Abstract
Plant–herbivore interactions and mycorrhizal symbiosis are important associations in grasslands that may interact due to their relationships with a common host plant and its resources. However, few studies have explored the effect of mycorrhizal symbiosis on plant–herbivore interactions. An understanding of the complex interactions between host plants, mycorrhizal fungi, and insect herbivores is enhanced by in situ examinations of the three parties acting simultaneously. The objective of this study was to quantify the effects of mycorrhizal symbiosis on insect herbivory and the host-plant community in tallgrass prairie. We used long-term mycorrhizal and fungicide-treated plots at Konza Prairie Biological Station (Manhattan, KS) to determine whether insect herbivory is altered by suppression of mycorrhizal fungi. Herbivory on plants in mycorrhizal control plots was greater than in fungicide-treated plots, and for one plant guild, C3 graminoids, the difference was significant. In fungicide-treated plots, plant diversity was significantly greater and herbivory was negatively correlated with plant species diversity. Differences in productivity of plant functional groups due to mycorrhizal symbiosis may be a key factor in patterns of insect herbivory. The results indicate that mycorrhizal symbiosis has diverse consequences, influencing the abundance and the quality of host plants in tallgrass prairie, as well as their responses to consumers.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In grasslands, arbuscular mycorrhizal fungi (AMF) are the most important mutualistic symbiont of plants (Hartnett and Wilson 2002), and aboveground arthropods are an important guild of herbivores. Arthropods consume a large portion of total annual primary production and are responsible for reductions in plant biomass and cover (French et al. 1979; Ritchie and Tilman 1993; Meyer et al. 2002). Mycorrhizal fungi and arthropod herbivores interact in significant and complex ways with plants (Gehring and Bennett 2009), but there is little consensus on the effect of mycorrhizal symbiosis on arthropod herbivory through their shared host plants. The effects of AMF on herbivory depends on the insect feeding breadth and habit: generalist insects and chewing insects are generally affected negatively by mycorrhizal symbiosis (Hartley and Gange 2009; Koricheva et al. 2009).
Because mycorrhizal fungi both consume photosynthate and, at the same time, enhance host-plant nutrient acquisition, these symbionts affect individual plant-level chemistry which may influence insect herbivory through multiple mechanisms. Higher nutrient levels in mycorrhizal plants translate into improved host-plant growth rate, and by increasing individual plant vigor, mycorrhizae can have positive or negative effects on herbivore performance and host-plant tolerance of and defense against herbivory (Price 1991; Waring and Cobb 1992; Bennett et al. 2006). Additionally, plants growing in symbiosis with AM fungi have increased levels of phosphorous and nitrogen, specifically (George et al. 1995; Smith and Read 1997; Gange et al. 2005; Gange and Nice 1997), and these are limiting nutrients for herbivores (Mattson 1980; Wigglesworth 1984; Perkins et al. 2004). Alternatively, an increase in host-plant C:N ratio may affect plant investment in anti-herbivore defenses (Gange and West 1994; Gehring et al. 1997), and mycorrhizal symbiosis may result in an increase (Bennett et al. 2006; Pozo and Azcon-Aguilar 2007; Gange and West 1994) or decrease (Reidinger et al. 2012) in defensive chemicals. Studies of the effect of AM symbiosis on secondary chemical composition of host plants and effects on herbivores have found conflicting results (Gehring and Bennett 2009).
AM fungi have been shown to alter plant species composition, increase dominance, and reduce plant species diversity in tallgrass prairie communities (Hartnett and Wilson 1999; Wilson et al. 2001; Hartnett and Wilson 2002), and these community-level changes provide an alternative mechanism for the effect of mycorrhizal symbiosis on herbivores (Hartley and Gange 2009). An increase in number of plant species or functional groups, plant community diversity, and total plant biomass have been reported to positively influence arthropod abundance, species diversity, and consumption (Brown and Southwood 1983; Power 1992; Siemann et al. 1999; Ritchie 2000; Haddad et al. 2001; Pfisterer et al. 2003), and grasshoppers consume more plant cover in species-rich plant communities (Pfisterer et al. 2003). Furthermore, in studies of grass species, over half found negative effects of AMF on insect herbivores (Hartley and Gange 2009).
Co-occurring host-plant species in tallgrass prairie vary significantly in their growth responsiveness to mycorrhizal colonization, which can affect their relative abundances (Wilson and Hartnett 1998). Thus, in addition to influencing host responses to herbivory at the individual plant level (Kula et al. 2005), AMF can alter plant community structure and diversity (Hartnett and Wilson 1999), which can also affect host-plant selection by herbivores.
Studying the effect of mycorrhizal fungi on herbivory within a plant community context is important (Reidinger et al. 2012). Our study makes a significant contribution because it was conducted in a field setting utilizing naturally occurring mycorrhizal fungal, plant, and herbivore communities and, therefore, places the interactions in a broader community context, as called for by Gehring and Bennett (2009) and Hartley and Gange (2009). Furthermore, using an in situ field experiment ensures that host plants are colonized by the unique suite of mycorrhizal species with which they are naturally associated (Koricheva et al. 2009). No previous studies have examined the preference of the aboveground herbivorous arthropod community for mycorrhizal status of host plants growing within intact natural plant communities (see Cahill et al. (2008) for a study of pollinator preferences for mycorrhizal or fungicide-treated plants).
The objective of this study was to quantify the effects of mycorrhizal symbiosis on insect herbivory in native tallgrass prairie communities. We used mycorrhizal and fungicide-treated plots within long-term transects to determine if insect herbivory is altered by suppression of mycorrhizal fungi. Although it has potential effects on other soil and plant microflora, our approach using the fungicide benomyl has the fewest drawbacks of all potential methods of AM fungal suppression and has been used successfully in a wide array of greenhouse and field studies (Fitter 1986; Fitter and Nichols 1988; Carey et al. 1992; Hartnett et al. 1993, 1994; Hetrick et al. 1994; Pederson and Sylvia 1997; Hartnett and Wilson 1999). Because insects often choose plants based on their nutritional quality (Mulkern 1967; Bernays and Chapman 1994), and mycorrhizal symbiosis typically increases nutrient acquisition by plants, we predicted that mycorrhizal plants will experience higher feeding damage by insect herbivores. Further we assessed mycorrhizal effects on plant abundance and community diversity because of the potential effects of plant community changes due to mycorrhizal suppression on herbivory and, therefore, explore a possible mechanism for the effects of mycorrhizae on aboveground arthropod herbivores. We predicted higher herbivory on plants in plots with higher host-plant abundance and diversity.
Methods
Study site
This study was conducted at Konza Prairie Biological Station (KPBS), a 3547 ha tallgrass prairie preserve located in the Flint Hills region of northeastern Kansas, 10-km south of Manhattan, KS. Dominant plants include C4 grasses Andropogon gerardii Vitman (big bluestem), Schizachyrium scoparium (Michx.; little bluestem), Sorghastrum nutans (L.) Nash (Indiangrass), and Panicum virgatum L. (switch grass) with many subdominant C4 and C3 graminoids, composites, legumes, and other forbs (Towne 2002). Mean annual precipitation is 835 mm (range 400–1500 mm annually), and approximately 75 % occurs during the growing season. In 2002, annual precipitation was 632 mm.
Experimental design
In 1999, six transects were established at KPBS in tallgrass prairie sites in replicate watersheds with similar soil type, slope, aspect, and previous land-use histories (1D and KIB). The transects were located on upland sites, typically with shallow soil, of the Florence series (Udic Argiustolls). Prescribed burning at KPBS began in 1972, and the transects were burned annually from 1972 until 2001. In 2002, however, watershed K1B was not burned.
Each of the six transects studied consisted of nine pairs of fungicide- or water-treated (control), 2 m × 2 m plots. We experimentally manipulated mycorrhizal fungal colonization through application of the fungicide benomyl. Benomyl is effective in reducing AM fungal colonization (Fitter and Nichols 1988; Sukarno et al. 1993; Hartnett and Wilson 2002), has minimal effects on other soil microflora in tallgrass prairie (Smith et al. 2000), does not result in significantly different results from inoculation experiments (Koricheva et al. 2009), and has no direct effects on a wide range of plants (Paul et al. 1989). In addition, several lines of evidence from studies conducted in our KPBS study site indicate that the use of benomyl is a conservative approach, and the primary, although not exclusive effect of benomyl is suppression of mutualistic mycorrhizal associations (Hetrick et al. 1986; Smith et al. 2000; Hartnett and Wilson 2002; Wilson et al. 2006; Wilson and Williamson 2008). Benomyl was applied as a soil drench (7.5 l per plot) at the rate of 1.25 g/m2 (active ingredient) to one plot in each pair bi-weekly throughout each growing season (April through October) beginning in 1999. The control plots received no fungicide, but an equivalent volume of water (7.5 l) was applied at the same time as fungicide applications.
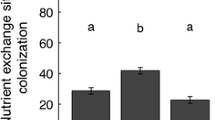
To evaluate mycorrhizal colonization, three soil cores (2.5 cm diameter × 14 cm deep) were removed during October 2002 from adjacent fungicide-treated and control plots. Roots were washed free of soil, stained with trypan blue using the method of Koske and Gemma (1989), and scored for arbuscular mycorrhizal colonization using the magnified gridline intersect method developed by McGonigle et al. (1990).
Herbivory assessment
Six study plots per transect (N = 36 plots, 18 control and 18 fungicide-treated) were sampled August 13–16, 2002 at the peak of the growing season. To assess aboveground herbivory, up to the first 25, individuals of each species present were haphazardly selected and examined for herbivory within the inner 1 m2 of each plot. For species with fewer than 25 stems, all stems were assessed. Mean herbivory level for each species in each plot was calculated. Herbivory level was determined by estimating the percent leaf area removed using a scale (0 = 0 % removed; 1 = 1–10 % removed; 2 = 11–20 %; 3 = 21–30 %; 4 = 31–40 %; 5 = 41–50; 6 = 61–75 %; 7 = 76–99 %). We employed a categorical method of herbivory assessment (rather than attempting to estimate exact percentages) to avoid investigator bias and increase the precision of the estimates, as in Kula et al. (2005). By using this type of herbivory assessment, we quantified herbivory primarily by chewing insects (e.g., grasshoppers, caterpillars).
Biomass sampling
The inner 1 m2 of 2 m × 2 m plots immediately adjacent to herbivory assessment plots were sampled so that herbivory plots were not destructively sampled. In late August 2002, after herbivory estimates were complete, all aboveground material was clipped, sorted by species, dried for at least 48 h at 60 °C and weighed. Mean biomass for each species in each plot was calculated. Plant species diversity (Shannon’s H’; see Hartnett and Wilson 1999) was determined for each plot using relative biomass as the measure of the abundance of each species.
Statistical analyses
Mixed model ANOVAs were used to examine herbivory, total standing crop biomass and diversity between mycorrhizal treatment, plant type and the interaction between mycorrhizae and plant type. The watershed by transect interaction was included as a random factor. Pairwise effects of mycorrhizae on herbivory and biomass were tested within each plant type. We also used mixed models to test the effect of mycorrhizae on feeding across plant species. Because some of the mean herbivory levels were less than 1, we added 1 to each value before inverse transforming all assessments to meet the assumption of normality for ANOVA. We tested for a correlation between feeding and both plant biomass and diversity for mycorrhizal and fungicide-treated plots separately because we wanted to test for the effect of biomass and diversity on feeding without confounding mycorrhizal effects on biomass and diversity. Species that were not encountered in all plots were not included in analyses. Differences were considered significant when P < 0.05. All statistical analyses were completed using SAS, version 9.2 (SAS Institute).
Results
Benomyl fungicide treatment successfully suppressed mycorrhizal symbiosis. Mycorrhizal colonization of fungicide-treated plots was significantly lower and approximately one-third that of control plots (Table 2).
Of the 15 species that were encountered in every plot, six species were warm season (C4) grasses (Table 1). Two cool season (C3) graminoids, Carex spp. and Dichanthelium oligosanthes, occurred in the plots. The remaining seven species were forbs (Table 1).
Herbivory
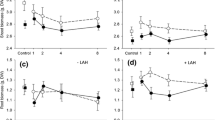
Overall, herbivory intensity was significantly higher on mycorrhizal plants (Table 2). Herbivory intensity varied significantly on individual plants across plant types (F 2,266 = 13.37, P < 0.0001). Warm season grasses and forbs experienced approximately 25 percent greater feeding levels than cool season graminoids (mean ± SE, warm season grasses = 1.12 ± 0.05, forbs = 1.01 ± 0.1, cool season graminoids = 0.768 ± 0.06). There was a significant interaction between plant type and mycorrhizal treatment for herbivory (F 2,266 = 4.01, P = 0.0192) (Fig. 1). For cool season grasses only, the difference in herbivory between mycorrhizal and fungicide-treated plants was significant (F 1,52 = 13.63, P = 0.0005). We also found large inter-specific differences in intensity of herbivory (F 14,240 = 7.79, P < 0.0001). Mean feeding levels on individual grass species ranged from 0.59 to 1.27 (Fig. 3a), and for forb species, the feeding levels ranged from 0.51 to 1.9 (Fig. 3b).
Plant community
Total standing crop biomass was not significantly different between mycorrhizal treatments (Table 2). For plant functional groups across experimental units, mean biomass of warm season grasses was more than five times greater than biomass of cool season graminoids and forbs per plot (mean ± SE, warm season grasses = 27.24 g ± 2.51, cool season graminoids = 4.59 g ± 0.54, forbs = 3.97 g ± 0.52; F 2,294 = 63.22, P < 0.0001). There was a significant interaction between mycorrhizal treatment and plant type (F 2,294 = 5.25, P = 0.0058) (Fig. 2). For warm season grasses, biomass was significantly higher in mycorrhizal plots, but for cool season graminoids and forbs, biomass was similar between plots in the two treatments (Fig. 3).
Plant species diversity differed significantly between control and fungicide-treated plots and was significantly greater in fungicide-treated plots (Table 2).
Plant community and herbivory
Some plant community characteristics were correlated with insect feeding levels. For fungicide-treated plots, insect feeding was significantly negatively correlated with plant species diversity (r = −0.473, P = 0.0473), but was not correlated with plant biomass (r = 0.220, P = 0.3810). In mycorrhizal plots, neither plant biomass (r = −0.116, P = 0.6467) nor plant species diversity (r = 0.042, P = 0.868) were correlated with herbivory level.
Discussion
Mycorrhizal fungi, host plants and arthropod herbivores are expected to interact in complex ways, but rarely have these interactions been investigated in a field setting with the natural community of aboveground herbivores and where intact plant communities with a natural suite of mycorrhizal fungi are compared to those where the fungus is reduced. We found, in the tallgrass prairie plant community, insect herbivory was greater in plots with active host-plant-mycorrhizal associations. Plant species diversity was higher in fungicide-treated plots likely due to due to suppression of the dominant, obligately mycotrophic warm-season grasses (Hartnett and Wilson 1999), but aboveground standing biomass of plants was similar between treatments. In fungicide-treated plots, herbivory was negatively correlated with plant species diversity. These results demonstrate (1) the role of mycorrhizal symbiosis in shaping plant–herbivore interactions, in particular through mycorrhizal-induced changes to plant community composition, and (2) the complexities of these multiple simultaneous interactions.
Our results are in agreement with a meta-analysis showing that chewing insect herbivory or consumption was higher on mycorrhizal host plants (Koricheva et al. 2009). There are important differences between our study and others, thereby lending to the generality of the results. Perhaps most importantly, we were interested in the response of the aboveground arthropod chewing insect herbivores as a whole. We conducted our study within tallgrass prairie where the plant community includes unequal abundance in each plant species guild. There is much natural variation in feeding preferences among host-plant species, resulting in a large range of herbivory levels among plant species, as demonstrated by our current and previous research (Kula et al. 2005). Chewing insect herbivores, in particular grasshoppers, choose to feed on host plants differentially based on their nutritional status (Behmer and Joern 2008). Other studies have experimentally demonstrated differences between insect guilds (chewing vs. sucking insects) and between species of different feeding preferences (specialists vs. generalist herbivores), whereas our study examined the integrated response of arthropod herbivores in a natural community context.
There are several possible plant community-based mechanisms that may account for the increase in feeding associated with mycorrhizal symbiosis. Plants in mycorrhizal plots experienced higher growth which likely influenced insect herbivory. The resource availability theory of plant defense predicts that plant resource acquisition and growth rates are the primary factors influencing patterns of herbivory and plant defense (Coley et al. 1985), and by influencing patterns of variability in both plant growth and nutrient status among species, mycorrhizal symbiosis may affect patterns of herbivory via changes to plant community composition. Therefore, our study provides suggestive evidence for the nutritional quantity hypothesis in which increased growth or nutrient status of host plants results in increased food quantity and higher herbivory (Bennett et al. 2006). Indeed, increases in plant biomass result in higher arthropod abundance (Brown and Southwood 1983, Power 1992, Haddad et al. 2001). Significantly higher biomass of C4 grasses in mycorrhizal plots may be especially attractive, as these grasses are usually the tallest of herbaceous tallgrass prairie plants, and grasshoppers, a large component of tallgrass prairie arthropod community, tend to orient vertically (Williams 1954).
Plant abundance is an important factor determining plant apparency (Feeny 1976; Dirzo 1984) which determines the likelihood of host-plant encounter by herbivores, and therefore plant species or functional groups with higher relative abundance should experience greater herbivory. However, insect herbivores must accept their host plants on a chemical basis after finding their potential food source. If plant species or functional types with higher abundance experience feeding levels that are lower than expected based on abundance, then plant acceptability (i.e., quality) may be low. The C3 graminoids had higher abundance in fungicide-treated plots and, therefore, these plants should have higher herbivory levels than mycorrhizal C3 graminoids. We found the opposite results, indicating that mycorrhizal C3 plants may have higher quality that results in higher than predicted herbivory compared to C3 graminoids with suppressed mycorrhizal symbiosis.
By altering plant community structure, mycorrhizal fungi may alter the attractiveness of different host-plant species to arthropod herbivores. The resource concentration hypothesis (Root 1973) predicts that insect herbivores are more likely to remain in plant patches with high density stands of individual species. We found that mycorrhizal plots, which had lower plant diversity and were dominated by a few tall C4 grass species, had higher levels of herbivory. Also, among the fungicide-treated plots there was a significant negative relationship between plant species diversity and intensity of herbivory. These patterns contrast with previous studies showing positive effects of increased plant species diversity on arthropod diversity and consumption (Siemann et al. 1999; Pfisterer et al. 2003) and suggest that the resource concentration hypothesis applies to tallgrass prairie plant–insect herbivore interactions.
Within tallgrass prairie plant communities, different plant guilds (C4 graminoids, C3 graminoids, forbs) and different species vary considerably in their growth responsiveness to mycorrhizal colonization and their dependency on the symbiosis (Wilson and Hartnett 1998). The dominant C4 grasses are mostly obligate mycotrophs with high growth responses to AMF colonization whereas C3 grasses are mostly facultative mycotrophs, and forbs show high inter-specific variation in mycotrophy. Among host-plant guilds, mycorrhizal symbiosis resulted in the greatest increase in feeding on C3 graminoids—the group with the lowest mean mycorrhizal responsiveness. Differences in host-plant mycotrophy result in different nutrient uptake and storage strategies. Bromus inermis, a C3 grass native to tallgrass prairie, stores phosphorous up to high levels without a corresponding increase in biomass, whereas A. gerardii, a C4 obligate mycotroph, has large increases in growth but maintains stable tissue P concentrations after P fertilization (Hetrick et al. 1994). Furthermore, there is evidence that herbivores perceive differences in plants with high versus low mycotrophy (Gange et al. 2002).
In addition to affecting host-selection by herbivores, mycorrhizal symbiosis may also increase host-plant tolerance to herbivory (Bennett et al. 2006), and our previous work showed that mycorrhizal suppression reduces the compensatory growth capacity of tallgrass prairie plants after defoliation, hence their herbivory tolerance (Kula et al. 2005). In the present study, the mycorrhizal plots experienced 28 % greater herbivory yet no reduction in final plant biomass compared to fungicide-treated plots, providing additional support for the hypothesis that mycorrhizal symbiosis increases host-plant tolerance to herbivory.
Conclusions
We assessed mycorrhizal effects on interactions between plants and insect herbivores in the field. We found higher herbivory on mycorrhizal plants, and a combination of improved plant quality and higher production are most likely the reasons for the feeding pattern. Dominant C4 grasses are more productive on mycorrhizal plots and four times more productive than other plant types overall. With higher productivity, insects perceive mycorrhizal communities differently and may be attracted to their structure. Significantly lower feeding on fungicide-treated C3 graminoids is likely due to changes in plant quality when mycorrhizal fungi are suppressed because there was not a significant difference in biomass between treatments. In summary, the patterns observed in this study suggest that mycorrhizal symbiosis may affect plant–herbivore interactions via multiple mechanisms including altering host-plant abundance and/or quality, host-plant species relative abundances and community structure, and host-plant herbivory tolerance. Dirzo’s (1984) phytocentric conceptual model of plant–herbivore interactions noted that different organism traits and environmental conditions can alter three processes including (a) the probability of a plant being found by a herbivore, (b) the probability of it being consumed by the herbivore, and (c) the plant’s response to consumption. Our study suggests that plant-mycorrhizal fungal associations can influence all three of these processes.
References
Behmer ST, Joern A (2008) Coexisting generalist herbivores occupy unique nutritional feeding niches. Proc Natl Acad Sci 105(6):1977–1982
Bennett AE, Alers-Garcia J, Bever JD (2006) Three-way interactions among mutualistic mycorrhizal fungi, plants, and plant enemies: hypothesis and Synthesis. Am Nat 167(2):141–152
Bernays EA, Chapman RF (1994) Host-plant selection by phytophagous insects. Chapman and Hall, New York
Brown VK, Southwood TRE (1983) Trophic diversity, niche breadth and generation times of exopterygote insects in a secondary succession. Oecologia 56:220–225
Cahill JF, Elle E, Smith GR, Shore BH (2008) Disruption of a below ground mutualism alters interactions between plants and their floral visitors. Ecology 89(7):1791–1801
Carey PD, Fitter AH, Watkinson AR (1992) A field study using the fungicide benomyl to investigate the effect of mycorrhizal fungi on plant fitness. Oecologia 90:550–555
Coley PD, Bryant JP, Chapin FS III (1985) Resource availability and plant antiherbivore defense. Science 230:895–899
Dirzo R (1984) Herbivory: a phytocentric overview. In: Dirzo R, Sarukhan J (eds) Perspectives on plant population ecology. Sinauer Associates Inc, Sunderland, pp 141–165
Feeny PP (1976) Plant apparency and chemical defense. In: Wallace J, Mansell R (eds) Biochemical interactions between plants and insects. Recent advances in phytochemistry, vol 10. Plenum Press, New York, pp 1–40
Fitter AH (1986) Effect of benomyl on leaf phosphorous concentration in alpine grassland: a test of mycorrhizal benefit. New Phytol 103:767–776
Fitter AH, Nichols R (1988) The use of benomyl to control infection by vesicular-arbuscular mycorrhizal fungi. New Phytol 110:201–206
French NR, Steinhorst RK, Swift DM (1979) Grassland biomass trophic pyramids. In: French NR (ed) Perspectives in grassland ecology. Springer-Verlag, New York, pp 59–87
Gange AC, Nice HE (1997) Performance of the thistle gall fly, Urophora cardui, in relation to host plant nitrogen and mycorrhizal colonization. New Phytol 137:335–343
Gange AC, West HM (1994) Interactions between arbuscular mycorrhizal fungi and foliar-feeding insects in Plantago lanceolata L. New Phytol 128:70–87
Gange AC, Bower E, Brown VK (2002) Differential effects of insect herbivory on arbuscular mycorrhizal colonization. Oecologia 131:103–112
Gange AC, Brown VK, Aplin DM (2005) Ecological specificity of arbuscular mycorrhizae: evidence from foliar- and seed-feeding insects. Ecology 86:603–611
Gehring C, Bennett A (2009) Mycorrhizal fungal-plant-insect interactions: the importance of a community approach. Environ Entomol 38(1):93–102
Gehring CA, Cobb NS, Whitham TG (1997) Three-way interaction among ectomycorrhizal mutualists, scale insects, and resistant and susceptible pinyon pines. Am Nat 149:824–841
George W, Marschner H, Jakobsen I (1995) Role of arbuscular mycorrhizal fungi in uptake of phosphorous and nitrogen from soil. Crit Rev Biotechnol 15:257–270
Haddad NM, Tilman D, Haarstad J, Ritchie M, Knops JMH (2001) Contrasting effects of plant richness and composition on insect communities: a field experiment. Am Nat 158:17–35
Hartley SE, Gange AC (2009) Impacts of plant symbiotic fungi on insect herbivores: mutualism in a multitrophic context. Annu Rev Entomol 54:323–342
Hartnett DC, Wilson GWT (1999) Mycorrhizae influence plant community structure and diversity in tallgrass prairie. Ecology 80:1187–1195
Hartnett DC, Wilson GWT (2002) The role of mycorrhizas in plant community structure and dynamics: lessons from grasslands. Plant Soil 244:319–331
Hartnett DC, Hetrick BAD, Wilson GWT, Gibson DJ (1993) VA-mycorrhizal influence on intra- and interspecific neighbor interactions among co-occurring prairie grasses. J Ecol 81:787–795
Hartnett DC, Samanus RJ, Fischer LE, Hetrick BAD (1994) Plant demographic responses to mycorrhizal symbiosis in tallgrass prairie. Oecologia 99:21–26
Hetrick BAD, Kitt DG, Wilson GWT (1986) The influence of phosphorus fertilization, drought, fungus species and non-sterile soil on mycorrhizal growth responses in tallgrass prairie plants. Can J Bot 54:1199–1203
Hetrick BAD, Hartnett DC, Wilson GWT, Gibson DJ (1994) Effects of mycorrhizae, phosphorous availability, and plant density on yield relationships among competing tallgrass prairie grasses. Can J Bot 72:168–176
Koricheva J, Gange AC, Jones T (2009) Effects of mycorrhizal fungi on insect herbivores: a meta-analysis. Ecology 90(8):2088–2097
Koske RE, Gemma JN (1989) A modified procedure for staining roots to detect mycorrhizas. Mycol Res 92:486–488
Kula AAR, Hartnett DC, Wilson GWT (2005) Mycorrhizal symbiosis and insect herbivory in tallgrass prairie microcosms. Ecol Lett 81:61–69
Mattson WJ (1980) Herbivory in relation to plant nitrogen content. Annu Rev Ecol Syst 11:119–161
McGonigle TP, Miller MH, Evans DG, Fairchild GL, Swan JA (1990) A new method which gives an objective measure of colonization of roots by vesicular-arbuscular mycorrhizal fungi. New Phytol 115:495–501
Meyer CK, Whiles MR, Charlton RE (2002) Life history, secondary production, and ecosystem significance of acridid grasshoppers in annually burned and unburned tallgrass prairie. Am Entomol 48(1):52–61
Mulkern GB (1967) Food selection by grasshoppers. Annu Rev Entomol 12:59–78
Paul ND, Aryes PG, Wyness LE (1989) On the use of fungicides for experimentation in natural vegetation. Funct Ecol 3:759–769
Pederson CT, Sylvia DM (1997) Limitations to using benomyl in evaluating mycorrhizal functioning. Biol Fertil Soils 25:163–168
Perkins MC, Woods HA, Harrison JF, Elser JJ (2004) Dietary phosphorus affects the growth of larval Manduca sexta. Arch Insect Biochem Physiol 55:153–168
Pfisterer AB, Diemer M, Schmid B (2003) Dietary shifts and lowered biomass gain of a generalist herbivore in species-poor experimental plant communities. Oecologia 135:234–241
Power ME (1992) Top-down and bottom-up forces in food webs: do plants have primacy? Ecology 73:733–746
Pozo MJ, Azcon-Aguilar C (2007) Unraveling mycorrhiza-induced resistance. Curr Opin Plant Biol 10:393–398
Price PW (1991) The plant vigor hypothesis and herbivore attack. Oikos 62:244–251
Reidinger S, Eschen R, Gange AC, Finch P, Martijn Bezemer T (2012) Arbuscular mycorrhizal colonization, plant chemistry, and aboveground herbivory on Senecio jacobaea. Acta Oecologia 38:8–16
Ritchie ME (2000) Nitrogen limitation and trophic vs. abiotic influences on insect herbivores in a temperate grassland. Ecology 81:1601–1612
Ritchie ME, Tilman D (1993) Predictions of species interactions from consumer-resource theory: experimental tests with grasshoppers and plants. Oecologia 94:516–527
Root RB (1973) Organization of a plant-arthropod association in simple and diverse habitats: the fauna of collards (Brassica oleracea). Ecol Monogr 43(1):95–124
Siemann E, Tilman D, Harrstad J, Ritchie M (1999) Experimental tests of the dependence of arthropod diversity on plant diversity. Am Nat 152:738–750
Smith SE, Read DJ (1997) Mycorrhizal symbiosis. Academic Press, San Diego
Smith MD, Hartnett DC, Rice CW (2000) Effects of long-term fungicide application on microbial processes in tallgrass prairie soils. Soil Biol Biochem 32:935–946
Sukarno N, Smith SE, Scott ES (1993) The effect of fungicides on vesicular-arbuscular mycorrhizal symbiosis. I. The effects on vesicular-arbuscular mycorrhizal fungi and plant growth. New Phytol 25:139–147
Towne EG (2002) Vascular plants of Konza prairie biological station: an annotated checklist of species in a kansas tallgrass prairie. Sida 20:269–294
Waring G, Cobb NS (1992) The impact of plant stress on herbivore population dynamics. In: Bernays EA (ed) Insect-plant interactions, vol 4. CRC Press, Boca Raton, pp 168–226
Wigglesworth VB (1984) Insect physiology. Chapman and Hall, New York
Williams LH (1954) The feeding habits and food preferences in Acridinae and the factors which determine them. Trans R Entomol Soc Lond 105:423–454
Wilson GWT, Hartnett DC (1998) Interspecific variation in plant responses to mycorrhizal colonization in tallgrass prairie. Am J Bot 85:1732–1738
Wilson GWT, Williamson MM (2008) Topsin-M: the new benomyl for mycorrhizal suppression experiments. Mycologia 100:548–554
Wilson GWT, Hartnett DC, Smith MD, Kobbeman K (2001) Effects of mycorrhizae on growth and demography of tallgrass prairie forbs. Am J Bot 88:1452–1457
Wilson GWT, Hartnett DC, Rice CW (2006) Mycorrhizal-mediated phosphorus transfer between the tallgrass prairie plants Sorghastrum nutans and Artemisia ludoviciana. Funct Ecol 20:427–435
Acknowledgments
We thank Gail W.T. Wilson for logistical support and for quantifying root colonization. This research was partially supported by grants from the National Science Foundation’s Division of Environmental Biology (DEB-9873654) and Long-Term Ecological Research Program (IBN-9632851).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Gavin Kernaghan.
Rights and permissions
About this article
Cite this article
Kula, A.A.R., Hartnett, D.C. Effects of mycorrhizal symbiosis on aboveground arthropod herbivory in tallgrass prairie: an in situ experiment. Plant Ecol 216, 589–597 (2015). https://doi.org/10.1007/s11258-015-0461-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11258-015-0461-0