Abstract
Urbanization in the Sahel is constantly competing with and greatly affecting the woody flora in urban areas. Urbanization can replace the species mix, leading to changes in plant community composition; however, there are limited studies that assess these attributes in Sahelian cities. This study assessed urban forests in Niamey and Maradi, two important cities of Niger, West Africa for differences between the cities and across LULC types in terms of species diversity and stand structure. Woody species were inventoried in 357 plots in urban forests located across seven land use/land cover types. Stem diameter at breast height of trees and shrubs ≥2.5 cm, total height and crown diameter were measured. Eighty-six species belonging to 69 genera with 33 families were inventoried in Niamey, while in Maradi 91 species belonging to 70 genera with 30 families were enumerated. Fabaceae was the dominant family in both cities. Azadirachta indica accounted for 41 and 54% of all stems in Niamey and Maradi. Residential areas had a higher species richness than other land uses. The majority of the species documented in the two cities were exotics representing 52% of all species encountered. Mean Shannon diversity index (H′) and standard deviation was 2.31 ± 0.43 for Niamey and 2.14 ± 0.74 for Maradi. The similarity index of the two cities was 70%. Urban forests of Maradi had significantly higher levels of species richness and evenness as well as tree density and canopy cover than urban forests of Niamey. Across LULC types, canopy cover varied significantly (p < 0.05) but basal area had a marginal variation whilst, stem density was not significantly different. There were significant positive relationships (p < 0.05) between species richness, basal area and canopy cover across the LULC types in both cities. The study shows that land use/land cover types influence the characteristics of urban forests and differences exist in diversity and stand structural characteristics of urban forests in the two cities. Future development of urban forests in the two cities must increase the planting of native species to improve their value for biodiversity conservation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Rapid urbanization and changes in peri-urban land use are the leading causes of shifts in vegetation cover. Urbanization causes the loss of agricultural lands (Bolca et al. 2007), destruction of natural habitats (He et al. 2014) and is responsible for loss of forest cover and introduction of new species to cities (Elmqvist et al. 2013). Vegetation in peri-urban areas is often cleared for construction of roads, settlements or industries (Su et al. 2014). Subsequently, agricultural land is converted into residential and other land uses (He et al. 2014) leading to displacement of agricultural frontiers to marginal areas (McKinney 2002).
The need for more sustainable urbanization has led to increased research on urban forests (Arnberger et al. 2016; Kabisch et al. 2015). This is due to the potential of urban forests to mitigate impacts of land use change in peri-urban areas and urban lands (Elmqvist et al. 2013) and provide a variety of ecosystem services (Boukili et al. 2017; Dingaan and du Preez 2010; Nowak et al. 2013). Trees and shrubs in urban forests improve air quality, and provide food, habitat for animals, aesthetic appeal, as well as places where people go to socialize, exercise, or connect with nature (Borelli and Conigliaro 2018). They also mitigate climate change through carbon sequestration, create microclimates and reduce floods (Greene and Millward 2017; Livesley et al. 2014, 2016). The challenge then is what scientific information is needed to facilitate the development and management of urban forests on a sustainable basis.
Some empirical studies across the world report of urban areas being rich in tree populations and species diversity. In peri-urban areas in North America, high tree densities and species richness are documented for many cities such as the metropolitan area in Minnesota (Berland 2012) and the City of Syracuse in New York (Nowak et al. 2016). Similar findings are reported for cities in other continents such as Beijing in Asia (Yan and Yang 2017; Yang 2016) and Adelaide in Australia (Tait et al. 2005). Urban forests in African cities also have a diversity of woody species as reported in Lomé in Togo (Raoufou et al. 2011), Abuja in Nigeria (Agbelade et al. 2017), Kumasi in Ghana (Nero and Callo-concha 2018), Nairobi in Kenya (Nyambane et al. 2016), in Bloemfontein, Free State, South Africa (Dingaan and du Preez 2010), in Eastern Cape towns, South Africa (Gwedla and Shackleton 2017) and in in selected towns of the Eastern Cape, South Africa (Kuruneri-Chitepo and Shackleton 2011). These studies reported on woody plant species used in urban forests, their abundance, growth characteristics and diversity, species coexistence and functional relations with green spaces. These studies of urban forests in African cities do not however, provide information on the extent to which species diversity in urban forests are linked to their structural characteristics. Also lacking in the literature is the inclusion of peri-urban vegetation in urban forestry analysis as well as differences in urban forests between cities. There are no reports in the international literature on the composition and structure of urban forests in the Sahelian region. This impedes effort to generalise urban forest characteristics across different climatic zones as well as regions with contrasting cultures and socio-economic development.
Niamey and Maradi are two important cities in Niger, a country which lies right in the heart of the Sahel. With a population growth rate of 3.9% and mass migration of people from rural to urban areas (INS, 2016), Niger’s urban population has increased from 7.5% of the country’s total population in 1967 to 19% in 2017 (INS, 2016). Faced with the challenges of limited access to housing, problems of urban mobility and employment, the two cities are expanding through development of formal and informal settlements and infrastructure (Ynaut 1987). The resultant zoning and infrastructural development of such cities is posited to alter woody species richness and density, resulting in either homogenization or depletion of biodiversity (Blair 1999; Mckinney 2006) or enhanced heterogeneity of the landscape over time (Nowak and Dwyer 2007; Yan and Yang 2017). Assessment of the woody flora in these cities can provide data that address these uncertainties. Focusing on woody species, this study aimed to determine the differences in floristic composition and structure of urban forests between Niamey and Maradi and across seven land use/land cover (LULC) types. The objectives of the study were to determine (i) differences in biodiversity indices, and stand structure of urban forests of Niamey and Maradi and (ii) the variation in diversity and structural characteristics of urban forests across the various LULC types in the two cities. The study tested the following hypotheses: (i) Urban forests in Niamey and Maradi are rich in woody biodiversity which is associated with stand structure (ii) there are differences between the species diversity and stand structure of urban forests of Niamey and Maradi, (iii) there are differences in urban forest richness and structural parameters (basal area and canopy cover) in the two cities across the LULC types.
Materials and methods
Study area
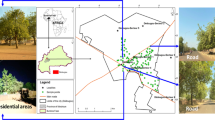
The study was carried out in the cities of Niamey and Maradi in Niger. The two cities are situated in the Sahel Zone, where the mean annual rainfall varies from 150 mm to 350 mm and is distributed in a unimodal pattern (CNEDD 2016). Niamey is the political capital and largest city in Niger with an extended area of over 552.27 km2, an urbanized area of 297.46 km2, and a population of 1,026,848 people (INS 2017). It is located at latitude 13°20′-13°35’N and longitude 2°00′-2°15′E in western Niger (Fig. 1). The mean annual temperature in Niamey is 29.2 °C. Maradi is Niger’s centre of commerce and the third largest city with 326,804 inhabitants and covers 86 km2 (INS, 2016) (Fig. 1). Maradi is located at latitude 13°29′- 13°49’N and longitude 7°5′ -7°09′E in south-central Niger. The mean annual temperature of the city is 27.3 °C.
Agriculture and trade are the economic mainstays in and around Niamey and Maradi. Agricultural production systems consist of rain-fed and irrigated agriculture under urban and peri-urban crop and livestock husbandry. Apart from local land use, anthropogenic activities that impact on vegetation in Niamey and Maradi are overexploitation and species introductions or removals (Moussa et al. 2019). Vegetation cover across the two cities is made up of a mosaic of steppe and short grass savanna, tree plantations, greenbelts and gardens (Saadou 1990). Remnant natural vegetation is dominated by Combretaceae, shrubs and savanna grassland (Wezel et al. 2000). There are also patches of gallery forests, although much of the woody cover has been lost due to drought and cutting trees for firewood (Saadou 1990). The droughts of 1970 and 1984 (Nicholson et al. 1998) and historical factors such as prior land use and colonial administration have influenced urban flora in Niamey and Maradi (Saadou Mahamane personal communication ,2018). For example, species such as Delonix regia, Averrhoa carambola and Plumeria rubra were introduced in Niamey and Maradi during the colonial period (Yacouba 1999). The creation of Niamey Green Belt in 1965 and subsequent programs aimed at fighting desertification saw widespread establishment of Azadirachta indica or neem tree, Eucalyptus camaldulensis, Khaya senegalensis and Prosopis juliflora (Yacouba 1999).
The two cities (Niamey and Maradi) chosen for this study have peculiar characteristics that could influence the urban forests diversity and structure distribution. Niamey is a political capital of Niger, largest city in terms of urban population and most urbanised city in Niger (Niamey has no land to sell as it has already exhausted all lands in its administrative territory (Niger’s Ministry of Habitat and Urbanization, 2018). In addition Niamey has 10 major public green spaces (one green belt of 2500 ha and nine green spaces of 42 ha) (Ministère de l’Environnement du Niger 2010) and some public botanical gardens. Niamey also has some water networks. For example, it is traversed by Niger River. Maradi is the economic capital and third largest city of Niger in terms of urban human population (Institut National de la Satistique du Niger (INS) 2016). It is the second city after Niamey in terms of physical urbanization and urban green spaces. It has consumed its administrative territory (Regional direction of Habitat and urbanization of Maradi, 2018). Maradi possesses 11 bloc forest plantations according to Gambo, (2013). Maradi has two botanical gardens (2.69 ha) in the Regional Agricultural Research Centre of Maradi (INRAN) and the newly created botanical garden of Université Dan Dicko Dankoulodo, Maradi. There exist also some water bodies in Maradi. Niamey and Maradi face the challenge of massive migration from the rural areas of Niger. The above characteristics have been reported as some of the determinant factors of urban forest development (Alvey 2006; Cilliers et al. 2013; Fan et al. 2019) thus justifying the choice of the two cities.
Survey of floristic composition and vegetation structure
A stratified random sampling approach was used for the inventory. In each city the study area was stratified into seven LULC types:(1) commercial area that included market, shops, restaurants and garages; (2) forested areas consisting of urban agricultural plots, green belt, urban wetlands, irrigated urban farmlands and botanical gardens; (3) residential areas covering houses, mosques and churches; (4) schools covering private and public training and learning institutions such as primary schools, secondary schools, universities, polytechnics, training colleges; (5) administrative areas such as governments offices and private offices; (6) roads covering the main streets and boulevards. These six LULCs formed the built areas or urban forests in the two cities. (7) peri-urban forest areas consisting of peri-urban croplands, peri-urban wetlands and pastoral field. This LULC type started from the end of the city (where there were no buildings) to a distance of 4 km away into the non-built area. The LULC types were randomly selected from five communes in Niamey and three communes in Maradi. A random list for sampling was prepared from an inventory of schools, administrative posts, urban green spaces, roads, markets and residential compounds obtained from directorates of education, environment, urban equipment and habitat of the communes in Niamey and Maradi. Floristic composition and vegetation structure were assessed through a survey of plants in plots of 50 m × 50 m, consistent with guidelines for inventories in the Sahel (Thiombiano et al. 2016). The plot size varied in some cases and was less than 0.25 ha if a randomly selected point could not allow a plot of a 50 m × 50 m to be demarcated without getting into another LULC type. In such cases, two smaller plots were used in place of one large plot. All woody plants with diameter at breast height (DBH) ≥ 2.5 cm within a plot were inventoried. The minimum DBH of 2.5 cm is recommended for dryland areas (Pearson et al. 2007) where the landscape is dominated by small trees, and some of the species may not attain large DBH at maturity (Thiombiano et al. 2016). DBH was measured at 1.30 m from the ground using callipers or a diameter tape. Woody plants with forks below 1.3 m were considered as multi-stemmed; their individual stems were measured separately and the DBH calculated as the square root of the sum of squares of the individual stems (Thiombiano et al. 2016). Crown diameter was measured crosswise with measuring tape. The longest diameter (d1) and the diameter perpendicular to it (d2) were measured. Species name of all the measured woody species was recorded. The position of each plot was recorded with Global Positioning System. Specimen and photographs were collected for plants whose identity could not be established in the field for later identification at the “Laboratoire de Biologie Garba Mounkaila”, University of Niamey and at Department of Biology of University Dan Dicko Dankoulodo of Maradi. Angiosperm Phylogeny Group IV (2016) classification was used. For the leguminosae, the new classification, LPWG ( 2017) (The Legume Phylogeny Working Group (LPWG) 2017) was used.
Data analysis
Shannon-Wiener diversity (H′), richness (S) and evenness (J) were calculated as indices of species diversity within and across different LULC types in the two cities. Richness was calculated as the total number of tree and shrub species in each LULC type. Shannon diversity index was calculated as the proportion of the species relative to the total number of species using the formula: H′ = −∑ [(ni/N)*Ln (ni/N)], where ni is the number of individuals of species i, N is the total number of individuals per LULC types and Ln is the natural logarithm. Pielou’s evenness (J’) was used as a measure of relative abundance of tree and shrub species, and was calculated using the formula: J = H′/Ln(S), where H′ is the Shannon diversity index and S is the number of species. Sorensen index was calculated for comparing the similarity between Niamey and Maradi using the formula described in (Thiombiano et al. 2016): K = 2C/(2C + A + B), where A is the number of species in one area (e.g. Niamey), B is the number of species in another area (e.g. Maradi) and C is the number of species common to the study sites. Basal area, density and dominance were used to describe the vegetation structure. Basal area was calculated as BA (m2) = D2 * (π/4), where π = 3.14, and D is the DBH. Plant density was calculated as the total number of individuals of all species per unit area. Species density was also determined as the total number of individuals of a species per unit area. Crown area (ca) was calculated assuming an elliptical crown shape, (ca) (m2) = π{(d1/2)x(d2/2), where d1 is the largest crown diameter (m) and (d2) is the diameter perpendicular to the larger crown diameter (m). Canopy cover (%) = ca(100)/Plot size (m2).
Statistical analysis
Prior to the statistical analysis, the Ryan-Joiner test and Levene’ test were used to check the normality and homogeneity of the data. A two-way ANOVA and Duncan Tests were used to examine the difference in the biodiversity characteristics (Shannon, evenness), origin of species (exotic or native) and structural characteristics (basal area, density, canopy cover, number of shrubs and trees) in each city and between cities across the LULCs at alpha 0.05 level of significance. Chi-square test was used to check whether the number of exotic and native stems, trees, shrubs and species were associated with LULCs in each of the city. A linear regression was used to determine the relationship between urban forest species richness and basal area and canopy cover in the two cities across the LULCs. Descriptive statistics was also used to determine the proportion of the number of exotic and native stems, trees, shrubs and species. Minitab version 17 and Excel were used for the statistical analysis.
Results
Floristic composition (trees and shrubs) of urban Forest stands in the two cities
Table 1 shows the diversity characteristics of urban forests of the various LULC types in the two cities. A total of 115 tree and shrub species belonging to 82 genera in 35 families were documented in 50.95 ha across the two study sites. In Niamey, 86 species belonging to 69 genera with 33 families were inventoried while in Maradi 91 woody species belonging to 70 genera with 30 families were enumerated. Mean Shannon diversity index (H′) and standard deviation was 2.31 ± 0.43 for Niamey and 2.14 ± 0.74 for Maradi.
The majority of the species documented in the two cities were exotics representing 52% of all species encountered (p < 0.001). At the city level, Niamey had 52% of species being exotic, while equal numbers of exotic and native species were found in Maradi. In terms of the distribution of exotics and native species there was no significant difference between urban forests of the two cities (X2 = 0.0042, df = 1 p = 0.94). However, at the city level, the distribution of indigenous and exotic species was found to be associated with LULC type in Niamey (X2 = 12.613, df = 5, p = 0.027) and in Maradi (X2 = 58.720, df = 5, p = 0.000). Residential areas had the highest exotic species richness in Niamey (23 exotic species) and in Maradi (35 exotic species). While the lowest number of exotic woody species were observed in peri-urban areas in Maradi (two exotic species) and in commercial areas and peri-urban areas in Niamey with six exotic species respectively. For the native species richness, the highest number was observed in forested areas in both cities. The similarity index of the woody floras in the built areas of the two cities was 70% i.e., there were 62 species that were common to the two cities, 29 species were specific to Maradi while 24 were found only in Niamey.
The diversity characteristics (richness and evenness) varied significantly (p < 0.05) in each of the two cities across the LULC types (Table 1). The overall Shannon index was greater than 2 in each city (Table 1) though there were no significant differences in the mean Shannon index across the LULC types (F = 4.14, df = 1, p = 0.06 in Niamey and F = 4.52, df = 1, p = 0.05 in Maradi). Residential areas had the highest species richness in Niamey (45 species) and in Maradi (56 species) (Table 1). A particular example was a residence in Niamey with 10 species, the highest number of species found in a single plot within a land use. The lowest diversity indices were found in commercial areas for Niamey (H = 1.78) and in schools (H = 0.84) for Maradi (Table 1). The dominant families were Fabaceae, Combretaceae and Verbenaceae with 24, 5 and 6 species in Niamey, and Annonaceae Fabaceae, Euphorbiaceae, Arecaceae, and Combretaceae with 24, 5, 4 and 4 species in Maradi (Fig. 2). In built up areas in Niamey, the percentage of exotic species ranged from 6% to 23% whilst in peri-urban areas it was 6%. In Maradi, the range was from 12% to 29% in built up areas and 2% in peri urban forests.
Structural characteristics of urban forests in the two cities
A total of 4689 individual trees and shrubs (2137 in Niamey and 2552 in Maradi) were documented over an area of 50.95 ha (Table 2). There were more trees than shrubs in both cities (92% in Niamey and 90% in Maradi). With the exception of basal area, there were significant differences (p < 0.05) in the mean structural characteristics (density and canopy cover) between the two cities. The mean stem density (with standard deviation) in Maradi (142.9 ± 62.08 stems/ha) was higher than 70.60 ± 20.36 stems/ha obtained for Niamey (F = 11.585, df = 1, p < 0.05). The mean canopy cover was also higher in Maradi (53.67% ± 27.16) than in Niamey (37.90% ± 14.56) (F = 6.57, df = 1, p < 0.05). However, the stand mean basal area for Maradi (11.26 ± 5.70 m2 /ha) was not significantly different from that for Niamey (8.60 ± 3.80 m2 /ha) (F = 2.88, df = 1, p > 0.05).
Across LULC types in Niamey, stem density ranged from a high of 108 stems/ha along roads to a low of (48 stems/ha) in peri-urban areas. In Maradi, stem density was highest in residential areas (216 stems/ha) and lowest (43 stems/ha) in peri-urban areas. Considering the two cities stem density was not significantly different across the LULC types (F = 1.704, df = 6, p > 0.05). Basal area was marginally significantly different across LULC types (F = 4.258, df = 6, p = 0.051). Its highest value (19.36 m2/ha) was observed in urban forests along roads in Maradi and the lowest (3.65 m2/ha) in peri-urban forests of the same city.
There was significant effect of species richness on basal area in the urban forests in Niamey (R2 = 0.75, Fsig = 0.08, p = 0.005) and in Maradi (R2 = 0.80, Fsig = 0.04, p = 0.02). There was also significant effect of species richness on canopy cover in Niamey (R2 = 0.81, Fsig = 0.004, p = 0.002 and in Maradi (R2 = 0.77, Fsig = 0.06, p = 0.00). For canopy cover, urban forests in commercial areas and along roads in Maradi had the highest values (87.87% and 82.56% respectively) whilst peri-urban forests of the two cities recorded the lowest values (17.8–15.37%). Canopy cover had significant variation across LULC types in the two cities (F = 6.574, df = 6, p < 0.05). The distribution of the number of trees and shrubs was associated with LULC types in Niamey (X2 = 79.171, df = 12, p = 0.000) and in Maradi (X2 = 206.113, df = 12, p = 0.000). Schools were more associated with trees than other LULC in both cities. However, forested areas were more associated with shrubs.
In terms of relative abundance, (Table 2) more exotic than native tree and shrub were enumerated in Niamey (62%, N = 1319) as well as in Maradi (77%, N = 1967). Statistically urban forests in Maradi were associated with higher levels of trees of exotic origin than urban forests in Niamey (X2 = 295.94, df = 1, p < 0.05). Schools had the higher individual exotic trees and shrubs than other LULC types. They had 27% and 29% respectively of all stems of exotic trees in urban forests of Niamey and Maradi respectively. The number of species by families across the city is presented in Table 3.
Azadiractha indica had the highest number of individuals in Niamey and Maradi, accounting for 41% and 54% of the total number of trees and shrubs documented, respectively from peri-urban to the built areas. In Niamey, other dominant species were Balanites aegyptiaca, Terminalia mantaly, Faidherbia albida, accounting for 8, 6 and 4%, respectively of the total number of trees and shrubs (Fig. 3). In Maradi, the most abundant species next to A. indica were T. mantaly, Vachellia nilotica and B. aegyptiaca accounting for 3, 4 and 3%, respectively of the trees and shrubs enumerated in the built area. F. albida (21%) was the dominant peri-urban forest species in Maradi while B. aegyptiaca dominated in Niamey peri-urban forests.
Uncommon species, (i.e. those with a mean of less than one individual per species) were 21 in Niamey and 16 in Maradi in the built areas. Ten of them in Niamey were exotic while nine were native to Africa. In Maradi, eight uncommon species were exotic; the rests were native to Africa. These species were specific to the cities, and only Vitellaria paradoxa was common to both cities. In Niamey, Mangifera indica, Ziziphus mauritiana, Vitex doniana, Adansonia digitata, and Hyphaene thebaica were the five most common fruit species with a combined frequency of 8%. In Maradi, the five most common fruit species were M. indica, Citrus lemon, A. digitata, Annona squamosa, and Moringa oleifera, with a combined frequency of 7%.
Discussion
The results show a high species diversity in Niamey and Maradi. The Shannon diversity index for the two cities was greater than 2.0, which indicates medium to high diversity (Magurran 2004). This demonstrates the importance of the two cities in woody species conservation. These results confirm the findings of (Gillespie et al. 2017; Ortega-Álvarez et al. 2011; Pickett et al. 2001) who reported that urban areas are home to a diversity of woody species. Among the species recorded in the two cities are some of the IUCN’s list of vulnerable species such as Khaya senegalensis and endangered species such as Combretum glutinosum, Senna singueana, Boswellia odorata, Prosopis africana, Anogeissus leiocarpus, and Daniella oliveri in Niger. The presence of these species in the cities’ urban forests lends credence to Conservation Visions’ philosophy that cities can play a significant role in the conservation of threatened plant species (Elmqvist et al. 2013). The high species diversity may have been achieved as a result of the Operation Green Sahel program that aimed at fighting desertification and during which many exotic species were introduced (Yacouba 1999). It could also be related to the diversity of ethnic groups with different cultures found in the two cities. The area being cosmopolitan is likely to host different ethnic groups with diverse preferences of tree and shrub species (Dwyer et al. 1991). It emerged from the field discussion with residents that some trees were introduced from outside Niger by migrants for reasons such as aesthetics e.g. (Coccoloba uvifera) and medicinal purposes (Senna alata). Hope et al. (2003) reported that diversity increases with urbanization because of human influences such as irrigation and ornamental landscaping. The high diversity determined for the two cities may contribute to ecosystem resilience (Kendal et al. 2014) and provision of multiple ecosystem services (Nowak and Dwyer 2007). Urban forests with low diversity tend to be vulnerable to ecological disturbances, such as the effects of pests and diseases (Alvey 2006) and impacts of climate change (Elmqvist et al. 2013). Between the two cities, significant differences exist in species richness, Shannon index and evenness of the urban forests, with Maradi having higher values than Niamey except in the case of Shannon index. This may probably be explained by the fact that being the capital city, Niamey may have its tree planting being more formal and centrally controlled than Maradi. In that case, individual residents’ influence on species choice will be limited in Niamey resulting in fewer species being planted than in Maradi.
With the exception of basal area, there were significant differences (p < 0.05) in the mean structural characteristics (density and canopy cover) between the two cities. This implies there were many smaller trees in Maradi and few larger trees in Niamey. It may be a reflection of age differences in the urban forests of the two cities with Niamey having an older history of urban forests than Maradi. However, other possibilities such as trees growing faster in Niamey than Maradi cannot be ruled out.
The study shows that the species richness varied significantly across the LULC types. For instance, residential urban forests had the highest species diversity in both cities suggesting that residents promote the number of woody species by choosing various woody species for their residential areas. Thus, in Niamey and Maradi, people plant multipurpose trees in their homes, and their preferences may vary according to ethnic, religious, and socioeconomic backgrounds or educational levels (Fan et al. 2019; Hungerford and Moussa 2017; Nitoslawski et al. 2016). Studies of other cities show that people maintain a variety of trees on their compounds for various reasons (Cilliers et al. 2013; Clark and Nicholas 2013; du Toit et al. 2018; Park et al. 2019; Raoufou et al. 2011). This supports the results of Hope et al. (2003); Hungerford and Moussa (2017); Wang et al. (2015) who reported that the household is a driver of plant species diversity.
The values of the Shannon diversity index were low in schools’ urban forests in both cities This is an important observation because species diversity in schools has been found to impact on students’ performance (Kweon et al. 2017; Sivarajah et al. 2018). Therefore, school authorities have to address the problem and this could be an opportunity for youth urban forestry education in both cities. Lower Shannon index values were observed in the commercial areas in Niamey which confirmed the finding of (Ortega-Álvarez et al. 2011) who observed fewer species diversity in commercial areas in Mexico city. Lower diversity seems not to be a surprise due to lack of space for planting trees in commercial areas especially markets. However, innovative ways can be found to improve the diversity by planting different woody species in the few spaces available.
The study recorded the presence of Neem (Azadirachta indica) trees in peri-urban forests of the two cities and Prosopis juliflora in peri-urban forests of Niamey where the native woody species are supposed to be naturally occurring. This may be the beginning of the invasion of peri-urban forests of Niger by the two species. Neem is noted as an invasive species in forest fragments of Southern Togo (Radji et al. 2010) whilst P. juliflora has invaded South Africa and India forests (Kumar and Mathur 2014; Shackleton et al. 2015).
Fabaceae and A. indica were the dominant family and species respectively in both cities. The family Fabaceae is widespread in semi-arid areas on a wide range of soils and climate (Mudzengi et al. 2014) and this may explain its abundance in the cities. The dominance of Fabaceae is a good proxy for overall angiosperm diversity in many habitats (Raes et al. 2013). The list of Fabaceae species included some multipurpose species such as Faidherbia albida, Tamarindus indica, Acacia senegal, Dialium guineense and Parkia biglobosa. The multipurpose nature of these species may also contribute to the family’s dominance in the urban forests. The dominance of the Neem tree may be traced to its role as a major species for the Sahel greening under various tree planting programs (Ministère de l’Environnement du Niger 2010) and its multi-purpose nature. Neem tree is used for construction, pest and disease control, traditional medicine and provision of shade among others worldwide (Ezzat et al. 2018; Raj and Sahu 2013).
Of the stand structural characteristics, canopy cover was the one that varied most across the LULC types followed by basal area whilst stem density was not significantly different across the LULC types. This may be explained by the unequal distribution of shrubs and trees in the LULC types such that types with more trees will have higher canopy cover than those with relatively few trees. It may also be accounted for by the fact that in LULC types where trees are planted mostly for shade preference will be given to large crown trees, which provide high canopy cover. This preference may then create a difference in canopy cover among the LULC types even where they have similar tree density. The study shows that stand basal area varied significantly with species richness suggesting that diversity increases stand basal in the two cities. This matches the findings of (Erskine et al. 2006; Liang et al. 2007) who reported that increases in species richness augment the stand basal area or mean individual tree basal area.
Implications of the high number of woody species in the urban forests of Niamey and Maradi
The study shows that in terms of species presence, there is statistically equal distribution of natives and exotics in both cities besides, some LULC types were more associated with exotics than natives. More exotic species were found in residential areas while native species were more in forested areas. Studies in the city of Sheffield, South Yorkshire, UK (Smith et al. 2006) and in Mexico city (Ortega-Álvarez et al. 2011) have shown that exotic species are preferred in residential areas because of the provision of different ecosystem services that may not be provided by indigenous species (Davies et al. 2011; Riley et al. 2018). It emerged from the field discussion with residents that some exotic trees are used in both cities for various purposes. For example, Azadirachta indica and, Terminalia mantaly are used for shading purpose, Citrus spp., mango tree, Anacardium occidentale, Annona spp. are used for fruit production, Bougainvillea spectabilis, Cola cordifolia, Nerium oleander for ornamental purpose, leaves of moringa spp. are used for human food, and the Neem tree, Senna alata are used for medicinal purpose in both cities. Further, the Neem trees planted in the mosques are used for burial purpose in Maradi. In addition to that, the Neem trees planted in secondary schools in both cities provide a wood that is used for the construction of hut classrooms (Classes paillotes in French). Nevertheless, other residents reported some ecosystem disservices delivered by exotic tree species in the two cities such as allergy (for example, someone told us that he was allergic to Neem tree pollen), Terminalia mantaly destroys walls, tap water installations, toilets, gutters in both cities through is root systems. Conflict between neighbours due to the branches of some exotic species falling on to neighbouring houses, walls, cars has been reported during the field discussion with residents in the two cities. The leaves of some deciduous exotic species such as Terminalia mantaly fall during dry spell make houses and neighbouring houses dirty has also been reported as exotic species disservices in both cities. In addition to that, the green belt of Niamey in which Neem tree is dominant specie has become the zone of human insecurity. Discussion with green belt of Niamey neighbouring residents reported that many people were killed in the green belt of Niamey city and it is dumping refuses of all types of wastes such as plastic wastes. Many of the exotic species were introduced in Maradi and Niamey during the colonial period, and also within the national afforestation programs of 1965 and 1975 (Ministère de l’Environnement du Niger 2010). Their high number suggests that many of the introduced species are well adapted to the Sahel climatic conditions. The presence of a variable number of exotic and native species in the cities, indicate the mixed nature of the urban forests and may come with some advantages. For instance, the presence of exotic species can be crucial for urban food production (Riley et al. 2018) and the fight against urban human malnutrition (FAO 2016). Nowak et al. (2017) reported the mix urban forest is important to mitigate urban air pollution and the presence of exotics can contribute to this species mix. However, the dominance of exotic species has implications for native species conservation. Ikin et al. (2012) reported more native birds and native invertebrates using native trees in urban areas showing the potential danger associated with having relatively few indigenous species in urban forests.
Conclusions
The findings show that Niamey and Maradi have high tree species richness but more than half of the woody flora population is made up of individuals of exotic origin. Between the two cities, significant differences exist in species richness, Shannon index and evenness as well as some stand structural characteristics (tree density and canopy cover) of the urban forests, with Maradi having higher values than Niamey except in the case of Shannon index. Maradi can therefore be said to possess better urban forests than Niamey in terms of these forest parameters. Across LULC types, species diversity and stand structure of the urban forests showed significant differences thus the location of urban forests within the city has influence on the species composition and basic stand structure. The results provide a baseline against which future assessments and monitoring can be done.
References
Agbelade, A.D., Onyekwelu, J.C., Oyun, M.B., 2017. Tree species richness, diversity, and vegetation index for Federal Capital Territory, Abuja, Nigeria. Int J Forest Res 12. https://doi.org/10.1155/2017/4549756
Alvey AA (2006) Promoting and preserving biodiversity in the urban forest. Urban Forest Urban Greening 5:195–201. https://doi.org/10.1016/j.ufug.2006.09.003
Arnberger A, Brigitte A, Eder R, Ebenberger M, Wanka A, Kolland F, Wallner P, Hutter H-P (2016) Elderly resident’s uses of and preferences for urban green spaces during heat periods. Urban For Urban Green 21:102–115. https://doi.org/10.1016/j.ufug.2016.11.012
Berland A (2012) Long-term urbanization effects on tree canopy cover along an urban-rural gradient. Urban Ecosyst 15:721–738. https://doi.org/10.1007/s11252-012-0224-9
Blair RB (1999) Birds and butterflies along an urban gradient : surrogate taxa for assessing biodiversity? Ecol Appl 9:164–170. https://doi.org/10.1890/1051-0761(1999)009[0164:BABAAU]2.0.CO;2
Bolca M, Turkyilmaz B, Kurucu Y, Altinbas U, Esetlili MT, Gulgun B (2007) Determination of impact of urbanization on agricultural land and wetland land use in Balçovas’ Delta by remote sensing and GIS technique. Environ Monit Assess 131:409–419. https://doi.org/10.1007/s10661-006-9486-0
Borelli, S., Conigliaro, M., 2018. Urban forests in the global context.http://www.fao.org/3/i8707en/I8707EN.pdf. Unasylva 69, 3–79
Boukili VKS, Bebber DP, Mortimer T, Venicx G, Lefcourt D, Chandler M, Eisenberg C (2017) Assessing the performance of urban forest carbon sequestration models using direct measurements of tree growth. Urban For Urban Green 24:212–221. https://doi.org/10.1016/j.ufug.2017.03.015
Cilliers S, Cilliers J, Lubbe R, Siebert S (2013) Ecosystem services of urban green spaces in African countries-perspectives and challenges. Urban Ecosyst 16:681–702. https://doi.org/10.1007/s11252-012-0254-3
Clark KH, Nicholas KA (2013) Introducing urban food forestry: a multifunctional approach to increase food security and provide ecosystem services. Landsc Ecol 28:1649–1669. https://doi.org/10.1007/s10980-013-9903-z
CNEDD, 2016. Troisieme Communication Nationale a la Conference des Parties de la Convention Cadre des Nations Unies sur les Changements Climatiques. 1-157P. https://unfccc.int/sites/default/files/resource/nernc3_0.pdf. Niamey
Davies ZG, Edmondson JL, Heinemeyer A, Leake JR, Gaston KJ (2011) Mapping an urban ecosystem service: quantifying above-ground carbon storage at a city-wide scale. J Appl Ecol 48:1125–1134. https://doi.org/10.1111/j.1365-2664.2011.02021.x
Dingaan MNV, du Preez PJ (2010) Floristic composition and species diversity of urban vegetation in Bloemfontein, Free State, South Africa. Bothalia-African Biodiversity Conserv 47(1–15). https://doi.org/10.4102/abc.v47i1.2244
Direction Régionale de l’Institut National de la Statistique (INS) de Niamey (2017). Annuaire Statistique Regional de Niamey 2012–2016. 1-94p. www.statniger.org/statistique/.../Regions/Niamey. Niamey
du Toit MJ, Cilliers SS, Dallimer M, Goddard M, Guenat S, Cornelius SF (2018) Urban green infrastructure and ecosystem services in sub-Saharan Africa. Landsc Urban Plan 180:249–261. https://doi.org/10.1016/j.landurbplan.2018.06.001
Dwyer JF, Schroeder HW, Gobster PH (1991) The Significane of urban trees and forests: toward a deeper understanding of values. J Arboric 17:276–284
Elmqvist T, Fragkias M, Goodness J, Marcotullio PJ, McDonald RI, Parnell S, Schewenius M, Sendstad M, Seto KC, Wilkinson C (2013) Urbanization, Biodiversity and Ecosystem Services: Challenges and Opportunities. Springer, New York
Erskine PD, Lamb D, Bristow M (2006) Tree species diversity and ecosystem function : can tropical multi-species plantations generate greater productivity ? For Ecol Manag 233:205–210. https://doi.org/10.1016/j.foreco.2006.05.013
Ezzat HN, Jawad HS, Abood SS (2018) A review on the effects of neem (Azadirachta indica) as feed additive in poultry production. J Entomol Zool Stud 6:1331–1333
Fan C, Johnston M, Darling L, Scott L, Liao FH (2019) Land use and socio-economic determinants of urban forest structure and diversity. Landsc Urban Plan 181:10–21. https://doi.org/10.1016/j.landurbplan.2018.09.012
FAO (2016) Guidelines on urban and peri-urban forestry, by F. Salbitano, S. Borelli, M. Conigliaro and Y. Chen. FAO Forestry Paper No. 178. Rome, Food and Agriculture Organization of the United Nations. www.fao.org/3/a-i6210e.pdf
Gambo S (2013) Méthodologies d’évaluation et d’inventaire des ressources forestières et agro forestières urbaines et périurbaines dans la zone d’intervention : Projet Promotion de la Foresterie Urbaine et Péri Urbaine au Niger (FUPU). Niamey
Gillespie TW, De Goede J, Aguilar L, Jenerette GD, Fricker GA, Avolio ML, Pincetl S, Johnston T, Clarke LW, Pataki DE (2017) Predicting tree species richness in urban forests. Urban Ecosyst 20:839–849. https://doi.org/10.1007/s11252-016-0633-2
Greene CS, Millward AA (2017) Getting closure : the role of urban forest canopy density in moderating summer surface temperatures in a large city. Urban Ecosyst 20:141–156. https://doi.org/10.1007/s11252-016-0586-5
Gwedla N, Shackleton CM (2017) Population size and development history determine street tree distribution and composition within and between eastern cape towns, South Africa. Urban Forest Urban Greening 25:11–18. https://doi.org/10.1016/j.ufug.2017.04.014
He C, Liu Z, Tian J, Ma Q (2014) Urban expansion dynamics and natural habitat loss in China: a multiscale landscape perspective. Glob Chang Biol 20:2886–2902. https://doi.org/10.1111/gcb.12553
Hope D, Gries C, Zhu W, Fagan WF, Redman CL, Grimm NB, Nelson AL, Martin C, Kinzig A (2003) Socioeconomics drive urban plant diversity. Proc Natl Acad Sci U S A 100:8788–8792. https://doi.org/10.1073/pnas.1537557100
Hungerford H, Moussa Y (2017) Seeing the (urban) forest through the trees : governance and household trees in Niamey , Niger. African Geographic Rev 36:1–19. https://doi.org/10.1080/19376812.2016.1226909
Ikin K, Knight E, Lindenmayer DB, Fischer J, Manning AD (2012) The influence of native versus exotic streescape vegetation on the spatial distribution of birds in subsurbs and reserves. Divers Distrib:1–13. https://doi.org/10.1111/j.1472-4642.2012.00937.x
Institut National de la Satistique du Niger (INS) (2016) Le Niger en Chiffres 2016. 1-84p. www.stat-niger.org/statistique/file/.../NigerEnChiffres2016.pdf. Niamey
Kabisch N, Qureshi S, Haase D (2015) Human – environment interactions in urban green spaces — a systematic review of contemporary issues and prospects for future research. Environ Impact Assess Rev 50:25–34. https://doi.org/10.1016/j.eiar.2014.08.007
Kendal D, Dobbs C, Lohr VI (2014) Global patterns of diversity in the urban forest: is there evidence to support the 10/20/30 rule? Urban For Urban Green 13:411–417. https://doi.org/10.1016/j.ufug.2014.04.004
Kumar S, Mathur M (2014) Impact of invasion by Prosopis juliflora on plant communities in arid grazing lands. Trop Ecol 55:33–46 https://doi.org/http://tropecol.com/pdf/open/PDF_55_1/03-Kumar%20&%20Mathur.pdf
Kuruneri-Chitepo C, Shackleton CM (2011) The distribution, abundance and composition of street trees in selected towns of the eastern cape, South Africa. Urban Forest Urban Greening 10:247–254. https://doi.org/10.1016/j.ufug.2011.06.001
Kweon BS, Ellis CD, Lee J, Jacobs K (2017) The link between school environments and student academic performance. Urban Forest Urban Greening 23:35–43. https://doi.org/10.1016/j.ufug.2017.02.002
Liang J, Buongiorno J, Monserud RA, Kruger EL, Zhou M (2007) Effects of diversity of tree species and size on forest basal area growth, recruitment, and mortality. For Ecol Manag 243:116–127. https://doi.org/10.1016/j.foreco.2007.02.028
Livesley SJ, Baudinette B, Glover D (2014) Rainfall interception and stem flow by eucalypt street trees – the impacts of canopy density and bark type. Urban For Urban Green 13:192–197. https://doi.org/10.1016/j.ufug.2013.09.001
Livesley SJ, McPherson EG, Calfapietra C (2016) The urban Forest and ecosystem services: impacts on urban water, heat, and pollution cycles at the tree, street, and City scale. J Environ Qual 124:119–124. https://doi.org/10.2134/jeq2015.11.0567
Magurran, A.E., 2004. Measuring biological diversity: rejoinder, Environmental and Ecological Statistics. Blackwell Science L.td. https://doi.org/10.2989/16085910409503825
McKinney ML (2002) Urbanization, biodiversity, and conservation. BioScience 52:883–890. https://doi.org/10.1641/0006-3568(2002)052[0883:UBAC]2.0.CO;2
Mckinney ML (2006) Urbanization as a major cause of biotic homogenization. Biol Conserv 3:247–260. https://doi.org/10.1016/j.biocon.2005.09.005
Ministère de l’Environnement du Niger (2010) Projet de Promotion de la Foresterie Urbaine et Péri-urbaine dans la Lutte contre les Changements Climatiques au Niger. 1-11p. Niamey. https://doi.org/Unpbublished
Moussa S, Kyereh B, Tougiani A, Kuyah S, Mahamane S (2019) West African Sahelian cities as source of carbon stocks: evidence from Niger. Sustain Cities Soc 50:101653. https://doi.org/10.1016/j.scs.2019.101653
Mudzengi C, Kativu S, Dahwa E, Poshiwa X, Murungweni C (2014) Effects of Dichrostachys cinerea (l.) Wight &amp; Arn (Fabaceae) on herbaceous species in a semi-arid rangeland in Zimbabwe. Nat Conserv 7:51–60. https://doi.org/10.3897/natureconservation.7.5264
Nero BF, Callo-concha D (2018) Structure , Diversity , and Carbon Stocks of the Tree Community of Kumasi , Ghana. Forests 9:1–17. https://doi.org/10.3390/f9090519
Nicholson SE, Tucker CJ, Ba MB (1998) Desertification, drought, and surface vegetation: an example from the west African Sahel. Bull Am Meteorol Soc 79:815–829. https://doi.org/10.1175/1520-0477(1998)079<0815:DDASVA>2.0.CO;2
Nitoslawski SA, Duinker PN, Bush PG (2016) A review of drivers of tree diversity in suburban areas: research needs for north American cities. Environ Rev 24:471–483. https://doi.org/10.1139/er-2016-0027
Nowak DJ, Dwyer JF (2007) Understanding the benefits and costs of urban Forest ecosystems. Springer, Dordrecht:25–45. https://doi.org/10.1007/978-1-4020-4289-8
Nowak DJ, Green EJ, Hoehn RE, Lapoint E (2013) Carbon storage and sequestration by trees in urban and community areas of the United States. Environ Pollut 178:229–236. https://doi.org/10.1016/j.envpol.2013.03.019
Nowak DJ, Hoehn RE, Bodine AR, Greenfield EJ, Neil-dunne JO (2016) Urban forest structure, ecosystem services and change in Syracuse, NY. Urban Ecosyst 19:1455–1477. https://doi.org/10.1007/s11252-013-0326-z
Nowak DJ, Mcgovern M, Pasher J (2017) Air pollution removal by urban forests in Canada and its effect on air quality and Human Health Urban Forestry & Urban Greening air pollution removal by urban forests in Canada and its e ff ect on air quality and human health. Urban For Urban Green 29:40–48. https://doi.org/10.1016/j.ufug.2017.10.019
Nyambane DO, Njoroge JB, Watako AO (2016) Assessment of tree species distribution and diversity in the major urban green spaces of Nairobi city , Kenya. J Horticult Forest 8:12–23. https://doi.org/10.5897/JHF2016.0439
Ortega-Álvarez R, Rodríguez-Correa HA, MacGregor-Fors I (2011) Trees and the city: diversity and composition along a neotropical gradient of urbanization. Int J Ecol 2011:8. https://doi.org/10.1155/2011/704084
Park H, Kramer M, Rhemtulla JM, Konijnendijk CC (2019) Urban food systems that involve trees in northern America and Europe: a scoping review. Urban Forest Urban Greening 45:126360. https://doi.org/10.1016/j.ufug.2019.06.003
Pearson TRH, Brown SL, Birdsey RA (2007) Measurement guidelines for the sequestration of Forest carbon. Northern Res Station 42
Pickett STA, Cadenasso ML, Grove JM, Nilon CH, Pouyat RV, Zipperer WC, Costanza R (2001) Urban Ecological Systems: Linking Terrestrial Ecological, Physical,and Socioeconomic Components of Metropolitan Areas. Annu Rev Ecol Syst 32:127–157. https://doi.org/10.1146/annurev.ecolsys.32.081501.114012
Radji R, Klu K, Kokou K (2010) Forest invasion by alien plant species : The case of neem tree (Azadirachta indica A . Juss .) in Southern. Int J Biodivers Conserv 2:300–307 https://doi.org/https://academicjournals.org/journal/IJBC/article-full-text-pdf/8C7B54B16718
Raes N, Saw LG, van Welzen PC, Yahara T (2013) Legume diversity as indicator for botanical diversity on Sundaland, South East Asia. S Afr J Bot 89:265–272. https://doi.org/10.1016/j.sajb.2013.06.004
Raj, A., , Sahu, K.P., 2013. Neem- a tree for solving global problem Manoj Kumar Jhariya. Indian J Appl Res 3, 1–3. https://doi.org/https://www.slideshare.net/AbhishekRaj77/neem-50266637
Raoufou R, Kouami K, Koffi A (2011) Woody plant species used in urban forestry in West Africa : case study in Lomé, capital town of Togo. J Horticult Forest 3:21–31 https://doi.org/https://academicjournals.org/article/article1379496813_Raoufou%2520et%2520al%2520Pdf.pdf
Riley CB, Herms DA, Gardiner MM (2018) Exotic trees contribute to urban forest diversity and ecosystem services in inner-city Cleveland, OH. Urban Forest Urban Greening 29:367–376. https://doi.org/10.1016/j.ufug.2017.01.004
Saadou M (1990) La végétation des milieux drainés nigériens à l’est du fleuve Niger. Thèse de doctorat, Université Niamey, Niger, 393p
Shackleton RT, Le DC, Richardson DM (2015) Prosopis invasions in South Africa : population structures and impacts on native tree population stability. J Arid Environ 114:70–78. https://doi.org/10.1016/j.jaridenv.2014.11.006
Sivarajah S, Smith SM, Thomas SC (2018) Tree cover and species composition effects on academic performance of primary school students. PLoS One 13:1–11. https://doi.org/10.1371/journal.pone.0193254
Smith RM, Thompson K, Hodgson JG, Warren PH, Gaston KJ (2006) Urban domestic gardens (IX): composition and richness of the vascular plant flora, and implications for native biodiversity. Biol Conserv 129:312–322. https://doi.org/10.1016/j.biocon.2005.10.045
Su S, Wang Y, Luo F, Mai G, Pu J (2014) Peri-urban vegetated landscape pattern changes in relation to socioeconomic development. Ecol Indic 46:477–486. https://doi.org/10.1016/j.ecolind.2014.06.044
Tait CJ, Daniels CB, Hill RS (2005) Changes in species assemblages within the Adelaide metropolitan area, Australia, 1836-2002. Ecol Appl 15:346–359. https://doi.org/10.1890/04-0920
The Legume Phylogeny Working Group (LPWG) (2017) A new subfamily classification of the leguminosae based on a taxonomically comprehensive phylogeny. Taxon. https://doi.org/10.12705/661.3
Thiombiano AR, Kakai RLG, Bayen P, Boussim IJ (2016) Méthodes et dispositifs d’inventaires forestiers en Afrique de l’Ouest : état des lieux et propositions pour une harmonisation. Annales des sciences agronomiques, FSA/UAC, 15, 31
Wang H, Qureshi S, Knapp S, Ross C, Hubacek K (2015) A basic assessment of residential plant diversity and its ecosystem services and disservices in Beijing , China. Appl Geogr 64:121–131. https://doi.org/10.1016/j.apgeog.2015.08.006
Wezel, A., Bohlinger, B., Böcker, R., 2000. Vegetation zones in Niger and Benin–present and past zonation. Atlas of natural and agronomic resources of Niger and Benin. Germany: University of Hohenheim. Atlas of natural and agronomic resources of Niger and Benin. https://doi.org/https://projekte.uni-hohenheim.de/atlas308/a_overview/a3_1/html/english/a31ntext.htm
Yacouba H (1999) Inventaire des Plantes Exotiques de la Ville de Niamey. Memoire de D.E.A. Universite de Ouagadougou, Burkina Faso. 1-117p
Yan P, Yang J (2017) Species diversity of urban forests in China. Urban For Urban Green 28:160–166. https://doi.org/10.1016/j.ufug.2017.09.005
Yang J (2016) Urban biodiversity in China : who are winners? Who are losers? Sci Bull 61:1631–1633. https://doi.org/10.1007/s11434-016-1187-7
Ynaut, J., 1987. Privilège urbain:conditions de vie et santé au Niger. Revue-urbanites 42–52. https://doi.org/http://www.politique-africaine.com/numeros/pdf/028042.pdf
Acknowledgements
Our sincere appreciation goes to the Federal Ministry of Education and Research (BMBF) and West African Science Centre on Climate Change and Adapted Land Use (WASCAL) for providing the scholarship and financial support for this program. Shem Kuyah acknowledges the support of Alexander von Humboldt Foundation through the International Climate Protection Fellowship. He also acknowledges his host Prof. Dr. Eike Lüdeling, and the enabling environment enjoyed at the department of Horticultural Sciences, University of Bonn.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Moussa, S., Kuyah, S., Kyereh, B. et al. Diversity and structure of urban forests of Sahel cities in Niger. Urban Ecosyst 23, 851–864 (2020). https://doi.org/10.1007/s11252-020-00984-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11252-020-00984-6