Abstract
Communication systems rely upon specific social contexts and environments that permit effective transmission, and thus, are particularly vulnerable to disruption by anthropogenic disturbance. The acoustic environments of cities may affect conspecific interactions by altering the transmission or reception of song in ways that might ultimately influence fitness, however, the evolutionary and ecological consequences of altered songs remain poorly understood. We hypothesized that the relationship between bird song attributes and fitness metrics would be landscape-dependent, differing between urban and rural habitats. We investigated this hypothesis by measuring vocal and breeding behavior of 54 male Northern Cardinals (Cardinalis cardinalis) at nine sites distributed across a rural to urban landscape gradient in Columbus, Ohio in 2011. Interestingly, we found evidence that relationships between song attributes and male quality differed among landscapes. Shorter and slower songs were associated with larger males in more preferred territories (i.e. with denser vegetation), but only in rural landscapes. Across all landscapes, males that sang at high frequencies had nestlings in poorer condition, but otherwise song attributes were not associated with reproductive output or male provisioning ability. Our results suggest that urban landscapes change the function of song as a signal of quality and could reduce the usefulness of song as a predictor of reproductive performance. This is one of few studies to investigate signal relationships and potential fitness consequences of song variation in natural urban systems, thereby providing insight into micro-evolutionary processes operating within novel environments.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The degree to which sexually-selected traits serve as reliable signals of quality can vary spatially with environmental and social context (Dunn et al. 2008; Higginson and Reader 2009; Cornwallis and Uller 2010; Rodewald et al. 2011). Heterogeneity in signal reliability should be most pronounced for traits that vary widely in the effectiveness of signal transmission among environments. Avian song is an excellent example of a trait for which (1) the effectiveness of signal transmission is strongly mediated by social and environmental context (Ryan and Brenowitz 1985; Bradbury and Vehrencamp 1998; Slabbekoorn et al. 2007) and (2) fitness consequences, as related to territory and/or mate quality, can be substantial (Catchpole 1987). In a time of rapid land transformation, understanding how selective environments for song are shaped by anthropogenic disturbance is necessary to anticipate the ecological and evolutionary responses of native species.
The dramatic changes imposed by urban development provide a unique opportunity to study variable selective pressures on sexual traits. For example, urban forests are characterized by abundant invasive vegetation (Borgmann and Rodewald 2005), high densities of generalist species (Shochat et al. 2006), high levels of predator activity (Rodewald and Kearns 2011), and substantial anthropogenic noise (Slabbekoorn and Peet 2003). Consequently, urban pressures have the potential to alter the expression of ornamental traits such as plumage (Yeh 2004; Isaksson and Andersson 2007; Jones et al. 2010) and vocal behavior (Slabbekoorn and den Boer-Visser 2006; Badyaev et al. 2008). Indeed, a growing number of studies have described a pattern of urban-associated song alterations. The most documented change being a rise in song frequency (Hz) that may overcome masking effects of low frequency (<2 kHz) anthropogenic noise (Roca et al. 2016). Urban birds may also change the temporal features of vocalizations, such as song length or rate, which may improve probability of detection in noise (Hamao et al. 2011), or be a response to an increase in social interactions (Narango and Rodewald 2015).
Yet even within cities, sufficient variation in song traits among individuals remains for sexual selection to operate; in other words, cities do not “fix” the traits. Thus, if sexual traits are changing within urban environments, they may no longer act as reliable sources of information. Evolutionary traps can develop in contexts where conspecifics prefer traits that have recently become associated with unreliable information such as poor territories (Rodewald et al. 2011) or mates of lower quality (Schlaepfer et al. 2002).
Song features can signal many components of quality such as age (Ballentine 2009), size (Ballentine 2009), condition (Rehsteiner et al. 1998), dominance rank (Rehsteiner et al. 1998), reproductive success (Conner et al. 1986; Mennill et al. 2006), territory quality (Conner et al. 1986; Van Oort et al. 2006) and parental investment (Buchanan and Catchpole 2000). However, the extent to which urbanization influences the usefulness of song as an informative signal remains poorly understood. Higher than average frequencies of bird song are prevalent in cities, however, preference for lower or higher frequencies is inconsistent in passerines; dependent on whether a species’ song features signal body size or performance ability (reviewed in Cardoso 2011). It’s possible that increasing frequency may reduce the quality of a song, given that high frequency songs tend to elicit weaker territorial responses (Mockford and Marshall 2009; Ripmeester et al. 2010; Luther and Derryberry 2012), are poor at attracting females (Halfwerk et al. 2011), and may simultaneously affect other song features that signal quality due to physiological limits in performance (Luther et al. 2016). Higher minimum frequencies that are not preferred by females could potentially reduce the likelihood of pairing success or extra-pair paternity in noisy areas (Gross et al. 2010; Wisner 2011).
To better understand the consequences of altered acoustic environments within cities, we investigated the utility of song as an informative signal across a rural-urban landscape gradient. Using the cosmopolitan Northern Cardinal (Cardinalis cardinalis) as a model species, we examined relationships between song traits and fitness parameters (i.e., reproductive output) as well as indicators of male quality (morphology, provisioning rates, nestling condition and territory vegetation). We hypothesized that relationships among song traits, fitness and quality parameters would change with urbanization, such that song characteristics indicating high fitness and quality in rural landscapes would be different than those in urban landscapes. Specifically, we predicted that males using song characteristics that, based on the literature, presumably transmit most efficiently in urban environments (e.g., increases in frequencies and longer, faster songs) would have higher fitness and associated quality traits.
Methods
Study area
This study was conducted between March–September2011 in nine mature riparian forests (≥ 250 m long and ≥100 m wide) along three rivers (Olentangy, Darby and Alum Creek) in and around Columbus, Ohio. Prior to this study, landscape composition was quantified using orthophotos to determine land use within a 1 km radius from the site, and these metrics were used in a principal components analysis to create an index of urbanization for each site (see Rodewald and Shustack 2008 for more details). In their study, index values were correlated with surrounding composition such that values loaded positively with number of buildings, and percent cover by roads, pavement and lawn and negatively with percent cover by agriculture. Hereafter, positive values are considered sites within an ‘urban’ landscape and negative values are embedded within a ‘rural’ landscape although analysis is conducted on a continuous scale. Sites were separated by at least 2 km and were similar in size and shape and differed primarily in respect to surrounding landscape matrix.
Study species
We used Northern cardinals (hereafter: cardinal) as a model synanthropic species to investigate consequences of song variation. Cardinals are a non-migratory resident, generalist species (Halkin and Linville 1999) that readily use resources associated with human developement (e.g. bird feeders, exotic shrubs) and as such are positively associated with landscapes with low to moderate urbanization (Leston and Rodewald 2006). Individual males were captured using mist-nets and fitted with a unique color band combination for individual identification. The few individuals that escaped capture could still be followed because of reliable song perches and/or were mated with banded females. Upon capture, tarsus and wing were measured to indicate body size.
Cardinals have a complex song repertoire with documented flexibility in the performance of different song and note types. Typical song contains 1 to 2 syllable types, but individuals possess repertoires containing up to 13 syllable types organized into different song types (Lemon 1965). Cardinals sing with eventual variation meaning that one song type is repeated many times before switching to a new song type (Lemon 1965; Ritchison 1988). Cardinals share songs within a local area but high variation in the song structural features exists between males in a neighborhood (Ritchison 1988), suggesting song characteristics may indicate condition or status of the singer. Furthermore, song performance plays an important role in the acquisition of mates and territory in cardinals (Conner et al. 1986; Ritchison 1988).
Song recording and analysis
During visits, songs were recorded for 5 h following sunrise between March and June during the breeding season when peak cardinal singing occurs. Territories were monitored weekly for 30 min, during which one observer (DLN) attempted to record all song bouts. We alternated the order of visits to territories in order to vary the time in the morning that individuals were recorded. Identity of singing individuals was confirmed by color bands (most observations) or associations with paired color-banded females and known nests. All recordings were made with a Marantz PMD 670 digital recorder and a Seinheiser directional microphone. Songs were sampled at 44.1 kHz with 16 bit resolution, and saved as uncompressed .wav files.
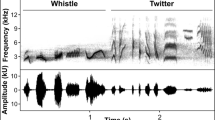
We created digital spectrograms of cardinal song with Raven 1.4 Pro (Cornell lab of Ornithology) using Hann sampling and a discrete Fourier transform of 256 and 1024 for frequency elements. Song characteristics were measured manually by the same observer (D.L.N., see Narango and Rodewald 2015 for additional spectrogram details). Songs were visually inspected to measure the best recording of each song type during the observation period. Songs were only included in spectrogram analysis if they were composed of more than three syllables, and could be assigned to a territory with complimentary breeding season data for subsequent analyses. We chose a three syllable cutoff because three was the fewest number of syllables performed in repeated song types, and excludes partial ‘songs’ of 1–2 syllables that are sometimes performed prior to a full bout of repeated song types (Narango personal observation). Our goal was to capture the variation across song types; hence to account for song types that were used more often, we measured the best recording of each song type, per visit. Songs of the same type, from the same individual, that were recorded on different visits were considered unique samples. Each song type was measured for minimum and maximum frequency (Hz), frequency bandwidth (range), frequency at the highest amplitude (‘peak frequency’), song duration (s), number of syllables, syllable rate (# of syllables/s), and number of syllable types. We chose these measurements because they are features that commonly differ in urban areas and may signal cardinal male quality (Conner et al. 1986). Mean measures of song characteristics per individual male are used in analyses here (8.28 ± 4.55 songs measured per male). Song length and number of syllable types were highly correlated with number of syllables (>0.8), therefore we chose to include number of syllables to describe ‘song length’ to reduce redundancy in the data.
Although low-frequency noise could impact the ability to visually measure frequency characteristics (Zollinger et al. 2012), measurement error in automatic measurements can be avoided by using manual measurements with high frequency resolution when the signal to noise ratio is small, as is common in urban habitats (Cardoso and Atwell 2012; Job et al. 2016). Thus we only used clean recordings for measurements (songs recorded at close range, with minimal reverberations, and were not overlapped by any other biological or ambient sounds). In addition, the potential error in cursor measurement due to noise (22–49 Hz via Verzijden et al. 2010) is much smaller than the relative observed differences between song types in this study (>1.5 kHz) therefore our comparisons are biologically meaningful.
Sample size was limited by the number and quality of the recordings, and some males were dropped because of insufficient recording samples (<3 song types). These males were mostly from urban landscapes due to accidental recordings of males adjacent to focal territories (n = 4), or overall poor recording quality from unusually high ambient noise (>70 dBA, 1 male). Because our samples were more limited by rural than urban males, these deletions likely do not bias results.
Annual reproductive success
From mid-March to September, we closely monitored territories of males to determine annual reproductive success. We visited territories every 2–3 days to locate and check nest contents until completion. Nesting attempts were considered successful if young were visually observed following day of fledging. Fledges from multiple nesting attempts were summed for each male for seasonal reproductive success. The number of young fledged over the course of a season was considered a reasonable estimate of paternity because cardinals exhibit low extra-pair fertilizations and high levels of mate-guarding (Ritchison et al. 1994).
Territory quality
Territory quality was quantified by measuring vegetative attributes known to be strongly preferred by cardinals, particularly high density of understory shrubs and exotic shrubs (Conner et al. 1986; Leston and Rodewald 2006). We described vegetation composition and structure by selecting one early-season and one late-season nest along with one random point within the territory for sampling (25 m in a random direction from the ‘early-season’ nest sampled). Vegetation was measured in an 11.3 m–radius circle centered on the nest or random point. Trees and large woody shrubs were identified and classified in 4 size classes (3–8, 8–26, 26–38, and 38+ cm diameter breast height (dbh). A large proportion of vegetation in the 3–8 cm dbh size class was comprised by native tree saplings and Amur honeysuckle (Lonicera maackii) > 3 m high. Shrubs <3 m were quantified by percent cover within the circle.
Parental care
We estimated male parental care by quantifying provisioning visits during the nestling stage. Nests were observed for one hour between days 5–7 post-hatching to control for variation at different nestling ages. Age was estimated from known hatch date or by nestling development (during the first subsequent visit following hatch day). During an observation, the observer was positioned in a concealed location >10 m from the nest. Observations were completed in the afternoon, during times of clear weather, and only when adults were not disturbed by the observer’s presence. The observer counted number of feeding visits by each adult as well as behaviors around the nest area. The number of nestlings was visually confirmed following each observation and adult feeding visits were divided by nestling number to obtain feeding visits per nestling by the male which describes the effort that each nestling is receiving independent of brood size.
For nests within reach (<4 m high), nestlings were weighed to the nearest 0.01 g and tarsi measured to the nearest 0.1 mm. Tarsus length was highly correlated with nestling age (r > 0.7) so tarsus was only included as a measure of size. Condition was obtained by regressing mass by tarsus and time (Jakob et al. 1996). Since nestlings from the same brood cannot be considered independent, mean nestling condition was calculated for each nest.
Statistical analysis
We used a principal components analysis to reduce the number of variables associated with song (see supplemental materials) and retained components with eigenvalues >1. Principal component 1 (PC1) indicated changes in temporal features whereas principal component 2 (PC2) and principal component 3 (PC3) indicated changes in frequency. PC1 had factors loadings that reflected decreases in temporal features of song (number of syllables; −0.84 and syllable rate: −0.90), such that increases in this component reflects songs that are increasing in speed and duration. PC2 loaded positively toward peak and maximum frequency such that increases in PC2 described increases frequency (peak frequency: 0.79, maximum frequency: 0.73). PC3 was loaded negatively toward minimum frequency such that increases in PC3 described decreases in minimum frequencies (minimum frequency:-0.92). Hereafter we call these variables the temporal component (PC1), peak/max frequency (PC2) and minimum frequency (PC3).
We also used a principal components analysis to reduce the number of variables in body size and vegetation characteristics (supplemental materials). One component that explained >50% of the variation was retained to represent ‘body size’. This component loaded positively toward both wing and tarsus (wing: 0.73, tarsus: 0.73). Because cardinals prefer territories with dense vegetation, we only used the first component from our vegetation PCA, ‘shrub and sapling density’, in subsequent analyses. This component loaded positively toward shrub density and number of 3–8 dbh trees (shrubs: 0.76, 3–8 dbh trees: 0.78).
Finally, we used generalized linear mixed-effect models (GLMM) to test for the relationships between fitness parameters and song parameters. Changes in the relationship between urban and rural landscapes were tested using the interaction between song and a continuous index of urbanization. As such, all full models included one of the song components as a fixed factor (temporal, peak/max frequency, minimum frequency components), urbanization and the interaction between song and urbanization. Because multiple males were recorded at each site and we were interested in the effect of surrounding urbanization, site was used as a random effect in the model. Because every fitness parameter was not collected for each male, sample sizes differ between models and we had to run separate models for each fitness parameter. If the interaction term was non-significant (p > 0.1) this term was deleted and re-run with the subsequent simpler model (Zurr et al. 2009). Model fit assumptions were validated by plotting and visually inspecting residuals against fitted values and a normal quantile plot of residuals (Zurr et al. 2007). The fledge count model was run with a Poisson distribution with a loglink (Zurr et al. 2009; O’Hara and Kotze 2010) using the glmmPQL function in R (package MASS, version 7.3–16, Venables and Ripley 2002). All other models were run with a Gaussian distribution using the lme function in R (package nlme, version 3.1–102, Pinheiro et al. 2017).
Results
Relationships between quality attributes and song
Interestingly, the relationship between temporal component (PC1) and body size was dependent upon the surrounding landscape (Temporal x Urban: β = −0.27 ± 0.11, F 7,32 = 6.72, p = 0.01; Table 1). Males with the shortest and slowest songs had the largest body size in rural landscapes, but this pattern was absent in urban landscapes (Fig. 1). No significant relationship existed between body size and the peak/max frequency and minimum frequency components, urbanization, nor the interaction between these traits and urbanization (Table 1).
Relationship between body size and temporal song characteristics between urban (urban index >0) and rural (urban index <0) landscapes for male cardinals in central Ohio, 2011 (n = 42). Males that sing shorter, slower songs are larger, but only in rural landscapes. Landscapes are graphically displayed as categories here, but analyses were conducted on the continuous urban index
The relationship between the temporal song component and territory quality also varied by landscape (Temporal x Urban: β = −0.23 ± 0.11, F 7,35 = 4.79, p = 0.04; Table 1), such that males singing shorter, slower songs had territories with the highest density of shrubs and saplings, but only in rural landscapes (Fig. 2). The opposite was true in urban landscapes, where males singing faster, longer songs had territories with the highest density of shrubs and saplings. However, shrub and sapling density increased over the urban gradient for all models (Table 1), largely due to the increase in Amur honeysuckle (Lonicera maackii) densities in urban compared to rural sites. The minimum frequency component (PC3) was also positively related to shrub and sapling densities, such that males singing higher frequency songs inhabited territories with the highest densities of shrubs and saplings (Minimum Frequency: β = 0.30 ± 0.15, F 7,35 = 5.97, p = 0.02; Table 1). Territory quality was not related to peak/max frequency, nor to the interaction between peak/max frequency and urbanization.
Relationship between territory quality and the temporal song component between urban (urban index >0) and rural (urban index <0) landscapes for male cardinals in central Ohio, 2011 (n = 42). Males that sing shorter, slower songs have territories with high densities of shrubs and saplings, but only in rural landscapes. A negative relationship between short, slow songs and vegetation density occurs in urban landscapes. Landscapes are graphically displayed as categories here, but analyses were conducted on the continuous urban index
The minimum frequency component was positively related to nestling condition such that males singing songs with lower minimum frequencies had nestlings in better condition, irrespective of landscape type (Minimum Frequency: β = 0.64 ± 0.26, F 6,17 = 6.90, p = 0.02; Table 1; Fig. 3). There was no significant relationship between nestling condition and the temporal or peak/max frequency component, nor the interactions (Table 1). There was no significant relationship between male feeding rates and song components, urbanization, nor the interaction between song and urbanization. (Table 1).
Relationship between nestling condition and the minimum frequency song component between urban (urban index >0) and rural (urban index <0) landscapes for male cardinals in central Ohio, 2011 (n = 26). In both landscapes, males singing at lower minimum frequencies are have nestlings in better condition
Relationships between reproductive fitness and song
Contrary to predictions, annual fledgling success was unrelated to any song component or the effect of urbanization. In addition, there was no significant interaction between song features and urbanization between landscapes for reproductive output (Table 1).
Discussion
Our study provides evidence that urban environments can alter the usefulness of song as a signal of male quality for some, but not all, fitness attributes. Relationships among body size, vegetation and song characteristics were landscape dependent, such that males with the shortest and slowest songs were larger and occupied more preferred territories, but only in rural forests. Other relationships were consistent across landscapes, including the pattern that males singing at high frequencies had nestlings in poorer condition and occupied preferred territories with high densities of vegetation. We found no evidence that song attributes were related to either annual reproductive success or male feeding rates.
Whereas numerous previous studies have described song differences between urban and rural birds (Roca et al. 2016), few have evaluated the functional consequences of urban-associated changes in song despite pleas for further study (Patricelli and Blickley 2006; Gil and Brumm 2013; Halfwerk and Slabbekoorn 2013; Read et al. 2014; Wong and Lowry 2016). To our knowledge, this study is the first to show that relationships between song and other quality traits may differ in rural and urban landscapes. Both female (Halfwerk et al. 2011; des Aunay et al. 2014) and male (Luther and Magnotti 2014) birds respond less favorably to song in the presence of urban noise under experimental conditions, but relationships between song and male quality in urban systems have not been tested.
Our finding that short and simple songs were positively associated with male quality, though only in rural landscapes, is consistent with previous work on cardinal song. Conner et al. (1986) demonstrated that males in non-developed landscapes that sang short songs of low complexity secured better territories and fledged more young. Others have shown that large body size provides fitness advantages in interspecific competition, survival and reproduction (Kodric-Brown and Brown 1984; Jennions et al. 2001). Nevertheless, we found that the relationships between song and either body morphology or territory quality were landscape-dependent, such that song failed to signal information about body size and density of vegetation in urban landscapes.
In addition to contributing to background levels of spatial heterogeneity in signal selection (Cornwallis and Uller 2010), variable environments may decouple relationships between sexual traits and the information they once signaled (Higginson and Reader 2009). Indeed, the degree to which sexual traits are associated with morphology or reproductive output varies widely across geographic ranges and landscapes (Yeh 2004; Badyaev et al. 2008; Jones et al. 2010; Vortman et al. 2011). One potential explanation for variation in the usefulness of a sexual trait is that males in poor habitat reduce the quality of advertisement when resources are low (Van Oort et al. 2006). Alternatively, features in a novel environment (e.g. anthropogenic resource subsidies or exotic plants) may select for ornamental traits that do not necessarily signal individual quality (Rodewald et al. 2011). Concordantly, song that provides unreliable information about a signaler’s condition or intent may unnecessarily increase time spent defending the territory against intruders (Conner et al. 1986) or reduce mating opportunities with prospective females (Catchpole 1987; Swaddle and Page 2007; Halfwerk et al. 2011). Based on prior work in this system, urban birds sang songs that were longer and faster due to increased conspecific densities (Narango and Rodewald 2015). Given that short songs are more attractive in this species, but relationships between song and body size is unreliable in urban environments, smaller males could secure more preferred territories and experience higher fitness further disassociating the usefulness of song as a signal of quality in urban landscapes.
Although other studies have consistently shown that minimum song frequencies increase in loud urban environments (reviewed in Roca et al. 2016), few have explicitly evaluated relationships between urban-associated frequency variation and other aspects of quality (Wong and Lowry 2016). Contrary to our predictions, we did not find support that increases in frequency (either peak/max or minimum frequency) provided any reproductive benefits, or consequences, in urban systems. Rather, males with high minimum frequencies had nestlings in lower condition – a relationship not explained by feeding rates, which were unrelated to frequency (Pearson’s correlation, r = 0.17, p = 0.41). Males with higher minimum frequencies also tended to have territories with high densities of vegetation in both landscapes, although total density of vegetation was different between urban and rural landscapes. Our proposed explanation for these relationships is that males singing at high minimum frequencies have secured territories in habitat that 1) are in disturbed areas dominated by invasive plants and 2) provide poor habitat for nestlings, given that noisy territories are often located close to high anthropogenic activity (e.g. roads) irrespective of landscape type. Noise itself can reduce nestling begging and the ability of adults to communicate to hear nestling signals (Warren et al. 2006; Leonard and Horn 2012). However, noise may also be correlated with habitat quality, as roads and edges tend to be dominated by invasive plants (Borgmann and Rodewald 2005) that are known to support fewer and lower quality arthropod prey items that are essential for nestling growth (Tallamy 2004; Magura et al. 2006; Isaksson and Andersson 2007). In this system, male cardinals tend to prefer territories with dense exotic plants, despite reductions in fitness (Leston and Rodewald 2006; Rodewald et al. 2011). These results suggests that the highest quality males may be singing high frequency song that is better able to secure mates, albeit at the expense of attracting females to suboptimal habitat. Further investigation of the interplay between noise, vegetation and other forms of avian behavior is needed to elucidate mechanistic causes from correlation.
One important caveat to this study is that we used an observational approach to investigate natural song behavior in urban birds. In urban areas, bird song structure may vary due to a several concurrent changes in the environment such as noise (Roca et al. 2016), conspecific density (Narango and Rodewald 2015), and habitat structure (Dowling et al. 2012; Job et al. 2016). However, adjustments may occur in the long-term via signal evolution (Derryberry 2007) or in the short-term by improving transmission with proximate adjustments in song structure (Hu and Cardoso 2009), types (Halfwerk and Slabbekoorn 2009), amplitude (Brumm and Slabbekoorn 2005) or activity (Díaz et al. 2011). Birds that prosper in urban areas may also be more behaviorally plastic. For example, in some species, females evaluate mates by evaluating the complexity in vocalizations because this feature could indicate an individual’s cognitive ability (Boogert et al. 2008). If structural components of song are not related to direct fitness benefits, song plasticity, as an indicator of behavioral flexibility, could be a more useful indicator of male quality. Future studies should utilize an experimental approach to test whether an individual’s ability to respond to urban-associated changes in the acoustic environment (LaZerte et al. 2016) is related to reproductive benefits as well.
Also important to note is that frequency may not be an important signal of quality for cardinals, unlike other species (Gil and Gahr 2002; Christie et al. 2004; Halfwerk et al. 2011, but see Luther and Magnotti 2014). Observable increases in minimum frequency could be an artifact of singing louder in noisy environments (Nemeth et al. 2012) and lack any functional information. However, because our minimum frequency measurements increased independently of peak and maximum frequency, this is unlikely to be the case (Cardoso and Atwell 2012). Regardless, Conner et al. (1986) found no positive correlations between cardinal song frequency and reproduction. For this species, individual fitness appears to be primarily related to song temporal structure or by plumage (Wolfenbarger 1999), as well as prey availability and predation pressure, which was outside the scope of this study. Although the association we found between song frequency and territory quality (based on preferences) might initially suggest frequency is a useful cue, our previous work indicates that nests in those territories often fail when territories contain exotic invasive honeysuckle – thereby amounting to an ecological trap for high quality males (Rodewald et al. 2011).
A growing paradigm in behavioral ecology is that relationships between sexually selected traits and fitness attributes are not static, but can vary in heterogeneous environments (Cornwallis and Uller 2010). In this way, the rapid expansion of urbanization may provoke rapid changes in selective environments within cities. Our study is one of the first to provide evidence that the usefulness of song as an indicator of quality may break down in cities, as evidenced by the decoupled relationships among temporal song elements, morphology, and territory quality. In contrast, the complex social use of song, coupled with high behavioral plasticity, may make vocal signals less susceptible to significant changes in fitness consequences. Future studies should combine experimental and observational based designs to further elucidate possible mechanisms behind dynamic sexual selection in human-dominated systems and the influence of urban-associated habitat changes on the development and maintenance avian behavior.
References
Badyaev AV, Young RL, Oh KP et al (2008) Evolution on a local scale: developmental, functional, and genetic bases of divergence in bill form and associated changes in song structure between adjacent habitats. Evol Evol 62(1951–1964):1964. https://doi.org/10.1111/j.1558-5646.2008.00428.x
Ballentine B (2009) The ability to perform physically challenging songs predicts age and size in male swamp sparrows, Melospiza Georgiana. Anim Behav 77:973–978
Boogert NJ, Giraldeau LA, Lefebvre L (2008) Song complexity correlates with learning ability in zebra finch males. Anim Behav 76:1735–1741
Borgmann KL, Rodewald AD (2005) Forest restoration in urbanizing landscapes: interactions between land uses and exotic shrubs. Restor Ecol 13:334–340. https://doi.org/10.1111/j.1526-100X.2005.00042.x
Bradbury JW, Vehrencamp SL (1998) Principles of animal communication. Sinauer Associates, Sunderland
Brumm H, Slabbekoorn H (2005) Acoustic communication in noise. In: Advances in the Study of Behavior. Academic, pp 151–209
Buchanan KL, Catchpole CK (2000) Song as an indicator of male parental effort in the sedge warbler. Proc R Soc B Biol Sci 267:321–326
Cardoso GC (2011) Paradoxical calls: the opposite signaling role of sound frequency across bird species. Behav Ecol 23(2):237–241
Cardoso GC, Atwell JW (2012) On amplitude and frequency in birdsong: a reply to Zollinger et al. Anim Behav 84:e10–e15. https://doi.org/10.1016/j.anbehav.2012.08.012
Catchpole CK (1987) Bird song, sexual selection and female choice. Trends Ecol Evol 2:94–97
Christie PJ, Mennill DJ, Ratcliffe LM (2004) Pitch shifts and song structure indicate male quality in the dawn chorus of black-capped chickadees. Behav Ecol Sociobiol 55:341–348
Conner RN, Anderson ME, Dickson JG (1986) Relationships among territory size, habitat, song, and nesting success of northern cardinals. Auk 103:23–31
Cornwallis CK, Uller T (2010) Towards an evolutionary ecology of sexual traits. Trends Ecol Evol 25:145–152. https://doi.org/10.1016/j.tree.2009.09.008
Derryberry EP (2007) Evolution of bird song affects signal efficacy: an experimental test using historical and current signals. Evolution 61(8):1938–1945
Des Aunay GH, Slabbekoorn H, Nagle L et al (2014) Urban noise undermines female sexual preferences for low-frequency songs in domestic canaries. Anim Behav 87:67–75
Díaz M, Parra A, Gallardo C (2011) Serins respond to anthropogenic noise by increasing vocal activity. Behav Ecol 22:332–336. https://doi.org/10.1093/beheco/arq210
Dowling JL, Luther DA, Marra PP (2012) Comparative effects of urban development and anthropogenic noise on bird songs. Behav Ecol 23(1):201–209
Dunn PO, Whittingham LA, Freeman-Gallant CR, DeCoste J (2008) Geographic variation in the function of ornaments in the common yellowthroat Geothlypis trichas. J Avian Biol 39:66–72
Gil D, Brumm H (2013) Acoustic communication in the urban environment: patterns, mechanisms, and potential consequences of avian song adjustments. Avian Urban Ecol Behav Physiol Adapt 69
Gil D, Gahr M (2002) The honesty of bird song: multiple constraints for multiple traits. Trends Ecol Evol 17:133–141
Gross K, Pasinelli G, Kunc HP (2010) Behavioral plasticity allows short-term adjustment to a novel environment. Am Nat 176:456–464
Halfwerk W, Slabbekoorn H (2009) A behavioural mechanism explaining noise-dependent frequency use in urban birdsong. Anim Behav 78:1301–1307
Halfwerk W, Slabbekoorn H (2013) The impact of anthropogenic noise on avian communication and fitness. Avian Urban Ecol Behav Physiol Adapt 84
Halfwerk W, Bot S, Buikx J et al (2011) Low-frequency songs lose their potency in noisy urban conditions. Proc Natl Acad Sci 108:14549–14554. https://doi.org/10.1073/pnas.1109091108
Halkin SL, Linville SU (1999) Northern cardinal (Cardinalis Cardinalis). The Birds of North America 440:32
Hamao S, Watanabe M, Mori Y (2011) Urban noise and male density affect songs in the great tit Parus major. Ethol Ecol Evol 23(2):111–119
Higginson AD, Reader T (2009) Environmental heterogeneity, genotype-by-environment interactions and the reliability of sexual traits as indicators of mate quality. Proc R Soc B Biol Sci 276:1153–1159
Hu Y, Cardoso GC (2009) Are bird species that vocalize at higher frequencies preadapted to inhabit noisy urban areas? Behav Ecol 20:1268–1273. https://doi.org/10.1093/beheco/arp131
Isaksson C, Andersson S (2007) Carotenoid diet and nestling provisioning in urban and rural great tits Parus Major. J Avian Biol 38:564–572
Jakob EM, Marshall SD, Uetz GW (1996) Estimating fitness: a comparison of body condition indices. Oikos 77:61–67
Jennions MD, Moller AP, Petrie M (2001) Sexually selected traits and adult survival: a meta-analysis. Q Rev Biol 76:3–36
Job JR, Kohler SL, Gill SA (2016) Song adjustments by an open habitat bird to anthropogenic noise, urban structure, and vegetation. Behav Ecol arw105
Jones TM, Rodewald AD, Shustack DP (2010) Variation in plumage coloration of northern cardinals in urbanizing landscapes. Wilson J Ornithol 122:326–333
Kodric-Brown A, Brown JH (1984) Truth in advertising: the kinds of traits favored by sexual selection. Am Nat 124:309–323
LaZerte SE, Slabbekoorn H, Otter KA (2016) Learning to cope: vocal adjustment to urban noise is correlated with prior experience in black-capped chickadees. Proc R Soc B The Royal Society 283:20161058
Lemon RE (1965) The song repertoires of cardinals (Richmondena cardinalis) at London, Ontario. Can J Zool 43(3):559–569
Leonard ML, Horn AG (2012) Ambient noise increases missed detections in nestling birds. Biol Lett 8:530–532. https://doi.org/10.1098/rsbl.2012.0032
Leston LFV, Rodewald AD (2006) Are urban forests ecological traps for understory birds? An examination using northern cardinals. Biol Con 131:566–574
Luther DA, Derryberry EP (2012) Birdsongs keep pace with city life: changes in song over time in an urban songbird affects communication. Anim Behav 83:1059–1066
Luther D, Magnotti J (2014) Can animals detect differences in vocalizations adjusted for anthropogenic noise? Anim Behav 92:111–116. https://doi.org/10.1016/j.anbehav.2014.03.033
Luther DA, Phillips J, Derryberry EP (2016) Not so sexy in the city: urban birds adjust songs to noise but compromise vocal performance. Behav Ecol 27:332–340. https://doi.org/10.1093/beheco/arv162
Magura T, Tóthmérész B, Lövei GL (2006) Body size inequality of carabids along an urbanisation gradient. Basic Appl Ecol 7:472–482
Mennill DJ, Badyaev AV, Jonart LM, Hill GE (2006) Male house finches with elaborate songs have higher reproductive performance. Ethology 112(2):174–180
Mockford EJ, Marshall RC (2009) Effects of urban noise on song and response behaviour in great tits. Proc R Soc B Biol Sci 276:2979–2985
Narango DL, Rodewald AD (2015) Urban-associated drivers of song variation along a rural–urban gradient. Behav Ecol 27:608–616. https://doi.org/10.1093/beheco/arv197
Nemeth E, Zollinger SA, Brumm H (2012) Effect sizes and the integrative understanding of urban bird song. Am Nat 180(1):146–152
O’Hara RB, Kotze DJ (2010) Do not log-transform count data. Methods Ecol Evol 1:118–122
Patricelli GL, Blickley JL (2006) Avian communication in urban noise: causes and consequences of vocal adjustment. Auk 123:639–649
Pinheiro J, Bates D, DebRoy S, Sarkar D, R Core Team (2017) _nlme: Linear and Nonlinear MixedEffects Models_. R package version 3.1-131. https://CRAN.R-project.org/package=nlme>
Read J, Jones G, Radford AN (2014) Fitness costs as well as benefits are important when considering responses to anthropogenic noise. Behav Ecol 25:4–7
Rehsteiner U, Geisser H, Reyer HU (1998) Singing and mating success in water pipits: one specific song element makes all the difference. Anim Behav 55:1471–1481
Ripmeester EA, Mulder M, Slabbekoorn H (2010) Habitat-dependent acoustic divergence affects playback response in urban and forest populations of the European blackbird. Behav Ecol 21:876–883
Ritchison G (1988) Song repertoires and the singing behavior of male northern cardinals. The Wilson Bulletin 100:583–603
Ritchison G, Klatt PH, Westneat DF (1994) Mate guarding and extra-pair paternity in northern cardinals. Condor 96:1055–1063
Roca IT, Desrochers L, Giacomazzo M et al (2016) Shifting song frequencies in response to anthropogenic noise: a meta-analysis on birds and anurans. Behav Ecol 27:1269–1274. https://doi.org/10.1093/beheco/arw060
Rodewald AD, Kearns LJ (2011) Shifts in dominant nest predators along a rural-to-urban landscape gradient. Condor 113(4):899–906
Rodewald AD, Shustack DP (2008) Urban flight: understanding individual and population-level responses of Nearctic–Neotropical migratory birds to urbanization. J Anim Ecol 77:83–91
Rodewald AD, Shustack DP, Jones TM (2011) Dynamic selective environments and evolutionary traps in human-dominated landscapes. Ecology 92:1781–1788
Ryan MJ, Brenowitz EA (1985) The role of body size, phylogeny, and ambient noise in the evolution of bird song. Am Nat 126:87–100
Schlaepfer MA, Runge MC, Sherman PW (2002) Ecological and evolutionary traps. Trends Ecol Evol 17:474–480
Shochat E, Warren PS, Faeth SH, McIntyre NE, Hope D (2006) From patterns to emerging processes in mechanistic urban ecology. Trends Ecol Evol 21:186–191
Slabbekoorn H, den Boer-Visser A (2006) Cities change the songs of birds. Curr Biol 16:2326–2331. https://doi.org/10.1016/j.cub.2006.10.008
Slabbekoorn H, Peet M (2003) Birds sing at a higher pitch in urban noise. Nature 424:267
Slabbekoorn H, Yeh P, Hunt K (2007) Sound transmission and song divergence: a comparison of urban and forest acoustics. Condor 109:67–78
Swaddle JP, Page LC (2007) High levels of environmental noise erode pair preferences in zebra finches: implications for noise pollution. Anim Behav 74:363–368. https://doi.org/10.1016/j.anbehav.2007.01.004
Tallamy DW (2004) Do alien plants reduce insect biomass? Conserv Biol 18:1689–1692. https://doi.org/10.1111/j.1523-1739.2004.00512.x
Van Oort H, Otter KA, Fort KT, Holschuh CI (2006) Habitat quality, social dominance and Dawn chorus song output in black-capped chickadees. Ethology 112:772–778
Venables WN, Ripley BD (2002) Modern Applied Statistics with S. Fourth Edition. Springer, New York
Verzijden MN, Ripmeester EAP, Ohms VR et al (2010) Immediate spectral flexibility in singing chiffchaffs during experimental exposure to highway noise. J Exp Biol 213:2575–2581. https://doi.org/10.1242/jeb.038299
Vortman Y, Lotem A, Dor R et al (2011) The sexual signals of the East-Mediterranean barn swallow: a different swallow tale. Behav Ecol 22:1344–1352
Warren PS, Katti M, Ermann M, Brazel A (2006) Urban bioacoustics: it’s not just noise. Anim Behav 71:491–502
Wisner EM (2011) The consequences of anthropogenic disturbance on communication and the operation of sexual selection in the eastern bluebird (Sialia sialis). Doctoral dissertation, Syracuse University
Wolfenbarger LLR (1999) Red coloration of male northern cardinals correlates with mate quality and territory quality. Behav Ecol 10:80
Wong BBM, Lowry H (2016) The struggle to be heard in an increasingly noisy world: a comment on Roca et al. Behav Ecol 27:1275–1276. https://doi.org/10.1093/beheco/arw102
Yeh PJ (2004) Rapid evolution of a sexually selected trait following population establishment in a novel habitat. Evolution 58:166–174. https://doi.org/10.1111/j.0014-3820.2004.tb01583.x
Zollinger SA, Podos J, Nemeth E et al (2012) On the relationship between, and measurement of, amplitude and frequency in birdsong. Anim Behav 84:e1–e9. https://doi.org/10.1016/j.anbehav.2012.04.026
Zurr A, Ieno E, Smith GM (2007) Analysing ecological data (statistics for biology & health). Theatr Rec 67:02
Zurr AF, Ieno EN, Walker NJ, Saveliev AA, Smith GM (2009) Mixed effects models and extensions in ecology with R. Springer Verlag, New York
Acknowledgements
This work was funded by the National Science Foundation (DEB-0340879, DEB-0639429) and the Ohio Division of Wildlife and U.S. Fish and Wildlife Service through the State Wildlife Grants Program to A.D. Rodewald. We thank numerous field technicians and graduate students for help with data collection, especially L. Kearns, B. Padilla, L. Rowse, S. Rose, M. McDermott, Z. Züst. We also thank D. Nelson, A. Poesel, E. Szeyller-Macolley and S. Wright for help with acoustic analysis. This manuscript was greatly improved by the advice supplied by two anonymous reviewers.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All animal handling was approved and conducted under IACUC# IS00000448 under The Ohio State University’s Animal Care and Use Program.
Conflict of interest
The authors have no conflict of interests to report
Electronic supplementary material
ESM 1
(DOCX 20 kb)
Rights and permissions
About this article
Cite this article
Narango, D.L., Rodewald, A.D. Signal information of bird song changes in human-dominated landscapes. Urban Ecosyst 21, 41–50 (2018). https://doi.org/10.1007/s11252-017-0698-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11252-017-0698-6