Abstract
Private gardens comprise a large component of greenspace in cities and can offer substantial conservation opportunities. There has been strong advocacy from researchers, policymakers, and conservation practitioners to engage householders in wildlife-friendly gardening practices to increase the quantity, quality and connection of habitat resources for urban wildlife. Despite this call to action, there remains limited knowledge on the use and benefit of some wildlife-friendly structures within gardens, such as artificial refuges and water sources. In collaboration with 131 citizen scientists in southwestern Australia, we examined the use of seven wildlife-friendly structure types by four vertebrate taxa groups. Following 2841 wildlife surveys undertaken between 31 July 2022 and 22 February 2023, we found that all structures were used primarily by target taxa, water sources were often used by relatively common species, certain structures such as possum shelters were used by rare and threatened species (e.g. western ringtail possum), and that there was evidence of animals making use of the wildlife-friendly structures for reproduction (e.g. bird eggs in nest boxes and tadpoles in water sources). Water sources were used more frequently and by a greater diversity of wildlife than artificial refuges. In particular, bird baths were used by the highest number of species (mainly birds) while ponds were used by the greatest variety of taxa (birds, reptiles, frogs, mammals). Our findings provide evidence-based support for the advocacy of wildlife-friendly gardening practices and further highlight the role of residential gardens for biodiversity conservation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Urbanisation leads to degradation, fragmentation and loss of natural habitat, impacting biodiversity on nearly all continents on earth (Fischer and Lindenmayer 2007). As urban land cover and the associated impacts continue to increase (Angel et al. 2011), so does the need to mitigate or ‘offset’ habitat loss through implementation of conservation strategies within cities (Miller and Hobbs 2002; Dunn et al. 2006; Soanes and Lentini 2019). Historically, efforts to preserve wildlife in urban areas have focused on protecting and enhancing the remaining patches of natural vegetation (Tulloch et al. 2016; Soanes et al. 2019). More recently, the value of smaller greenspaces for wildlife conservation has also been recognised (Rega-Brodsky et al. 2022) with evidence that parks, cemeteries, golf courses, and private gardens also support biodiversity (Gallo et al. 2017; Van Helden et al. 2020b; Lerman et al. 2021). Private gardens in particular could offer substantial conservation opportunities given that they collectively comprise a substantial component of the greenspace in cities (e.g. Loram et al. 2007; González-García and Sal 2008) and in some cases can support more biodiversity than other small urban greenspaces such as parks (Gallo et al. 2017; Lerman et al. 2021).
One approach to combat the impacts of urbanisation is to purposefully increase or enhance the resources available to wildlife; a strategy termed ‘wildlife-friendly gardening’, ‘ecological gardening’, or ‘naturalistic gardening’ (Lindemann-Matthies and Marty 2013). This practice has been widely implemented in remnant green spaces and residential gardens in urbanised areas across the globe (Goddard et al. 2010). Within residential gardens, wildlife-friendly gardening commonly includes activities such as installing artificial refuges for shelter or breeding sites; planting native vegetation for protection, connectivity, or food; providing water sources for bathing, feeding, drinking or breeding; and supplementary feeding where appropriate. Due to the collective impact that individual households could contribute within their home gardens, there is strong advocacy from researchers, policymakers, and conservation practitioners to engage householders in wildlife-friendly gardening practices (Goddard et al. 2010, 2013; van Heezik et al. 2012; Heezik et al. 2013; Larson et al. 2022).
The widespread adoption of wildlife-friendly gardening by residents has been shown to provide a substantial contribution of resources to wildlife (e.g. Lepczyk et al. 2004; Gaston et al. 2005b, 2007; Davies et al. 2009), and some studies have demonstrated a positive correlation between the presence of wildlife in gardens and the provision of wildlife-friendly features such as artificial shelters, water sources, supplementary food and planted vegetation (Daniels and Kirkpatrick 2006; Fuller et al. 2008; Gehlbach 2012; Van Helden 2020a). However, few studies have explored the direct use of wildlife-friendly features by wildlife, and most of these have focussed on the use of planted native vegetation by insect pollinators (e.g. Pawelek et al. 2009; Matteson and Langellotto 2011; Garbuzov and Ratnieks 2014). Evidence of the ecological benefits of the full range of wildlife-friendly gardening activities is surprisingly scarce and underrepresented in urban biodiversity literature (Rega-Brodsky et al. 2022; Delahay et al. 2023).
In particular, the ecological benefits of artificial refuges and water sources in gardens for wildlife have received comparatively little attention. Studies that have investigated the use of garden water sources by wildlife have demonstrated their use by a variety of native birds, frogs and invertebrates (Parris 2006; Hamer and Parris 2011; Hill et al. 2015, 2017; Cleary et al. 2016; Coetzee et al. 2018; Gibbons et al. 2023). In some cases, garden water sources can support a different assemblage of wildlife to existing urban blue spaces and may increase water dependent biodiversity across urbanised landscapes (Gibbons et al. 2023). Knowledge of the species that use artificial refuges (e.g. nest boxes, constructed shelters) is mostly derived from studies investigating their use in natural vegetation (see Cowan et al. 2021 for review) or in non-garden greenspaces of urban landscapes (e.g. Goldingay et al. 2015, 2020; Gryz et al. 2021). These studies have demonstrated that a range of native and introduced fauna including flying, arboreal and ground dwelling species will make use of artificial refuges (Harper et al. 2005; Mainwaring 2011; Cowan et al. 2020; Goldingay et al. 2020), but that refuge placement and design strongly influences its use (Goldingay and Stevens 2009; Goldingay et al. 2015). Exploration of artificial refuge use in gardens is notably rare, with near exclusive emphasis on single-species nest box studies (e.g. Gazzard and Baker 2022), while the use of other refuge types such as frog hotels or reptile shelters remains unexplored.
Importantly, the knowledge of how fauna respond to and use wildlife-friendly structures within urban bushland remnants and other greenspaces may not be applicable to structures within residential gardens. The use of wildlife-friendly structures by wildlife within residential gardens may be unique, as gardens have distinct ecological characteristics compared to other types of greenspaces such as urban bushland, golf courses and parks (Threlfall et al. 2016). For example, gardens are typically floristically richer with a higher proportion of exotic species, possess fewer large living or hollow-bearing trees, have limited bare soil and leaf litter, and reduced understorey vegetation volume (Threlfall et al. 2016). Consequently, it remains unclear which wildlife species are supported by wildlife-friendly structures within residential gardens, and how these species may use or benefit from different types of structures, particularly installed artificial refuges and water sources. Given this limited current knowledge, combined with the increasing advocacy for householders to install wildlife-friendly structures, there is a pressing need to investigate whether these structures are of use and benefit to native wildlife within gardens.
In this study, we explored the animal use of multiple wildlife-friendly structures in residential gardens located across multiple cities and towns of southwestern Australia. This geographic region was selected as it is experiencing significant and highly centralised population growth, has lost more than 70% of natural vegetated habitats through agriculture and urban expansion and is experiencing significant climate warming and drying. These regional attributes suggest native fauna may particularly benefit from wildlife-friendly gardening practices. We aimed to identify the diversity of vertebrate species that use seven types of wildlife-friendly structures (two water source and five artificial refuge types) within private gardens in the biodiversity hotspot of southwestern Australia. Additionally, we aimed to assess the frequency at which these structures were used by fauna to gain further understanding of their role in supporting animal wildlife. In doing so, the study provides new knowledge of the potential benefits of wildlife-friendly gardening for native wildlife within urban landscapes.
Methods
Study area and study sites
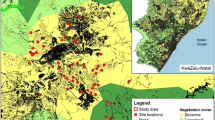
We conducted this study within 131 gardens in southwestern Australia (Fig. 1), an area that covers approximately 44,000 km2 and supports a population of around 2.5 million (ABS 2021). Southwestern Australia is divided into six regions: Perth and Peel Regions which contain Western Australia’s capital city and surrounding areas of Perth; as well as the Wheatbelt, Southwest, Great Southern, and Goldfields Regions which contain other major regional cities including Bunbury (108,000 people), Geraldton (39,500 people), and Albany (38,800 people) (Fig. 1) (ABS 2021). Despite its large area (2.65 million km2), Western Australia has one of the nations most centralised populations with around 80% of the population (approx. 2.1 million people) residing in the capital city of Perth (ABS 2021). It is also currently experiencing the nation’s greatest population growth, estimated at 2.3% (national average 1.9%) (ABS 2021).
Location of residential garden sites (blue dot, N = 131) within regions of southwestern Australia where citizen scientists monitored use of wildlife-friendly structures between 31 July 2022 and 22 February 2023. Inset shows location of southwestern Australia. Photos illustrate examples of the seven types of wildlife-friendly gardening structures: (A) bird bath, (B) pond, (C) reptile shelter, (D) frog hotel, (E) bird box, (F) possum shelter and (G) bat box. Photos e-g provided by Simon Cherriman, the remainder taken by BEVH
Southwestern Australia is a globally recognised biodiversity hotspot (Myers et al. 2000) that supports approximately 3620 endemic plant species, a nationally significant vertebrate fauna with high levels of endemism, many of which are threatened (Rix et al. 2015; Stewart et al. 2018) and has experienced over 70% loss of natural vegetation through agricultural and urban land use change (Halse et al. 2003). Urbanised landscapes within the region are relatively ‘green’ (e.g. Albany; Van Helden et al. 2021a) and, like other cities, native animal wildlife distributions overlap urban areas (Ives et al. 2016). This region is characterised by a Mediterranean climate, with cool wet winters (June–August) and hot dry summers (December–February). Southwestern Australia has been warming and drying for at least five decades, with a reduction of ~ 20% mean precipitation since 1975 (Bates et al. 2008; Hope et al. 2010). This significant climate warming and drying, has been implicated in the decline or contraction of geographic ranges of numerous vertebrate species in the region (Stewart et al. 2018).
Garden sites were identified using an online questionnaire (Qualtrics, Provo, UT), developed as part of a broader study that identified respondents willing to participate in wildlife surveys as citizen scientists. The questionnaire was circulated through social media, newspaper releases, email lists, radio interviews and newsletters of a variety of local businesses and community organisations. We targeted a range of organisations, including those not associated with conservation or natural resource management. Residents that agreed to be involved were subsequently contacted and trained in the wildlife monitoring methods before the project began (see Wildlife surveys for further detail) and were asked to provide specific site information on their garden. A total of 131 citizen scientists with artificial refuges and/or water sources within their residential gardens were identified through this process and approximately 94% of residents submitted site data for their garden.
Based on the cohort of residents that submitted garden site data (N = 123), gardens were located in medium-density urban areas (~ 8–15 dwellings per hectare, 61.8%), low-density urban areas (~ 1–7 dwellings per hectare, 30.1%) or rural areas of southwestern Australia ( ~ < 1 dwelling per hectare, 8.1%) (Fig. 1). The highest proportion of gardens was found in urban areas within the Perth metropolitan and Peel regions (55.3%), followed by the Southwest region (21.1%) and the Great Southern region (19.5%, Fig. 1). Perth and Albany cities had the highest proportion of participating gardens (53.7% and 13.8%, respectively), with the remaining gardens located in 18 other cities or townships, each of which represented less than 4% of the total garden sites. The majority of gardens had less than 50% tree (76.4%) and shrub (69.9%) cover but over half of the garden plants were native to Australia in most gardens (60.2%) (Table 1). Gardens were commonly within 2.5 km of a patch of native vegetation (87%) and within 2.5 km of a natural water source such as a wetland or river (81.3%).
Wildlife-friendly structures
We examined wildlife use of seven wildlife-friendly structure types; bat boxes, bird baths, bird boxes, frog hotels, possum shelters, ponds and reptile shelters (Fig. 1). We chose to focus on artificial refuges and water sources rather than food resources (e.g. planting plants and supplementary feeding) as their utility has received comparatively less attention in the existing literature (e.g. Fuller et al. 2008; Galbraith et al. 2015; Garbuzov and Ratnieks 2014; Matteson and Langellotto 2011; Pawelek et al. 2009) and because in Western Australia supplementary feeding is considered an offence without a license under the Biodiversity Conservation Act 2016. Bird baths exhibit a variety of designs and styles, catering to both the needs of birds and the aesthetic preferences of bird enthusiasts. They vary significantly in surface area and depth, with common designs including traditional pedestal bird baths (Fig. 1A), hanging bird baths, ground-level bird baths, multi-tiered bird baths, and bird bath fountains. Similarly, ponds vary extensively in shape, size and depth. They can be elevated or at ground level (Fig. 1B), often featuring a variety of aquatic plants, decorative elements like rocks, waterfalls or fountains, and can include introduced fish (both native and exotic).
Reptile shelters can be in the form of rock or wood piles, tiles/pavers (Fig. 1C), or commercially available ‘hiding caves’ placed on the ground. Frog hotels consist of a collection of PVC pipes, usually of varying diameters and lengths (< 1m) buried upright in the ground (Fig. 1D) or in a water source (e.g. pond). These hotels serve as cool, moist resting areas for tree frogs.
Bat boxes, possum shelters and bird boxes are typically made of wood to provide nesting and resting spaces for wildlife. Bird boxes, featuring forward-facing entrance holes, are available in various sizes and shapes to accommodate a variety of bird species, including owls, ducks, parrots, and pardalotes (Cherriman 2022; Fig. 1E). Possum shelters are typically wooden boxes (Fig. 1F) or circular human-made dreys (possum nests) often constructed using hanging planter baskets. These can be built to accommodate small possums such as the western pygmy possum (Cercartetus concinnus) or relatively large possums such as the western ringtail possum (Pseudocheirus occidentalis) and common brushtail possum (Trichosurus vulpecula) (Cherriman 2022). In southwestern Australia, bat boxes are designed as roosting spots for insectivorous micro-bats and can come in both single-chambered (Fig. 1G) and multi-chambered designs, often with downward-facing entrances at the base (Cherriman 2022).
The majority of these structures are commercially available in Australia (except frog hotels) and there are abundant resources, both in print and online, that offer guidelines for constructing homemade versions of all structures. As a result, garden structures monitored by citizen scientists varied in size, design and construction. As our focus was on the use of wildlife-friendly structures in typical garden settings we did not account for the potential variations in designs that may be present among gardens.
Wildlife surveys
All surveys of wildlife-friendly structures were undertaken by citizen scientists (i.e. the residents within their own gardens) between 31 July 2022 and 22 February 2023. Residents selected which wildlife-friendly structure surveys they wished to complete in their garden. To standardise the monitoring methods and increase accuracy of species identification (Mason and Arathi 2019) all citizen scientists were given comprehensive training in (1) species identification, (2) the monitoring methods, (3) general scientific principles (including the need to report surveys where no animals were recorded), and (4) animal ethics protocols and procedures. Training was delivered through a combination of face-to-face workshops, online training videos and written manuals that provided step-by-step instructions of the monitoring methods (Van Helden et al. 2022) and images of species likely to be encountered in gardens (Gulliver et al. 2022). Citizen scientists were also able to ask experienced ecologists questions at any point during the study to help them with their monitoring and species identification. Residents were encouraged to undertake a weekly survey for each structure they had in their garden during specific periods of the day when structures would most likely be occupied or used (see below for structure-specific detail). However, this weekly inspection was not always achieved, and as a result the survey effort at each property was variable. In total, 131 citizen scientists participated in the study and contributed 2841 surveys of 7 wildlife-friendly structure types.
Citizen scientists were instructed to record all vertebrate species (except for bats, see below) observed using their wildlife-friendly structures, including both ‘target’ and ‘non-target’ species (Table 2). Individual animals that could not be identified to species, but that were clearly distinguishable from other observed species, were assigned a unique name (e.g., ‘Bird A’, ‘Bird B’) and the total number of each of these was recorded during the surveys. If the animal could not be clearly observed or identified, and was therefore indistinguishable from another species, it was recorded as an ‘unknown animal’. The total number of all ‘unknown animals’ observed using structures was also recorded. The following provides detailed information on each monitoring method undertaken by citizen scientists (see Van Helden et al. 2022 for further detail).
Artificial refuge inspections
Bat boxes and surrounding ground were inspected for the presence of guano between 0900 and 1700 h. This method allowed determination of general bat presence (i.e., not identified to species) because residents were unable to inspect inside the boxes (box lids were often fastened shut due to downward facing entrances). The bat box was recorded as ‘used’ if any guano or guano stains were observed during each survey. Any guano observed during individual inspections was removed so that new guano could be easily identified on the subsequent survey. In total, 100 inspections of 10 bat boxes occurred at 7 properties, with citizen scientists inspecting between one and three boxes on their property (median = 1, N = 7).
Bird boxes were inspected between 1000 and 1400 h by first lightly tapping the box to flush adult birds, and then inspecting the inside of the box for eggs, chicks, unflushed birds or other wildlife. If eggs or chicks were observed in the box, inspections ceased for four weeks to reduce potential disturbance to breeding birds and nestlings. The species and number of adult individuals, as well as the presence or absence of chicks or eggs were recorded for each inspection. In total, 142 inspections of 27 bird boxes occurred at 13 properties, with citizen scientists inspecting between one and six boxes on their property (median = 1, N = 13).
The inside of possum shelters (possum boxes or artificial dreys) were inspected between 0900 and 1700 h. The species and number of individuals, as well as the presence or absence of non-target adult, eggs or nestling birds were recorded for each inspection. In total, 136 inspections of 21 possum shelters occurred at 12 properties, with citizen scientists inspecting between one and four shelters on their property (median = 1, N = 12).
Frog hotels and reptile shelters were inspected between 1000 and 1400 h and the species and number of individuals were recorded for each inspection. Frog hotels were inspected by looking on and inside the tubes of the frog hotel for the presence of wildlife, whilst reptile shelters were inspected by looking for wildlife on top or under the structure. For frog hotels, 269 inspections of 23 frog hotels occurred at 11 properties, with citizen scientists inspecting between one and five frog hotels on their property (median = 4, N = 11). For reptile shelters, 502 inspections of 44 reptile shelters occurred at 20 properties, with citizen scientists inspecting between one and six shelters on their property (median = 2, N = 20).
Water source surveys
For ponds and bird baths, citizen scientists undertook timed animal wildlife surveys between either sunrise and 1000 h or between 1400 h and sunset. For each survey, citizen scientists positioned themselves 5–10 m from the water source and documented the species that visited the water, along with the total number of visits made by each species during a 20-minute observation period. A visit was defined as an individual interacting directly with the water (i.e., drinking, bathing etc.). All visits to the water were counted, including if the same individual returned to the water source numerous times within the survey period. On completion of each 20-minute survey, ponds were visually inspected for the presence of tadpoles. Citizen scientists surveyed only one pond and/or bird bath on their property and in total completed 179 surveys of 23 ponds (42.3 h of observation) and 1513 surveys of 119 bird baths (504.3 h of observation).
Data analysis
With the exception of bat boxes, we calculated species richness for each wildlife-friendly structure, the number of visitations to each structure (both collectively and per species), and how frequently each structure was used by target and non-target wildlife (i.e. count of observations where at least one animal was seen divided by the total number of observations). For bat boxes, we were only able to determine the frequency that the structure was used by bats as residents were unable to inspect inside the boxes to identify the species using the structure. For ponds and bird baths we also calculated the mean species richness per survey and the mean visitation of wildlife per hour. We calculated mean visitation per hour rather than per survey, as this is a widely used metric in similar studies (see for example, Gibbons et al. 2023) making our estimates comparable. For this reason, it is important to note that our visitation rates were calculated from periods of the day when structures were presumably most used. Citizen scientists were not instructed to record invertebrate species during their monitoring, so all records of invertebrates were excluded from analysis. Fish were also excluded from analysis given that they were likely introduced into ponds by residents. Any species that could not be identified to species level was excluded from species richness calculations, however, species that had been identified to class level (e.g. ‘bird sp.’) were included when calculating the frequency of structure use. Species where the class was unclear (e.g. named ‘unknown animal.’) were excluded from frequency analyses as we were unable to determine whether it was a target or non-target species.
Results
Diversity of wildlife using structures
In total, citizen scientists completed 2841 wildlife-friendly structure surveys at 131 residential properties in southwestern Australia. A total of 77 species were observed using wildlife-friendly structures during the study including three threatened species and four introduced species (Table 3). Collectively, structures were used by 55 bird species, 4 frog species, 5 mammal species and 13 reptile species (Table 3). Frog hotels were used by 3 species, bird boxes were used by 6 species, possum shelters were used by 2 species, reptile shelters were used by 17 species, ponds were used by 14 species and bird baths were used by 57 species (Table 3).
Frequency of structure use
Artificial refuges
Bat boxes had evidence of bat guano on 1% of inspections (N = 100) with no other records of non-target wildlife recorded (Fig. 2). Bird boxes were used by birds on 19.7% of inspections and chicks or eggs were recorded on 15.5% of inspections (N = 142, Table 3; Fig. 2). Non-target wildlife such as possums were recorded in bird boxes on 2.1% of inspections (Table 3; Fig. 2). In terms of total animals seen, 39 animals were recorded during bird box inspections. Bird boxes were more frequently used by birds (92.3% of animal records) compared to other taxa (Table 4), with striated pardalotes (Pardalotus striatus) accounting for 53.8% of animal sightings in bird boxes (N = 39, Supplementary Information). Possum shelters were only used by possums and were occupied on 19.9% of inspections (N = 136, Table 3; Fig. 2). Of the total 41 possums recorded, the common brushtail possum and the Critically Endangered western ringtail possum accounted for 9.8% and 90.2% of animal records respectively (Tables 3 and 4; Supplementary Information).
Bars represent the percentage of wildlife refuge inspections (green) or water source surveys (blue) during which target animals were detected. Icons (bats, birds, frogs, reptiles, and mammals) represent the taxa groups that were identified using each wildlife-friendly structure type. Coloured icons represent the target taxa and grey icons represent the non-target taxa recorded using each structure type
Frog hotels were used by frogs on 5.9% of inspections (N = 269) and by non-target animals such as reptiles on 0.4% of inspections (Table 3; Fig. 2). A total of 21 animals were recorded during inspections. Frog hotels were used most frequently by frogs (95.2% of animal records, N = 21) (Table 4), with one species, the motorbike frog (Litoria moorei), accounting for 90.5% of animal records (Supplementary Information). Reptile shelters were used by reptiles on 14.5% of inspections and by non-target wildlife such as frogs on 1.0% of inspections (N = 502, Table 3; Fig. 2). A total of 183 animals were recorded at reptile shelters, of which most (97.8%) were reptiles (Table 4). The most common species detected at reptile shelters were the two-toed earless skink (Hemiergis quadrilineata; 42.1% of animal records) and the western worm lerista (Lerista praepedita; 18.6% of animal records) (Supplementary Information).
Water sources
Bird baths were used by birds on 53.3% of surveys and by non-target wildlife such as frogs and mammals on 0.1% of surveys (N = 1513, Table 3; Fig. 2). Mean visitation to bird baths was 12.8 ± 1.8 visits per hour (± SE, N = 1513) and mean species richness was 1.3 ± 0.05 species per survey (N = 1513, 504.3 h of observation). In total, 7018 animal visitations were recorded at bird baths (N = 1513, 504.3 h of observation), and these were most commonly by birds (92.3% of animal visits, Table 4). New Holland honeyeaters (Phylidonyris novaehollandiae; 37.4%) and silvereyes (Zosterops lateralis; 12.5%) accounted for half of all visits to bird baths (N = 7018; Supplementary Information).
Ponds were used by target wildlife on 45.3% of pond surveys (N = 179, Table 3; Fig. 2). Tadpoles were recorded on 21.8% of survey occasions (N = 179). Mean visitation to ponds was 5.6 ± 2.8 visits per hour (N = 179) and mean species richness was 0.52 ± 0.05 species per survey (N = 179, 42.3 h of observation). In total, 267 visitations were recorded at ponds (N = 179, 42.3 h of observation) with frogs being the most frequent visitor (82% of animal visits), although birds also visited relatively frequently (16.5% of animal visits, Table 4). The motorbike frog accounted for 77.5% of animal visits to ponds (N = 267, Supplementary Information).
Discussion
Despite strong advocacy from researchers, policymakers and conservation practitioners to engage householders in wildlife-friendly gardening practices (Goddard et al. 2010; van Heezik et al. 2012; Larson et al. 2022), evidence of the use and benefits of wildlife-friendly structures within gardens remains surprisingly scarce. While some previous studies have documented the use of individual structures both in gardens and other urban greenspaces (e.g. ponds, bird baths; Hamer and Parris 2011; Cleary et al. 2016), our study is the first to evaluate the use of a suite of wildlife-friendly structures within gardens by a diversity of vertebrate species. We demonstrated that collectively, wildlife-friendly structures were used frequently by a high diversity of species, including threatened species, and taxa for which relatively little is known about their presence in urban areas (e.g. herptiles, Magle et al. 2012; Collins et al. 2021; Rega-Brodsky et al. 2022). Our study showed that the frequency of use and the taxa supported varied substantially among wildlife-friendly structure types. Structures were generally used by the taxa that they were intended to support, although most structures were also used by non-target species, albeit on rare occasions. Our study adds to a growing body of research demonstrating the value of residential gardens for supporting a variety of urban wildlife, particularly those already occurring within urban landscapes. In particular, the use of structures for drinking, bathing, sheltering and nesting reported in our study demonstrates benefit to individual animals. In urban environments where the availability of these resources limit population numbers, these benefits may extend beyond the individual and provide broader population benefits through supporting critical life history processes and increasing population numbers.
Use of wildlife-friendly structures in residential gardens
Our finding that different types of wildlife-friendly structures exhibited varying levels of usage (both in terms of visitation and taxa diversity), suggests that certain structures may offer greater conservation benefits than others. For example, we found that water sources (bird baths and ponds) were used more frequently than artificial refuges, with ponds supporting the greatest cross-taxa richness. In contrast, bat boxes were used only once, and frog hotels were used by frogs on less than 6% of inspections. This suggests if the goal is to create a wildlife-friendly garden capable of supporting a variety of wildlife, water supplementation may be the most advantageous approach, at least in relatively hot and dry climate regions similar to that experienced in our study area. Alternatively, if the objective is to target and support specific taxa, then opting for wildlife-friendly structures that cater specifically to these species may be more useful. For example, some gardens in our study overlap the distribution of the Critically Endangered western ringtail possum (Van Helden et al. 2021b) and possum shelters were used nearly exclusively by this species. Our findings illustrate that to support this species through wildlife-friendly gardening, the provision of possum shelters would likely be most beneficial. Examining the applicability of these findings to diverse regions and climates and assessing whether the presence of multiple wildlife-friendly structures exhibit synergistic positive effects warrants further investigation to gain a more comprehensive understanding of the relative benefits associated with each wildlife-friendly structure type. Overall, our findings suggest that gardens with numerous structure types will provide more biodiversity benefit than gardens with only a single structure type by supporting both a broad and specific array of taxa.
Collectively, our results demonstrate that artificial refuges and water sources are used by a variety of native wildlife, providing valuable habitat resources for native species within the urban landscape. First, our findings add to a growing body of evidence suggesting that supplemented water sources, such as bird baths and ponds, can be an effective conservation strategy in urban gardens (Parris 2006; Hill et al. 2015, 2017; Gibbons et al. 2023). The frequent use of bird baths by a high diversity of bird species in our study supports the findings of previous investigations that demonstrate the value of garden bird baths (Gehlbach 2012; Miller et al. 2015; Cleary et al. 2016; Gibbons et al. 2023). Our study also provides new evidence of the use of ponds by wildlife, demonstrating that they can also support terrestrial fauna, particularly birds, in addition to their well-established benefit to a variety of invertebrate (Gaston et al. 2005a; Hill et al. 2015, 2017) and amphibian (Parris 2006; Hamer and Parris 2011) taxa.
Furthermore, our study is one of the first to contribute knowledge on the use of frog hotels and reptile shelters within residential gardens. Despite strong promotion and abundant information on the construction and installation of frog hotels, we found these structures were not commonly used by frogs. Rather, the majority of frog sightings were recorded at ponds, suggesting that the installation of ponds may be a more effective way to support frog taxa. Whether frog hotels are used more frequently when placed adjacent to ponds is not something we examined during this study, but worthy of further investigation given anecdotal evidence from our citizen scientists of increased frog use when hotels were in closer proximity to areas of moisture (e.g. ponds or bogs). Interestingly, reptile shelters were used quite frequently (~ 15%) by reptiles and on occasion also by frogs and mammals. This aligns with studies from natural environments that demonstrate reptiles will readily occupy and make use of artificial refuges (Webb and Shine 2000; Grillet et al. 2010), a fact also known from cultural practices of building lizard traps by First Nations people in Australia (Cramp et al. 2022). This is a noteworthy finding as it suggests reptile shelters, which are usually inexpensive, simple structures such as a paver or rock piles, could be easily deployed by householders to provide a useful and effective shelter for a variety of wildlife.
Finally, our study provides further insights into the utility of artificial refuges that simulate tree hollow environments by providing resting, sleeping or nesting locations for wildlife (Menkhorst 1984; Goldingay and Stevens 2009). We extend the already demonstrated use of these structures in woodlands and non-garden sites of Australia (Durant et al. 2009; Goldingay et al. 2018, 2020) to their use in residential gardens where we found they are frequently used by birds and arboreal marsupials for shelter and nesting. In contrast, despite the widespread occurrence of bats in residential areas of southwestern Australia (Van Helden et al. 2020b), including species known to use boxes in eastern Australia, we found their use of bat boxes was considerably lower than studies undertaken in other non-garden areas of Australia (Goldingay and Stevens 2009; Rhodes and Jones 2011; Griffiths et al. 2019). It is unclear whether the low use of bat boxes is specific to residential gardens, the bat assemblage found in southwestern Australia, the length of time the bat boxes had been installed, or due to poor bat box design which is known to affect bat box use (Rueegger et al. 2019). Further investigation of bat box usage in residential gardens is required to better understand their utility for native southwestern Australian bat fauna.
Use of structures by threatened and introduced species
Wildlife-friendly gardening practices are often implemented to support native wildlife taxa, particularly rare or threatened species, however, can be detrimental to biodiversity if they support introduced species or aggressive common species. For example, there has been some contention on the utility of shelter boxes (i.e. bird, possum and bat boxes), as highlighted by Goldingay et al. (2020), as they can mostly benefit common species, be predominantly used by exotic species and infrequently used by species of conservation concern. Interestingly, we found that bird boxes and possum shelters were not used by any exotic species, and generally supported less-common or threatened species such as the striated pardalote and Critically Endangered western ringtail possum, respectively. It remains uncertain whether this finding stems from the design of nest boxes deployed in our study gardens, which may feature small entrance holes that deter more aggressive introduced species like rainbow lorikeets (Trichoglossus moluccanus) and common species like brushtail possums, or if it reflects the unique wildlife assemblage in southwestern Australia. Regardless, our research demonstrates in some circumstances, shelter boxes can provide resources suitable for use by threatened and less common species in residential gardens.
The potential of wildlife-friendly structures to benefit exotic or common species is also a pertinent consideration for the other wildlife-friendly structures we examined in this study. Encouragingly, we found that reptile shelters, frog hotels, ponds and bird baths were infrequently used by exotic species and occasionally used by threatened species such as the endangered Carnaby’s cockatoo (Calyptorhynchus latirostris) and Baudin’s cockatoo (Calyptorhynchus baudinii). However, we did also find that common species such as the New Holland honeyeater, motorbike frog and numerous common skinks were the most frequent visitors to water sources, frog hotels and reptile shelters. This is not surprising given urban landscapes can filter biodiversity and contribute to biotic homogenisation, where wildlife assemblages can be dominated by generalist, common species (e.g. Blair 2001; Devictor et al. 2007). Nonetheless, as with nest boxes, our study demonstrates that in some circumstance wildlife-friendly structures support threatened species within residential gardens and that their use by exotic species is limited, indicating their value for native wildlife.
Use of structures for reproduction and life history cycles
One of the significant knowledge gaps surrounding the value of urban landscapes is how they impact upon the longer-term sustainability of wildlife populations. While evidence continues to grow on the prevalence of wildlife within urbanised habitats (Daniels and Kirkpatrick 2006; Gallo et al. 2017; Soanes and Lentini 2019; Van Helden et al. 2020b), our understanding of critical life history processes, animal health and survivorship within these areas remain poorly understood. It is clear from the literature that depending on a complex set of landscape, environmental and species-specific characteristics, urban habitats can be both detrimental and beneficial for biodiversity (see Shochat et al. 2006; Grimm et al. 2008; Concepción et al. 2015). For example, while some urban landscapes are seemingly capable of supporting substantial proportions of biodiversity (e.g. Gregory and Baillie 1998; Van Helden et al. 2020b), other studies demonstrate that urban areas can act as ecological traps by increasing mortality (e.g. Vlaschenko et al. 2019) or reducing reproductive output (e.g. Boal 1997; Sumasgutner et al. 2014). Whilst we did not directly address this concern, the presence of chicks or eggs on 16% of bird box inspections, and tadpoles on 22% of pond inspections provides valuable evidence of fauna reproduction in residential gardens (Bland et al. 2004; Van Helden et al. 2020b; Helden et al. 2021b). Evidence of offspring provides optimism that gardens may support reproduction and therefore contribute to population persistence within urban landscapes, further highlighting the value of residential landscapes and wildlife-friendly gardening for conservation (see Delahay et al. 2023 for review). Further studies that examine reproduction, survival and recruitment of wildlife in residential gardens would further advance our understanding of their value for wildlife, while also considering the risk of gardens acting as ‘ecological traps’ (Dwernychuk and Boag 1972).
Future research directions
While this study provides valuable new evidence of the use of wildlife-friendly garden structures by fauna, additional research is needed to fully understand the value these structures provide for biodiversity conservation. Firstly, our study did not consider potential seasonal variations in use of structures and is based on a relatively small number of properties and structures. Given that the influence of garden features on wildlife presence is known to vary seasonally (Van Helden et al. 2021a), investigating how seasonal variations impact structure usage may provide a more comprehensive understanding of which species benefit from these structures and when. Secondly, further research is needed that explores how the design and placement of wildlife-friendly structures influences their use within residential gardens. This should include examination of the influence of design and placement in preventing unintended negative impacts, including heightened disease transmission, elevated predation risk, and decreased survival rates of offspring (e.g. see Zhang et al. 2023) while maximising use by target native wildlife (e.g. Goldingay and Stevens 2009; Hamer and Parris 2011; Goldingay et al. 2015; Cowan et al. 2020; Gazzard and Baker 2022; Gibbons et al. 2023). Thirdly, it is unclear from our study whether multiple individuals of a single species are using the structure, or a single individual is using the structure repeatedly. Studies that examine the number of individuals using structures would clarify whether the structures support animal residency or supplement the resource needs of numerous individuals, both of which suggest the structures offer valuable contributions for urban wildlife conservation. Fourthly, due to our relatively small number of properties, combined with variable survey effort, we were unable to robustly investigate whether features of the garden (e.g. vegetative cover and nativeness, presence of pets, etc.) or garden location within the urban landscape influenced use of wildlife-friendly structures. This knowledge could be used to identify gardens where incorporation of wildlife-friendly structures would have maximum benefit. Lastly, while our study suggests that wildlife-friendly structures may be capable of increasing the diversity and abundance of wildlife within gardens, there are currently very few studies that experimentally test the ability of newly added structures to increase biodiversity (Delahay et al. 2023). This would be an area worthy of investigation to further understand the role of wildlife-friendly gardening for biodiversity conservation and management (Rega-Brodsky et al. 2022; Delahay et al. 2023).
Conclusion
Our study contributes important insights into the use and potential benefits of wildlife-friendly structures within gardens, addressing a significant knowledge gap amidst the growing advocacy for wildlife-friendly gardening practices. In collaboration with 131 citizen scientists, we examined an array of artificial refuges and water sources, revealing their utilisation by a surprisingly wide range of species, including those of conservation concern. The findings highlight that water sources may be particularly valuable for promoting a broad array of urban biodiversity. Future research should explore seasonal variations, consider diverse geographic regions, and evaluate structure design and placement effects on usage. Clarifying the dynamics of species residency and occupancy within the structures and conducting experimental tests to assess the capacity of new structures to enhance biodiversity will further advance our understanding of the role of wildlife-friendly gardening for conservation. Our findings provide evidence-based support for the advocacy of wildlife-friendly gardening practices by householders and further highlight the role of residential gardens for biodiversity conservation (Goddard et al. 2010; Van Helden et al. 2020a; Helden et al. 2020b; Delahay et al. 2023).
Data availability
No datasets were generated or analysed during the current study.
References
Angel S, Parent J, Civco DL, Blei A, Potere D (2011) The dimensions of global urban expansion: estimates and projections for all countries, 2000–2050. Prog Plann 75:53–107. https://doi.org/10.1016/j.progress.2011.04.001
Australian Bureau of Statistics (ABS) (2021) Region data summary http://www.abs.gov.au/. Accessed 12 December 2023
Bates BC, Hope P, Ryan B, Smith I, Charles S (2008) Key findings from the Indian Ocean Climate Initiative and their impact on policy development in Australia. Clim Change 89:339–354
Blair RB (2001) Creating a homogeneous avifauna. Avian ecology and conservation in an urbanizing world. Kluwer Academic, Boston, pp 459–486
Bland RL, Tully J, Greenwood JJD (2004) Birds breeding in British gardens: an underestimated population? Bird Study. 51:97–106. https://doi.org/10.1080/00063650409461340
Boal CW (1997) An urban environment as an ecological trap for Cooper’s hawks. Dissertation, The University of Arizona
Cherriman S (2022) Hollowed out? A story of tree-hollows, habitat loss and how nest-boxes can help wildlife in south-western Australia. Australia. ISBN: 978-0-6453357-1-2
Cleary GP, Parsons H, Davis A, Coleman BR, Jones DN, Miller KK, Weston MA (2016) Avian assemblages at bird baths: a comparison of urban and rural bird baths in Australia. PLoS ONE 11:e0150899
Coetzee A, Barnard P, Pauw A (2018) Urban nectarivorous bird communities in Cape Town, South Africa, are structured by ecological generalisation and resource distribution. J Avian Biol 49:jav–01526. https://doi.org/10.1111/jav.01526
Collins MK, Magle SB, Gallo T (2021) Global trends in urban wildlife ecology and conservation. Biol Conserv 261:109236
Concepción ED, Moretti M, Altermatt F, Nobis MP, Obrist MK (2015) Impacts of urbanisation on biodiversity: the role of species mobility, degree of specialisation and spatial scale. Oikos 124:1571–1582
Cowan MA, Dunlop JA, Turner JM, Moore HA, Nimmo DG (2020) Artificial refuges to combat habitat loss for an endangered marsupial predator: how do they measure up? Conserv. Sci Pract 2:e204. https://doi.org/10.1111/csp2.204
Cowan MA, Callan MN, Watson MJ, Watson DM, Doherty TS, Michael DR, Dunlop JA, Turner JM, Moore HA, Watchorn DJ, Nimmo DG (2021) Artificial refuges for wildlife conservation: what is the state of the science? Biol. Rev 96:2735–2754. https://doi.org/10.1111/brv.12776
Cramp S, Murray S, Knapp L, Coyne H, Eades A, Lullfitz A, Speldewinde P, Hopper SD (2022) Overview and investigation of Australian Aboriginal Lizard traps. J Ethnobiol 42:400–416. https://doi.org/10.2993/0278-0771-42.4.400
Daniels GD, Kirkpatrick JB (2006) Does variation in garden characteristics influence the conservation of birds in suburbia? Biol Conserv 133:326–335
Davies ZG, Fuller RA, Loram A, Irvine KN, Sims V, Gaston KJ (2009) A national scale inventory of resource provision for biodiversity within domestic gardens. Biol Conserv 142:761–771
Delahay RJ, Sherman D, Soyalan B, Gaston KJ (2023) Biodiversity in residential gardens: a review of the evidence base. Biodivers Conserv 32:4155–4179. https://doi.org/10.1007/s10531-023-02694-9
Devictor V, Julliard R, Couvet D, Lee A, Jiguet F (2007) Functional homogenization effect of urbanization on Bird communities. Conserv Biol 21:741–751. https://doi.org/10.1111/j.1523-1739.2007.00671.x
Dunn RR, Gavin MC, Sanchez MC, Solomon JN (2006) The pigeon paradox: dependence of global conservation on urban nature. Conserv Biol 20:1814–1816
Durant R, Luck GW, Matthews A (2009) Nest-box use by arboreal mammals in a peri-urban landscape. Wildl Res 36:565–573. https://doi.org/10.1071/WR09058
Dwernychuk LW, Boag DA (1972) Ducks nesting in association with gulls—an ecological trap? Can. J Zool 50:559–563. https://doi.org/10.1139/z72-076
Fischer J, Lindenmayer DB (2007) Landscape modification and habitat fragmentation: a synthesis. Glob Ecol Biogeogr 16:265–280. https://doi.org/10.1111/j.1466-8238.2007.00287.x
Fuller RA, Warren PH, Armsworth PR, Barbosa O, Gaston KJ (2008) Garden bird feeding predicts the structure of urban avian assemblages. Divers Distrib 14:131–137. https://doi.org/10.1111/j.1472-4642.2007.00439.x
Galbraith JA, Beggs JR, Jones DN, Stanley MC (2015) Supplementary feeding restructures urban bird communities. Proc. Natl. Acad. Sci. 112: E2648-E2657. https://doi.org/10.1073/pnas.1501489112
Gallo T, Fidino M, Lehrer EW, Magle SB (2017) Mammal diversity and metacommunity dynamics in urban green spaces: implications for urban wildlife conservation. Ecol Appl 27:2330–2341. https://doi.org/10.1002/eap.1611
Garbuzov M, Ratnieks FLW (2014) Quantifying variation among garden plants in attractiveness to bees and other flower-visiting insects. Funct Ecol 28:364–374. https://doi.org/10.1111/1365-2435.12178
Gaston KJ, Smith RM, Thompson K, Warren PH (2005a) Urban domestic gardens (II): experimental tests of methods for increasing biodiversity. Biodivers Conserv 14:395–413. https://doi.org/10.1007/s10531-004-6066-x
Gaston KJ, Warren PH, Thompson K, Smith RM (2005b) Urban Domestic Gardens (IV): the extent of the resource and its Associated features. Biodivers Conserv 14:3327–3349. https://doi.org/10.1007/s10531-004-9513-9
Gaston KJ, Fuller RA, Loram A, MacDonald C, Power S, Dempsey N (2007) Urban domestic gardens (XI): variation in urban wildlife gardening in the United Kingdom. Biodivers Conserv 16:3227–3238
Gazzard A, Baker PJ (2022) What makes a house a home? Nest box use by west European hedgehogs (Erinaceus europaeus) is influenced by nest box placement, resource provisioning and site-based factors. PeerJ 10:e13662
Gehlbach FR (2012) Eastern screech-owl responses to suburban sprawl, warmer climate, and additional avian food in Central Texas. Wilson J Ornithol 124:630–633
Gibbons EK, Close PG, Van Helden BE, Rooney NJ (2023) Water in the city: visitation of animal wildlife to garden water sources and urban lakes. Urban Ecosyst 1–13
Goddard MA, Dougill AJ, Benton TG (2010) Scaling up from gardens: biodiversity conservation in urban environments. Trends Ecol Evol 25:90–98. https://doi.org/10.1016/j.tree.2009.07.016
Goddard MA, Dougill AJ, Benton TG (2013) Why garden for wildlife? Social and ecological drivers, motivations and barriers for biodiversity management in residential landscapes. Ecol Econ 86:258–273. https://doi.org/10.1016/j.ecolecon.2012.07.016
Goldingay RL, Stevens JR (2009) Use of artificial tree hollows by Australian birds and bats. Wildl Res 36:81–97. https://doi.org/10.1071/WR08064
Goldingay RL, Rueegger NN, Grimson MJ, Taylor BD (2015) Specific nest box designs can improve habitat restoration for cavity-dependent arboreal mammals. Restor Ecol 23:482–490. https://doi.org/10.1111/rec.12208
Goldingay RL, Thomas KJ, Shanty D (2018) Outcomes of decades long installation of nest boxes for arboreal mammals in southern Australia. Ecol Manag Restor 19:204–211. https://doi.org/10.1111/emr.12332
Goldingay RL, Rohweder D, Taylor BD (2020) Nest box contentions: are nest boxes used by the species they target? Ecol. Manag Restor 21:115–122. https://doi.org/10.1111/emr.12408
González-García A, Sal AG (2008) Private urban greenspaces or patios as a key element in the urban ecology of tropical central America. Hum Ecol 36:291–300
Gregory RD, Baillie SR (1998) Large-scale habitat use of some declining British birds. J Appl Ecol 35:785–799. https://doi.org/10.1046/j.1365-2664.1998.355349.x
Griffiths SR, Lumsden LF, Bender R, Irvine R, Godinho LN, Visintin C, Eastick DL, Robert KA, Lentini PE (2019) Long-term monitoring suggests bat boxes may alter local bat community structure. Aust Mammal 41:273–278. https://doi.org/10.1071/AM18026
Grillet P, Cheylan M, Thirion J-M, Doré F, Bonnet X, Dauge C, Chollet S, Marchand MA (2010) Rabbit burrows or artificial refuges are a critical habitat component for the threatened lizard, Timon lepidus (Sauria, Lacertidae). Biodivers Conserv 19:2039–2051. https://doi.org/10.1007/s10531-010-9824-y
Grimm NB, Faeth SH, Golubiewski NE, Redman CL, Wu J, Bai X, Briggs JM (2008) Global change and the ecology of cities. Science 319:756–760
Gryz J, Jaworski T, Krauze-Gryz D (2021) Target species and other residents—an experiment with nest boxes for red squirrels in central Poland. Diversity 13:277
Gulliver HF, Van Helden BE, Pitman JJ, Close PG (2022) Wildlife Friendly Gardening in Southwestern Australia - Guide 1: Common Garden Wildlife of Southwestern Australia - Vertebrate species Identification Guide. Centre For Natural Resource Management, University of Western Australia and Perth NRM
Halse SA, Ruprecht JK, Pinder AM (2003) Salinisation and prospects for biodiversity in rivers and wetlands of south-west Western Australia. Aust J Bot 51:673–688
Hamer AJ, Parris KM (2011) Local and landscape determinants of amphibian communities in urban ponds. Ecol Appl 21:378–390. https://doi.org/10.1890/10-0390.1
Harper MJ, McCarthy MA, van der Ree R (2005) The use of nest boxes in urban natural vegetation remnants by vertebrate fauna. Wildl Res 32:509–516. https://doi.org/10.1071/WR04106
Hill MJ, Mathers KL, Wood PJ (2015) The aquatic macroinvertebrate biodiversity of urban ponds in a medium-sized European town (Loughborough, UK). Hydrobiologia 760:225–238. https://doi.org/10.1007/s10750-015-2328-8
Hill MJ, Biggs J, Thornhill I, Briers RA, Gledhill DG, White JC, Wood PJ, Hassall C (2017) Urban ponds as an aquatic biodiversity resource in modified landscapes. Glob Chang Biol 23:986–999. https://doi.org/10.1111/gcb.13401
Hope P, Timbal B, Fawcett R (2010) Associations between rainfall variability in the southwest and southeast of Australia and their evolution through time. Int J Climatol 30:1360–1371
Ives CD, Lentini PE, Threlfall CG, Ikin K, Shanahan DF, Garrard GE, Bekessy SA, Fuller RA, Mumaw L, Rayner L (2016) Cities are hotspots for threatened species. Glob Ecol Biogeogr 25:117–126
Larson KL, Lerman SB, Nelson KC, Narango DL, Wheeler MM, Groffman PM, Hall SJ, Grove JM (2022) Examining the potential to expand wildlife-supporting residential yards and gardens. Landsc Urban Plan 222:104396. https://doi.org/10.1016/j.landurbplan.2022.104396
Lepczyk CA, Mertig AG, Liu J (2004) Assessing landowner activities related to birds across rural-to-urban landscapes. Environ Manag 33:110–125. https://doi.org/10.1007/s00267-003-0036-z
Lerman SB, Narango DL, Avolio ML, Bratt AR, Engebretson JM, Groffman PM, Hall SJ, Heffernan JB, Hobbie SE, Larson KL, Locke DH, Neill C, Nelson KC, Padullés Cubino J, Trammell TLE (2021) Residential yard management and landscape cover affect urban bird community diversity across the continental USA. Ecol Appl 31:e02455. https://doi.org/10.1002/eap.2455
Lindemann-Matthies P, Marty T (2013) Does ecological gardening increase species richness and aesthetic quality of a garden? Biol. Conserv 159:37–44. https://doi.org/10.1016/j.biocon.2012.12.011
Loram A, Tratalos J, Warren PH, Gaston KJ (2007) Urban domestic gardens (X): the extent & structure of the resource in five major cities. Landsc Ecol 22:601–615
Magle SB, Hunt VM, Vernon M, Crooks KR (2012) Urban wildlife research: past, present, and future. Biol Conserv 155:23–32
Mainwaring MC (2011) The use of nestboxes by roosting birds during the non-breeding season: a review of the costs and benefits. Ardea 99:167–176
Mason L, Arathi HS (2019) Assessing the efficacy of citizen scientists monitoring native bees in urban areas. Glob Ecol Conserv 17:e00561
Matteson KC, Langellotto GA (2011) Small scale additions of native plants fail to increase beneficial insect richness in urban gardens. Insect Conserv Divers 4:89–98. https://doi.org/10.1111/j.1752-4598.2010.00103.x
Menkhorst PW (1984) Use of Nest boxes by Forest vertebrates in Gippsland: Acceptance, Preference and demand. Wildl Res 11:255–264. https://doi.org/10.1071/WR9840255
Miller JR, Hobbs RJ (2002) Conservation where people live and work. Conserv Biol 16:330–337
Miller KK, Blaszczynski VN, Weston MA (2015) Feeding wild birds in gardens: a test of water versus food. Ecol Manag Restor 16:156–158. https://doi.org/10.1111/emr.12157
Myers N, Mittermeier RA, Mittermeier CG, da Fonseca GAB, Kent J (2000) Biodiversity hotspots for conservation priorities. Nature 403:853–858. https://doi.org/10.1038/35002501
Parris KM (2006) Urban amphibian assemblages as metacommunities. J Anim Ecol 75:757–764. https://doi.org/10.1111/j.1365-2656.2006.01096.x
Pawelek J, Frankie GW, Thorp RW, Przybylski M (2009) Modification of a Community Garden to attract native Bee pollinators in Urban San Luis Obispo, California. Cities and the Environment. 2:: Article 7–21
Rega-Brodsky CC, Aronson MFJ, Piana MR, Carpenter E-S, Hahs AK, Herrera-Montes A, Knapp S, Kotze DJ, Lepczyk CA, Moretti M (2022) Urban biodiversity: state of the science and future directions. Urban Ecosyst 25:1083–1096
Rhodes M, Jones D (2011) The use of bat boxes by insectivorous bats and other fauna in the greater Brisbane region. The Biology and Conservation of Australasian bats. Royal Zoological Society of New South Wales, Mosman, pp 424–442
Rix MG, Edwards DL, Byrne M, Harvey MS, Joseph L, Roberts JD (2015) Biogeography and speciation of terrestrial fauna in the south-western Australian biodiversity hotspot. Biol Rev 90:762–793. https://doi.org/10.1111/brv.12132
Rueegger N, Goldingay RL, Law B, Gonsalves L (2019) Limited use of bat boxes in a rural landscape: implications for offsetting the clearing of hollow-bearing trees. Restor Ecol 27:901–911. https://doi.org/10.1111/rec.12919
Shochat E, Warren PS, Faeth SH, McIntyre NE, Hope D (2006) From patterns to emerging processes in mechanistic urban ecology. Trends Ecol Evol 21:186–191
Soanes K, Lentini PE (2019) When cities are the last chance for saving species. Front Ecol Environ 17:225–231. https://doi.org/10.1002/fee.2032
Soanes K, Sievers M, Chee YE, Williams NSG, Bhardwaj M, Marshall AJ, Parris KM (2019) Correcting common misconceptions to inspire conservation action in urban environments. Conserv Biol 33:300–306
Stewart BA, Ford BM, Van Helden BE, Roberts JD, Close PG, Speldewinde PC (2018) Incorporating climate change into recovery planning for threatened vertebrate species in southwestern Australia. Biodivers Conserv 27:147–165. https://doi.org/10.1007/s10531-017-1426-5
Sumasgutner P, Nemeth E, Tebb G, Krenn HW, Gamauf A (2014) Hard times in the city – attractive nest sites but insufficient food supply lead to low reproduction rates in a bird of prey. Front Zool 11:48. https://doi.org/10.1186/1742-9994-11-48
Threlfall CG, Ossola A, Hahs AK, Williams NSG, Wilson L, Livesley SJ (2016) Variation in vegetation structure and composition across urban green space types. Front Ecol Evol 4:66
Tulloch AIT, Barnes MD, Ringma J, Fuller RA, Watson JEM (2016) Understanding the importance of small patches of habitat for conservation. J Appl Ecol 53:418–429
van Heezik Y, Dickinson K, Freeman C (2012) Closing the gap: communicating to change gardening practices in support of native biodiversity in urban private gardens. Ecol Soc 17
van Heezik Y, Freeman C, Porter S, Dickinson KJM (2013) Garden size, householder knowledge, and socio-economic status influence plant and bird diversity at the scale of individual gardens. Ecosystems 16:1442–1454
Van Helden BE, Close PG, Steven R (2020a) Mammal conservation in a changing world: can urban gardens play a role? Urban Ecosyst. 23:555–567
Van Helden BE, Close PG, Stewart BA, Speldewinde PC, Comer SJ (2020b) An underrated habitat: residential gardens support similar mammal assemblages to urban remnant vegetation. Biol Conserv 250:108760
Van Helden BE, Close PG, Stewart BA, Speldewinde PC (2021a) Managing gardens for wildlife: features that predict mammal presence and abundance in gardens vary seasonally. Ecosphere 12:e03453. https://doi.org/10.1002/ecs2.3453
Van Helden BE, Close PG, Stewart BA, Speldewinde PC, Comer SJ (2021b) Critically endangered marsupial calls residential gardens home. Anim Conserv 24:445–456
Van Helden BE, Gulliver HF, Pitman JJ, Close PG (2022) Wildlife Friendly Gardening in Southwestern Australia - Guide 2: Wildlife Monitoring Manual. Centre for Natural Resource Management. University of Western Australian and Perth NRM
Vlaschenko A, Kovalov V, Hukov V, Kravchenko K, Rodenko O (2019) An example of ecological traps for bats in the urban environment. Eur J Wildl Res 65:20
Webb JK, Shine R (2000) Paving the way for habitat restoration: can artificial rocks restore degraded habitats of endangered reptiles? Biol Conserv 92:93–99. https://doi.org/10.1016/S0006-3207(99)00056-7
Zhang L, Ma X, Chen Z, Wang C, Liu Z, Li X, Xing X (2023) Negative effects of artificial nest boxes on birds: a review. Avian Res 100101. https://doi.org/10.1016/j.avrs.2023.100101
Acknowledgements
This study forms part of the ‘Turning Gardeners into Conservationists: Using Gardens to Conserve Wildlife’ project funded by the Australian Government’s Inspiring Australia Science Engagement Programme (grant number: CSGII000225). We acknowledge the Noongar people of southwestern Australia as the Traditional Custodians of the country upon which this study occurred and pay our respects to Elders past and present. We thank the citizen scientists who generously donated their time to monitor wildlife and our Scientific Steering Committee (G. Barret, O. Nevin, B. Webber, C. Ramalho, N. Pauli) for their guidance. We also thank J. Pitman, K. Greenop and H. Gulliver for their contribution to citizen scientist recruitment and training, and our project partners for their in-kind support of the project: Armadale Gosnells Landcare Group, BirdLife WA, City of Albany, City of Armadale, City of Canning, City of Cockburn, Department of Biodiversity, Conservation and Attractions, Oyster Harbour Catchment Group, Torbay Catchment Group, Town of Victoria Park, South Coast NRM, South East Regional Centre for Urban Landcare, South West NRM, and Western Australian Biodiversity Science Institute. This research was approved under the University of Western Australia’s Human Ethics Committee (2021/ET001057) and Animal Ethics Committee (2021/ET000799).
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions. This work was supported by the Australian Government’s Inspiring Australia Science Engagement Programme (grant number: CSGII000225).
Open Access funding enabled and organized by CAUL and its Member Institutions
Author information
Authors and Affiliations
Contributions
Conceived and designed the research: BVH, PGC. Funding acquisition: BVH, PGC. Coordinated citizen scientist data collection: BVH, LMS. Prepared the data for analysis: BVH. Prepared the first draft of the manuscript: BVH. Reviewed and revised the manuscript: PGC, LMS, BVH. Approved final manuscript: PGC, LMS, BVH.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
This research was approved under the University of Western Australia’s Human Ethics Committee (2021/ET001057) and Animal Ethics Committee (2021/ET000799).
Consent to participate
Participation of citizen scientists in this research was voluntary. Participation was supported by a Participant Information Form for citizen scientist recruitment. Completion of the recruitment questionnaire and wildlife surveys was considered evidence of consent to participate.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Van Helden, B.E., Skates, L.M. & Close, P.G. Use of wildlife-friendly structures in residential gardens by animal wildlife: evidence from citizen scientists in a global biodiversity hotspot. Urban Ecosyst 27, 1493–1507 (2024). https://doi.org/10.1007/s11252-024-01530-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11252-024-01530-4