Abstract
Mollusc communities are getting endangered in the aftermath of urban sprawl because artificial structures do not surrogate natural substrates. In this study, we compared the diversity, community and trophic arrangements of molluscs among different models of artificial substrate and their adjacent natural rock, to detect relationships between some abiotic variables and the mollusc communities. Complexity, chemical composition and age were tested as potential drivers of the community. Diversity, community and trophic structure differed between natural and artificial substrates. Complexity at the scale of cm was detected as the most important factor driving the community structure. In addition, a chemical composition based on silica and/or scarce calcium carbonates seems to be relevant for molluscs, as well as for the secondary substrate where they inhabit. However, age did not seem to be a driving factor. Among the different artificial structures, macroscale complexity was detected as the main factor diverging a drastically poor community at seawall from other artificial structures. In this context, macro and microscale complexity, chemical composition and mineral type are variables to consider in future designs of artificial substrates.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Biodiversity on natural coastal habitats is under threat by many causes, mainly: coastal artificialization, exploitation of renewable (fisheries) and non-renewable (mineral and energy extraction) resources, pollutant discharge and marine debris (Dulvy et al. 2003; Jacob et al. 2018).
In the Bay of Sydney (Australia), 50% of the natural coastline is replaced by artificial substrates (Chapman 2006; Dafforn et al. 2015) and around 22.000 Km2 of European coasts are covered with concrete or asphalt (European Environment Agency Report 2006; Airoldi and Beck 2007). This coastal transformation, the so-called ‘urban sprawl’ (Firth et al. 2016), is being boosted by shore erosion due to more frequent stormy events and the sea-level rise (Bouma et al. 2014; Bulleri and Chapman 2010), altogether, threatening intertidal ecosystems. Intertidal communities are diverse and complex due to the broad range of biotic and abiotic interactions that occur on intertidal natural substrates (Chapman 2013).
For example, wave and tide action (Southward and Orton 1954), desiccation or top-down processes (predation, competition, grazing, etc.) modulate both the sessile and vagile biota, promoting the development of rich and ecologically important communities.
Molluscs are one of the most abundant taxa in the intertidal zone, providing important ecosystem services (see Table 2 in Firth et al. 2016). They are considered early colonizers of substrates (Underwood and Chapman 2013) and play important roles in C and Si cycles (Meysman and Montserrat 2017). Sessile filter molluscs can contribute to clean water and improve nutrient uptake for algae (Eriksson et al. 2017) and together with other sessile organisms, they serve as engineers (Melero et al. 2017; Commito et al. 2018) setting up a secondary substrate for many different species. Also, mobile grazers can feed on macrophytes, cleaning areas for subsequent colonization of many species (Firth et al. 2016). Previous studies have reported a negative impact of artificial substrates on intertidal molluscs. For example, Moreira et al. (2006) suggested that seawall do not sustain viable populations of limpets. Furthermore, complexity/heterogeneity (e.g. micro-roughness) can affect the abundance of chitons (Moreira et al. 2007) or limpets (Rivera-Ingraham et al. 2011) on artificial substrates. In fact, substrate complexity is one of the biggest drivers of intertidal biodiversity. Concrete-made artificial substrates usually lack microhabitats (crevices, rock pools, etc.), preventing refuge from stressful conditions such as desiccation or predation, and are largely responsible for the biodiversity deficit of artificial substrates compared with the natural rocky shore (Firth et al. 2016 and references therein). Therefore, eco-engineering actions that added habitat complexity at different scales have been related with a higher number of taxa (Kefi et al. 2015; Strain et al. 2018) and enhanced recruitment and survival of sessile and mobile macrofauna (Atilla and Finelli 2005).
Substrate composition, like minerals and elements, is also known to be an important factor affecting communities developing on artificial structures (Coombes et al. 2015; Sempere-Valverde et al. 2018). For example, acidic siliceous quartz from sandstone may cause oxidative stress and hold less diverse and mature community when compared to limestone (Bavestrello et al. 2000; Cattaneo-Vietti et al. 2005). The mineralogical composition usually varies from artificial substrates (normally made from concrete) to natural substrates (Ido and Shimrit 2015; Ponti et al. 2015). Concrete may liberate toxic metals and carbonates that enhance alkalinity (pH ~ 13) producing stress on individuals (Ido and Shimrit 2015). In the case of molluscs, higher saturation of aragonite can facilitate a higher occurrence of burrowing bivalves (Mos et al. 2019) and alkaline concrete surfaces may increase oysters’ recruitment (Anderson 1996).
Although ecological succession may not occur in a parallel manner on artificial and natural substrates (Burt et al. 2011), the age of substrates has been considered as an important factor explaining the differences between artificial and natural substrates (Glasby and Connell 1999a, b). Some authors have estimated that it takes from 5 to 20 years for artificial structures to reach climax communities (Coombes 2011; Hawkins et al. 1983; Pinn et al. 2005), while others suggest that communities on low crested structures never reach climax (Gacia et al. 2007) or take more than 100 years (Perkol-Finkel et al. 2005). Consequently, we decided to consider the date of substrates deployment in the present study, together with substrate composition and complexity, to study mollusc diversity associated with artificial substrates.
Furthermore, changes in the community structure of epifaunal organisms associated with artificial substrates can cause trophic shifts (Sedano et al. 2020a). Artificial substrates are known to affect prey resources (Munsch et al. 2015), limiting the diet of some mollusc species (Burgos-Rubio et al. 2015) and ultimately restricting the diversity of trophic strategies. For example, the reduced primary productivity on seawalls has been related to the scarcity of herbivore grazers (Lai et al. 2018). These effects, among others, call for an ecological evaluation of coastal artificial substrates to prevent the decline of intertidal habitats (Dafforn et al. 2015; Firth et al. 2016) and promote other ecological services (García-Gómez et al. 2014; Dearborn and Kark 2010). Taking into account that molluscs are diverse and contribute highly to this habitat (Ricciardi et al. 1997), we decided to study the community of molluscs as a model to detect relationships between the abiotic features of the man-made intertidal substrate and the associated fauna. We focused on habitat complexity, substrates composition and age intending to identify which factors are driving the differences in molluscs taxonomic and trophic structure between artificial substrates and natural substrates. In this regard, we hypothesized that:
-
1.
Substrate complexity and composition would be the main drivers differentiating artificial from natural substrates, given the differences in complexity and composition between artificial and natural substrates in our study area. Additionally, we hypothesized that the mollusc community at rip-raps (an artificial substrate made from natural rock) would be the most similar to natural substrates.
-
2.
Trophic community structure would vary among different artificial substrates and between artificial and natural substrates.
-
3.
Age will be a driver structuring intertidal molluscs’ community on artificial substrates.
Methods
Study area
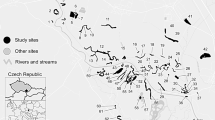
Our study area was located in the Algeciras Bay (Cadiz, Spain), which achieves 400 m in depth and occupies 73 Km2 of area. This deep bay is found next to one of the most relevant marine regions in the world, the Strait of Gibraltar. It is a marine area with high biodiversity due to its location and structure, which is placed between Africa and Europe and between two water bodies, the Atlantic Ocean and the Mediterranean Sea (Usero et al. 2016). Algeciras Bay contains five different substrates (four artificial and their nearest natural rocky shore), very close to each other and under very similar environmental conditions. We selected four nearby artificial substrates (acropods, cubes, rip-raps and seawall) and compared the molluscan assemblages and trophic structure among them and with the nearest natural substrate. Given the difficulty to find different artificial substrates next to each other, we limited our study area to this single Bay (Fig. 1).
Abiotic analysis
To identify possible drivers of the differences between substrates, we measured the physicochemical features of each substrate. The variables included macro and microscale complexity, elemental composition, minerals, crystallinity, calcination percentage (C.P) and age. Complexity measures were divided into macroscale complexity (m) and microscale complexity (cm). In both cases (macro and microscale), substrate roughness was calculated as in Rivera-Ingraham et al. (2011) using the equation by Blanchard and Bourget (1999): Roughness or topographical heterogeneity index (THI) = Tr / Ts, where Tr is the “effective” distance between two points “A-B” (measuring the contour between A-B) and Ts is the linear distance between A-B. Macroscale roughness was calculated over 15 m length transects. Three transects were selected at each substrate and a flexible meter was laid directly over it, trying to conform as closely as possible to all contours of the bare substrate. Regarding microscale roughness, three 15 cm profile gauges with 0.5 mm pins were pushed onto the bare rock to record the surface of each substrate (Frost et al. 2005). The resulting profiles were photographed, and the images were digitally processed with Adobe Photoshop to obtain two coloured images. The length of the contour of the profile was obtained with ImageJ software. The elemental composition and calcination percentage, mineralogical absorption spectra, crystallinity and lithology composition of each sample were obtained from Sedano et al. (2019). All chemical composition was characterized using three powdered fragments of each substrate. Age of the substrate was based on the date of construction and resulting from the difficulty of dating age of the natural substrate, the oldest possible date in the same order of magnitude compared to the oldest artificial substrate was used instead. Also, wave exposure was quantified at each substrate using a combination of the maximum fetch and the modified effective fetch (Fe) index developed by (Howes et al. 1994): Fe = [∑ (cos ɵi) X Fi/∑cos ɵi], where ɵi is the angle between the shore-normal, and the directions 0º, 45º left and 45º right and Fi is the fetch distance in Km along the relevant vector. To determine if substrates differed physico-chemically and to detect the most relevant abiotic components that separate the substrates, we performed a Principal Component Analyses (PCA) using macro, microscale complexity, elemental composition (calcium, silicon and magnesium), crystallinity and age. Data were normalized before analyses.
Biotic analysis
Community and trophic structure, as well as biodiversity indices (richness, Pielou’s Evenness and Shannon’s diversity), were compared among artificial and natural substrates. Three different sites were randomly selected within each of the five substrates (natural, cubes, acropods, rip-raps and seawall). At each site, three replicate quadrats of 20 × 20 cm were scraped (3 sites × 3 replicates × 5 substrates = 45 samples). The samples were collected during low tide and within the lower intertidal zone (5–30 cm over the lowest tidal level). We scraped the biotic substrate (secondary substrate) and the associated fauna and preserved it in 96% ethanol until laboratory analyses. At the laboratory, associated molluscs were sorted out from the rest of sessile and vagile biota, identified down to species level whenever possible and quantified in terms of their abundance. Since the secondary substrate (sessile biota developing on the hard primary substrate) can influence the associated fauna (Chapman et al. 2005), all sessile fauna and flora that conform the secondary substrate were volumetrically quantified at each replicated site to control this variable (used as a covariate in the analyses). Besides, percentages of the most abundant species of the secondary substrate were recorded as well, to detect possible differences between substrates.
To identify possible trophic shifts, we grouped the different species into trophic categories and compared the trophic structure among substrates. Species were assigned and grouped according to their trophic strategies (see Table 1 in Donnarumma et al. 2018), with slight modifications to better represent the feeding strategy of the species in our study. We assigned them into different groups depending on what they feed on, and the way to obtain the food (trophic guilds) (Table 1). Despite being known to feed on larvae of animals and detritus (Burgos-Rubio et al. 2015), limpets were considered herbivores. We also considered omnivores all animals that feed on suspended organic-matter or by filtering particles in the water column.
After species identification, three biodiversity indexes were measured (Badalamenti et al. 2002): species richness, Shannon–Wiener diversity and Pielou evenness for each replicated site in all substrates. To test for biodiversity differences among substrates, we performed a nested ANOVA for each biodiversity index using GMAV5 (Underwood et al. 2002). A Student Newman Keuls (SNK) test was also conducted to elucidate differences among pairs of substrates. The statistic design had two factors: “Substrate” (fixed), with five levels: Natural, Cubes, Acropods, Rip-raps and Seawall, and “Sites” (random), which was nested in the substrate and had three levels (1,2,3). Cochran’s test was performed to confirm the homoscedasticity of biotic data.
Metric multidimensional scaling (MDS) based on the similarity of Bray Curtis matrix was performed on the community and trophic structure (see Table 1 with six categories). We made an additional CLUSTER and SIMPROF test to group the substrates depending on their dissimilarity. Furthermore, a permutational multivariate analysis of variance (PERMANOVA) was also carried out to test if the community and trophic structure of molluscs varied significantly among substrates and sites. Previously, data were square-root transformed and the analysis was carried out on a Bray Curtis triangular matrix. When a significant source of variance was detected, a pair-wise test between pairs of substrates was also computed to obtain the corresponding p-values.
Correlation analyses
Correlation tests between the community and abiotic matrix were made to explore potential relationships between community structure and the abiotic variables. Multicollinearity among abiotic factors was previously tested with a Draftsman’s plot based on Pearson correlations and only one abiotic factor was used when there were high pairs of correlation (Pearson correlation limit was set a 0.80) (See Fig. 5 in Sedano et al. 2019). Variance inflance factor (VIF) was also perfomed to avoid multicollinearity. Furthermore, a distance-based redundancy analysis (dbRDA) was computed using a fourth root transformed biotic matrix paired with a normalized abiotic matrix, to give similar weight to variables measured on different units. DbRDA was portrayed into a bidimensional representation. A BIOENV routine (Clarke and Ainsworth 1993) was done to detect the best set of variables that better suit the response data. This method calculates correlation coefficients between response variables (community matrix) and predictor variables (abiotic matrix) (Balkenhol et al. 2009). RELATE routine (Clarke and Warwick 2001) was carried out to detect the correlation coefficient between both, community and abiotic dissimilarity matrix. All multivariant and correlation analyses were carried out with PRIMER + PERMANOVA 6 using 9999 permutations (Anderson et al. 2008).
Results
Abiotic analyses
The results of the fetch index indicate that all the substrates belong to a semi-exposed wave exposure class, whereas the age of origin was different for each substrate (Table 2). Regarding substrate complexity, microscale complexity was higher at natural substrate and cubes comparing with the rest of substrates, being very low at seawall and acropods. In contrast, macroscale complexity was higher at acropods and cubes than rip-raps and natural substrate (Table 2). From a chemical point of view, elemental composition differed between natural and artificial substrates and among all substrates. Silica (SiO2) concentration was higher at natural substrate compared to artificial substrates, which were characterized by a higher concentration of calcium oxide (CaO) at all samples (Table 3).
According to the mineralogical composition, natural substrate was very different from the artificial ones, and differences were also found within the artificial substrates. The natural substrate was composed of high percentages of quartz, while cubes and rip-raps were mostly composed by quartz and CaO in a carbonated form, calcite (CaCO3). Acropods presented high levels of magnesium oxide (MgO) and their mineralogical composition was based on dolomite (CaMg(CO3)2) (Table 3) (full mineralogical composition in supplementary files of Sedano et al. 2019).
Figure 2 represent these values bidimensionally. In addition, crystallinity was positively correlated with silica and negatively correlated with calcium oxide and calcination percentage.
Age causes the separation of cubes and natural samples from the rest of substrate samples, correlating with micro-scale complexity.
Biotic analyses
A total of 3198 molluscan specimens were identified during this study, corresponding to 3 classes, 20 orders, 35 families, 41 genera and 46 species (supplementary material Table 1). A total of 1855 specimens were collected from 4 artificial substrates (4 × 9 = 36 samples) and 1343 from 1 natural substrate (9 samples). Abundance was drastically lower at seawall (28) and moderately lower at cubes (447), acropods (705) and rip-raps (675).
Ellisolandia elongata was the dominant secondary substrate at natural substrate (83.5%) and rip-raps (85.4%); Perforatus perforatus predominated at cubes (66.9%) and acropods (47.5%) and Mytillus galloprovincialis predominated at seawall (Fig. 3).
Regarding trophic groups, natural substrate contained all the groups measured, and scavenger was exclusive for this substrate. This group was formed by a single species, Tritia tingitana, and its abundance was 2. On the other hand, mostly all artificial substrates lacked the group detritus feeder except for seawall. Shared species per substrate are shown in Fig. 4. The differences in the percentages of groups between substrates were also remarkable.
In terms of percentages, more macro and micro grazers appeared at artificial substrates compared to natural substrate. In contrast, predators and detritus feeders appeared in higher percentage at natural substrate, with the exception of seawall, where the percentage of predators and detritus feeders was higher than that of natural substrate.
There were also differences between artificial substrates because the percentage of filter feeders was higher at rip-raps and cubes compared to natural substrate and acropods, while more macro and micro grazers appeared at acropods compared to the rest of substrates. Finally, seawall had a very heterogeneous trophic structure among samples (Fig. 5, supplementary material Table 1).
Shannon’s diversity and richness varied significantly among substrates (p < 0,001) (Table 4). According to SNK test, Shannon’s diversity was greater on the natural substrate compared to the artificial substrate. Among artificial substrates, acropods showed higher Shannon’s diversity values than seawall, cubes and rip-raps. In contrast, cubes, rip-raps and seawall did not differ significantly on Shannon’s diversity. Similarly, SNK test showed that natural substrate had higher species richness than the artificial substrates. Among artificial substrates, acropods were richer than cubes, rip-raps (p < 0,05) and seawall (p < 0,01). cubes and rip-raps did not differ between them but both were richer than Seawall (p < 0,05). Finally, Pielou’s evenness did not differ among substrates (Fig. 6).
MDS analysis for the community structure showed three groups: 1 = natural, 2 = seawall and 3 = acropods, rip-raps and cubes. These groups were statistically supported by the SIMPROF test (p < 0.05). The natural group appeared homogeneous and different from the rest of the substrates. Seawall group was heterogeneous, but it also appeared clearly segregated. The third group, formed by acropods, cubes and rip-raps was homogeneous but distinct from natural substrate and seawall groups (Fig. 7).
MDS for the trophic structure revealed three groups: 1 = seawall, 2 = seawall and 3 = natural, acropods, cubes, rip-raps and seawall. These groups were statistically supported by the SIMPROF test (p < 0.05). Seawall was the most heterogeneous substrate diverging into three groups, while acropods, rip-raps, natural substrate and cubes were more similar. However natural substrate was homogeneous and was significantly different from the rest at a level of similarity of 80% (Fig. 8).
PERMANOVA test indicated significant differences in community structure between substrates and among sites. According to the pair-wise tests, the community at natural substrate differed from the rest. When comparing among artificial substrates, seawall differed from rip-raps and acropods (p < 0,01), but not from cubes. Cubes, acropods and rip-raps seemed to have a similar community structure (Table 5).
PERMANOVA test indicated differences in trophic structure among substrates but not among sites. The pair-wise test revealed that trophic structure on the natural substrate was different from the artificial substrates. Among artificial substrates, seawall differed from rip-raps (< 0,05) and acropods (p < 0,01) but not from cubes. Also, acropods, rip-raps and cubes had a similar trophic community (Table 6). The volume of the secondary substrate using as a covariable was significant for taxonomical PERMANOVA but not for the trophic one.
Correlation analyses
A dbRDA analysis revealed a relationship between the physical–chemical composition and the molluscan community. The associated community of molluscs at natural substrate was highly correlated with a low CaO concentration and carbonated nature, a high microscale complexity and older age. On the other hand, the community at artificial substrates was correlated with a high CaO concentration and carbonated nature, low microscale complexity and younger age. Because SiO2 appeared negatively collineated with CaO and C.P, these parameters were not included in the analyses. Among artificial substrates, the seawall community was also correlated with a low macroscale complexity, and therefore they have clustered apart from the more heterogeneous artificial substrates (acropods, cubes and rip-raps) (Fig. 9). The BIOENV analyses showed that the most correlated variables (p < 0.01) were macro, microscale complexity and crystallinity (Rho = 0,669). The RELATE test showed a significant correlation of abiotic with taxonomic matrices (Rho = 0,518; p < 0,01).
Discussion
The molluscs community structure and diversity seemed to be significantly different between artificial and natural substrates. Among our studied variables, substrate complexity (macro and microscale roughness) and chemical composition appeared to be the main drivers of those differences. Besides, the trophic structure also seemed to be different between artificial and natural substrates.
By our results, habitat complexity in terms of the relative abundance of microhabitats such as crevices (Evans et al. 2016), rockpools and macrophytes is considered as one of the most influencing factors on intertidal communities (Warfe et al. 2008). Higher heterogeneity at the scale of centimeters increases recruitment of spores and larvae (Sempere-Valverde et al. 2018) due to a higher number of refugees (Kostylev et al. 2005; Coombes et al. 2010). This can be particularly important for intertidal molluscs since they can not only find shelter against environmental stress (Meager et al. 2011; Harley and Helmuth 2003; Loke et al. 2015), but also against predation (Warfe et al. 2008) and competition (Huston 1979) by finding crevices that fit their shell size (Loke and Todd 2016), determining community structure and diversity. Moreover, algae turfs that cover the rocky substrate can influence the abundance and biodiversity of associated fauna, playing biogenic roles, similar to sessile animals such as barnacles or annelids who play the role of “ecological engineers”, providing the secondary substrate where many species live (Simboura et al. 1995; Bavestrello et al. 2000). The greater abundance of the calcareous algae Ellisolandia elongata on natural substrate can influence the associated fauna via increasing both the habitat volume and habitat complexity (Guerra-García et al. 2012; Veiga et al. 2014; Torres et al. 2015). Moreover, it can decrease desiccation and temperature stress by providing shelter for mobile fauna (Singh et al. 2013; Kefi et al. 2015).
In our study, 15 species were exclusive of natural substrate. Natural substrate had high microscale complexity, but they were also highly covered by the calcareous algae Ellisolandia elongata, altogether possibly boosting the higher occurrence of more taxa. Species sensitive to disturbance such as the bivalves Irus irus, Parvicardium vroomi and gastropods such as Skeneopsis planorbis only appeared at natural substrate. For example, S. planorbis and P. vroomi are known to be well represented along the Algeciras Bay associated with the algae Halopteris sp. (Sánchez-Moyano et al. 2000), a highly complex algae (as E. elongata) that can support rich associated communities (Navarro-Barranco et al. 2018). In addition, P. vroomi has shown preference for the algae Halopteris filiscina (Avila 2003). Similarly, more abundance of sea snails and bivalves appeared at natural substrate. For example, Cerithiopsis tubercularis is usually restricted to live on algae that are associated with its food (sponges), as it is the case of the branched Ellisolandia spp. and its association with the sponges Halichondria and Hymeniacidon (Fretter and Manly 1977). Given the close association between algae and molluscs that certain species can present, the absence or very low abundances of these species at artificial substrates, where the cover of algae was very scarce, highlights the importance of calcareous algae in supporting richer communities of molluscs on artificial substrates at this area. In contrast, a lower microscale complexity and scarce algae canopy, probably lead to a lower abundance of the less competitive bivalves and sea snails because fewer microhabitats are available (Underwood and Fairweather 1989; Hills 1996; Strain et al. 2018) as it happens at seawall. Also, the sandstone porosity of natural substrates increases algae settlement (Green et al. 2012), probably generating positive cascading effects.
However, a higher complexity at the scale of meters increases recruitment of propagules and the dissipation of water energy (Vieira et al. 2020) on cubes, acropods and rip-raps, boosting the abundance, richness and diversity of associated fauna, on these substrates, in comparison with seawall. However, these species were mostly “limpets” as Fissurela nubeluca, Siphonaria pectinata and Patella caerulea and chitons. The increment of this taxa is possibly related to the fact that artificial substrates are better habitat for sedentary species, such as limpets and chitons, rather than strictly vagile gastropods (Rivera-Ingraham et al. 2011; Cha et al. 2013), probably by suffering lower predation and being more resilient to wave action. In fact, non-native species of Siphonaria and barnacles have been recorded on seawalls at Plymouth and Singapore (Hsiung et al. 2020).
Seawalls have a small intertidal area for recruitment but, as it happens in our study, seawalls harbour abundant beds of mussels (Chapman et al. 2005) and barnacles on the secondary substrate, associated with lower biodiversity values in comparison with natural substrates (People 2006; Sedano et al. 2020b). The community at seawall was very scarce and had the lowest diversity. Chapman (2006) suggested that seawalls lack microhabitats for many species and limit the life strategies of specialized intertidal fauna, such as limpets and chitons. For example, as it has been recorded in the chiton belonging to Ischiochiton genera that inhabits underneath the boulders as habitat-specialist (Grayson and Chapman 2004). On the other hand, the pulmonate limpet Siphonaria pectinata was absent at seawall, in accordance with Moreira et al. (2006) who detected a relation among living on seawall and a reduction on the reproductive output of this limpet. However, these results contrast with Hsiung et al. (2020) who recently detected non-native species of Siphonaria guanemensis and barnacles on seawalls at Plymouth and Singapore.
Chemical composition was also identified as a possible driver of the community, mainly differentiating communities settled on natural or artificial substrates, since the natural rock was mainly pure quartz (SiO2), while artificial substrates had great amount of carbonated minerals, with high levels of the calcite (CaCO3). The effect of quartz on natural substrates and the carbonated mineralogy at artificial substrates could affect the associated fauna. For example, it has been reported that quarzitic radicals inhibit the settlement of first recruits of secondary substrates such as the hydroid Eudendrium glomeratum (Bavestrello et al. 2000) or the sponge Clionia sp. (Cerrano et al. 2007) while they are neutral for algae settlement. Moreover, in the present study, associated fauna was more abundant and more diverse at natural substrate, where more Ellisolandia elongata appeared, possibly due to a reduction in competition with other sessile biota affected by the toxicity of silicon radicals (Cerrano et al. 1999). In addition, facilitation by calcium hydroxides that are liberated by concrete artificial substrates to the substrate surface, alkalinizing the pH, also contributes to the settlement of bivalves (Anderson 1996; Soniat and Burton 2005; Burt et al. 2009) and barnacles (Guilbeau et al. 2003) on the sessile substrate, as it occurs in the concrete substrates in this study (acropods and seawall).
The concrete substrates are also rich in magnesium oxides and other minerals, which could influence the presence of exclusive species. For example, aragonite has been related with improving boring bivalves’ settlement (Green et al. 2013), being more soluble on water than calcite (Cornelis and Cornelius 2007), and acropods that are composed by this material showed the presence of the boring species Leisonelus aristatus.
Therefore, a combination of a carbonated nature and a lower microscale complexity at artificial substrates possibly promotes a different community of molluscs and increase the dominance of the most ‘colonizer’ species of the secondary substrate (mussels and barnacles) at cubes, acropods and seawall (Miller and Etter 2008; Underwood and Chapman 2013), all disturbing the associated fauna of molluscs.
According to the trophic structure, natural substrate seems to be more diverse, mostly because they contain all the trophic groups measured, while acropods, rip-raps and cubes lack of suspension feeder and scavengers. Contrarily, seawall had all the measured groups except scavengers. Another interesting difference was the different percentage of groups among substrates because detritus feeder and predator were more abundant at natural substrate than at acropods, rip-raps and cubes, where more percentage of macro and micro-grazer appeared. Natural substrate hold the majority of detritus feeders, a fact that could be related to higher sediment retention by macrophytes (Melero et al. 2017; Casoli et al. 2019). In contrast, the detritus feeder Barleeia unifasciata appeared at seawall. In fact, littorinid snails have been related to breakwaters, with lower crevice availability (Aguilera et al. 2014). In addition, predators were exclusive from natural substrate, probably because intraguild predation (Janssen et al. 2007) and the number of preys has been reported to be lower at structures with less complexity. In contrast, macro and micro grazers were highly abundant at artificial substrates, mostly derived from an increment in species of limpets and chitons (see the first part of Discussion).
Limpets are known to control the volume of macrophytes in concert with sea-urchins by their grazing activity (Piazzi et al. 2016) and, in this area, they have been recorded as omnivorous and very generalist (Burgos-Rubio et al. 2015). This could explain the lower volume of the secondary substrate at cubes, rip-raps and acropods when compared to natural substrate and seawall. In addition, the higher volume of secondary substrate on seawall, a substrate with a low abundance of molluscs, sustains the hypothesis that grazers control these sessile populations at acropods, rip-raps and cubes, in special the Ellisolandia elongata, as has been observed at rip-raps. The idea that these grazers could be controlling the associated fauna at artificial substrates should be considered.
Several authors had pointed out that biodiversity could be driven by the age of substrate (Perkol-Finkel et al. 2005; Glasby and Connell 1999a, b), and others have reported that temporal heterogeneity among artificial and natural substrates is a relevant factor driving communities (Glasby and Connell 1999a). For example, on artificial substrates, first recruits as ephemeral algae, sponges and bivalves occur fast in less than a year on but later, as a consequence of a low microscale complexity, dominant species outcompete the first colonizers (Burt et al. 2011). Nevertheless, among the artificial substrates studied in the present work, age did not appear as a driver of the community, because the community at cubes (80y) and rip-raps (20y) was similar, and community of seawall (20y) and acropods (20y) differed, independently of age.
Conclusions and future approach
As expected in our first hypothesis, mollusc community and diversity differed between artificial and natural substrates. Distortion in the bottom-up interactions between, a combination of low microscale complexity and a carbonated nature, rich in calcium, of the artificial substrates and the mollusc community, seems to impact over many species of molluscs and the common calcareous algae Ellisolandia elongata they inhabit in, comparing with the natural substrate. Moreover, macroscale complexity seems to influence the community of molluscs, increasing recruitment of species at acropods, rip-raps and cubes in comparison with seawall, but mostly benefiting limpets, chitons and bivalves, and also barnacles on the secondary substrate.
As for our second hypothesis, physico-chemical factors seem to alter the trophic community increasing the percentage of macro and micro grazers and filter feeders on artificial substrates. In contrast with our third hypothesis, age did not appear as a driver of the mollusc community.
We suggest that, according to previous studies, increasing habitat heterogeneity by means of increasing crevices (Archambault and Bourget 1996) and rock-pools (Ostalé-Valriberas et al. 2018) and microscale complexity is fundamental in the future designs of artificial substrates. On the other hand, the chemical structure should be included as an important topic of research in new models of artificial substrates, possibly depending on the geology and chemistry of the surrounding lands (Moschella et al. 2005).
In the case of Algeciras, rip-raps and cubes were the most similar to natural substrate in relation to abiotic features. At the same time, in relation to community and trophic structure, these substrates seem to be less disturbed. Even though the study was replicated within substrates, the lack of several types of substrates under similar environmental factors on a higher spatial scale represents a limitation. In this sense, further studies re-analyzing already published data and/or meta-analysis at higher spatial scales will provide valuable insights on the role of artificial substrates in structuring coastal assemblages. Furthermore, more research should clarify how molluscs or macrophytes recruitment and survivance are influenced by chemical and other physical issues of the substrate. Finally, biological interactions among secondary substrates and associated fauna should be also explored in future designs.
Data and code availability
The data manufactured can be made available upon request via email to the author. Compliance with ethical standards. All computational work was carried out using PRIMER & PERMANOVA 6, GMAV 5. and R-Studio.
Change history
14 June 2022
A Correction to this paper has been published: https://doi.org/10.1007/s11252-022-01249-0
References
Aguilera MA, Broitman BR, Thiel M (2014) Spatial variability oin community composition on a granite breakwater versus natural rocky hsores: Lack of microhabitats suppreses intertidal biodiversity. Mar Polut Bull 87(1–2):257–268. https://doi.org/10.1016/j.marpolbul.2014.07.046
Airoldi L, Beck MW (2007) Loss, status and trends for coastal marine habitats of Europe. Oceanogr Mar Biol 45:345–405. https://doi.org/10.1201/9781420050943.ch7
Anderson MJ (1996) A chemical cue induces settlement of sydney rock oysters, saccostrea commercialis, in the laboratory and in the field. Biol Bull 190(3):350–358. https://doi.org/10.2307/1543027
Anderson M, Gorley RN, Clarke RK (2008) PERMANOVA+ for PRIMER: Guide to Software and Statistical Methods. Primer-E Limited, Plymouth
Archambault P, Bourget E (1996) Scales of coastal heterogeneity and benthic intertidal species richness, diversity and abundance. Mar Ecol Prog Ser 136:111–121. https://doi.org/10.3354/meps136111
Atilla N, Finelli MC (2005) Effects of habitat complexity and hydrodynamics or the abundance and diversity on small invertebrates. J Mar Sci Technol 63(6):1151–1172. https://doi.org/10.1357/002224005775247580
Avila SP (2003) The littoral molluscs (Gastropoda, Bivalvia and Polyplacophora) of Sao Vicente, Capelas (Sao Miguel island, Azores): ecology and biological associations to algae. Iberus 21(1): 11–33. Retrieved from: https://www.researchgate.net/publication/232084705_The_littoral_molluscs_Gastropoda_Bivalvia_and_Polyplacophora_of_Sao_Vicente_Capelas_Sao_Miguel_Island_Azores_Ecology_and_biological_associations_to_algae
Badalamenti F, Chemello R, D’Anna G, Henriquez Ramos P, Riggio S (2002) Are reefs comparable to neighbouring rocky areas? A mollusc case study in the Gulf of Castellammare (NW Sicily). ICES J Mar Sci 59:127–131. https://doi.org/10.1006/jmsc.2002.1265
Balkenhol N, Waits LP, Dezzani RJ (2009) Statistical approaches in landscape genetics: an evaluation of methods for linking landscape and genetic data. Ecography 5(32):818–830. https://doi.org/10.1111/j.1600-0587.2009.05807.x
Bavestrello G, Bianchi CN, Calcinai B, Cattaneo-Vietti R, Cerrano C, Morri C, Puce S, Sarà M (2000) Biomineralogy as a structuring factor for marine benthic communities. Mar Ecol Prog Ser 193: 241–249. Retrieved from: http://www.jstor.org/stable/24855698
Blanchard D, Bourget E (1999) Scales of coastal heterogeneity: Influence on intertidal community structure. Mar Ecol Prog Ser 179:163–173. https://doi.org/10.3354/meps179163
Bouma TJ, van Belzen J, Balke T, Zhu Z, Airoldi L, Blight AJ, Davies AJ, Galvan C, Hawkins SJ, Hoggart SPG, Lara JL, Losada IJ, Maza M, Ondiviela B, Skov MW, Strain EM, Thompson RC, Yang S, Zanuttigh B, Zhang L, Peter Herman PMJ (2014) Identifying knowledge gaps hampering application of intertidal habitats in coastal protection: Opportunities and steps to take. Coast Eng 87:147–157. https://doi.org/10.1016/j.coastaleng.2013.11.014
Bulleri F, Chapman MG (2010) The introduction of coastal infrastructure as a driver of change in marine environments. J Appl Ecol 47(1):26–35. https://doi.org/10.1111/j.1365-2664.2009.01751.x
Burgos-Rubio V, De la Rosa J, Altamirano M, Espinosa F (2015) The role of patellid limpets as omnivorous grazers: a new insight into intertidal ecology. Mar Biol 162(10):2093–2106. https://doi.org/10.1007/s00227-015-2739-0
Burt J, Bartholomew A, Bauman A, Saif A, Sale PF (2009) Coral recruitment and early benthic community development on several materials used in the construction of artificial reefs and breakwaters. J Exp Mar Biol Ecol 373(1):72–78. https://doi.org/10.1016/j.jembe.2009.03.009
Burt J, Bartholomew A, Sale PF (2011) Benthic development on large-scale engineered reefs: A comparison of communities among breakwaters of different age and natural reefs. Ecol Eng 37(2):191–198. https://doi.org/10.1016/j.ecoleng.2010.09.004
Casoli E, Bonifazi A, Ardizzone G, Gravina MF, Russo GF, Sandulli R, Donnarumma L (2019) Comparative analysis of mollusc assemblages from different hard bottom habitats in the central Tyrrhenian Sea. Diversity 11(74):1. https://doi.org/10.3390/d11050074
Cattaneo-Vietti R, Albertelli G, Bavestrello G, Nike-Bianchi C, Cerrano C, Mariachiara C, Gaggero L, Morri C, Schiaparelli S (2005) Can rock composition affect sublittoral epibenthic communities? Mar Ecol 23:65–77. https://doi.org/10.1111/j.1439-0485.2002.tb00008.x
Cerrano C, Arillo A, Bavestrello G, Benatti U, Calcinai B, Cattaneo-Vietti R, Cortesogno L, Gaggero L, Giovine M, Puce S, Sara M (1999) Organism-quartz interactions in structuring benthic communities: towards a marine bio-mineralogy? Ecol Lett 2(1):1–3. https://doi.org/10.1046/j.1461-0248.1999.00041.x
Cerrano C, Sambolino F, Azzini F, Calcinai B, Bavestrello, (2007) Growth of the massive morph of Cliona nigricans (Schmidt 1862) (Porifera, Clionaidae) on different mineral substrata. Ital J Zool 74(1):13–19. https://doi.org/10.1080/11250000600929370
Cha JH, Kim KB, Song JN, Kim IS, Seo JB, Kwoun CH (2013) Comparative study on the fauna composition of intertidal invertebrates between natural and artificial substrata in the northeastern coast of Jeju Island. Ocean Sci J 48(4):319–328. https://doi.org/10.1007/s12601-013-0030-1
Chapman MG, People J, Bockley D (2005) Intertidal assemblages addociated with natural corallina turf and invasive mussel beds. Biodivers Conserv 14:1761–1776. https://doi.org/10.1007/s10531-004-0698-8
Chapman MG (2006) Intertidal seawall as habitats for molluscs. J Molluscan Stud 72(3):247–257. https://doi.org/10.1093/mollus/eyi069
Chapman MG (2013) Constructing replacement habitat for specialist and generalist molluscs-the effect of patch size. Mar Ecol Prog Ser 473:201–214. https://doi.org/10.3354/meps10074
Clarke KR, Ainsworth M (1993) A method of linking multivariate community structure to environmental variables. Mar Ecol Prog Ser 92:205–205. https://doi.org/10.3354/meps092205
Clarke KR, Warwick RM (2001) Changes in marine communities: an approach to statistical analysis and interpretation. 2nd Edition, PRIMER-E: Plymouth Marine Laboratory, UK, 176 p
Commito J, Jones B, Jodes M, Winders A, Como S (2018) What happens after mussels die? Biogenic legacy effects on community structure and ecosystem processes. J Exp Mar Biol Ecol 506:30–41. https://doi.org/10.1016/j.jembe.2018.05.004
Coombes MA, Naylor LA, Thompson RC, Roast SD (2010) Colonization and weathering of engineering materials by marine microorganisms: An SEM study. Earth Surf Process Landf 36(5):582–593. https://doi.org/10.1002/esp.2076
Coombes MA (2011) Biogeomorphology of Coastal Structures: Understanding interactions between hard substrata and colonising organisms as a tool for ecological enhancement. University of Exeter. Retrieved from http://hdl.handle.net/10036/3103
Coombes MA, La Marca EC, Naylor LA, Thompson RC (2015) Getting into the groove: opportunities to enhance the ecological value of hard coastal infrastructure using fine-scale surface textures. Ecol Eng 77:314–323. https://doi.org/10.1016/j.ecoleng.2015.01.032
Cornelis K, Cornelius S (2007) Manual de Mineralogía. Spain. Editorial Reverté, 4 Edition
Dafforn K, Glasby TM, Airoldi L, Rivero N, Mayer-Pinto M, Johnston E (2015) Marine urbanization: an ecological framework for designing multifunctional artificial structures. Front Ecol Environ 12(2):82–90. https://doi.org/10.1890/140050
Dearborn DC, Kark S (2010) Motivations for conserving urban biodiversity. Conserv Biology 24(2):432–440. https://doi.org/10.1111/j.1523-1739.2009.01328.x
Dulvy NK, Sadovy Y, Reynolds JD (2003) Extinction vulnerability in marine populations. Fish Fish 4(1):25–64. https://doi.org/10.1046/j.1467-2979.2003.00105.x
Donnarumma L, Sandulli R, Apolloni L, Sánchez-Lizaso JL, Russo F (2018) Assessment of structural and functional diversity of mollusc assemblages within vermetid bioconstructions. Diversity 10(3):96. https://doi.org/10.3390/d10030096
Eriksson BK, Westra J, Van Gerwen IV, Weerman E, van der Zee E, van der Heide T, van de Koppel J, Olff H, Piersma T, Donadi S (2017) Facilitation by ecosystem engineers enhances nutrient effects in an intertidal system. Ecosphere 8:1–12. https://doi.org/10.1002/ecs2.2051
European Environment Agency Report (2006) The changing faces of Europe’s coastal areas (6). Retrieved from: https://www.eea.europa.eu/publications/eea_report_2006_6
Evans J, Firth L, Hawkins S, Morris E, Goudge H, Moore P (2016) Drill-cored rock pools: an effective method of ecological enhancement on artificial structures. Mar Freshw Res 67:123–130. https://doi.org/10.1071/MF14244
Firth LB, White FJ, Schofield M, Hanley ME, Burrows MT, Thompson RC, Skov MW, Evans AJ, Moore JP, Hawkins SJ (2016) Facing the future: the importance of substratum features for ecological engineering of artificial habitats in the rocky intertidal. Mar Freshw Res 67:131–143. https://doi.org/10.1071/MF14163
Fretter V, Manly R (1977) Algal associations of Tricolia pullus, Lacuna vincta and Cerithiopsis tubercularis (Gastropoda) with special reference to the settlement of their larvae. J Mar Biolog Assoc U.k. 57(4):999–1017
Frost NJ, Burrows MT, Johnson MP, Hanley ME, Hawkins SJ (2005) Measuring surface complexity in ecological studies. Limnol Oceanogr Methods 3(4):203–210. https://doi.org/10.4319/lom.2005.3.203
Gacia E, Satta MP, Martín D (2007) Low crested coastal defence structures on the Catalan coast of the Mediterranean Sea: how they compare with natural rocky shores. Sci Mar 71(2):259–267. https://doi.org/10.3989/scimar.2007.71n2259
García-Gómez JC, Guerra-Garcia JM, Espinosa F, Maestre JM, Rivera-Ingraham G, Fa D, González AR, Ruíz-Tabares A, López-Fe CM (2014) Artificial Marine Micro-Reserves Networks (AMMRNs): an innovative approach to conserve marine litoral biodiversity and protect endangered species. Mar Ecol 36(3):259–277. https://doi.org/10.1111/maec.12167
Glasby TM, Connell SD (1999a) Do urban structures influence local abundance and diversity of subtidal epibiota? A case study from Sydney Harbour. Australia Mar Environ Res 47(4):373–387. https://doi.org/10.1016/s0141-1136(98)00126-3
Glasby TM, Connell SD (1999b) Urban structures as marine habitats. Ambio 7(28):595–598
Grayson JE, Chapman MG (2004) Patterns of distribution and abundance of chitons of the genus Ischnochiton in intertidal boulder fields. Austral Ecol 29(4):363–373. https://doi.org/10.1111/j.1442-9993.2004.01375.x
Green DS, Chapman MG, Bockley DJ (2012) Ecological consequences of the type of rock used in the construction of artificial boulder –fields. Ecol Eng 46:1–10. https://doi.org/10.1016/j.ecoleng.2012.04.030
Green MA, Waldbusser GG, Hubazc L, Cathcart E, Hall J (2013) Carbonate mineral saturation state as the recruitment cue for settling bivalves in marine muds. Estuar Coast 36:18–27. https://doi.org/10.1007/s12237-012-9549-0
Guerra-García JM, Ros M, Izquierdo D, Soler-Hurtado MM (2012) The invasive Asparagopsis armata versus the native Corallina elongata: differences in associated peracarid assemblages. J Exp Mar Biol Ecol 416:121–128. https://doi.org/10.1016/j.jembe.2012.02.018
Guilbeau BP, Harry FP, Gambrell RP, Knopf FC, Dooley KM (2003) Algae attachment on carbonated cements in fresh and brackish waters preliminary results. Ecol Eng 20(4):309–319. https://doi.org/10.1016/S0925-8574(03)00026-0
Harley H, Helmuth B (2003) Local and regional scale effects of wave exposure, thermal stress, and absolute versus effective shore level on patterns of intertidal zonation. Limnol Oceanogr 48(4):1498–1508. https://doi.org/10.4319/lo.2003.48.4.1498
Hawkins SJ, Southward AJ, Barrett RL (1983) Population structure of Patella vulgata L. during succession on rocky shores in Southwest England. Oceanologica Acta, Special Issue (0399–1784) (Gauthier-Villars)
Hills JM (1996) A multi-scale analysis of settlement density and pattern dynamics of the barnacle semibalanus balanoides. Mar Ecol Prog Ser 138:103–115. https://doi.org/10.3354/meps08309
Howes D, Harper J, Owens E (1994) Physical shore-zone mapping system for british columbia. technical report by the coastal task force of the resource inventory. Committee (RIC), R. I. C. Secretariat. Victoria, BC
Hsiung AR, Tan WT, Loke LHL, Firth LB, Heery EC, Ducker J, Clark V, Shona Pek Y, Birch WA, Ang ACF, Hartanto RS, Chai TFM, Todd P (2020) Little evidence that lowering the pH of concrete supports greater biodiversity on tropical and temperate seawall. Mar Ecol Prog Ser. https://doi.org/10.3354/meps13365
Huston M (1979) A general hyphotesis of species diversity. Am Nat 113(1):81–101
Ido S, Shimrit PF (2015) Blue is the new green–ecological enhancement of concrete based coastal and marine infrastructure. Ecol Eng 84:260–272. https://doi.org/10.1016/j.ecoleng.2015.09.016
Jacob C, Buffard A, Pioch S, Thorin S (2018) Marine ecosystem restoration and biodiversity offset. Ecol Eng 120:585–594. https://doi.org/10.1016/j.ecoleng.2017.09.007
Janssen A, Sabelis MW, Magalhães S, Montserrat M, van der Hammen T (2007) Habitat structure affects intraguild predation. Ecology 88:2713–2719. https://doi.org/10.1890/06-1408.1
Kefi S, Berlow EL, Wieters EA, Joppa LN, Wood SA, Brose U, Navarrete SA (2015) Network structure beyond food webs: mapping non-trophic and trophic interactions on Chilean rocky shores. Ecology 96(1):291–303. https://doi.org/10.1890/13-1424.1.sm
Kostylev VE, Erlandsson J, Yiu Ming M, Williams GA (2005) The relative importance of habitat complexity and surface area in assessing biodiversity: Fractal application on rocky shores. Ecol Complex 2:272–286. https://doi.org/10.1016/j.ecocom.2005.04.002
Lai S, Loke LHL, Bouma TJ, Todd PA (2018) Biodiversity surveys and stable isotope analyses reveal key differences in intertidal assemblages between tropical seawall and rocky shores. Mar Ecol Prog Ser 587:41–53. https://doi.org/10.3354/meps12409
Loke LH, Ladle RJ, Bouma TJ, Todd PA (2015) Creating complex habitats for restoration and reconciliation. Ecol Eng 77:307–313. https://doi.org/10.1016/j.ecoleng.2015.01.037
Loke LH, Todd PA (2016) Structural complexity and component type increase intertidal biodiversity independently of area. Ecology 97(2):383–393. https://doi.org/10.1890/15-0257.1
Meager JM, Schlacher TA, Green M (2011) Topographic complexity and landscape temperature patterns create a dynamic hábitat structure on a rocky intertidal shore. Mar Ecol Prog Ser 428:1–12. https://doi.org/10.3354/meps09124
Melero I, López-Velasco S, López E (2017) On the role of turf species as refuge in disturbedenvironments: A case study with polychaetes (Annelida: Polychaeta) in SW Mediterranean Sea. Med Mar Sci 18: 229–240. https://doi.org/10.12681/mms.2050
Meysman F, Montserrat F (2017) Negative CO2 emissions via enhanced silicate weathering in coastal environments. Biol Lett 13(4):20160905. https://doi.org/10.1098/rsbl.2016.0905
Miller R, Etter R (2008) Shading facilitates sessile invertebrate dominance in the rocky subtidal Gulf of Maine. Ecology 89(2):456–462. https://doi.org/10.1890/06-1099.1
Moreira J, Chapman MG, Underwood AJ (2006) Seawall do not sustain viable populations of limpets. Mar Ecol Prog Ser 322:179–188. https://doi.org/10.3354/meps322179
Moreira J, Chapman MG, Underwood AJ (2007) Maintenance of chitons on seawall using crevices on sandstone blocks as habitat in Sydney Harbour. Australia J Exp Mar Biol 347(1–2):134–143. https://doi.org/10.1016/j.jembe.2007.04.001
Mos B, Dworjanyn SA, Mamo LT, Kelaher BP (2019) Building global change resilience: Concrete has the potential to ameliorate the negative effects of climate-driven ocean change on a newly-settled calcifying invertebrate. Sci Total Environ 646:1349–1358. https://doi.org/10.1016/j.scitotenv.2018.07.379
Moschella PS, Abbiati M, Åberg P, Airoldi L, Anderson JM, Bacchiocchi F, Bulleri F, Dinesen GE, Frost M, Gacia E, Granhag L, Jonsson PR, Satta MP, Sundelöf A, Thompson RC, Hawkins SJ (2005) Low-crested coastal defence structures as artificial habitats for marine life: Using ecological criteria in design. Coast Eng 52(10–11):1053–1071. https://doi.org/10.1016/j.coastaleng.2005.09.014
Munsch SH, Cordell JR, Toft JD (2015) Effects of seawall armoring on juvenile Pacific salmon diets in an urban estuarine embayment. Mar Ecol Prog Ser 535:213–229. https://doi.org/10.3354/meps11403
Navarro-Barranco C, Florido M, Ros M, González-Romero P, Guerra-García JM (2018) Impoverished mobile epifaunal assemblages associated with the invasive macroalga Asparagopsis taxiformis in the Mediterranean Sea. Mar Environ 141:44–52. https://doi.org/10.1016/j.marenvres.2018.07.016
Ostalé-Valriberas E, Sempere-Valverde J, Coppa S, García-Gómez JC, Espinosa F (2018) Creation of microhabitats (tidepools) in ripraps with climax communities as a way to mitigate negative effects of artificial substrate on marine biodiversity. Ecol Eng 120:522–531. https://doi.org/10.1016/j.ecoleng.2018.06.023
People J (2006) Mussel beds on different types of structures support different macroinvertebrate assemblages. Austral Ecol 31(2):271–281. https://doi.org/10.1111/j.1442-9993.2006.01585.x
Perkol-Finkel S, Shashar N, Barneah O, Ben-David-Zaslow R, Oren U, Reichart T, Benayahu Y (2005) Fouling reefal communities on artificial reefs: Does age matter? Biofouling 21(2):127–140. https://doi.org/10.1080/08927010500133451
Piazzi L, Bulleri F, Ceccherelli G (2016) Limpets compensate sea urchin decline and enhance the stability of rocky subtidal barrens. Mar Environ Res 115:49–55. https://doi.org/10.1016/j.marenvres.2016.01.009
Pinn EH, Mitchell K, Corkill J (2005) The assemblages of groynes in relation to substratum age, aspect and microhabitat. Estuar Coast Shelf Sci 62(1–2):271–282. https://doi.org/10.1016/j.ecss.2004.09.002
Ponti M, Fava F, Perlini RA, Giovanardi O, Abbiati M (2015) Benthic assemblages on artificial reefs in the northwestern Adriatic Sea: Does structure type and age matter? Mar Environ Res 104:10–19. https://doi.org/10.1016/j.marenvres.2014.12.004
Ricciardi A, Whoriskey FG, Rasmussen JB (1997) The role of the zebra mussel (Dreissena polymorpha) in structuring macroinvertebrate communities on hard substrata. Can J Fish Aquat Sci 54(11):2596–2608. https://doi.org/10.1139/f97-174
Rivera-Ingraham GA, Espinosa F, Garcia-Gomez JC (2011) Conservation status and updated census of Patella ferruginea (Gastropoda, Patellidae) in Ceuta: distribution patterns and new evidence of the effects of environmental parameters on population structure. Anim Biodivers Conserv 34:83–99. Retrieved from: https://idus.us.es/handle/11441/17098
Sánchez-Moyano JE, Estacio FJ, García-Adiego EM, García-Gómez JC (2000) The molluscan epifauna of the alga Halopteris scoparia in Southern Spain as a bioindicator of coastal environmental conditions. J Molluscan Stud 66(4):431–448. https://doi.org/10.1093/mollus/66.4.431
Singh GG, Markel RW, Martone RG, Salomon AK, Harley CD, Chan KM (2013) Sea otters homogenize mussel beds and reduce habitat provisioning in a rocky intertidal ecosystem. PLoS One 8(5):e65435. https://doi.org/10.1371/journal.pone.0065435
Sedano F, Tierno de Figueroa JM, Navarro-Barranco C, Ortega E, Guerra-García J, Espinosa F (2020a) Do artificial structures cause shifts in epifaunal communities and trophic guilds across different spatial scales? Mar Environ Res. https://doi.org/10.1016/j.marenvres.2020.104998
Sedano F, Navarro-Barranco C, Guerra-García JM, Espinosa F (2019) Habitat quality assesment of coastal defence structures; a multi-community approach. Mar Poll Bull
Sedano F, Navarro-Barranco C, Guerra-García JM, Espinosa F (2020b) Understanding the effects of coastal defence structures on marine biota: The role of substrate composition and roughness in structuring sessile, macro and meiofaunal communities. Mar Poll Bull 157:111334. https://doi.org/10.1016/j.marpolbul.2020.111334
Sempere-Valverde J, Ostalé-Valriberas E, Farfán GM, Espinosa F (2018) Substratum type affects recruitment and development of marine assemblages over artificial substrata: A case study in the Alboran Sea. Estuar Coast Shelf Sci 204:56–65. https://doi.org/10.1016/j.ecss.2018.02.017
Simboura N, Zenetos M, Thessalou-Legaki M, Nicolaidou A (1995) Benthic communities of the infralittoral in the N. Sporades (Aegean Sea): variety of biotopes encountered and analysed. Mar Ecol 16(4):283–306
Soniat TM, Burton GM (2005) A comparison of the effectiveness of sandstone and limestone as cultch for oysters, Crassostrea Virginica. J Shellfish Res 24(2):483–485. https://doi.org/10.2983/07308000(2005)24[483:ACOTEO]2.0.CO;2
Southward BAJ, Orton JH (1954) The effects of wave-action on the distribution and numbers of the commoner plants and animals living on the Plymouth breakwater. J Mar Biolog Assoc U.k. 33:1–19
Strain EMA, Morris RL, Coleman RA, Figueira WF, Steinberg PD, Johnston EL, Bishop MJ (2018) Increasing microhabitat complexity on seawall can reduce fish predation on native oysters. Ecol Eng 120:637–644. https://doi.org/10.1016/j.ecoleng.2017.05.030
Torres AC, Veiga P, Rubal M, Sousa-pinto I (2015) The role of annual macroalgal morphology in driving its epifaunal assemblages. J Exp Mar Biol Ecol 464:96–106. https://doi.org/10.1016/j.jembe.2014.12.016
Underwood AJ, Chapman MG, Richards SA (2002) GMAV-5 for Windows. University of Sydney, Australia, An analysis of variance programme
Underwood AJ, Fairweather PG (1989) Supply-side ecology and benthic marine assemblages. Trends Ecol Evol 4(1):16–20. https://doi.org/10.1016/0169-5347(89)90008-6
Underwood AJ, Chapman MG (2013) Intertidal ecosystem. Encycl Biodivers (Second Ed.). 4:332−344.https://doi.org/10.1016/B978-0-12-384719-5.00156-8
Usero JA, Rosado D, Usero J, Morillo J (2016) Environmental quality in sediments of Cádiz and Algeciras Bays based on a weight of evidence approach (southern Spanish coast). Mar Poll Bull 110(1):65–74. https://doi.org/10.1016/j.marpolbul.2016.06.078
Veiga P, Rubal M, Sousa-Pinto I (2014) Structural complexity of macroalgae influences epifaunal assemblages associated with native and invasive species. Mar Environ Res 101:115–123. https://doi.org/10.1016/j.marenvres.2014.09.007
Vieira F, Cavalcante G, Campos E, Taveira-Pinto F (2020) Wave energy flux variability and trend along the United Arab Emirates coastline based on a 40-year hindcast. Renew Energ 160(C):1194–1205. https://doi.org/10.1016/j.renene.2020.07.072
Warfe DM, Barmuta LA, Wotherspoon S (2008) Quantifying habitat structure: surface convolution and living space for species in complex environments. Oikos 117:1764–1773. https://doi.org/10.1111/j.1600-0706.2008.16836.x
Acknowledgements
We want to thank our colleagues Rafael Espinar for helping in data collection, Carlos Navarro for helping with data analyses, Jose Manuel Guerra for identification assistance and Marta Florido for her tips with the writing. We also express our gratitude to the anonymous reviewers that have contributed to improve the early version of the manuscript.
Funding
X-raY analyses were funded by Ministerio de Economía y Competitividad, Spain (Project CGL2017-82739-P co-financed by Agencia Estatal de Investigacion-AEI- and Fondo Europeo de Desarrollo Regional -FEDER-).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval/ consent to participate
"Not applicable to this study."
Consent for publication
The author gives Urban Ecosystems consent for publication.
Conflicts of interests
"Not applicable to this study."
Additional information
The original online version of this article was revised due to a retrospective Open Access cancellation.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ortega-Jiménez, E., Sedano, F. & Espinosa, F. Molluscs community as a keystone group for assessing the impact of urban sprawl at intertidal ecosystems. Urban Ecosyst 25, 819–834 (2022). https://doi.org/10.1007/s11252-021-01192-6
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11252-021-01192-6