Abstract
Purpose
Epithelioid angiomyolipoma (EAML) is a rare variant of renal angiomyolipoma with malignant potential, and the cytogenetic and clinical behavior of EAML remains a challenging issue.
Methods
We retrospectively analyze the clinical courses of five EAML, the use of everolimus on metastatic EAML, and next-generation sequencing (NGS) and polymerase chain reaction (PCR) studies to investigate the gene mutation of TSC and the impact of PI3K/Akt/mTOR signaling pathway in metastatic EAML.
Results
The mean age was 37.8 years, mean tumor size was 13 cm, all patients received radical nephrectomy, one stage IV patient received neoadjuvant mTOR inhibitor management, and one patient with high mitotic activity developed metastasis 1 year after nephrectomy. NGS assay showed a frameshift gene mutation of TSC2 in chromosome 16. PCR array for the mRNA alterations in PI3K/Akt/mTOR signaling pathway of EAML showed high expression of PIP3, AKT, TSC1, mTOR, PDK1, P70, 4E-BP1 and elF4E.
Conclusion
EAML of the kidney is a specific type of renal AML with malignant potentials, where around 22% of the patients present with invasion or metastasis. Higher mitotic activities indicate a greater metastatic potential, with radical nephrectomy as the treatment of choice, and mTOR inhibitors such as everolimus either as neoadjuvant or adjuvant targeted therapy can lead to a better clinical outcome. NGS to explore the mTOR signaling pathway may help us to better understand the pathogenesis and progression of EAML.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Renal angiomyolipoma (AML) is a benign mesenchymal tumor composed of abnormal thick-walled blood vessels, smooth muscle cells and adipose tissue [1, 2]. Most renal AMLs are sporadic, but some are associated with tuberous sclerosis complex (TSC). TSC is an autosomal dominant disorder that leads to the activation of mammalian target of rapamycin (mTOR), characterized by the development of multiple benign tumors throughout the body, with around half of the TSC patients eventually developing variable sizes of renal AMLs [3, 4]. The epithelioid angiomyolipoma of the kidney (EAML) is a rare variant of AML with malignant potential, with one-third of reported EAML cases developing metastasis [5,6,7]. Sensitivity to chemotherapy reagents remains controversial for invasive or metastatic EAML [8, 9]. Everolimus is one of the mTOR inhibitors. Apart from being used for cancer target therapy, it is also used in the treatment of TSC-related renal AML [10, 11] and some cases with EAML. The role of TSC on renal AML development is well established, demonstrating a loss of heterozygosity (LOH) of the TSC complex [12, 13]; however, the cytogenetic and metastatic features of EAML have not been extensively studied. Given the potentially poor prognosis associated with invasive EAML, the clinical and molecular studies of this rare entity is essential.
Materials and methods
With the approval from the Institutional Research Board (IRB), we retrospectively reviewed invasive EAML cases in our institution. Here we present the clinical courses of these cases, the use of everolimus on metastatic EAML, and next-generation sequencing (NGS) and polymerase chain reaction (PCR) studies to investigate the gene mutation of TSC and the impact of PI3K/Akt/mTOR signaling pathway in metastatic EAML.
DNA extraction and next-generation sequencing (NGS) analysis
Genomic DNA is extracted from tumor tissues by QIAmp DNA Mini kit (QIAGEN Science, Germantown, MD, USA) according to manufacturer’s instructions. The concentration and purity of DNA were measure using ND-1000 spectrophotometer (OD260/OD280 ratio between 1.8 and 2.0) as well as checked by electrophoresis on 1% agarose gel. Subsequently, 1.0 μg DNA per sample was used as input material for the DNA sample preparations. Sequencing libraries were generated using Truseq Nano DNA HT Sample Preparation Kit (Illumina, San Diego, CA, USA) according to manufacturer’s instructions, and index codes were added to attribute sequences to each sample. The genomic DNA is randomly fragmented to a size of 350 bp by Covaris cracker, and then DNA fragments were end-polished, A-tailed and ligated with the full-length adapter for Illumina sequencing with further PCR amplification. PCR products were purified using Agencourt AMPure XP system (Beckman Coulter Life Sciences, Indianapolis, IN, USA), and libraries were analyzed for size distribution by Agilent 2100 Bioanalyzer (Agilent Technologies, Santa Clara, CA, USA) and quantified using real-time PCR. Finally, the sequencing strategy is Illumina HiSeq 4000 (150 bp, paired-end) was applied to generate 1 Gb for each sample.
PI3K-AKT signaling pathway PCR array
Changes in PI3K-AKT-related genes expression were detected by using Human PI3K-AKT Signaling Pathway RT2 Profiler™ PCR Array (PAHS-058ZF-12; Qiagen). Firstly, total RNA from tumor tissues was extracted using the guanidinium–phenol–chloroform extraction method. Total RNA was reverse-transcribed into cDNA using RT2 First Strand Kit (Qiagen), followed by conducting qPCR using the RT2SYBR Green qPCR Master Mix (Qiagen) according to manufacturer’s instructions. The PCR was performed with Roche LightCycler 480 apparatus (Roche Applied Science, Mannheim, Germany). Finally, gene expression was analyzed by using the Web-based RT2 Profiler PCR Array Data Analysis version 3.5 software.
Case EAML-1
A 35-year-old man suffered from dry cough for weeks and a low-grade fever for 1 day. He visited our emergency department in September 2014. There was no hematuria or flank pain. Physical examination revealed a chronically ill male with a palpable left upper quadrant mass and grade 3 left varicocele. Chest and abdominal computed tomography (CT) showed multiple lung masses, a huge mediastinal mass and a 17-cm hypervascular left renal tumor (Fig. 1a, c). Laboratory tests showed that his hemoglobin was 14.9 g/dL, serum creatinine 0.73 (eGFR 122 mL/min/1.73 m2), albumin 4.7 g/dL, LDH 219 U/L, AST 25 U/L, ALT 22 U/L, ALK-P 79 U/L and calcium 9.4 mg/dL. Under the suspicion of renal tumor with mediastinum and lung metastasis, CT-guided kidney biopsy was performed which disclosed solid sheets of epithelioid tumor cells with abundant eosinophilic granular or vesicular cytoplasm, moderate pleomorphic nuclei and focal necrosis. Immunohistochemical (IHC) staining was positive for vimentin, HMB-45, Melan-A and cytokeratin (AE1/AE3); partially positive for actin, CD10; and negative for CK7, PAX-8, RCC, S-100 protein and desmin. The diagnosis of metastatic EAML was thus confirmed. The patient was then started on 10 mg everolimus daily since October 2014 with grade 1–2 mucositis and fatigue. Follow-up CT scans in January 2015 showed a decrease in size of the renal tumor (from 17 to 12 cm) and mediastinal mass (from 6.6 to 3.4 cm) and a regressive change with a decrease in size and number of bilateral lung lesions (Fig. 1b, d). A cytoreductive left nephrectomy was performed in March 2015, where the tumor mitotic count was 1–2 per 10 high-power field (HPF). Postoperative course was uneventful, and the patient restarted everolimus 1 month after nephrectomy with the dose reduced to 5 mg daily because of grade 2–3 nausea, vomiting, anorexia, fatigue, malaise and muscle aching. Follow-up chest CT showed a further decrease in size of the lung nodules. To investigate the kind of sequence variants on a genomic scale in the tumor tissue, NGS assay of the tumor region and nonmalignant kidney tissue from this patient was performed. PCR array for the mRNA alterations in PI3K/Akt/mTOR signaling pathway was determined.
Case EAML-2
A 58-year-old male presented to our hospital with chronic cough and body weight loss of more than 15 kg over the past 6 months. Imaging studies including abdominal CT and bone scans revealed a 13-cm right kidney tumor with IVC tumor thrombus, with a tentative staging of T3bN0M0. Right nephrectomy and thrombectomy with cardiopulmonary bypass were performed in April 2015. The IHC staining of the tumor was positive for HMB-45, Melan-A and vimentin, and the mitotic count was 6 per 10 HPF. Postoperative follow-up with CT scans showed free of recurrence until March 2016 where local recurrence and IVC tumor thrombosis appeared, and multiple metastases involving the liver and bone. Ten milligram everolimus daily was prescribed since then. Six months later, in October 2016, CT showed a regression of the IVC thrombus, from 18.4 to 15.9 cm, but left renal vein encasement was found.
Case EAML-3
An 18-year-old female presented with a sudden onset of diffuse abdominal pain in February 2005 where a 17-cm left renal tumor with IVC invasion was found. Left radical nephrectomy with IVC thrombectomy and cardiopulmonary bypass was performed, and postoperative recovery was uneventful. IHC staining was positive for HMB-45, smooth muscle actin and vimentin. Mitotic activity was rare. She has been regularly followed up at our hospital since then, with no recurrence till now, 12 years after surgery.
Case EAML-4
A 38-year-old female suffered from painless hematuria in July 2014 and was found to have a 4.8-cm tumor in her right kidney, where right renal vein and IVC tumor thrombi were noted. She underwent right nephrectomy and thrombectomy without cardiopulmonary bypass. IHC staining was positive for HMB-45, smooth muscle actin and vimentin, and the mitotic activity was 0–1 per 10 HPF. The patient was disease-free at the time of writing this paper.
Case EAML-5
A 40-year-old male suffered from hematuria and flank pain for 2 weeks, there was 5 kg body weight loss in 3 months, and CT image found a 12-cm left renal tumor. He received left radical nephrectomy in February 2016 that disclosed a 12.3-cm tumor with over 70% of atypical epithelioid area and left renal vein tumor thrombus (pT3aN0). The mitotic activity was 2 per 10 HPF, and necrosis area was 50% of the tumor specimen. IHC staining was positive for HMB-45, Melan-A, smooth muscle actin, and negative for AE1/AE3. Follow-up abdominal CT in October 2017 was free of recurrence.
Results
Clinical characteristics
From the chart and image reviews of 23 EAML cases in our institution, five patients (22%) presented as locally advanced or metastatic. As shown in Table 1, the mean age of these five patients was 37.8 years; the presenting symptoms include hematuria, body weight loss, cough and abdominal pain. Four-fifth of the EAML tumor was huge, with the size ranging from 12 to 17 cm. One patient was stage IV when diagnosed. All patients received radical nephrectomy, one received neoadjuvant mTOR inhibitor management, and two IVC thrombectomies were performed with cardiopulmonary bypass. One patient (EAML-2) with high mitotic activity (6 per 10 HPF in the EAML tumor and 3 per 10 HPF in the IVC tumor thrombus) had recurrence and developed multiple metastases 1 year after nephrectomy. Another patient (EAML-3) whose tumor mitotic activity was very rare had been followed up over 12 years without any evidence of recurrence.
Molecular studies
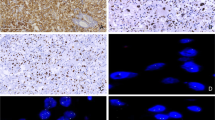
The NGS assay result for EAML-1 is shown in Table 2, where 33 genes were identified in the chromosomes, including 28 genes with single-point mutation. It is noteworthy that gene mutation of TSC2 was observed in chromosome 16 that had been represented with frameshift deletion. Meanwhile, nonsynonymous SNV at IRS2 formed stop codon, let translation of amino acid early stopped and caused dysfunction. Subsequently, we also demonstrated that deletion mutation certainly occurred in TSC2, but not in IRS by Sanger sequencing. It may be speculated that frequency of IRS mutation was too low to be detected by Sanger sequence, but only measured by NGS analysis. As shown in Fig. 2, the result of gene sequencing analysis revealed that tumor sample certainly existed with deletion of G and nucleotide C left-shift compared with that of the paired normal sample (at arrow site) (Fig. 2). PCR array for the mRNA alterations in PI3K/Akt/mTOR signaling pathway of EAML indicated that the mRNA levels of PI3K, AKT, TSC1, mTOR, PDK1, P70, 4E-BP1 and elF4E were >twofold in tumor tissue compared to the paired normal tissue in the same patient (Table 3). Among them, 4E-BP1 was 20.6-fold high expression. Our IHC staining result revealed that the level of mTOR phosphorylation in EAML-1–EAML-5 cases had higher than this in AML-1–AML-5 (Fig. 5). Using Western blot, the protein expressions of PI3K/Akt/mTOR signaling pathway-related factors, including TSC1, TSC2, p70 and S6 kinase in renal tissues of EAML-1 and two cases of renal AML, are illustrated in Fig. 4. The renal TSC1 and TSC2 expressions were reduced, but S6K and total phosphorylated forms of p70 were increased in the tumor region of EAML-1 compared with normal sample from the same case. This suggests that mTOR inhibitors may provide therapeutic benefit in the treatment of invasive or metastatic EAML by upregulation of 4E-BP1 expression.
Discussion
Renal AML is a common benign neoplasm characterized by abnormal thick-walled blood vessels, smooth muscles and adipose tissue. Most renal AMLs are sporadic and asymptomatic if the tumor size is small. As the tumor grows, it can cause serious problems such as bleeding because of its abnormal vasculature and can lead to the need for immediate management such as nephrectomy, partial nephrectomy or transcatheter arterial embolization (TAE) [14, 15]. Tuberous sclerosis complex (TSC) is an autosomal dominant disorder characterized by multiple benign tumors throughout the body, including the brain, skin, lungs and kidneys. Renal manifestations in TSC cause significant burdens. In contrast to classical AMLs, epithelioid AML (EAML), one of the AML variants, has a distinct common morphology and immunohistochemical features [16]. In 2004, the World Health Organization (WHO) classification of renal neoplasms defines EAML as a potentially malignant mesenchymal neoplasm, characterized by the proliferation of predominantly epithelioid cells with metastatic potentials. EAML behaves much more aggressively and is capable of malignant transformation, recurrence, metastasis and fatal outcome. Most EAMLs might be initially misdiagnosed as renal cell carcinoma (RCC) or sarcoma, especially if they morphologically appear without the adipose component [17]. Radical nephrectomy remains the standard of care for this aggressive disease, and the cytotoxic chemotherapy response remains controversial [8, 9]. Not all the EAML develops metastasis, large tumor size, tumor necrosis, and the presence of venous invasion, and high mitotic activity (>2 mitotic figures/10 HPF) was highly associated with malignant behavior [18, 19]. In our observation, the tumors with rare mitotic activity (EAML-3 and EAML-4) were disease-free after radical nephrectomy even though they initially presented with locally advanced status, but the patient with high mitotic activity (3–6 mitotic activity/10 HPF) in tumor tissue developed multiple metastases 1 year after nephrectomy. Aggressive treatment for high-risk EAML should consider adjuvant therapy including either chemotherapy or mTOR inhibitor. For metastatic EAML, mTOR inhibitors or multikinase inhibitors were reported to have their clinical efficacy [20, 21].
In the target therapy era, there are still lacks of sufficient evidence to support neoadjuvant treatments for large unresectable renal tumors, with high-level vascular invasion and venous thrombus [22]. Neoadjuvant therapy may sometimes be used to downsize and downstage large and/or unresectable tumors [23], not only in hepatic tumor but also may change radical nephrectomy to nephron-sparing surgery [24].
Genetic analysis showed that the three types of AMLs, including the AML, EAML and TSC-related renal AML, shared the same allelic loss on chromosome arm 16p [6, 25,26,27]. However, evidence for the relationship between the AML, EAML and the malignant transformation is still lacking. The TSC1 and TSC2 genes encode for harmartin and tuberin, respectively, and are tumor suppressor genes. Both are involved in the Rheb/mTOR/p70S6 kinase signaling pathway. Mutation in these genes result in an mTORC1(mammalian target of rapamycin complex-1)-dependent activation of the hypoxia-inducible factor (HIF) and upregulate the expression of vascular endothelial factor, which will increase the angiogenesis and tumor growth [28, 29]. Our current results from the mRNA (Fig. 3) and protein (Fig. 4) expression of PI3K/Akt/mTOR signaling cascades in renal tissue of an EAML case found that there is frameshift mutation of TSC2 and downregulation of TSC1/TSC2 protein level, subsequently inducing increased levels of mTOR. Therefore, treating this case with everolimus clinically may reasonably lead to two phenomena in tumor tissues. One, the upregulation of 4E-BP1 suppresses the translational process and results in reduced tumor cell proliferation, and the other is presented with the increase of total and phosphorylated form of p70 protein in our study (Fig. 5).
Schematic representation of PI3K/Akt/mTOR signaling pathway. The expression patterns of PI3K/Akt/mTOR signaling pathway molecules in EAML-1 were determined by PI3K-AKT signaling PCR array. The mRNA expression in tumor tissue with more than twofold overexpression as compared to the paired normal tissue is labeled in red [31]. The 4E-BP1 was 20.6-fold high expression
Based on these findings, we speculated that the mechanism on the decrease of tumor size, cell proliferation and translational process was due to the increase of 4E-BP1 expression in the renal tumor of the EAML patient after mTOR inhibitor treatment.
The functional relationship between TSC1/TSC2 and mTORC1 has led to the use of mTOR inhibitors in several clinical manifestations of TSC, including TSC-associated renal AMLs [30]. Everolimus is an oral, active analogue of rapamycin, which acts as a signal transduction inhibitor, and targets the activity of mTOR. Its efficacy has been established in TSC-related AMLs, neuroendocrine tumors and renal cell carcinomas [9, 10]. Finding out whether there is a common signaling pathway in the development of these different types of AMLs or each of them having their own TSC genetic mutation, or whether the mTOR pathway participates in advanced/metastatic EAML, as well as the TSC gene status, may help us explore the pathogenesis of EAML, malignant transformation and their management. The TSC gene status should also be considered in EAML.
Conclusions
Epithelioid angiomyolipoma (EAML) of the kidney is a specific type of renal AML with malignant potentials, where around 22% of the patients present with invasion or metastasis. The most common metastatic sites are lungs, liver and lymph nodes. For a young patient with a huge renal mass and less fat content, the diagnosis of EAML should be considered, and renal biopsy along with positive IHC staining for HMB-45 can confirm the diagnosis. Higher mitotic activities in EAML tumor specimen indicate a greater metastatic potential, with radical nephrectomy as the treatment of choice, and mTOR inhibitors such as everolimus either as neoadjuvant or as adjuvant targeted therapy can lead to a better clinical outcome. It would also be valuable to perform next-generation sequencing on more EAML samples and explore the mTOR signaling pathway in more detail in order for us to better understand the pathogenesis and progression of EAML.
References
Amin MB (2004) Epitheloid angiomyolipoma. In: Eble JN, Sauter G, Epstein JI, Sesterhenn IA (eds) WHO classification of tumours. Pathology and genetics. Tumors of the urinary system and male genital organs. IARC Press, Lyon, pp 68–69
Eble JN, Sauter G, Epstein JI, Sesterhenn IA (2004) Pathology and genetics of tumours of the urinary system and male genital organs. Lyon IARC 90:109
Van Slegtenhorst M, De Hoogt R, Hermans C, Nellist M, Janssen B, Verhoef S, Lindhout D, Van den Ouweland A, Halley D, Young J (1997) Identification of the tuberous sclerosis gene TSC1 on chromosome 9q34. Science 277(5327):805–808
Consortium ECTS (1993) Identification and characterization of the tuberous sclerosis gene on chromosome 16. Cell 75(7):1305–1315
Brimo F, Robinson B, Guo C, Zhou M, Latour M, Epstein JI (2010) Renal epithelioid angiomyolipoma with atypia: a series of 40 cases with emphasis on clinicopathologic prognostic indicators of malignancy. Am J Surg Pathol 34(5):715–722
Martignoni G, Pea M, Bonetti F, Zamboni G, Carbonara C, Longa L, Zancanaro C, Maran M, Brisigotti M, Mariuzzi G (1998) Carcinoma-like monotypic epithelioid angiomyolipoma in patients without evidence of tuberous sclerosis: a clinicopathologic and genetic study. Am J Surg Pathol 22(6):663–672
Huang K-H, Huang C-Y, Chung S-D, Pu Y-S, Shun C-T, Chen J (2007) Malignant epithelioid angiomyolipoma of the kidney. J Formos Med Assoc 106(2):S51–S54
Yamamoto T, Ito K, Suzuki K, Yamanaka H, Ebihara K, Sasaki A (2002) Rapidly progressive malignant epithelioid angiomyolipoma of the kidney. J Urol 168(1):190–191
Cibas ES, Goss GA, Kulke MH, Demetri GD, Fletcher CD (2001) Malignant epithelioid angiomyolipoma (sarcoma ex angiomyolipoma’) of the kidney: a case report and review of the literature. Am J Surg Pathol 25(1):121–126
Bissler JJ, Kingswood JC, Radzikowska E, Zonnenberg BA, Frost M, Belousova E, Sauter M, Nonomura N, Brakemeier S, de Vries PJ (2016) Everolimus for renal angiomyolipoma in patients with tuberous sclerosis complex or sporadic lymphangioleiomyomatosis: extension of a randomized controlled trial. Nephrol Dial Transp 31(1):111–119
Sampson JR (2009) Therapeutic targeting of mTOR in tuberous sclerosis. Portland Press Limited, London
Sepp T, Yates J, Green AJ (1996) Loss of heterozygosity in tuberous sclerosis hamartomas. J Med Genet 33(11):962–964
Pan C-C, Jong Y-J, Chai C-Y, Huang S-H, Chen Y-J (2006) Comparative genomic hybridization study of perivascular epithelioid cell tumor: molecular genetic evidence of perivascular epithelioid cell tumor as a distinctive neoplasm. Hum Pathol 37(5):606–612
Chang Y-H, Wang L-J, Chuang C-K, Wong Y-C, Wu C-T, Hsieh M-L (2007) The efficacy and outcomes of urgent superselective transcatheter arterial embolization of patients with ruptured renal angiomyolipomas. J Trauma Acute Care Surg 62(6):1487–1490
Yip K, Peh W, Tam P (1998) Spontaneous rupture of renal tumours: the role of imaging in diagnosis and management. Br J Radiol 71(842):146–154
Delgado R, Bojorge BdL, Albores-Saavedra J (1998) Atypical angiomyolipoma of the kidney. Cancer 83(8):1581–1592
Švec A, Velenská Z (2005) Renal epithelioid angiomyolipoma—a close mimic of renal cell carcinoma. Report of a case and review of the literature. Pathol Res Pract 200(11):851–856
He W, Cheville JC, Sadow PM, Gopalan A, Fine SW, Al-Ahmadie HA, Chen Y-B, Oliva E, Russo P, Reuter VE (2013) Epithelioid angiomyolipoma of the kidney: pathological features and clinical outcome in a series of consecutively resected tumors. Mod Pathol 26(10):1355–1364
Nese N, Martignoni G, Fletcher CD, Gupta R, Pan C-C, Kim H, Ro JY, Hwang IS, Sato K, Bonetti F (2011) Pure epithelioid PEComas (so-called epithelioid angiomyolipoma) of the kidney: a clinicopathologic study of 41 cases: detailed assessment of morphology and risk stratification. Am J Surg Pathol 35(2):161–176
Shitara K, Yatabe Y, Mizota A, Sano T, Nimura Y, Muro K (2011) Dramatic tumor response to everolimus for malignant epithelioid angiomyolipoma. Jpn J Clin Oncol 41(6):814–816
Lee D-W, Chang H, Kim Y-J, Kim K-M, Lee H-J, Lee J-S (2014) Sorafenib-induced tumor response in a patient with metastatic epithelioid angiomyolipoma. J Clin Oncol 32(11):e42–e45
Schrader AJ, Steffens S, Schnoeller TJ, Schrader M, Kuczyk MA (2012) Neoadjuvant therapy of renal cell carcinoma: a novel treatment option in the era of targeted therapy? Int J Urol 19(10):903–907
Bergamo F, Maruzzo M, Basso U, Montesco MC, Zagonel V, Gringeri E, Cillo U (2014) Neoadjuvant sirolimus for a large hepatic perivascular epithelioid cell tumor (PEComa). World J Surg Oncol 12(1):46
Borregales LD, Adibi M, Thomas AZ, Wood CG, Karam JA (2016) The role of neoadjuvant therapy in the management of locally advanced renal cell carcinoma. Ther Adv Urol 8(2):130–141
Martignoni G, Pea M, Rigaud G, Manfrin E, Colato C, Zamboni G, Scarpa A, Tardanico R, Roncalli M, Bonetti F (2000) Renal angiomyolipoma with epithelioid sarcomatous transformation and metastases: demonstration of the same genetic defects in the primary and metastatic lesions. Am J Surg Pathol 24(6):889–894
Carbonara C, Longa L, Grosso E, Mazzucco G, Borrone C, Garrè ML, Brisigotti M, Filippi G, Scabar A, Giannotti A (1996) Apparent preferential loss of heterozygosity at TSC2 over TSC1 chromosomal region in tuberous sclerosis hamartomas. Genes Chromosom Cancer 15(1):18–25
Henske EP, Neumann HP, Scheithauer BW, Herbst EW, Short MP, Kwiatkowski DJ (1995) Loss of heterozygosity in the tuberous sclerosis (TSC2) region of chromosome band l6p13 occurs in sporadic as well as TSC-associated renal angiomyolipomas. Genes Chromosom Cancer 13(4):295–298
Karbowniczek M, Henske EP (2005) The role of tuberin in cellular differentiation: Are B-Raf and MAPK involved? Ann N Y Acad Sci 1059(1):168–173
Brugarolas JB, Vazquez F, Reddy A, Sellers WR, Kaelin WG (2003) TSC2 regulates VEGF through mTOR-dependent and-independent pathways. Cancer Cell 4(2):147–158
El-Hashemite N, Zhang H, Henske EP, Kwiatkowski DJ (2003) Mutation in TSC2 and activation of mammalian target of rapamycin signalling pathway in renal angiomyolipoma. Lancet 361(9366):1348–1349
Lam JS, Pantuck AJ, Belldegrun AS, Figlin RA (2007) Protein expression profiles in renal cell carcinoma: staging, prognosis, and patient selection for clinical trials. Clin Cancer Res 13(2):703s–708s. doi:10.1158/1078-0432.Ccr-06-1864
Acknowledgements
We thank the National Science Council of the Republic of China, Chang Gung Molecular Medicine Research Center and Chang Gung Memorial Hospital for financial aid: NSC 105-2314-B-182-024-MY3, CMRPG1A0051-2, CMRPG3A0811-3.
Funding
This study was funded by National Science Council of Taiwan (NSC 105-2314-B-182-024-MY3) and Chang Gung Memorial Hospital (CMRPG1A0051-2, CMRPG3A0811-3).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
We declare that we have no conflict of interest.
Ethical approval
This study was approved by IRB of Chang Gung Medical Foundation (IRB 104-7555C). All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards.
Rights and permissions
About this article
Cite this article
Chuang, CK., Lin, H.C.A., Tasi, HY. et al. Clinical presentations and molecular studies of invasive renal epithelioid angiomyolipoma. Int Urol Nephrol 49, 1527–1536 (2017). https://doi.org/10.1007/s11255-017-1629-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11255-017-1629-4