Abstract
The pantropical genus Ixora is highly diverse, with several species endemic to the Philippines. Owing to their endemic nature, many of these species are endangered and little is known about their basic biology. This study aimed to establish baseline information about the bioclimatic niches of Ixora species endemic to the Philippines, determine suitable areas and potential range shifts under future climate conditions, and identify priority areas for conservation and future research. Locality records of 12 endemic Ixora species from the Philippine archipelago were analyzed, with a particular focus on the five most abundant species I. auriculata, I. bartlingii, I. cumingiana, I. macrophylla, and one island endemic species, Ixora palawanensis. Bioclimatic variables from the WorldClim database at 2.5′ resolution were used, with a focus on annual means and seasonality of temperature and precipitation as well as precipitation of the warmest quarter. Analysis of the relationships of the species locations with the bioclimatic variables showed that the bioclimatic niches of the five focal Ixora species generally had narrow temperature and wider precipitation niches. Species distribution modeling with the model Maxent suggested that I. auriculata and I. bartlingii will likely shift their geographic distributions southwards under predicted levels of climate change, while I. cumingiana and I. macrophylla were found to likely expand their ranges. Ixora palawanensis, in contrast, was predicted to decrease its potential distribution with future climate change. Further, results of species distribution modeling for the rare endemic Ixora species I. bibracteata, I. chartacea, I. ebracteolata, I. inaequifolia, I. longistipula, I. luzoniensis, and I. macgregorii were presented, which, however, had much less observation points and therefore only provide a first estimate of potential species distributions. The generated potential habitat suitability maps can assist policy makers in designing conservation strategies for the species and in identifying areas with potential to withstand climate change until at least 2080.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Climate is considered a primary factor constraining distributions of plant species (Cox and Moore 2000; Woodward 1996; Vetaas 2002). Changes in climate are expected to affect distributions of plant species negatively if climatic conditions diverge from climatic optima (Miller and Urban 1999; Ferranini 2012). Over the coming decades, the global climate is predicted to warm significantly due to increasing greenhouse gas concentrations (Parry et al. 2007) and will most likely cause shifts in species composition of plant communities (Niu and Wan 2008; Yang and Rudolf 2010). These shifts can result from differential growth responses to higher temperature, from changes in plant interactions, or a combination of both (Llorens et al. 2004). Warming directly affects the rate of plant respiration, photosynthesis, and other biogeochemical processes and could limit plant growth directly via heat stress (White et al. 2000) or indirectly via water shortage resulting from increased evapotranspiration (De Boeck et al. 2008). All these effects may cause range shifts of various individual species (Huntley 1991; Prentice and Jolly 2000; Davis and Shaw 2001; Walther et al. 2005; Parmesan 2006; Thuiller et al. 2008; Trisurat et al. 2011) and consequently lead to changes in the composition of plant communities in a warmer environment (Llorens et al. 2004).
Future climate change is likely to intensify endangerment and extinction particularly of species with limited ranges or narrow ecological niches, and limited dispersal capabilities (Convention on Biological Diversity [CBD] 2010). Among species with narrow ranges, endemic species are of particular concern since they are confined to a particular geographic area and may have small population sizes associated with relatively high risk of extinction (Ndayishimiye et al. 2012; Fischlin et al. 2007; Vié et al. 2008). Endemic species are more likely dispersal limited than other species, and may be less able to respond rapidly to changing climate (Morueta-Holme et al. 2010). It has been estimated that 20–30 % of plant and animal species, globally, will be at higher risk of extinction due to global warming and that a significant proportion of endemic species may become extinct by 2050 or 2100 as a consequence of the increase in global mean temperatures exceeding 2–3 °C above pre-industrial levels (CBD 2010; Fischlin et al. 2007). Endemic species are particularly sensitive to climate change, as such, an understanding of climatic and ecological requirements and distribution constraints of endemic species is crucial in developing effective conservation strategies that are robust to future climate changes (Svenning and Skov 2007; Ohlemüller et al. 2008; Fløjgaard et al. 2010; Morueta-Holme et al. 2010).
The Philippines (Fig. 1) is identified as one of the world’s biologically richest countries and ranks second among the biodiversity hotspots in the world, next to Madagascar (Myers et al. 2000). The country hosts about five percent of the world’s species of flora; more than half of its 10,000 vascular and 14,000 non-vascular plants are endemic to the archipelago (Ong et al. 2002). For the Rubiaceae family alone, 83 % of the 535 species found in the Philippines are endemics (Davis et al. 2009). However, due to anthropogenic activities as well as natural disturbances, the country continues to lose its rich biodiversity resources (Conservation International [CI] 2012). Aside from anthropogenic habitat alteration, climate change has been identified as one of the major threats facing biodiversity worldwide (CBD 2010; Parry et al. 2007). In 2009, Yusuf and Francisco identified the Philippines, together with Indonesia and Malaysia, as among the climatically most vulnerable countries in Southeast Asia, due to the high exposure to frequent droughts, cyclones, landslides, and floods. As evidenced by the increasing mean temperature observed over time, climate change is apparently affecting the Philippines and future climate change will likely have a substantial impact on the Philippine flora and fauna (Philippines Atmospheric, Geophysical, and Astronomical Services Administration [PAGASA] 2011). Hence, there is a greater risk of extinction for species that are already vulnerable, particularly those with strict habitat requirements and restricted ranges (CBD 2010).
The pantropical genus Ixora L. was chosen as focus in this study for a number of reasons. The genus is almost exclusively restricted to the rain forest, which implies narrow ecological preferences and sufficiently limited dispersal potential of the genus (De Block 1998). This could mean that the endemic species of Ixora may be particularly sensitive to future climate change and calls for increased efforts to conserve these species. The genus Ixora belongs to the family Rubiaceae, which shows great species diversity across tropical Asia (Davis et al. 2009; Mouly et al. 2009), and is present in the Philippines with 25 endemic species (Banag et al. in prep.). The Philippine Ixora species are of particular interest due to their ornamental value as garden plants and their importance as a potential source of a wide variety of pharmaceuticals (Inouye et al. 1988; Wen et al. 2011; Wahab et al. 2012; Yoga Latha et al. 2012; Rajendra et al. 2013).
In 2007, the Department of Environment and Natural Resources of the Philippines (DENR Administrative Order No. [DAO] 2007-01) established the official list of threatened plant species and identified 99 critically endangered (CR) species, 187 endangered (EN) species, 176 vulnerable (VU) and 64 other threatened species (OTS) of plants in the Philippines. However, no Ixora species was included in this list despite the high number of endemic species and the continuing decline in their natural habitat. This could be attributed to the lack of basic information on the Philippine Ixora species, concerning their geographic distribution, habitat status, and ecological requirements. Knowing the current and potential distribution and understanding the responses to climate change of endemic Philippine Ixora species can inform and help policy makers to facilitate appropriate decision-making for the conservation of Ixora.
Preliminary studies on the distribution of Philippine endemic Ixora using herbarium records show that most species are single island endemics or are restricted to few islands and only few species are widely distributed across the country. To date, only three studies dealing with potential effects of climate change have been carried out in the Philippines focusing particularly on forest ecosystems (Cruz 1997; Lasco et al. 2008) and selected threatened forest tree species (Garcia et al. 2013). However, no studies have investigated potential impacts of climate change on Philippine endemic Ixora species.
With this research gap in mind, and given the potentially drastic climate change in the Philippines, our study aims to establish baseline information about bioclimatic niches of endemic Philippine Ixora species and identify the effects of future climatic conditions on Philippine endemic Ixora. In particular, our questions are: (1) How does the occurrence of endemic Ixora species relate to bioclimatic conditions? and (2) How will climate change affect the distribution of potential habitat of endemic Philippine Ixora species?
Materials and methods
The present study aims to derive information on bioclimatic requirements of 12 endemic Ixora species of the Philippines. Five focal species (I. auriculata, I. bartlingii, I. cumingiana, I. macrophylla, I. palawanensis) were selected for the main investigation, since these species had the highest number of observations (≥20) and therefore allowed to draw most robust conclusions. The remaining 7 endemic species (I. bibracteata, I. chartacea, I. ebracteolata, I. inaequifolia, I. longistipula, I. luzoniensis, and I. macgregorii) (with ≤5 observations) were investigated with the same methods, with results shown in the Online Resource 1.
The analysis began with the investigation of the distributions of species records in the climate space to provide basic information on species’ climatic niches, followed by the modeling of the potential species distributions under present and future climatic conditions with the model Maxent (Phillips et al. 2006; Phillips and Dudík 2008; Elith et al. 2011).
Study region and distribution data
Locality data of the naturally occurring Philippine Ixora were derived from three sources. Herbarium specimens were surveyed at the following herbaria: A, BK, BR, C, CAHUP, K, L, NY, P, PNH, PPC, SAN, and US (Thiers 2013). Data were also downloaded from Co’s Digital Flora of the Philippines (Pelser et al. 2011). Finally, data from the first authors’ own field surveys during 2008–2012 were included. Owing to the large spatial extent and topographic complexity of the study region, no systematic surveys are available; records were considered as presence-only data with negligible sampling bias with respect to bioclimatic conditions.
In total, 410 locality records of endemic Ixora species were collated (Online Resource 2). Records lacking coordinates were georeferenced using World Gazetteer (http://worldgazetteer.com) and Geonames (http://www.geonames.org/export/web-services.html). All coordinates were carefully checked for plausibility and/or error-corrected using Google Earth (version 7; Google 2013). Notably, a few common species (I. bartlingii, I. auriculata, I.cumingiana, I. macrophylla, I. palawanensis) dominated these records. Ixora bibracteata, I. chartacea, I. ebracteolata, I. inaequifolia, I. longistipula, I. luzoniensis, and I. macgregorii had between 5 and 16 observations each. Since Pearson et al. (2007) reported good modeling results with Maxent for as few as 5 observation points, we also analyzed the potential distribution for these species (results are shown in Fig. S2a, b, Online Resource 1).
Environmental data
For analyzing species’ distributions, bioclimatic variables from the WorldClim database at 2.5′ resolution (approx. 3.5 km in the Philippine archipelago) for the period 1950–2000 were considered (http://www.worldclim.org; see also Hijmans et al. 2005).
A preliminary analysis also explored human population density in the year 2000 (Center for International Earth Science Information Network [CIESIN] 2013) as an additional predictor. Given the known association of Ixora with undisturbed rainforests (De Block 1998), we assumed that human impact would affect the probability of species presence negatively. However, the low spatial resolution of the data (town level) and a possible sampling bias with respect to human impact (sampling concentrated near settled and accessible areas) resulted in unreasonable results indicating higher probability of presence with increasing human population density. We therefore chose to exclude effects of human presence and focus instead on bioclimatic niche space only.
In order to limit the potential effect of collinearity between bioclimatic variables which can reduce predictive power and cause difficulties in the interpretation (Dormann et al. 2013), predictor pairs with Pearson’s correlation coefficient |r| > = 0.7 were identified for the 19 bioclimatic variables from the WorldClim database (Table 1).
Since some of the endemic Ixora species included in the Maxent analysis had as few as five observation points, we chose to limit the number of bioclimatic variables to five to avoid model overfitting. Five variables with pairwise Pearson’s correlation coefficients |r| > = 0.7 were chosen that had good ecological interpretability and were highlighted by the PAGASA report (2011) on climate change in the Philippines for their future importance: (1) annual mean temperature (AMT) and (2) annual mean precipitation (AMP), as the basic variables determining the climatic niche. (3) Temperature seasonality (TS) and (4) precipitation seasonality (PS), as indicators for seasonal variability, which are of particular importance in a monsoon climate. (5) Precipitation of the warmest quarter (PwarmQ) as an indicator integrating both water and temperature effects.
We visualized the occurrence of each of the species within the climatic space by plotting their relationship between annual means, as well as seasonality values for temperature and precipitation. For better comparability, smoothed density curves were plotted for each climatic variable, using the function density (“Kernel Density Estimation” with a “SJ” smoothing bandwidth, R-internal package stats, R Development Core Team 2014). All analyses were performed using R Version 3.1.2 (R Development Core Team, 2014) with the packages raster (Hijmans 2014), dismo (Hijmans et al. 2014), rgdal (Bivand et al. 2014), sp (Pebesma and Bivand 2005), rJava (Urbanek 2013), and gplots (Warnes et al. 2013).
For future climate projections, two scenarios (A2 and B1) for the year 2080 from the GCM data portal (http://www.ccafs-climate.org, “HadCM3” model results) were chosen. These two scenarios represent a range from low (B1) to high (A2) levels of expected climate change. Scenario B1 assumes the storyline of “clean and efficient technologies” and represents an estimated increase of 1.8 °C global temperature rise. In contrast, Scenario A2 assumes the storyline of a “heterogeneous world” with “slower technological change”, resulting in an increase of 3.4 °C (at 2090–2099 compared to reference period 1980–1999; Table TS6 of Parry et al. 2007). In the Philippines, future mean temperature are predicted to rise by 0.9–1.1 °C in 2020 and by 1.8–2.2 °C in 2050, together with a change in temperature seasonality with largest temperature increases predicted for the summer season (June, July, August). Precipitation is predicted to decrease in most parts of the country during the summer season, but will increase during the southwest monsoon season. Drought conditions during the dry months of March to May are likely to become more severe and the wet months of June to November are likely to become wetter. A reduction in rainfall for all seasons is expected across Mindanao, and stronger southwest monsoon winds are anticipated for Luzon and the Visayas (PAGASA 2011). For further details of the IPCC scenarios, see Parry et al. 2007, and PAGASA 2011 for details about climate predictions for the Philippines. The maps of the five bioclimatic predictors (AMT, AMP, TS, PS, PWarmQ) for present and future conditions are shown in Figure S1 (Online Resource 1).
Species distribution modeling
Models were developed for I. auriculata, I. bartlingii, I. cumingiana, I. macrophylla and I. palawanensis using Maxent version 3.3.3 k (http://www.cs.princeton.edu/~schapire/maxent/). Maxent is a machine-learning method based on the maximum entropy algorithm (Phillips et al. 2006; Phillips and Dudík 2008; Elith et al. 2011). Maxent estimates the relative density of species occurrences, based on the environmental conditions at occurrence locations, representing the used environmental conditions and at background locations, representing the available environmental conditions (Phillips et al. 2006). It thereby provides an estimate of the species’ realized niche, and projects it into geographic space (Phillips et al. 2006). For predicting future climate scenarios, the same relationships between environmental conditions and relative species density are applied to maps of future climate, in order to estimate the species potential distribution under these new conditions.
Maxent is among the most frequently used methods for species distribution modeling and has been shown to generally perform better than many other species distribution models (Phillips et al. 2006; Elith et al. 2006; Fischer et al. 2011). Maxent has furthermore been found to have good predictive ability at low number of species records, making it a suitable model choice for the prediction of distributions for rare species (Hernandez et al. 2006; Pearson et al. 2007; Wisz et al. 2008; Garcia et al. 2013). A detailed description of Maxent is given in Phillips et al. (2006) and Phillips and Dudík (2008).
It has to be noted, that our models only consider bioclimatic variables, further potentially important aspects such as soil conditions, dispersal barriers, and biotic interactions are not taken into account.
Performance of fitted models was assessed using the area under the receiver operator curve (AUC, see also Hanley and McNeil 1982). The AUC provides a measure of a model’s discriminatory ability between suitable and unsuitable areas for a species and is a well-established approach for model evaluation (Reineking and Schröder 2006).
Maxent models were run with 5 replicates (based on the lowest number of locality records for the included Ixora species). The model settings were kept at default values, except of ‘threshold’ and ‘hinge’ features, which were disabled. Model runs were performed with a Jackknife test of variable importance (which allows to identify the variables with the most useful information by itself, Elith et al. 2011).
Since simpler models can be more appropriate for modeling range-shifting species (Elith et al. 2010), a stepwise backward selection of variables was performed for each Ixora species. Starting with all five bioclimatic predictors (AMT, AMP, TS, PS, and PWarmQ), the model was progressively simplified by removing the variable with the lowest test gain (according to the Jackknife test) until the exclusion of a variable did not further improve the Test-AUC, i.e., Test-AUC was maximized.
With these species-specific predictor sets, Maxent simulations were run for present and future climate scenarios B1 and A2 (see previous description and Online Resource S1). Model performance was evaluated with a fivefold cross validation. The result of the selection procedure and the model performance is given in Table 1 for the five main Ixora species.
Since model extrapolation into novel climates (which are outside the range of the reference set) can be problematic (Elith et al. 2010), Multivariate Environmental Similarity Surfaces (MESS) were calculated. The MESS calculation provides an index of similarity of a point to a reference set of points for a set of predictor variables (Elith et al. 2010). Sites with at least one environmental variable outside the range of environments over the reference set receive negative values, which indicate novel environments.
All R-Code used to perform the Maxent analysis, the evaluation of model performance and the MESS calculation is provided in the Online Resources 3.
Results
Climatic niche differentiation
Analysis of the relationships of the four widely distributed species (≥20 number of observations records), I. auriculata, I. bartlingii, I. cumingiana and I. macrophylla and the single island endemic I. palawanensis, with the five bioclimatic variables, mean annual temperature (AMT), mean annual precipitation (AMP), temperature seasonality (TS), precipitation seasonality (PS), and precipitation of the warmest quarter (PwarmQ), shows that the bioclimatic niches of these endemic Ixora species generally have narrow temperature and wider precipitation niches (Fig. 2). The occurrences of these species are mainly concentrated in areas where the AMT is within 23–28 °C and AMP between 1500 and 4000 mm. Of these five species, I. bartlingii and I. cumingiana show generally similar niches in respect to the five bioclimatic variables. Ixora auriculata is mainly characterized by high TS and narrow PS while I. macrophylla is distributed along a rather wide range of temperature and precipitation regimes. Among these five endemic species, I. palawanensis is shown to have the narrowest bioclimatic niches.
Scatter plots and Kernel density plots of the five climatic variables for the five endemic Philippine species of Ixora. Gray background indicates the climatic space for the entire Philippine archipelago, based on the climatic conditions within each raster cell of the Bioclim-maps. Black points and black density curves show the distribution of the respective Ixora species within bioclimatic space. Temperature seasonality is shown as standard deviation (SD), precipitation seasonality as coefficient of variation (CoV) for all plots)
Species distribution modeling
The modeling with Maxent shows the most important bioclimatic variables for predicting species distributions of the five endemic species I. auriculata, I. bartlingii, I. cumingiana, I. macrophylla, and I. palawanensis (Table 2). Among the bioclimatic variables, TS has the highest percent contribution for I. auriculata and I. bartlingii followed by PS for I. auriculata and Precipitation of the warmest quarter (PwarmQ) for I. bartlingii. The main contributor among the bioclimatic variables for I. cumingiana is PS followed by PwarmQ, while for I. macrophylla, AMP, TS and AMT show the highest percent contribution. PwarmQ and AMP are the two most important bioclimatic variables for I. palawanensis.
Model performance for the five species are in an acceptable Test-AUC range (values > 0.7). According to the results of Maxent modeling, I. macrophylla, I. cumingiana, I. bartlingii and I. auriculata are found to benefit from future climate while I. palawanensis is predicted to experience a decline of suitable habitats.
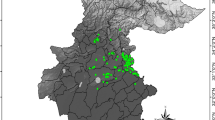
Suitable areas are identified for I. auriculata in northern Luzon and eastern Visayas (Fig. 3a, b). In the year 2080, under both climate scenarios, there will be an increase of some moderately suitable areas in Luzon and in the western and southern parts of the country (Fig. 3c, d). Current suitable climatic areas were identified for I. bartlingii mostly in central and southern Luzon and the Visayas, with isolated patches in northern Luzon (Cordillera Administrative Region) and Mindanao (Surigao del Sur) (Fig. 3e, f). However by 2080, under both climate scenarios, most areas in Luzon are predicted to be no longer suitable for the species in the future, and the model shows a general southward shift of suitable areas for I. bartlingii (Fig. 3g, h).
Maps of habitat suitability (expressed as probability of presence) of a–d Ixora auriculata, e–h Ixora bartlingii, i–l Ixora cumingiana, m–p Ixora macrophylla, q–t Ixora palawanensis under current conditions and predicted climate change conditions for the two emission scenarios (B1 and A2 from Parry et al. 2007) for 2080 using the model Maxent
The extent of suitable habitat areas for I. cumingiana and I. macrophylla are likely to increase in both climate scenarios by 2080. Some moderately suitable habitat areas for I. cumingiana (Fig. 3i, j) particularly in Zambales are predicted to become highly suitable for the species in the future. Furthermore, model results show an increase in moderately to highly suitable areas in some provinces of Mindanao for the B1 scenario but not in the more severe A2 climate scenario (Fig. 3k, l). Current suitable areas for I. macrophylla (Fig. 3m, n) are predicted to increase from moderately to highly suitable habitat for the species, particularly in the eastern part of the country with appearance of moderately suitable habitat in Mindanao for both climate scenarios (Fig. 3o, p). Ixora palawanensis is only found in Palawan and is shown to be the species that will be affected most by future climate change. Model results predict a decrease in habitat suitability on Palawan, but also show the potential of newly occurring suitable areas outside the areas (Fig. 3).
For the species with lower numbers of occurrences (and thus less robust model results), potential bioclimatic habitats tend to increase with future climate for seven species (I. bibracteata, I. chartacea, I. ebracteolata, I. inaequifolia, I. longistipula, I. luzoniensis, and I. macgregorii) as shown in Fig. S2a, b (Online Resource 1).
Similarity between current and projected climate for both B1 and A2 scenarios are analysed using Multivariate Environmental Similarity Surfaces (MESS) within Maxent (see Elith et al. 2010) and are presented in Fig. S3a, c (Online Resource 1). The highest similarity in projections is indicated for the southern part of the country in Mindanao as well as for eastern part of Luzon, whereas the lowest similarity exists for most parts of Luzon and eastern Visayas.
Discussion
Climatic niche of endemic Philippine Ixora
Our results provide for the first time an estimate of the bioclimatic relationships of the endemic Philippine Ixora species. As shown in Fig. 2, Ixora species appear to have a narrow temperature and wider precipitation niche. Because the genus Ixora is pantropical in distribution, it is not surprising that the Philippine endemic species are concentrated on areas with AMT between 23 and 28 °C. In the Philippines, essentially no difference in mean annual temperature occurs between Luzon, Visayas, or Mindanao. Based on the average of all weather stations in the Philippines, the mean annual temperature is 26.6 °C (excluding Baguio City with a mean annual temperature of 18.3 °C). January is the coolest month with a mean temperature of 25.5 °C while May is the warmest month with a mean temperature of 28.3 °C (PAGASA 2011). Thus, with respect to temperature requirements of Ixora [20–30 °C (according to Chen et al. 2003)], the Philippines provide an optimum condition for the genus.
Model results indicate an adaption of Ixora species to the monsoon seasons in the Philippines based on the species preference toward areas with higher annual precipitation seasonality. This is of particular importance as it was reported by PAGASA (2011) that the Philippines will most likely experience a substantial difference in terms of seasonal rainfall change in most parts of the country in the future, and climate change will probably lead to an active southwest monsoon in Luzon and Visayas. This will most likely be due to future increases in rainfall mostly in the months of June to August and a reduction in rainfall in most provinces during the summer season making the usually dry season drier, and increasing the likelihood of both droughts and floods (PAGASA 2011).
Looking at the individual species’ response to specific bioclimatic variables, most species showed a substantial niche overlap which suggests that Ixora species may react in a similar way to climate change. This result supports the assumption by Warren et al. (2008) that although some factors (e.g., range size, habitat and distribution) differ among species, closely related species tend to share similar climatic niches (Warren et al. 2008; Wiens et al. 2010; Pearman et al. 2014).
Information regarding the important bioclimatic variables of Philippine Ixora was to this date not available. Here, our results present a first estimate of the bioclimatic niches of endemic Ixora species with respect to AMT, AMP, TS, PS, and PwarmQ, however, it should be emphasized that these bioclimatic variables are not necessarily the only and main environmental factors determining the occurrence of Ixora in the Philippines, as species distributions are affected by further environmental factors and biotic interactions (Gaston 2003). Nevertheless, knowledge of the baseline information about climatic variables to which plants are adapted is necessary to design future experiments and model simulations investigating vulnerability of terrestrial plants to climate change. Furthermore, knowledge of the plants’ response to specific environmental conditions such as climatic variables is needed to explain their specialized traits that allow them to occupy and survive in a particular habitat (Körner 1998, 2003; Körner and Basler 2010). For the case of Rubiaceae, such studies on the ecological requirements of species are scarce and mainly restricted to Coffea (Teketay 1999). For Ixora, in situ experiments with the ornamental I. coccinea from India have revealed that floral initiation was promoted at temperatures between 10 and 20 °C and inhibited at 30 °C (Chen et al. 2003). Similar experimental approaches using the endemic Philippine species are suggested to elucidate the specific requirements of the Philippine endemic Ixora species.
Species distribution modeling
Four out of the five focal species of Ixora (I. auriculata, I. bartlingii, I. cumingiana, I. macrophylla) considered in the species distribution modeling (Fig. 3) have a wider distribution and have been recorded in several provinces across the country. Ixora palawanensis on the other hand, is considered as a single island endemic and is only found in Palawan. The other endemic species of Ixora (I. bibracteata, I. chartacea, I. ebracteolata, I. inaequifolia, I. longistipula, I. luzoniensis, and I.macgregorii, see Fig. S2a, b Online Resource 1) have limited distribution and are only found in one or few provinces or localities in the Philippines. When considering species distributions, it is important to take into account the crucial role of the areas that have been accessible to the species via dispersal over relevant periods of time (Barve et al. 2011). Given that the Ixora species included in the present study are Philippine endemics, our study considers the whole country as the area of these Ixora species and did not focus on a particular island or province.
According to the results of the Maxent modeling, the predicted current and future ranges of habitat of the endemic Ixora species are likely to be positively and negatively affected by future climate. Maxent predicted gains in suitable habitat for I. auriculata in northern parts of Luzon, in Cagayan, Isabela, and Nueva Vizcaya provinces (Fig. 3). Suitable habitats for I. bartlingii are predicted to increase in Mindanao, particularly in the provinces of Misamis Occidental, Zamboanga del Sur, and Zamboanga del Norte, but a loss of habitat in northern Luzon, in the Cordillera Administrative Region (CAR), and Pampanga, Bulacan, and Bataan provinces (Fig. 3). Both I. cumingiana and I. macrophylla are likely to experience an increase in suitable habitats in the future. Model results suggest generally a greater risk of loss of suitable habitats in Palawan for I. palawanensis.
For the species with lower numbers of occurrence points, the increasing potential habitat of the species I. bibracteata, I. ebracteolata, I. chartacea, I. inaequifolia, I. longistipula, I. luzoniensis, and I. macgregorii imply a similar positive effect of the predicted changes in climate. However, it has to be noted that due to the low sampling size, results for the species shown in the appendix are less robust and therefore need to be considered with much care. Further sampling efforts are clearly necessary in order to improve the understanding of the climatic niches of these endemic species.
Species projected to experience shrinkage or shift in geographical range under climate change are considered as the most sensitive (Midgley et al. 2003). The loss of suitable areas in Luzon and shift of potential distribution toward the western Visayas and Mindanao for I. bartlingii is an indication of the species’ sensitivity to climate change, as predicted with Maxent. Most of the predicted future suitable habitats for Philippine endemic Ixora species are found in Visayas and mostly in Mindanao. This finding is supported by recent analyses of climate projections in the Philippines, emphasizing that extreme rainfall is likely to increase in Luzon and Visayas only (PAGASA 2011). The expected decline of suitable areas in Luzon also coincides with results of a study by Yusuf and Francisco (2009), who point out that the northern Philippines will show highest vulnerability to climate change owing to exposure to tropical cyclones.
Furthermore, the key risks associated with future climate change include the occurrence of novel climates and disappearance of extant climates (Williams et al. 2007). The results of the MESS analysis reveal novel climates (portions of climate space that do not presently occur in the Philippines but may appear with future climate change) by the year 2080 that would affect the distribution of the Ixora species. Among the five focal species, I. macrophylla and I. palawanensis are predicted to be affected most by novel climates, thus conclusions about predicted range shifts need to be drawn with additional care.
In general, this study together with that of Garcia et al. (2013) on 14 threatened forest tree species, demonstrate that species distribution modeling with Maxent provides a strong tool to improve our understanding of climate related range shifts in areas like the Philippines.
Relevance to Conservation
Although scientific knowledge of the Philippine biota has improved substantially over the recent years, basic biological information for many species remains incomplete (Posa et al. 2008). These gaps become evident in the present study, as only 12 of the 25 Philippine endemic Ixora species had a sufficient number of known occurrences to permit species distribution modeling. More studies like the present one are required to understand the distributional potential of endemic species to improve information for better conservation management plans.
Despite the possible expansion of new suitable habitats for the endemic species, it is important to be aware of the likely loss of habitats in areas where species currently occur (e.g., I. palawanensis in Palawan). It is therefore necessary to establish measures that prevent the loss of the suitable habitats and to conduct future studies to ensure that these habitats can actually be occupied by the species. Aside from identifying these habitats, it is important to investigate whether the species will be able to overcome dispersal and other environmental barriers and whether they will be able to successfully compete with other species (Soberón 2007; Hirzel and Le Lay 2008; Olalla-Tarraga et al. 2011). The Philippine archipelago consists of several islands and the sea between them poses a potential dispersal barrier for successful population of suitable areas. In general, the dispersal potential of the genus Ixora is limited as their fruits are eaten by frugivorous birds and small mammals, which can only disperse the seeds within the local habitat (De Block 1998). The actual occurrence of the species may therefore deviate from the predicted suitable habitats since the model prediction did not account for landcover changes (e.g., deforestation, agriculture). Furthermore, niche saturation could prevent the species from establishing (Davis et al. 2012). Given the limited dispersal abilities of Ixora species and the archipelagic nature of the Philippines, assisted migration (Davis et al. 2012) may become a necessary future conservation strategy for Philippine endemic Ixora. However, field surveys and experiments should be conducted in advance to test for any adverse effects of introductions to a given area and to assess its probability of success.
Lastly, we hope that the assessments emerging from this study may be useful in future modeling studies and development of conservation strategies particularly for endemic species in the Philippines. Our model results can be used to assist the design of conservation strategies for the species, as they identify areas predicted to have potential to withstand climate change until at least 2080, and represent assessment priorities for in situ conservation.
Limitations of the study
Criticisms of the bioclimatic approach include the fact that many other factors beyond climate play a role in structuring species’ distributions, such as plant to plant interactions, pollinator availability, dispersal ability, and plant plasticity (Pearson and Dawson 2003; Heikkinen et al. 2006; Hijmans and Graham 2006; Oney et al. 2013). Other environmental variables that might be potentially important to Ixora like soil type, topography, vegetation type, and anthropogenic factors (in particular possible future changes in landuse) are therefore suggested to be considered in future modeling studies (Trisurat et al. 2011; Ndayishimiye et al. 2012). We therefore emphasize the need for improving the data situation at a sufficient spatial (and temporal) resolution in order to enhance model predictions for Ixora and other Philippine endemic species.
Owing to the paucity of locality records available for the Philippine endemic Ixora, only five species were considered in detail. Data points (especially of the rare species) most likely do not cover the entire niche of the species. This problem calls for a need of better primary documentation of plant distributions in the country. Thus, the resulting distribution maps should not be interpreted as representing the actual limits to the range of the species (Pearson et al. 2007), rather, these models tried to identify regions that have similar bioclimatic conditions where the species could possibly maintain populations with respect to the current and future climate conditions.
Finally, the present study did not consider the impact of population density, land use, and habitat fragmentation, so in the wake of strongly increasing land use and population in the future, the actual fraction of suitable areas is likely to be substantially lower. The improvement of the data situation, in particular in respect to species occurrence and human landuse changes, are recommended to test the implications from the present study and to draw more general conclusions on the ecology of endemic Ixora species on the Philippines.
References
Barve N, Barve V, Jimenez-Valverde A, Lira-Noriega A, Maher SP, Peterson AT, Soberon J, Villalobos F (2011) The crucial role of the accessible area in ecological niche modeling and species distribution modeling. Ecol Model 222:1810–1819
Bivand R, Keitt T, Rowlingson B (2014) rgdal: bindings for the geospatial data abstraction library. R package version 0.8-16. http://CRAN.R-project.org/package=rgdal. Accessed 08 Jan 2015
Center for International Earth Science Information Network (2013) Gridded Population of the World (GPW), v3. http://sedac.ciesin.columbia.edu/data/collection/gpw-v3. Accessed 06 Sept 2012
Chen L, Chu C, Huang M (2003) Inflorescence and flower development in Chinese Ixora. J Am Soc Hortic Sci 128:23–28
Conservation International (2012) Biodiversity hotspots: Philippines. http://www.conservation.org/How/Pages/Hotspots.aspx. Accessed 08 Jan 2015
Convention on Biological Diversity (2010) Global biodiversity outlook 3 (GBO-3). Montreal, Canada: secretariat of the convention on biological diversity. http://www.cbd.int/GBO3/. Accessed 08 Jan 2015
Cox CB, Moore PD (2000) Biogeography an ecological and evolutionary approach, 6th edn. Blackwell Science Ltd, Oxford
Cruz RVO (1997) Adaptation and mitigation measures for climate change: impacts on the forestry sector. In: Proceedings of the consultation meeting for the international conference on tropical forests and climate change. Environmental Forestry Programme, College of Forestry and Natural Resources, University of the Philippines Los Baños, College, Laguna
Davis MB, Shaw RG (2001) Range shifts and adaptive responses to quaternary climate change. Science 292:673–679
Davis AP, Govaerts R, Bridson DM, Ruhsam M, Moat J (2009) A global assessment of distribution, diversity, endemism, and taxonomic effort in the Rubiaceae. Ann Mo Bot Gard 96:68–78
Davis AP, Gole TW, Baena S, Moat J (2012) The impact of climate change on indigenous Arabica coffee (Coffea arabica): predicting future trends and identifying priorities. PLoS One. doi:10.1371/journal.pone.0047981
De Block P (1998) The African species of Ixora (Rubiaceae—Pavetteae). Opera Bot Belg 9:1–217
De Boeck HJ, Lemmens CMHM, Zavalloni C et al (2008) Biomass production in experimental grasslands of different species richness during three years of climate warming. Biogeosciences 5:585–894
Department of Environment and Natural Resources (2007) Establishing the national list of threatened Philippine plants and their categories, and the list of other wildlife species. Department of Environment and Natural Resources Administrative Order No. 2007-01, DENR, Quezon City. http://server2.denr.gov.ph/files/dao-2007-01_200.pdf. Accessed 08 Jan 2015
Dormann CF, Elith J, Bacher S, Buchmann C, Carl G, Carré G, Marquéz JRG, Gruber B, Lafourcade B, Leitão PJ, Münkemüller T, McClean C, Osborne PE, Reineking B, Schröder B, Skidmore AK, Zurell D, Lautenbach S (2013) Collinearity: a review of methods to deal with it and a simulation study evaluating their performance. Ecography 36:27–46
Elith J, Graham CH, Anderson RP, Dudik M, Ferrier S, Guisan A, Hijmans RJ, Huettmann F, Leathwick JR, Lehmann A, Li J, Lohmann LG, Loiselle BA, Manion G, Moritz C, Nakamura M, Nakazawa Y, Overton JM, Peterson AT, Phillips SJ, Richardson K, Scachetti-Pereira R, Schapire RE, Soberon J, Williams S, Wisz MS, Zimmermann NE (2006) Novel methods improve prediction of species’ distributions from occurrence data. Ecography 29:129–151
Elith J, Kearney M, Phillips S (2010) The art of modelling range-shifting species. Methods Ecol Evol 1:330–342
Elith J, Phillips SJ, Hastie T, Dudík M, Chee YE, Yates CJ (2011) A statistical explanation of MaxEnt for ecologists. Divers Distrib 17:43–57
Ferranini A (2012) I think different: models of climate warming impact on plant species are unrealistic. Environ Skept Crit 1:30–33
Fischer D, Thomas SM, Niemitz F, Reineking B, Beierkuhnlein C (2011) Projection of climatic suitability for Aedes albopictus Skuse (Culicidae) in Europe under climate change conditions. Glob Planet Change 78:54–64
Fischlin A, Midgley GF, Price JT, Leemans R, Gopal B, Turley C, et al. (2007). Ecosystems, their properties, goods and services. In: Parry ML, Canziani OF, Palutikof JP, van der Linden PJ, Hanson CE (eds.), Climate change 2007: impacts, adaptation and vulnerability. Contribution of working group II to the fourth assessment report of the intergovernmental panel of climate change. Cambridge University Press, Cambridge, pp 211e272. http://www.ipcc.ch/pdf/assessment-report/ar4/wg2/ar4-wg2-chapter4.pdf. Accessed 08 Jan 2015
Fløjgaard C, Normand S, Skov F, Svenning JC (2010) Deconstructing the mammal species richness pattern in Europe-towards an understanding of the relative importance of climate, biogeographic history, habitat heterogeneity and humans. Glob Ecol Biogeogr 20:218–230
Garcia K, Lasco R, Ines A, Lyon B, Pulhin F (2013) Predicting geographic distribution and habitat suitability due to climate change of selected threatened forest tree species in the Philippines. Appl Geogr 44:12–22
Gaston KJ (2003) The structure and dynamics of geographic ranges. Oxford University Press, Oxford
Hanley JA, McNeil BJ (1982) The meaning and use of the area under a receiver operating characteristic (ROC) curve. Radiology 143:29–36
Heikkinen RK, Luoto M, Araujo MB, Virkkala R, Thuiller W, Sykess MT (2006) Methods and uncertainties in bioclimatic envelope modelling under climate change. Prog Phys Geogr 30:1–27
Hernandez PA, Graham CH, Master LL, Albert DL (2006) The effect of sample size and species characteristics on performance of different species distribution modelling methods. Ecography 29:773–785
Hijmans RJ (2014) Raster: geographic data analysis and modeling. R package version 2.3-12. http://CRAN.R-project.org/package=raster. Accessed 08 Jan 2015
Hijmans RJ, Graham CH (2006) The ability of climate envelope models to predict the effect of climate change on species distributions. Glob Change Biol 12:2272–2281
Hijmans RJ, Cameron SE, Parra JL, Jones PG, Jarvis A (2005) Very high resolution interpolated climate surfaces for global land areas. Int J Climatol 25:1965–1978
Hijmans RJ, Phillips D, Leathwick J, Elith J (2014) dismo: species distribution modeling. R package version 1.0-5. http://CRAN.R-project.org/package=dismo. Accessed 08 January 2015
Hirzel AH, Le Lay G (2008) Habitat suitability modelling and niche theory. J Appl Ecol 45:1372–1381
Huntley B (1991) How plants respond to climate change: migration rates, individualism and the consequences for plant communities. Ann Bot 67:15–22
Warnes GR. Includes R source code and/or documentation contributed by: Bolker B, Bonebakker L, Gentleman R, Huber W, Liaw A, Lumley T, Maechler M, Magnusson A, Moeller S, Schwartz M and Venables B (2013) gplots: Various R programming tools for plotting data. R package version 2.11.0.1. http://CRAN.R-project.org/package=gplots. Accessed 08 Jan 2015
Inouye H, Takeda Y, Nishimura H, Kanomi A, Okuda T, Puff C (1988) Chemotaxonomic studies of rubiaceous plants containing iridoid glycosides. Phytochemistry 27:2591–2598
Körner C (1998) Tropical forests in a CO2-rich world. Clim Change 39:297–315
Körner C (2003) Limitation and stress—always or never? J Veg Sci 14:141–143
Körner C, Basler D (2010) Phenology under global warming. Science 327:1461–1462
Lasco R, Pulhin F, Cruz R, Roy S, Sanchez P (2008) Forest responses to changing rainfall in the Philippines. In: Leary N, Conde C, Kulkarni J, Nyong A, Pulhin J (eds) Climate change and vulnerability. Earthscan, London, pp 49–66
Llorens L, Peñuelas J, Estiarte M, Bruna P (2004) Contrasting growth changes in two dominant species of a Mediterranean shrubland submitted to experimental drought and warming. Ann Bot 94:843–853
Midgley GF, Hannah L, Millar D, Thuiller W, Booth A (2003) Developing regional and species-level assessments of climate change impacts on biodiversity in the cape floristic region. Biol Conserv 112:87–97
Miller C, Urban DL (1999) Forest pattern, fire, and climatic change in the Sierra Nevada. Ecosystems 2:76–87
Morueta-Holme N, Fløjgaard C, Svenning JC (2010) Climate change risks and conservation implications for a threatened small-range mammal species. PLoS One. doi:10.1371/journal.pone.0010360
Mouly A, Razafimandimbison S, Florence J, Jeremie J, Bremer B (2009) Paraphyly of Ixora and new tribal delimitation of Ixoreae (Rubiaceae): inference from combined chloroplast (rps16, rbcl, and trnT-F) sequence data. Ann Mo Bot Gard 96:146–160
Myers N, Mittermeier RA, Mittermeier CG, da Fonseca GAB, Kent J (2000) Biodiversity hotspots for conservation priorities. Nature 403:853–858
Ndayishimiye J, Greve M, Stoffelen P, Bigendako MJ, De Canniere C, Svenning J, Bogaert J (2012) Modelling the spatial distribution of endemic Caesalpinioideae in Central Africa, a contribution to the evaluation of actual protected areas in the region. Int J Biodivers Conserv 4:118–129
Niu S, Wan S (2008) Warming changes plant competitive hierarchy in a temperate steppe in northern China. J Plant Ecol 1:103–110
Ohlemüller R, Anderson BJ, Araújo MB, Butchart SHM, Kudrna O, Ridgely RS, Thomas CD (2008) The coincidence of climatic and species rarity: high risk to small-range species from climate change. Biol Lett 4:568–572
Olalla-Tarraga MA, McInnes L, Bini LM, Diniz-Filho JAF, Fritz SA, Hawkins BA, Hortal J, Orme CDL, Rahbeks C, Rodriguez MA, Purvis A (2011) Climatic niche conservatism and the evolutionary dynamics in species range boundaries: global congruence across mammals and amphibians. J Biogeogr 38:2237–2247
Oney B, Reineking B, O’Neill G, Kreyling J (2013) Intraspecific variation buffers projected climate change impacts on Pinus contorta. Ecol Evol 3:437–449
Ong PS, LE Afuang, Rosell Ambal RG (eds) (2002) Philippine biodiversity conservation priorities: a second iteration of the National Biodiversity Strategy and action plan. Department of Environment and Natural Resources-Protected Areas and Wildlife Bureau, Conservation International Philippines, Biodiversity Conservation Program-University of the Philippines Center for Integrative and Development Studies, and Foundation for the Philippine Environment, Quezon City
Parmesan C (2006) Ecological and evolutionary responses to recent climate change. Annu Rev Ecol Evol Syst 37:637–669
Parry ML, Canziani OF, Palutikof JP, van der Linden PJ, Hanson CE (2007) Technical Summary. In: Climate change 2007: impacts, adaptation and vulnerability. Contribution of working group II to the fourth assessment report of the intergovernmental panel on climate change. Cambridge University Press, Cambridge, pp 23–78
Pearman PB, Lavergne S, Roquet C, Wüest R, Zimmermann NE, Thuiller W (2014) Phylogenetic patterns of climatic habitat and trophic niches in a European avian assemblage. Glob Ecol Biogeogr 23:414–424
Pearson R, Dawson T (2003) Predicting the impacts of climate change on the distribution of species: are bioclimate envelope models useful? Glob Ecol Res 12:361–371
Pearson RG, Raxworthy CJ, Nakamura M, Peterson AT (2007) Predicting species distributions from small numbers of occurence records: a test case using cryptic geckos in Madagascar. J Biogeogr 34:102–117
Pebesma EJ, Bivand RS (2005) Classes and methods for spatial data in R. R News 5(2), http://cran.r-project.org/doc/Rnews/. Accessed 08 Jan 2015
Pelser PB, Barcelona JF, Nickrent DL (eds) (2011) onwards. Co’s digital flora of the Philippines. http://www.philippineplants.org. Accessed 02 June 2012
Philippine Atmospheric, Geophysical and Astronomical Services Administration (2011) Climate change in the Philippines. http://kidlat.pagasa.dost.gov.ph/climate-agromet/climate-change-in-the-philippines. Accessed 26 Nov 2012
Phillips SJ, Dudík M (2008) Modeling of species distributions with Maxent: new extensions and a comprehensive evaluation. Ecography 31:161–175
Phillips SJ, Anderson RP, Schapire RE (2006) Maximum entropy modeling of species geographic distributions. Ecol Model 190:231–259
Posa MRC, Diesmos AC, Sodhi NS, Brooks TM (2008) Hope for threatened tropical biodiversity: lessons from the Philippines. Bioscience 58:231–240
Prentice IC, Jolly D (2000) Mid-Holocene and glacial-maximum vegetation geography of the northern continents and Africa. J Biogeogr 27:507–519
R Development Core Team (2014) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Rajendra K, Nitin G, Mahavir G, Sudhir V, Mangesh K (2013) Evaluation on anti-inflammatory and antioxidant potential of Ixora coccinea, Linn ethanolic root extract. Int J Drug Dev Res 5:1–10
Reineking B, Schröder B (2006) Constrain to perform: regularization of habitat models. Ecol Model 193:675–690
Soberón J (2007) Grinellian and Eltonian niches and geographic distributions of species. Ecol Lett 10:1115–1123
Svenning JC, Skov F (2007) Ice age legacies in the geographical distribution of tree species richness in Europe. Glob Ecol Biogeogr 16:234–245
Teketay D (1999) History, botany and ecological requirements of Coffee. Walia J Ethiop Wildl Nat Hist Soc 20:28–50
Thiers B (2013) Index herbariorum: a global directory of public herbaria and associated staff. New York Botanical Garden Virtual Herbarium. http://sweetgum.nybg.org/ih/. Accessed 8 Sept 2014
Thuiller W, Albert C, Araujo MB, Berry PM, Cabeza M, Guisan A, Hickler T, Midgley GF, Paterson J, Schurr FM, Sykes MT, Zimmermann NE (2008) Predicting global change impacts on plant species distributions: future challenges. Perspect Plant Ecol Evol Syst 9:137–152
Trisurat Y, Shrestha RP, Kjelgren R (2011) Plant species vulnerability to climate change in Peninsular Thailand. Appl Geogr 31:1106–1114
Urbanek S (2013). rJava: low-level R to Java interface. R package version 0.9-6. http://CRAN.R-project.org/package=rJava. Accessed 08 Jan 2015
Vetaas OR (2002) Realized and potential climate niches: a comparison of four Rhododendron tree species. J Biogeogr 29:545–554
Vié J-C, Hilton-Taylor C, Stuart SN (2008) Wildlife in a changing world: a analysis of the 2008 IUCN red list of threatened species. International Union for Conservation of Nature, Gland
Wahab F, Subramaniam K, Suriyamoorthy S, Subburaj SP (2012) Phytochemical analysis and antagonistic activity of Ixora macrothyrsa on multidrug resistant bacteria. Asian Pac J Trop Biomed 2:1312–1316
Walther G-R, Berger S, Sykes MT (2005) An ecological ‘‘footprint’’ of climate change. Proc R Soc Lond B 272:1427–1432
Warren DL, Glor RE, Turelli M (2008) Environmental niche equivalency versus conservatism: quantitative approaches to niche evolution. Evolution 62:2868–2883
Wen K, Chiu H, Fan P, Chen C, Wu S, Chang J, Chiang H (2011) Antioxidant activity of Ixora parviflora in a cell/cell-free system and in UV-exposed human fibroblasts. Molecules 16:5735–5752
White TA, Campbell BD, Kemp PD, Hunt CL (2000) Sensitivity of three grassland communities to simulated extreme temperature and rainfall events. Glob Change Biol 6:671–684
Wiens JJ, Ackerly DD, Allen AP, Anacker BL, Buckley LB, Cornell HV, Damschen EI, Davies TJ, Grytnes JA, Harrison SP, Hawkins BA, Holt RD, Mccain CM, Stephens PR (2010) Niche conservatism as an emerging principle in ecology and conservation biology. Ecol Lett 13:1310–1324
Williams JW, Jackson ST, Kutzbach JE (2007) Projected distributions of novel and disappearing climates by 2100AD. Proc Nat Acad Sci 104:5738–5742
Wisz MS, Hijmans RJ, Li J, Peterson AT, Graham CH, Guisan A (2008) Predicting species distributions working group. Effects of sample size on the performance of species distribution models. Divers Distrib 14:763–773
Woodward FI (1996) Climate and plant distribution. Cambridge University Press, Cambridge
Yang LH, Rudolf VHW (2010) Phenology, ontogeny and the effects of climate change on the timing of species interactions. Ecol Lett 13:1–10
Yoga Latha L, Darah I, Jain K, Sasidharan S (2012) Pharmacological screening of methanolic extract of Ixora species. Asian Pac J Trop Biomed 2:149–151
Yusuf AA, Francisco HA (2009) Climate change vulnerability mapping for Southeast Asia. http://www.eepsea.org/o-k2/view-item/id-301/Itemid-385/. Accessed 26 Nov 2012
Acknowledgments
We are grateful to the following herbaria and their staff for providing loans and/or access to collections: A, BK, BR, C, CAHUP, K, L, NY, P, PNH, PPC, SAN, and US. Dr. A. Townsend Peterson, for his helpful comments in the earlier drafts of this paper. The present work is part of the doctoral dissertation of C.I. Banag, for which financial assistance was obtained from Philippine Commission on Higher Education (CHED), NAGAO Natural Environment Foundation (NEF), DAAD STIBET Grants for Doctoral Studies, and the Katholischer Akademischer Ausländer–Dienst (KAAD).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Joseph Paul Messina.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Banag, C., Thrippleton, T., Alejandro, G.J. et al. Bioclimatic niches of selected endemic Ixora species on the Philippines: predicting habitat suitability due to climate change. Plant Ecol 216, 1325–1340 (2015). https://doi.org/10.1007/s11258-015-0512-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11258-015-0512-6