Abstract
Few studies have quantified changes in riparian and adjacent forest across landscape units. In this study, the composition and structure of riparian and adjacent forest were compared in a humid and a semiarid ecoregion in northwestern Argentina: the Yungas forest and the Western Chaco. We expected that differences between riparian and adjacent zones could be less marked in humid than in semiarid regions. Ten sites were surveyed with a block design. An Importance Value Index, Rank-Abundance curves, and Analysis of Similarity and multivariate analyzes (NMDS) were performed to evaluate differences between forests. Stream and floodplain widths, lateral, and longitudinal slopes of streamside were analyzed by a principal components analysis (PCA). NMDS and PCA axes were correlated to analyze the relations among physical and biological arrangements. Results revealed that riparian forest may be very different from the adjacent in both ecoregions. Marked differences in geomorphological and physical features of streamsides were found between ecoregions and they were strongly associated with assemblage distribution. In Yungas forest, dominant species were different at all sites, according to the altitudinal stratification of this region. Within Western Chaco the species Salix humboldtiana Willd. and Tessaria integrifolia Ruiz and Pav., were commonly dominant in riparian sectors. The dominance of these species in both sectors by the widest rivers could indicate that the dimensions of the riparian zone in those sites are greater than those by the smaller streams. Our study reinforced the concept of riparian zones as dynamic ecosystems and we propose considering a landscape perspective in managerial decision making.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Riparian forests are considered an interface between terrestrial and aquatic ecosystems and are among the most vulnerable environments to both climate change and human impact (Capon et al. 2013). The importance of this land–water interface has been emphasized for many reasons: they are extremely dynamic environments in terms of structure, function, and diversity, and they reinforce abiotic–biotic feedbacks (Naiman et al. 1993; Corenblit et al. 2007, 2015; Pokrovsky 2016; Pinay et al. 2018). According to Naiman et al. (2005), riparian forest is defined as the vegetation directly adjacent to rivers and streams. This forest extends laterally from the active channel to the uplands, including active floodplains and the immediately adjacent terraces. Many authors have identified characteristic vegetation within riparian zones, with different compositions, structures, and functions from that of the adjacent vegetation (Gregory et al. 1991; Naiman et al. 1993; Tang and Montgomery 1995; Prach and Straskrabová 1996; Naiman and Décamps 1997). However, few studies over the last decades have quantified changes in riparian and adjacent forest relations across landscape units or ecoregions that have marked climatic or geographic differences (Pinay et al. 1990; Naiman et al. 1992; Cattaneo et al. 1995). In recent years, there has been a renewed interest in studying the relation between riparian vegetation and hydrogeomorphological processes from new conceptual frameworks (e.g., Steiger et al. 2005; Corenblit et al. 2007, 2015), although few studies have addressed this issue from a landscape perspective (Kim and Kupfer 2016; Kujanová et al. 2018).
The characteristics and dimensions of riparian zones could change according to variations in the hydrological and geomorphological features of fluvial ecosystems (Amoros and Bornette 2002; Gurnell et al. 2016). For instance, in riparian floodplains having an irregular topography, plant communities alternate between those in depressions adapted to long flooded periods and those on elevations with species also found in uplands (Salo et al. 1986; Brinson 1990; Mertes et al. 1995; Hupp and Rinaldi 2007). Furthermore, the size and position of the stream within the watershed will influence the width of the riparian zone (Décamps 1996; Junk et al. 1989; Naiman and Décamps 1990; Salo and Cundy 1987). For example, the riparian zone may be small in the headwater streams that are surrounded by forest, while in mid-sized streams; the riparian zone is larger, composed of particular and adapted vegetation. In contrast, the riparian zones of large rivers are characterized by wider, complex, and dynamic floodplains with extended periods of seasonal flooding and a diverse vegetation (Salo et al. 1986; Malanson 1993; Naiman et al. 2005; Kujanová et al. 2018). In addition, many authors identified that “within arid and semiarid regions riparian zones act as ‘ribbons’ of organization for the surrounding landscape because of the concentration of water, nourishment, and habitats as compared to the uplands” (Malanson 1993 in Naiman et al. 2005, p. 101). Ecosystem processes in the riparian forest of arid and semiarid regions may be limited by moisture availability (Ellis et al. 2002), and flooding could result in an important natural disturbance that fosters strong responses in the inundated floodplain (Vallet et al. 2005). Conversely, the contrasts between riparian and adjacent upland microclimates and vegetative communities are much less dramatic in mesic regions (Naiman et al. 2005).
Historically, research on major rivers of the planet has focused on the interactions between the rivers and their floodplains in lowland fluvial landscapes (Amoros et al. 1987; Décamps et al. 1988; Junk et al. 1989; Naiman et al. 2005; Sioli 1984; Welcomme 1985). The most widely studied riparian system in northeastern Argentina is the lowland riparian forest of the Eastern Chaco region. In this region, different studies have recognized particular forest formations in the riverside of the fluvial ecosystems, distinct from the adjacent forest and principally composed of the species Salix humboldtiana Willd. and Tessaria integrifolia Ruiz and Pav. (Neiff 1986; Reboratti and Neiff 1987). On the other hand, in mountain streams of northwestern Argentina, increasing interest is evident, mainly in riparian forest quality and its influence on fluvial ecosystems (Sirombra and Mesa 2010, 2012; Mesa 2014; Fernández et al. 2016; Garcia et al. 2017). Sirombra and Mesa (2010) studied the riparian forest of the Yungas subtropical cloud forest ecoregion and concluded that the composition of riparian vegetation was not different from that of the adjacent forest and that the riparian vegetation of the Yungas forest was not influenced by hydrometric fluvial fluctuations. Nevertheless, further studies on riparian forest are necessary within areas minimally affected by human activities to improve the knowledge of this ecosystem in such environmental conditions, and to establish reference conditions. Studies on riparian forests within an ecoregional context are important to identify differences among riparian ecosystems related to landscape features. Furthermore, characterizing riparian forests, defining their boundaries, and identifying their changes across the landscape are also of interest for decision-makers to establish riparian buffer areas (Rasmussen et al. 2011; Hunt et al. 2017) or develop and adapt riparian quality indices (Sirombra and Mesa 2012) for different riparian landscapes or ecoregions.
The Yungas forest and Western Chaco dry forest ecoregions are important components of the regional landscape, covering a great surface and including most of the urban centers and agro-industrial activities of northwestern Argentina. For this reason, many regional studies on fluvial ecosystems and water quality have focused on those ecoregions (Domínguez and Fernández 1998; Fernández et al. 2006; Molineri et al. 2009; Pero et al. 2019). The Yungas forest is characterized by mountainous and humid environments, while the Western Chaco forest is characterized by lowland semiarid environments. Accordingly, the main objective of the present study was to analyze the composition and structure of the riparian forest within and between the humid and semiarid ecoregions mentioned. Firstly, we compared the composition and structure of tree, bush, liana, and fern species between the forest zones located next to the river and those located farther away. Secondly, we analyzed the physical variables and the physiography of the streamside to compare the geomorphology among sites. Thirdly, we compared dissimilarities in forest sectors between ecoregions to analyze variations across landscapes. We expected the water availability in riparian zones in Western Chaco to allow the establishment of plant species different from those adapted to the semiarid conditions in the adjacent forest of that ecoregion. Conversely, the high levels of humidity in the Yungas forest would diminish the differences in environmental conditions between riparian and adjacent forests, allowing the establishment of common species between them (Sirombra and Mesa 2010).
Methods
Study area
The study area is located between 26°–28°S and 66°–64°W, including most of Tucumán province and their borders with Santiago del Estero province in Northwestern Argentina (Fig. 1). The area covers a wide zone with heterogeneous landscapes containing diverse ecosystems such as deserts, mountain cloud forests, dry forests, and grasslands (Brown and Pacheco 2006). In this study, we sampled streams located in two different ecoregions: the Yungas subtropical cloud forest and the Western Chaco dry forest.
The Yungas subtropical cloud forest (Yungas forest) is a narrow belt of mountain rainforest that ranges from 400 to > 3000 m a.s.l. (Brown 2000). The Yungas forest is part of a long chain of mountain cloud forests that extends along the east side of the Andes Mountains of South America from Venezuela to northwestern Argentina. The climate is warm and humid, with mean annual temperatures ranging from 14° to 26° C and rainfall from 1000 to 2500 mm (Hueck 1978). The Yungas forest is stratified into three vegetation floors or bands. The high montane forest (1500–3000 m a.s.l.) contains monospecific tree stands that are usually either Alnus acuminata or Podocarpus parlatorei. Rainfall reaches 1000 mm. The main human activity in this area is scattered cattle and fire to maintain pastures (Brown and Pacheco 2006). The low montane forest (700–1500 m a.s.l.) has the most diverse vegetation, with many evergreen species, and is dominated by Cinnamomum porphyrium and Blepharocalyx salicifolius. The low montane forest also has the highest precipitation (2000 mm annually) and the least seasonal hydrological regime. The foothill forest (400–700 m a.s.l.) contains deciduous trees and is dominated by Tipuana tipu and Enterolobium contortisiliquum. The annual rainfall on this floor varies between 1000–1500 mm during the wet season, and the 6-month dry season (50 mm rainfall) extends from June to November (Brown et al. 2001). This area is the one, most widely modified by human activities at present, with the main urban centers and industrial activities (sugar and citrus) located in it (Brown and Pacheco 2006).
The Western Chaco ecoregion is a vast sedimentary fluvial plain formed by the streams or rivers that run northwest to southeast and includes parts of northwestern Argentina, southeastern Bolivia, northwestern Paraguay, and southwestern Brazil (Great South American Chaco). The headwaters are located in the mountains, outside the region to the west, and they transport great quantities of sediments into the region. Mean annual temperatures range between 19 °C and 24 °C. Mean annual rainfall varies between 400 and 900 mm, with most precipitation falling in the summer and little falling in the winter (Minneti 1999). The vegetation is composed of dry forests and segregated grasslands. This ecoregion is classified into three sub-ecoregions: Arid Chaco, Semiarid Chaco, and Chaco Serrano (Brown and Pacheco 2006). Only the latter two are represented in the study area. The Chaco Serrano is part of the western border of the ecoregion and is characterized by low mountain topography. It is bordered in some places by the Yungas forest. The Semiarid Chaco occupies the greater portion of the ecoregion and is a continuous xerophytic and semi-deciduous forest. A wide transition zone occurs between the Western Chaco and the Yungas forest, which includes species common in both ecoregions (Cabrera 1976), although it is currently highly modified by agricultural use (Gasparri 2016).
Sampling design and methods
Ten sites were surveyed, each consisting of a stream or river reach of around 100 m in length (Fig. 1). Four sites were located in the Yungas forest ecoregion (Apeadero Muñoz [High montane forest], Las Conchas [Low montane forest], and El Sonador streams and Pueblo Viejo river [Foothill forest]) and the other six in the Western Chaco ecoregion (Tala and Salí [Chaco Serrano] and Chico, Marapa, and two sites in Urueña river [Semiarid Chaco]) (geographic coordinates in Online Resource 1). All the sites selected were minimally impacted by human activities. Nevertheless, the sites located in Western Chaco were closer to human settlements and had some sign of cattle presence in the area, such as dung. A block design was performed to minimize the differences among sites in the analyzes (Feinsinger 2001). Three longitudinal transects randomly distributed (left or right riparian margin) and situated in a perpendicular direction from the stream or river channel were surveyed in each sampling site (Fig. 1). Each transect was divided into sampling units (SU) of 5 m in length and 1 m in width, totaling 10 SU per transect. The first four SU (0 to 20 m) were considered a priori as the “riparian forest” sectors closest to the water course and the last four (30 to 50 m) were considered the “adjacent forest” sectors distant from the water course. The middle SU (20 to 30 m) were considered a buffer area between forest sectors and were therefore not included in the analyzes (Feinsinger 2001). The composition and structure of riparian and adjacent forests were surveyed through transects, totaling 120 m2 surveyed in each site (60 m2 per forest sector). In each transect, the identity, basal area (calculated using diameter at breast height, DBH) and height of each tree, bush, liana, and fern individual were registered. Only specimens with more than 50 cm in height and 1 cm in DBH were considered. Specimens were identified to species level following the South American catalog for vascular plants (Zuloaga et al. 1994; Zuloaga and Morrone 1996; Zuloaga and Morrone 1999). All species found were listed in a table (“Appendix”). In addition, at each transect, the lateral slope of the river margins was measured using a clinometer, which was aligned between two distant objects (1-m-high sticks) every 10 m to produce a physiographic lateral view of the margins. A longitudinal slope was obtained from a digital elevation map (ASTER DEM 30 × 30 m resolution) and calculated using Geographic Information Systems (GIS) software (QGIS, 2014). The widths of the wet channel and floodplain (the area between the wet channel banks and the base of the enclosing valley walls, Naiman et al. 2005) were measured with metric ruler in each site. Site C6 was not completed and only two transects were surveyed in it due to climatic conditions during the sampling work. Site Y4 had a canyon-constrained reach and it was therefore very difficult to survey the adjacent forest sectors completely. Sampling was carried out during October 2015 and May 2016.
Data analyzes
The Importance Value Index (IVI, Lamprecht 1990) was calculated for each species in each sample site, both for the riparian forest sector and for the adjacent forest sector. The IVI was formulated using three main variables of the species (xi) in the community: density (d), dominance (D), and frequency (f), IVI xi = dxi + Dxi + f xi. First, d was calculated as the number of individuals of the species (ni) over the area surveyed in each forest sector per site (60 m2), d = ni/60 m2. Second, D was calculated as the sum of normal section area (Xi) of all stems at DBH level, D = \(\sum_{{i}=1}^{{n}}{X}_{i}\). Third, the f was calculated as the number of SU in which the species was present (ni) over the total number of SU for the corresponding forest sector in each site (N), f = ni/N. Data from the total transects in each site were added up to calculate the IVI. A ranking list of IVI value for each species was obtained for both the riparian forest sector and the adjacent forest sector in each site.
We used rank-abundance (RA) curves (dominance–diversity curves) to compare how forest structure (species abundance as basal area) varied across the different sectors and ecoregions (Feinsinger 2001). For the comparison among sites, the species abundance matrix had a datum (the sum of abundance from the three transects) for each species (columns) at each forest sector from each ecoregion (rows). For the comparison between ecoregions, we analyzed average abundance from each species by sector. RA curves, in combination with species identity, can provide insight into specific patterns of species diversity, dominance, rarity, and composition (e.g., Andresen 2005; Cultid-Medina and Escobar 2016; Vidaurre et al. 2006). We used these analyzes to complement the IVI and multivariate analyzes and allow more detailed observations of compositional and structural differences among forests.
We used the species abundance matrix to calculate a dissimilarity value by applying Bray–Curtis index (BC, Bray and Curtis 1957) to evaluate and compare the composition and structure among riparian and adjacent forest sectors of the two ecoregions. As defined by Bray and Curtis, the index of dissimilarity is \({\rm BC}_{ij} = 1- \frac{2C_{ij}}{S_{i}+S_{j}},\)where Cij is the sum of the lesser values of only those species in common between both sites. Si and Sj are the total number of specimens counted at both sites. The BC is bounded between 0 and 1, where 0 means that the two sites have the same composition, and 1 means that the two sites do not share any species. We used ANOSIM (Legendre and Legendre 1998) to determine if forest composition based on abundance data differed statistically between sectors regardless of ecoregion (riparian or adjacent) and among sectors within each ecoregion. We also used multivariate analyzes to determine if differences in forest composition among sites were associated with sectors and ecoregions. We used Non-metric Multidimensional Scaling (NMDS) based on dissimilarity values obtained from abundance data to visualize if the positions of sites in species space were concordant with sectors and ecoregions. Average dissimilarity was also compared at ecoregional level to evaluate the degree of difference between forest sectors at this scale using confidence interval (CI) 95%. Non-overlapping CIs were considered to indicate statistically significant differences among treatments (Cumming et al. 2007; MacGregor-Fors and Payton 2013).
Physical variables were analyzed by a Principal Components Analysis (PCA) using the function “dudi.pca” in the ade4 R package (version 1.7–8/Dray et al. 2017). The lateral slope for each site was calculated by averaging the lateral slopes from both streamsides. In addition, we determined if forest assemblage positions along the NMDS axes were correlated (Pearson correlation coefficients) with environmental PCA axes, and then we accounted for multiple comparisons with a Bonferroni correction (Scheiner and Gurevitch 1993). Bray–Curtis, NMDS, PCA, and Pearson Correlation analyzes were produced via the R platform (version 3.3.0 2012, R Foundation for Statistical Computing, Vienna), and IVI index and RA curves via Microsoft Office Excel 2007.
Results
Importance Value Index
Variations were registered between the IVI ranking of riparian and adjacent sectors in most of the Western Chaco sites (Fig. 2 and Online Resource 1). Within the Yungas forest, variations in IVI ranking between riparian and adjacent sectors were not evident in all sites (Fig. 3 and Online Resource 1).
Rank-Abundance curves
Rank-Abundance curves revealed changes in composition and structure between riparian and adjacent forest sectors within (Fig. 4) and between ecoregions (Fig. 5). Within Western Chaco, most of the riparian sectors were dominated mainly by two species, S. humboldtiana and T. integrifolia (Fig. 4a). These species were also dominant in some adjacent sectors. Different species were dominant in the rest of the adjacent sectors (Fig. 4b). Consequently, these species had the highest average abundance in riparian sectors of Western Chaco (Fig. 5a, b). The slope of the curves (species evenness) was similar between sectors in Western Chaco. In addition, in the Yungas forest, variations in species dominance and composition were also evidenced between riparian and adjacent sectors (Fig. 4c, d), although the dominant species were different at all the sites. Two species were present in all riparian sectors in the Yungas forest, Urera caracasana (Jacq.) Gaudich. ex Griseb. and Vernonia fulta Griseb. The slope of the curves was less steep (higher species evenness) in adjacent than in riparian sectors in most of the Yungas sites (Fig. 4d). Some species that were more commonly found in Yungas were also found in the riparian forest in Western Chaco: Heimia montana (Griseb.) Lillo, Terminalia triflora (Griseb.) Lillo, U. caracasana, and V. fulta. The slope of Western Chaco curves was steeper (lower species evenness) than that of Yungas curves. Variations were noted in the community composition and structure between transects of the same sampling site. Some species commonly found in riparian transects were less abundant or even absent in some transects. On the contrary, other species more typically present in adjacent sectors were dominant in the riparian sectors of those transects. In addition, exotic invasive species were found in riparian sectors, such as Hedychium coronarium J. König in the Yungas forest, as well as Arundo donax L., Ricinus communis L., Morus sp. L., and Tamarix ramosissima Ledeb. in Western Chaco.
Rank-Abundance curves for riparian and adjacent forest sectors in each sampling site. Western Chaco: A = Riparian sectors and B = Adjacent sectors; Yungas forest: C = Riparian sectors and D = Adjacent sectors. See species code in “Appendix”
Rank-Abundance curves with average abundance for riparian and adjacent forests in each ecoregion. Western Chaco: A = Riparian sectors and B = Adjacent sectors; Yungas forest: C = Riparian sectors and D = Adjacent sectors. See species code in “Appendix”
Dissimilarity
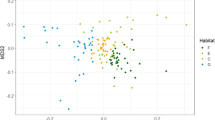
ANOSIM results (p = 0.001) showed that assemblages were significantly associated with sectors (riparian or adjacent) within each ecoregion (R = 0.50), but not with sectors in general (R = − 0.08). The overall structure of the forest assemblages was concordant with ecoregions. NMDS axis 1 segregated two groups: one composed of the Yungas assemblages and the other composed of those in Western Chaco. Within each group, an association between forest assemblages according to sectors (riparian or adjacent) was evidenced, although discrete groups were not clearly visualized (Fig. 6). Most of the sites showed high dissimilarity values between riparian and adjacent sectors (dissimilarity coefficient > 0.5), while only two sites had dissimilarity values lower than 0.5 (Table 1). In addition, at the ecoregional level, the difference between the average sector dissimilarities was slightly significant.
Physical variables and streamside physiography
Wet channel and floodplain widths were broader in Western Chaco than in Yungas forest, whereas lateral and longitudinal slopes were higher in Yungas forest than in Western Chaco (Table 2). PCA ordination of the sites using the physical variables (wet channel width, floodplain width, lateral, and longitudinal slopes) showed that sites clearly grouped following an ecoregional scheme (Fig. 7). PCA revealed that two main factors (PC-1 and PC-2) accounted for most (~ 93%) of the variation in the dataset. PC-1 (~ 76% of total variation) was negatively correlated with floodplain and wet channel widths and positively with lateral and longitudinal slopes. Western Chaco sites were located on the negative side of axis 1 (higher wet channel and floodplain widths and lower lateral and longitudinal slopes), while Yungas forest sites were situated on the positive side of the axis 1 (higher lateral and longitudinal slopes and lower wet channel and floodplain widths). These characteristics were observed and they coincided with the physiographic draft from each site (Fig. 8).
Correlations
Correlations among axes (Table 3) showed that NMDS-1 was significant and most strongly correlated with PC-1 (−) after they were adjusted for multiple comparisons (adjusted significance: p < 0.001). Thus, the segregation between the Yungas forest and Western Chaco assemblages was most strongly related to PC-1.
Discussion
Our results revealed that the riparian forest may be very different from the adjacent, mainly in species dominance. Our hypothesis that differences between riparian and adjacent zones would be less marked in humid than semiarid regions was not supported by the obtained results. However, marked differences in geomorphological and physical streamside features were found between ecoregions, and they were strongly associated with assemblage distribution. In the Yungas forest, dominant species were different at all sites, according to the altitudinal stratification of this region. On the other hand, Western Chaco showed lower slopes and the species S. humboldtiana and T. integrifolia were commonly dominant in riparian sectors, and could be considered a gallery forming corridors across the dry Chaco forest, which is similar to the gallery forest in savannas and grassland environments (Malanson 1993) or the Eastern Chaco floodplains (Neiff 1986; Reboratti and Neiff 1987). Furthermore, the dominance of these species in both riparian and adjacent sectors by the widest rivers could indicate that the dimensions of the riparian zone in those sites are greater than 50 m.
Our findings help us to understand how the relation between riparian and adjacent vegetation could vary when rivers flow through different ecoregional landscapes. Our result contrasted with a previous study of Sirombra and Mesa (2010), where only presence–absence data were used. These authors concluded that riparian vegetation in Yungas was not different from that in the adjacent forest because all species from the riparian zone had already been cited as typical of the ecoregion. Beyond this fact, the use of abundance data in our study allowed us to detect structural differences between riparian and adjacent sectors in Yungas. These results indicated that riparian vegetation could be influenced by environmental conditions near streams or rivers both in humid and semiarid regions. Therefore, riparian communities could be expected to share a set of functional traits among ecoregions regardless of their species identities. Consequently, according to Naiman et al. (2005) several studies suggested that closer to the wet channel it is common to find plants with a set of characteristics related with early successional stages, such as adaptation to low nutrient availability, and tolerance to high light levels, whereas at higher elevations, distant to the channel, vegetation communities are commonly composed of woody vascular plants with traits such as, long-lived, tolerance to shade, and usually low tolerance to long flooded periods. It would be interesting to evaluate if the differences between riparian and adjacent forests found could be related to functional aspects of the vegetation associated with the frequent occurrence of natural disturbance within riparian zones such as floods and sediment removal.
Our results reinforce the idea that geomorphology and hydrology are important factors influencing riparian characteristics. For example, the variation in species dominance and composition within Yungas is probably related to the typical altitudinal stratification of its vegetation. Altitudinal gradients also influence the geomorphic structure of riparian zones, as described by Ward et al. (2002), who observed that streams in high montane forest had typically constrained reaches, whereas streams and rivers in lowland forest had increasingly large floodplain reaches. These geomorphological and hydrological patterns could explain the presence and abundance of some hydrophilic plants in both riparian and some adjacent sectors of the Yungas foothill floor and Chaco Serrano. The presence of these species in adjacent sectors could indicate the location of old floodplains or water channels and the development of oxbows, which are water bodies typical of the braided to meandering transition zone of a river corridor, as proposed by Ward et al. (2002). Thus, riparian ecosystems in foothill forest and Chaco Serrano could be considered a transitional area, similar to that proposed by Naiman et al. (2005), who described mid-order streams as transitional areas between small streams and large rivers. In addition, rivers in Western Chaco had increasingly larger floodplain reaches, and the species that dominated their riparian forests were adapted to flooded soil conditions and survive with a long-lasting inundation phase in similar ecosystems, such as the Eastern Chaco (Casco et al. 2010). Lateral hydrologic exchange is concentrated near the river in constrained reaches, whereas it extends laterally in larger floodplains (Naiman et al. 2005). Hence, it would be important to evaluate the flooded soil/dry soil time ratio across the different ecoregions to check if this variable is related to the observed vegetation distribution.
The local site heterogeneity noted in physical and biological features between streamsides could influence the composition of the riparian biota. Some streamsides were located in a floodplain area and others in a more elevated surface or terrace. These types of streamsides could be very different in soil composition and flood conditions. Accordingly, other studies observed that variations in soil composition and flood conditions within riparian zones influenced species establishment and survival (Casco et al. 2010; Corenblit et al. 2015). Several authors have recognized the importance of geomorphic processes in shaping distributional patterns of vegetation and soil (e.g., Rot et al. 2000; Corenblit et al. 2007) and included related variables in the classification of riparian vegetation (van Coller et al. 1997). Furthermore, many authors suggested that this habitat heterogeneity within riparian forest probably influences the high biodiversity and production noted in these environments (Naiman and Décamps 1997; McClain et al. 2003). A number of studies in the Brazilian Cerrado forest, which has floristic and environmental similarities with Western Chaco, revealed a greater diversity of tree and shrub species in the riparian forest than in the adjacent Cerrado itself (Ramos 1995; Pereira et al. 1993). Thus similar diversity patterns could be found between the riparian and inner forest of the Western Chaco if a larger distance to the river is considered. On the other hand, Oliveira and Marquis (2002) proposed that this diversity pattern observed in the Brazilian Cerrado could be associated with the diverse floristic elements from which the communities of the riparian forest are derived. Consequently, we could hypothesize that the riparian forest allows some species, more commonly found in Yungas, to extend their distribution through the semiarid conditions of Western Chaco. These results also support the concept of riparian zones as biological corridors that permit the movement of species between habitats (Naiman et al. 1993; Paolino et al. 2018).
Other observations are considered important in terms of ecosystem management, such as the record of exotic invasive species in some riparian sectors, mostly in Western Chaco, some of them recently recorded for the first time in this ecoregion (Pero 2017). These findings must raise an alert, and further studies should be done on the ecological behavior of these alien species within riparian ecosystems. In addition, the knowledge obtained here about the composition of species inhabiting minimally impacted riparian forests could be useful in developing restoration programs for modified riparian ecosystems, and mainly in selecting which species to reintroduce.
The variations observed between riparian and adjacent sectors reinforced the concept of riparian zones as dynamic and diverse ecosystems (Naiman et al. 2005; Pokrovsky 2016). Furthermore, the changes across ecoregions supported that landscape features could influence the composition and structure of riparian forests. The dimensions and boundaries considered as defining these ecosystems could vary between ecoregions. These differences must be taken into account for the development and implementation of protection laws or riparian buffers. Finally, we propose that riparian forests must be studied also from a landscape perspective to improve their study, conservation, and management.
References
Amoros C, Bornette G (2002) Connectivity and biocomplexity in waterbodies of riverine floodplains. Freshw Biol 47:761–776
Amoros C, Roux AL, Reygrobeller JL, Bravard JP, Pautou C (1987) A method for applied ecological studies of fluvial hydrosystems. Regulat Rivers 1:17–38
Andresen E (2005) Effects of season and vegetation type on community organization of dung beetles in a tropical dry forest. Biotropica 37:291–300
Bray JR, Curtis JT (1957) An ordination of upland forest communities of southern Wisconsin. Ecol Monogr 27:325–349
Brinson MM (1990) Riverine forests. In: Lugo AE, Brinson MM, Brown SL (eds) Forested wetlands. Ecosystems of the world, Elsevier, Amsterdam, pp 87–141
Brown AD (2000) Development threats to biodiversity and opportunities for conservation in the mountain ranges of the upper Bermejo river basin, NW Argentina and SW Bolivia. Ambio 29:445–449
Brown AD, Pacheco S (2006) Propuesta de actualización del mapa ecorregional de la Argentina. In: Brown A, Martínez Ortiz U, Acerbi M, Corcuera J (eds) La situación ambiental argentina 2005. Fundación Vida Silvestre, Buenos Aires, pp 28–31
Brown AD, Grau HR, Malizia LR, Grau A (2001) Argentina. In: Kapelle M, Brown AD (eds) Bosques Nublados del Neotrópico. INBio, Heredia, Costa Rica, pp 623–659
Cabrera AL (1976) Regiones Fitogeográficas Argentinas. Enciclopedia Argentina de Agricultura y Jardinería, 2nd edn. Editorial Acme S.A.C.I., Buenos Aires
Capon SJ, Chambers LE, Mac Nally R, Naiman RJ, Davies P, Marshall N, Pittock J, Reid M, Capon T, Douglas M, Catford J, Baldwin DS, Stewardson M, Roberts J, Parsons M, Williams SE (2013) Riparian ecosystems in the 21th century: hotspot for climate change adaptation? Ecosystems 16:359–381
Casco SL, Neiff JJ, Poi de Neiff A (2010) Ecological responses of two pionner species to a hydrological connectivity gradient in riparian forest of the lower Paraná River. Plant Ecol 209:167–177. https://doi.org/10.1007/s11258-010-9734-9
Cattaneo A, Salmoiraghi G, Gazzera S (1995) The rivers of Italy. In: Cushing CE, Cummins KW, Minshal GW (eds) River and stream ecosystems. Elsevier, Amsterdam, pp 479–505
Corenblit D, Tabacchi E, Steiger J, Gurnell AM (2007) Reciprocal interactions and adjustments between fluvial landforms and vegetation dynamics in river corridors: a review of complementary approaches. Earth Sci Rev 84:56–86
Corenblit D, Davies NS, Steiger J, Gibling MR, Bornette G (2015) Considering river structure and stability in the light of evolution: feedbacks between riparian vegetation and hydrogeomorphology. Earth Surf Proc Land 40:189–207
Cultid-Medina CA, Escobar F (2016) Assessing the ecological response of Dung Beetles in an agricultural landscape using number of individuals and biomass in diversity measures. Environ Entomol 45:310–319
Cumming G, Fidler F, Vaux DL (2007) Error bars in experimental biology. J Cell Biol 21:7–11
Décamps H (1996) The renewal of floodplain forests along rivers: a landscape perspective. Verh Int Ver Theor Angew Limnol 26:35–59
Décamps H, Fortune M, Gazelle F, Patou G (1988) Historical influence of man on the riparian dynamics of a fluvial landscape. Landsc Ecol 1:163–173
Domínguez E, Fernández HR (1998) Calidad de los ríos de la cuenca del Salí (Tucumán, Argentina) medida por un índice biótico. Conservación de la Naturaleza, 12. Fundación Miguel Lillo, Tucumán
Dray S, Dufour AB, Thioulouse J (2017) Package ade.4. Analysis of ecological data: Exploratory and Euclidean methods in environmental sciences. R-Project 20:31:18 UTC. https://pbil.univ-lyon1.fr/ADE-4
Ellis LM, Crawford CS, Molles MC Jr (2002) The role of flood pulse in ecosystem-level processes in southwestern riparian forest: a case study from the Middle Rio Grande. In: Middleton BA (ed) Flood pulsing in wetlands: restoring the natural hydrologic balance. Wiley, New York, pp 51–107
Feinsinger P (2001) Designing field studies for biodiversity conservation. In: Moreno CE (ed) The nature conservancy. Island Press, Washington, DC
Fernández HR, Domínguez E, Romero F, Cuezzo MG (2006) La calidad del agua y la bioindicación en los ríos de montaña del Noroeste Argentino. Serie Conservación de la Naturaleza, vol 16. Fundación Miguel Lillo, Tucumán.
Fernández RD, Ceballos SJ, González Achem AL, Fernández HR, Hidalgo MV (2016) Quality and conservation of riparian forest in a Mountain Subtropical Basin of Argentina. Int J Ecol 1:1–12. https://doi.org/10.1155/2016/4842165
Garcia AK, Fernández HR, Rolandi ML, Gultemirian L, Sanchez N, Pla L, Hidalgo MV (2017) Effect of diffuse pollution on water quality in mountain forest streams. For Res Eng Int J 1(1):00001. https://doi.org/10.1155/2016/4842165
Gasparri NI (2016) The transformation of Land-Use Competition in the Argentinean Dry Chaco Between 1975 and 2015. In: Niewöhner J, Bruns A, Hostert P, Krueger T, Nielsen JØ, Haberl H, Lauk C, Lutz J, Müller D (eds) Land use competition: ecological, economics and social perspectives. Springer, Berlin, pp 59–73
Gregory SV, Swanson FV, McKee WA, Cummins KW (1991) An ecosystem perspective of riparian zones. Bioscience 41:540–551
Gurnell AM, Corenblit D, García de Jalón D, Gonzaléz del Tánago M, Grabowski RC, O’Haref MT, Szewczyk M (2016) Conceptual model of vegetation-hydrogeomorphology interactions within rivers corridors. River Res Appl 32:142–163
Hueck K (1978) Los bosques de Sudamérica. Ecología, Composición e Importancia Económica. Sociedad Alemana de Cooperación Técnica (GTZ), Berlín
Hunt L, Marrochi N, Bonetto C, Liess M, Buss DF, Vieira da Silva C, Chiu MC, Resh VH (2017) Do riparian buffer protect stream invertebrate communities in South American Atlantic forest agricultural areas? Environ Manag. https://doi.org/10.1007/s00267-017-0938-9
Hupp CR, Rinaldi M (2007) Riparian vegetation patterns in relation to fluvial landforms and channel evolution along selected rivers from Tuscany (Central Italy). Ann Assoc Am Geogr 97(1):12–30
Junk WJ, Bayley PB, Sparks RE (1989) The flood pulse concept in river-floodplain systems. In: Dodge DP (ed) Proceedings of the international large river symposium. Canadian Special Publication of Fisheries and Aquatic Science, vol 106. Department of Fisheries and Oceans, Ottawa, pp 110–127
Kim D, Kupfer JA (2016) Tri-variate relationships among vegetation, soil, and topography along gradients fluvial biogeomorphic succession. PLoS ONE 11(9):e0163223
Kujanová K, Matausková M, Hosek Z (2018) The relationship between river types and land cover in riparian zones. Limonologica 71:29–43
Lamprecht H (1990) Silvicultura en los trópicos. GTZ, Eschborn
Legendre P, Legendre L (1998) Numerical ecology. Developments in environmental modelling, 2nd ed. Elsevier, Amsterdam
MacGregor-Fors I, Payton ME (2013) Contrasting diversity values: Statistical inferences based on overlapping confidence intervals. PLoS ONE 8(2):e56794
Malanson GP (1993) Riparian Landscapes. Cambridge University Press, Cambridge
McClain ME, Boyer EW, Dent CL, Gergel SE, Grimm NB, Groffman PM, Hart SC, Harvey JW, Johnston CA, Mayorga E, McDowell WH, Pinay G (2003) Biogeochemical hot spots and hot moments at the interface of terrestrial and aquatic ecosystems. Ecosystems 6:301–312
Mertes LAK, Daniel DL, Melack JM, Nelson B, Martinelli LA, Forsberg BR (1995) Spatial patterns of hydrology, geomorphology, and vegetation on the floodplain of the Amazon River in Brazil from a remote sensing perspective. Geomorphology 13:215–232
Mesa LM (2014) Influence of riparian quality on macroinvertebrate assemblages in subtropical mountain streams. J Nat Hist 1:1–12. https://doi.org/10.1080/00222933.2013.861937
Minneti JL (1999) Atlas climático del Noroeste Argentino. Laboratorio Climatológico sudamericano, Fundación Zon Caldenius, Tucumán
Molineri C, Romero F, Fernández HR (2009) Diversidad y Conservación de Invertebrados Acuáticos. In: Brown AD, Blendinger PG, Lomáscolo T, García Bes P (eds) Selva Pedemontana de las Yungas: Historia natural, Ecología y Manejo de un Ecosistema en Peligro. Ediciones del Subtrópico, Yerba Buena
Naiman RJ, Décamps H (1990) The ecology and management of aquatic-terrestrial ecotones. UNESCO, Paris
Naiman RJ, Décamps H (1997) The ecology of interfaces: Riparian zones. Ann Rev Ecol Syst 28:621–658
Naiman RJ et al (1992) Fundamental elements of ecologically healthy watershade in the Pacific Northwest coastal ecoregion. In: Naiman RJ (ed) Watershade management: balancing sustainability and environmental change. Springer, New York, pp 127–188
Naiman RJ, Décamps H, Pollock M (1993) The role of riparian corridors in maintaining regional biodiversity. Ecol Appl 3:209–212
Naiman RJ, Décamps H, McClain ME (2005) Riparia. Ecology, conservation and management of streamside communities. Elsevier Academic Press, London.
Neiff JJ (1986) Las grandes unidades de vegetación y ambientes insular del río Paraná en el tramo Candelaria-Itá Ibaté. Rev Cienc Nat Lit 17:7–30
Oliveira PS, Marquis RJ (2002) The Cerrados of Brazil: ecology and natural history of a Neotropical savanna. Columbia University Press, New York
Paolino RM, Royle JA, Versiani NF, Rodrigues TF, Pasqualotto N, Krepschi VG, Chiarelo AG (2018) Importance of riparian forest corridors for the ocelot in agricultural landscapes. J Mammal 99(4):874–884
Pereira BAS, Silva MA, Mendonça RC (1993) Reserva ecológica do IBGE (Brasília, DF): lista das plantas vasculares. IBGE, Rio de Janeiro
Pero EJI (2017) New records of Tamarix ramosissima Ledeb. (Tamaricaceae) in basins of Western Chaco dry forest, northwestern Argentina. Check List 13:925–930
Pero EJI, Hankel G, Molineri C, Domínguez E (2019) Correspondance between stream benthic macroinvertebrates and ecoregions in northwestern Argentina. Freshw Sci 38(1):64–76. https://doi.org/10.1086/701467
Pinay G, Décamps H, Chauvet E, Fustec E (1990) Functions of ecotones in fluvial systems. In: Naiman RJ, Décamps H (eds) The ecology and management of aquatic-terrestrial ecotones. Parthenon Publishing Group, Carnforth, pp 141–169
Pinay G, Bernal S, Abbott BW, Lupon A, Marti E, Sabater F, Krause S (2018) Riparian corridors: a new conceptual framework for assessing Nitrogen buffering across biomes. Frontiers in Environmental Science 6:47
Pokrovsky OS (2016) Riparian zones: characteristics, management practices and ecological impacts. Nova Science Publishers, New York
Prach K, Straskrabová J (1996) Restoration of degraded meadows: an experimental approach. In: Prach K, Jeník J, Large ARG (eds) Floodplain ecology and management. Central Europe SPB, Academic Publishing, Amsterdam, The Lunice River in the Trebon Biosphere Reserve, pp 87–93
Quantum GIS Development Team (2014) Quantum GIS geographic information system. Open Source Geospatial Foundation Project, Chicago
R Core Team (2012) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Ramos PCM (1995) Vegetation communities and soils in the National Park of Brasília. Dissertation, University of Edinburgh
Rasmussen JJ, Baattrup-Pedersen A, Wiberg-Larsen P, McKnight US, Kronvang B (2011) Buffer strip width and agricultural pesticide contamination in Danish lowland streams: implications for stream and riparian management. Ecol Eng 37:1990–1997. https://doi.org/10.1016/j.ecoleng.2011.08.016
Reboratti HJ, Neiff JJ (1987) Distribución de los alisales de Tessaria integrifolia (Compositae) en los grandes ríos de la Cuenca del Plata. Boletín de la Sociedad Argentina de Botánica 25:25–42
Rot BWL, Naiman RJ, Bilby RE (2000) Stream channel configuration, landform, and riparian forest structure in the Cascade Mountains, Washington. Can J Fish Aquat Sci 57:699–707
Salo EO, Cundy TW (1987) Streamside management: forestry and fishery interactions. Contribution 57. Institute of Forest Resources, University of Washington, Seattle
Salo J, Kalliola R, Häkkinen I, Mäkinen Y, Niemelä P, Puhakka M, Coley PD (1986) River dynamics and the diversity of Amazon lowland forest. Nature 322:254–258
Scheiner SM, Gurevitch J (1993) Design and analysis of ecological experiments. Chapman and Hall, New York
Sioli H (1984) The Amazon: limnology and landscape ecology of a mighty tropical river and its basin. Dr. W. Junk Publishers, Dordrecht
Sirombra MG, Mesa LM (2010) Composición florística y distribución de los bosques ribereños subtropicales andinos del Río Lules, Tucumán, Argentina. Rev Biol Trop 58:499–510
Sirombra MG, Mesa LM (2012) A method for assessing the ecological quality of riparian forests in subtropical Andean streams: QBRy index. Ecol Indic 20:324–331
Steiger J, Tabacchi E, Dufour S, Corenblit D, Peiry J-L (2005) Hydrogeomorphic processes affecting riparian habitat within alluvial channel-floodplain river systems: a review for the temperate zone. River Res Appl 21:719–737
Tang SM, Montgomery DR (1995) Riparian buffers and potentially unstable ground. Environ Manag 19:741–749
Vallet HM, Baker MA, Morrice JA, Crawford CS, Molles MC Jr, Dahm CN, Moyer DL, Thibault JR, Ellis ML (2005) Biogeochemical and metabolic responses to the flood pulse in a semiarid floodplain. Ecol 86(1):220–234
van Coller AL, Rogers KH, Heritage GL (1997) Linking riparian vegetation types and fluvial geomorphology along Sabie River within Kruger National Park, South Africa. Afr J Ecol 35:194–212
Vidaurre M, Pacheco LF, Roldán AI (2006) Composition and abundance of birds of Andean alder (Alnus acuminata) patches with past and present harvest in Bolivia. Biol Conserv 132:12–21
Ward JV, Tockner K, Arscott DB, Claret C (2002) Riverine landscape diversity. Freshw Biol 46:807–819
Welcomme RL (1985) River fisheries. Food and Agriculture Organization fisheries technical paper 262. United Nations Publications, Rome
Zuloaga FO, Morrone O (1996) Catálogo de las plantas vasculares de la República Argentina. I. Pteridophyta, Gymnospermae and Angiospermae (Monocotyledoneae). Monogr Syst Bot Missouri Bot Garden 60:1–323
Zuloaga FO, Morrone O (1999) Catálogo de las plantas vasculares de la República Argentina. II. Angiospermae (Dicotyledoneae). Monogr Syst Missouri Bot Garden 64:1–1269
Zuloaga FO, Nicora EG, Rúgolo de Agrasar ZE, Morrone O, Pensiero JF, Cialdella AM (1994) Catálogo de la familia Poaceae en la República Argentina. Monogr Syst Bot Missouri Bot Garden 47:1–178
Acknowledgements
We are grateful to Sofia Malcum, Mario Feylling, Nicolas Laguna, Sebastian Albanesi, Guillermo Hankel, Dante Loto, and Carlos Navarro for their assistance in sampling trips; to Luciana Cristobal for helping to edit the image of the study area; to Sergio Georgieff, Ignacio Gasparri, Carlos Cultid, Daniel Dos Santos, and Juan Pablo Juliá for their valuable comments; to Hugo Fernández and Eduardo Domínguez for a review of the manuscript; as well as to the three anonymous reviewers for their comments and suggestions which improved the manuscript. This study was supported by fellowships of ANPCyT (National Agency of Scientific and Technological Promotion) and CONICET (National Council of Scientific Research, Argentina) and the following grants: ANPCyT PICT 1067–2012, PIP-CONICET 0330, P-UE CONICET 0099, and Universidad Nacional de Tucumán POA2-2016/05.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Shayne Martin Jacobs.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Appendix: Species list
Appendix: Species list
Species | Family | Ecoregion | Abbreviation |
|---|---|---|---|
Abutilon niveum Griseb. | Malvaceae | C | An |
Acacia aroma Gillies ex Hook. and Arn | Fabaceae | C | Aa |
Achatocarpus praecox Griseb. | Achatocarpaceae | C | Ap |
Allophylus edulis (A. St.-Hil., A. Juss. and Cambess.) Hieron. ex Niederl. | Sapindaceae | Y | Ae |
Alnus acuminata Kunth | Betulaceae | Y | Al |
Anisocapparis speciosa (Griseb.) X. Cornejo and H.H. Iltis | Capparaceae | C | As |
Arundo donax L. | Poaceae | C | Ad |
Baccharis sp. | Asteraceae | C | Ba |
Bidens sp. | Asteraceae | Y | Bi |
Blepharocalyx salicifolius (Kunth) O. Berg | Myrtaceae | Y | Bs |
Bougainvillea stipitata Griseb. | Nyctaginaceae | C | Bo |
Bulnesia foliosa Griseb. | Zygophylaceae | C | Bf |
Caesalpinia paraguariensis (D. Parodi) Burkart | Fabaceae | C | Ce |
Capparicordis tweediana (Eichler) H.H. Iltis and X. Cornejo | Capparaceae | C | Ct |
Celtis iguanaea (Jacq.) Sarg. | Celtidaceae | Y | Cg |
Celtis tala Gillies ex Planch. = Celtis ehrenbergiana (Klotzsch) Liebm. var. ehrenbergiana | Celtidaceae | C | Cet |
Cestrum strigilatum Ruiz and Pav. | Solanaceae | Y | Cst |
Chamissoa altissima (Jacq.) Kunth | Amaranthaceae | Y | Cha |
Chenopodium sp. | Chenopodiaceae | C | Ch |
Chrysophyllum marginatum (Hook. and Arn.) Radlk. | Sapotaceae | Y | Cm |
Cinnamomun porphyrium (Griseb.) Kosterm. = Ocotea porphyria (Griseb.) van der Werff | Lauraceae | Y | Cpo |
Citrus aurantium L. | Rutaceae | Y | Ci |
Croton sp. | Euphorbiaceae | C | Cr |
Cupania vernalis Cambess. | Sapindaceae | Y | Cv |
Duranta serratifolia (Griseb.) Kuntze | Verbenaceae | Ds | |
Enterolobium contortisiliquum (Vell.) Morong | Fabaceae | Y | Eco |
Ephedra sp. | Ephedraceae | C | Ep |
Equisetum giganteum L. | Equisetaceae | C, Y | Eg |
Erythrina crista-galli L. | Fabaceae | C | Ec |
Eugenia uniflora L. | Myrtaceae | Y | Eu |
Geoffroea decorticans (Gillies ex Hook. and Arn.) Burkart | Fabaceae | C | Gd |
Hedychium coronarium J. König | Zingiberaceae | Y | He |
Heimia montana (Griseb.) Lillo | Lythraceae | C, Y | Hm |
Iresine diffusa Humb. and Bonpl. ex Willd. | Asteraceae | Y | Id |
Jacaranda mimosifolia D. Don | Bignoniaceae | Y | Jm |
Juglans australis Griseb. | Juglandaceae | Y | Ja |
Justicia sp. | Acanthaceae | C, Y | Ju |
Lantana canescens Kunth | Verbenaceae | C | La |
Lippia sp. | Verbenaceae | C | Li |
Ludwigia sp. | Onagraceae | Y | Lu |
Lycium sp.1 | Solanaceae | C | Ls |
Lycium sp.2 | Solanaceae | C | LsII |
Malva sp. | Malvaceae | C | Ma |
Maytenus vitis-idaea Griseb. | Celastraceae | C | Mv |
Melia azedarach L. | Meliaceae | C | Me |
Miconia ioneura Griseb. | Melastomataceae | Y | Mi |
Morus sp. L. | Moraceae | C | Mo |
Myrcianthes mato (Griseb.) McVaugh | Myrtaceae | Y | Mm |
Myrcianthes pungens (O. Berg) D. Legrand | Myrtaceae | Y | Mp |
Nicotiana glauca Graham | Solanaceae | C | Ng |
Opuntia quimilo K. Schum. | Cactaceae | C | Oq |
Parapiptadenia excelsa (Griseb.) Burkart | Fabaceae | Y | Pe |
Phenax laevigatus Wedd. | Urticaceae | Y | Pl |
Piper hieronymi C. DC. var. hieronymi | Piperaceae | Y | Ph |
Piper tucumanum C. DC. | Piperaceae | Y | Pt |
Prosopis alba Griseb. | Fabaceae | C | Pa |
Prosopis ruscifolia Griseb. | Fabaceae | C | Pv |
Prunus tucumanensis Lillo | Rosaceae | Y | Ptu |
Psycotria carthagenensis Jacq. | Rubiaceae | Y | Pc |
Pteridophyta indet. | – | C | Pte |
Randia micracantha (Lillo) Bacigalupo | Rubiaceae | Y | Rs |
Ricinus communis L. | Euphorbiaceae | C | Rc |
Rubus imperialis Cham. and Schltdl. | Rosaceae | Y | Ri |
Ruprechtia apetala Wedd. | Polygonaceae | C | Rt |
Salix humboldtiana Willd. | Salicaceae | C | Sa |
Sapium haematospermum Müll. Arg. | Euphorbiaceae | C | Sh |
Schinus bumelioides I.M. Johnst. | Anacardiaceae | C | Sb |
Schinus fasciculatus (Griseb.) I.M. Johnst. | Anacardiaceae | C | Sf |
Schinus gracilipes I.M. Johnst. | Anacardiaceae | Y | Sg |
Senna morongii (Britton) H.S. Irwin and Barneby | Fabaceae | C | Se |
Serjania marginata Casar. | Sapindaceae | C | Sm |
Sida rhombifolia L. | Asteraceae | C | Sr |
Solanum sp. | Solanaceae | C, Y | So |
Solanum palinacanthum Dunal = S. claviceps | Solanaceae | Y | Sc |
Solanum hieronymi Kuntze | Solanaceae | C | Sp |
Solanum riparium Pers. | Solanaceae | Y | Sri |
Tamarix ramosissima Ledeb. | Tamaricaceae | C | Tr |
Terminalia triflora (Griseb.) Lillo | Combretaceae | C, Y | Tt |
Tessaria dodoneifolia (Hook. and Arn.) Cabrera | Asteraceae | C | Td |
Tessaria integrifolia Ruiz and Pav. | Asteraceae | C, Y | Ti |
Thelypteris sp. | Thelypteridaceae | Y | Th |
Tipuana tipu (Benth.) Kuntze | Fabaceae | Y | Tti |
Trema micranta (L.) Blume | Urticaceae | Y | Tm |
Urera baccifera (L.) Gaudich. | Urticaceae | Y | Ub |
Urera caracasana (Jacq.) Gaudich. ex Griseb. | Urticaceae | C, Y | Uc |
Vallesia glabra (Cav.) Link | Apocynaceae | C | Vg |
Verbesina suncho (Griseb.) S.F. Blake | Asteraceae | C, Y | Vs |
Vernonia fulta Griseb. = Quechualia fulta (Griseb.) H. Rob. | Asteraceae | C, Y | Vf |
Xylosma pubescens Griseb. | Salicaceae | Y | Xp |
Ziziphus mistol Griseb. = Sarcomphalus mistol (Griseb.) Hauenschild | Ramnaceae | C | Zm |
Rights and permissions
About this article
Cite this article
Pero, E.J.I., Quiroga, P.A. Riparian and adjacent forests differ both in the humid mountainous ecoregion and the semiarid lowland. Plant Ecol 220, 481–498 (2019). https://doi.org/10.1007/s11258-019-00929-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11258-019-00929-w