (2005) Forest fragmentation effects on patch occupancy and population viability of herbaceous plant species. New Phytol 166:723–736. https://doi.org/10.1111/j.1469-8137.2005.01352.x
Hooftman DAP, van Kleunen M, Diemer M (2003) Effects of habitat fragmentation on the fitness of two common wetland species, Carex davalliana and Succisa pratensis. Oecologia 134:350–359. https://doi.org/10.1007/s00442-002-1096-0
Inouye D, Saavedra F, Lee-Yang W (2003) Environmental influences on the phenology and abundance of flowerin by Androsace septentrionalis (Primulaceae). Am J Bot 90:905–910. https://doi.org/10.3732/ajb.90.6.905
Kittlein MJ, Mora MS, Vassallo AI, de Durana F, Ricciardulli MG, Tizón FR (2004) Dunas del sureste bonaerense. In: Bilenca D, Miñarro F (eds) Identificación de Áreas Valiosas de Pastizal (AVPs) en las Pampas y Campos de Argentina, Uruguay y sur de Brasil. Fundación Vida Silvestre, Buenos Aires, pp 76–77
Lienert J (2004) Habitat fragmentation effects on fitness of plant populations—a review. J Nat Conserv 12:53–72. https://doi.org/10.1016/j.jnc.2003.07.002
López CL, Domic AI, Mayta C, García E, Gallegos SC (2018) Fenología reproductiva de la queñua (Polylepis incarum, Rosaceae) durante un ciclo anual en la puna mesofítica de La Paz, Bolivia. Ecol Austral 28:301–309
Lord JM, Norton DA (1990) Scale and the spatial concept of fragmentation. Conserv Biol 4(2):197–202
Matter SF, Roland J, Moilanen A, Hanski I (2004) Migration and survival of Parnassius smintheus: detecting effects of habitat for individual butterflies. Ecol Appl 14(5):1526–1534
McGarigal K, Cushman SA (2002) Comparative evaluation of experimental approaches to the study of habitat fragmentation effects. Ecol Appl 12(2):335–345
Monserrat AL (2010) Evaluación del estado de conservación de dunas costeras: dos escalas de análisis de la costa pampeana. Universidad de Buenos Aires, Buenos Aires
Morgan JW (1999) Effects of population size on seed production and germinability in an endangered, fragmented grassland plant. Conserv Biol 13(2):266–273
Nakagawa J (1999) Teste de vigor baseados no desempenho das plántulas. In: Krzyzanoski FC, Vieira RD, França Neto JB (eds) Vigor de sementes: conceitos e testes. ABRATES, Londrina, pp 1–24
Nora S, Albaladejo RG, González Martínez SC, Robledo-Arnuncio JJ, Aparicio A (2011) Movimiento de genes (polen y semillas) en poblaciones fragmentadas de plantas. Ecosistemas 20(2):35–45
Novara LJ (1993) Aportes Botánicos de Salta—Ser. Flora. ROSACEAE Juss. Herbario MCNS, Facultad de Ciencias Naturales, Universidad Nacional de Salta, Salta
Nuciari MC (2012) Senecio pulcher Hook & Arm., Asterácea nativa de las sierras de Tandilia con potencial ornamental. Multequina 21:39–46
Padilla Ruiz FM (2008) Factores limitantes y estrategias de establecimiento de plantas leñosas en ambientes semiáridos. Implic Para la Restaur Ecosistemas 17(1):155–159
Picasso G, Rodríguez E (2008) Bañados del Este. In: Schüttler E, Karez CS (eds) Especies exóticas invasoras en las Reservas de Biosfera de América Latina y el Caribe. UNESCO, Montevideo, pp 289–294
Pretelli MG, Isacch JP, Cardoni DA (2018) Species-area relationships of specialist versus opportunistic Pampas grassland birds depend on the surrounding landscape matrix. Ardeola 65(1):3–23
Rey PJ, Alcántara JM (2014) Effects of habitat alteration on the effectiveness of plant-avian seed dispersal mutualisms: consequences for plant regeneration. Perspect Plant Ecol Evol Syst 16:21–31. https://doi.org/10.1016/j.ppees.2013.11.001
Riginos C, Heschel MS, Schmitt J (2007) Maternal effects of drought stress and inbreeding in Impatiens capensis (Balsaminaceae). Am J Bot 94(12):1984–1991
Roach DA, Wulff RD (1987) Maternal effects in plants. Annu Rev Ecol Evol Syst 18:209–235. https://doi.org/10.1146/annurev.es.18.110187.001233
Rosch V, Tscharntke T, Scherber C, Batáry P (2013) Landscape composition, connectivity and fragment size drive effects of grassland fragmentation on insect communities. J Appl Ecol 50:387–394
Saunders DA, Hobbs RJ, Margules CR (1991) Biological consequences of ecosystem fragmentation: a review. Conserv Biol 5:18–32
Simberloff D, Nuñez MA, Ledgard NJ, Pauchard A, Richardson DM, Sarasola M, Vanwilgen B, Zalba SM, Zenni R, Bustamante R, Peña E, Ziller SR (2010) Spread and impact of introduced conifers in South America: lessons from other southern hemisphere regions. Austral Ecol 35:489–504. https://doi.org/10.1111/j.1442-9993.2009.02058.x
Soons MB, Heil GW (2002) Reduced colonization capacity in fragmented populations of wind-dispersed grassland forbs. J Ecol 90:1033–1043. https://doi.org/10.1046/j.1365-2745.2002.00729.x
Spector S, Ayzama S (2003) Rapid turnover and edge effects in dung beetle assemblages (Scarabaeidae) at a Bolivian Neotropical forest-savanna ecotone. Biotropica 35(3):394–404
Steibel P, Troiani HO (1999) Sinopsis de las Rosáceas (Rosaceae) nativas, naturalizadas y adventicias de la Provincia de La Pampa (República Argentina). Rev Fac Agron Univ Nac La Pampa 10(2):49–58
Suttie JM, Reynolds SG, Batello C (2005) Grasslands of the world. Food and Agriculture Organization of the United Nations (FAO), Rome
Torres C, Galetto L (2008) Importancia de los polinizadores en la reproducción de Asteraceae de Argentina Central. Acta Bot Venezuélica 31(2):473–494
Torres C, Galetto L (2011) Flowering phenology of co-occurring Asteraceae: a matter of climate, ecological interactions, plant attributes or of evolutionary relationships among species? Org Divers Evol 11:9–19
Wolf JB, Wade MJ (2009) What are maternal effects (and What are they not)? Phil Trans R Soc B 364:1107–1115. https://doi.org/10.1098/rstb.2008.0238
Wulff RD, Causin HF, Benitez O, Bacalini PA (1999) Intraspecific variability and maternal effects in the response to nutrient addition in Chenopodium album. Can J Bot 77(8):1150–1158. https://doi.org/10.1139/b99-124
Yezzi A, Nebbia AJ, Zalba SM (2018) Fragmentación de pastizales psamófilos por plantaciones de pinos: efectos sobre la riqueza y la composición vegetal. Ecol Austral 28(1):133–144
Yezzi A, Nebbia AJ, Zalba SM (2019) Interaction between fire and fragmentation in the successional stages of coastal dune grasslands of the southern Pampas, Argentina. Sci Rep. https://doi.org/10.1038/s41598-019-51595-x
Zalba SM, Villamil CB (2002) Woody plant invasion in relictual grasslands. Biol Invasions 4:55–72. https://doi.org/10.1023/A:1020532609792
Zar JH (1996) Biostatistical analysis. Prentice Hall, Upper Saddle River
Zardini EM (1973) Los géneros de Rosáceas espontáneos en la República Argentina. Bol Soc Argent Bot 15(2–3):209–228
Zuloaga F, Morrone O, Belgrano MJ (2019) Catálogo de las Plantas Vasculares del Cono Sur. Instituto de Botánica Darwinion. https://www2.darwin.edu.ar/proyectos/floraargentina/especies.asp. Accessed Mar 11 2019
Acknowledgements
We thank the land owners who allowed access to their properties for this study, to Mariano Kittler for his help during field work, and Dr. Pamela Geddes for the detailed review of English language. This work was supported by Universidad Nacional del Sur and CONICET (Consejo Nacional de Investigaciones Científicas y Técnicas), Argentina.
Funding
The work included in this paper received financial support from Universidad Nacional del Sur and CONICET (Consejo Nacional de Investigaciones Científicas y Técnicas), Argentina.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Data collection and experiments were performed by Alejandra L. Yezzi. All authors analyzed the results and contributed to the writing of the paper.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Michelle Tedder.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yezzi, A.L., Nebbia, A.J. & Zalba, S.M. Fragmentation and grassland plants: individual and transgenerational effects. Plant Ecol 221, 1275–1291 (2020). https://doi.org/10.1007/s11258-020-01081-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11258-020-01081-6