Abstract
Impact mitigation practices are currently one of the hottest topics in conservation and regarded as priorities worldwide. Forest bat populations are known to provide important ecosystem services such as pest control and bat boxes have become one of the most popular management options for counteracting the loss of roosts. However, bat boxes tend to be employed in non-native forests near highly humanized areas where human disturbance is higher. The aim of this study was to evaluate how the surrounding landscape composition affects bat box occupancy in urban non-native forests along the Mediterranean corridor in the Northeastern Iberian Peninsula. Two hundred wooden bat boxes were monitored in young non-native forests in the period 2004–2012. The influence of land cover on occupancy rate of bat boxes was analysed at the landscape level in a 5 km buffer around bat-box stations. In total, 1659 inspections were carried out, in which a 15 % occupancy rate was detected. Bat boxes hosted three different species (Pipistrellus pygmaeus, Pipistrellus kuhlii and Nyctalus leisleri). More than 70 % of the occupancy can be explained by habitat and spatial composition. The presence of urban areas around bat boxes tends to have a negative impact on bat occupation rates; by contrast, forest coverage has a positive effect, especially for the tree-dwelling bats. These patterns could be associated with the large number of available roosts in buildings, microhabitat or phylopatry. Thus, to increase success, we suggest that landscape composition should be considered when using bat boxes for conservation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
At a time when humans are witnessing the Earth’s sixth major extinction and global change is provoking massive biodiversity loss, eco-friendly practices and respectful natural resource management have become one of the hottest topics in science and conservation, and in general a priority for societies throughout the world. Bats are known to provide important ecosystem services such as pest control, pollination and seed dispersal (Kunz et al. 2011; Puig-Montserrat et al. 2015). However, many bat species – and especially forest bats – are seriously threatened by human activities that lead, for example, to habitat fragmentation and/or deforestation (Kunz and Parsons 2009).
Bats’ roosting requirements play a major role in their ecologies (Kunz and Lumsden 2003) and must be taken into account in conservation plans. Tree-dwelling species are considered to be especially sensitive and threatened by intensive forestry practices and deforestation given that they usually select hollow trees as cavities in which to roost (Kunz 1982; Brigham et al. 1997; Patriquin and Barclay 2003; Flaquer et al. 2007; Ruczynski and Bogdanowicz 2005). Most of these species (with the exception of those that also roost in caves and buildings) will switch roost sites, which further complicates conservation strategies since the survival of even small numbers of bats will depend on the availability of a good number of suitable trees (Russo et al. 2004, 2005, 2007).
Likewise, ubiquitous species (which tend to select a range of different roost types) are threatened by the alteration (e.g. house restorations) or destruction of human-made structures that are sometimes home to sizeable bat colonies (Pierson 1998; Russo and Ancillotto 2015). Despite occasional disagreements, most scientists believe biological conservation should focus on recovering and maintaining the biodiversity that would naturally be present in a certain region at a certain time (to mitigate as much as possible human impact on the ecosystem).
A popular management option for counteracting the loss of bat roosts in non-native plantations, damaged forests, secondary regrowth forests and young coppices (usually conifers) is the provision of artificial roosting sites such as bat boxes (Racey 1998; Geoffrey and Agnew 2002; Rueegger 2016). This practice could provide an effective way of increasing roost availability and a great opportunity for studying the distribution of certain bat species and their ecology. In fact, much valuable work carried out on bats worldwide is based on the use of bat boxes (Boyd and Stebbings 1989).
Tree-dwelling bat populations greatly benefit from the placement of bat boxes in otherwise inhospitable woodland areas (Chambers et al. 2002). Although often used as temporary roosts, bat boxes are also important during key phases of bats’ annual cycles such as mating, parturition and even hibernation (Flaquer et al. 2006; Rueeger 2016). Bat boxes are also a good substitute for natural or human-made roosts for maternity colonies excluded from buildings (Lourenço and Palmeirim 2004; Flaquer et al. 2006) and alternative sites for mating roosts for tree-dwelling bats (Benzal 1991; Flaquer et al. 2005). Factors such as box structure, material, height above the ground and colour can all influence the occupation rate (Kerth et al. 2001; Lourenço and Palmeirim 2004; Flaquer et al. 2006, 2014). Nevertheless, despite being widely used in several countries, there is little available information about how landscape composition affects bat box occupancy rates and thus whether or not this technique is suitable for use in all forests regardless of their degrees of maturity (Rueegger 2016).
Here we present the results of a nine-year bat box monitoring program, conducted in non-native forests surrounded by highly modified urban landscapes in the Mediterranean wildlife corridor in Catalonia (NE Spain), with the aim to evaluate how surrounding landscape structure and composition affects bat box occupancy.
Material and methods
Study area
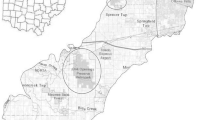
The project was run in the Barcelona Provincial Council’s network of natural parks (100,625 ha), an important corridor for wildlife that crosses the massive Barcelona Metropolitan Area (nearly 5 million inhabitants) on the shores of the Mediterranean Sea. The Mediterranean climate (mean annual temperatures of ca. 16 °C; average precipitation of 500–700 mm/year) ensures that holm oak (Quercus ilex) formations are the normal climax forest throughout much of this network of protected areas, although in many places these forests have been replaced by secondary coniferous forests of umbrella (Pinus pinea) and Aleppo (Pinus halepensis) pines.
Bat box stations
More than 200 open-sided wooden bat boxes (100-mm deep, 190-mm wide and 200-mm high; see Flaquer et al. 2006) were randomly placed in 2003–2005 in clusters of 3–5 boxes (n = 40 stations) in five natural parks (Fig. 1). All boxes in each cluster were placed 4 m above the ground with randomly chosen orientations (Fig. 2). Boxes were situated less than 50 m from each other and each cluster was at least 2 km from the next nearest one. Boxes were checked annually during the period 2004–2012 in summer and/or autumn. Every bat box was checked on average 8.49 ± 7.29 times. To avoid unnecessary disturbance, not all bats were examined during the surveys and some were classified only as Pipistrellus sp. when it was not possible to identify individuals of this genus to species level. Although occupancy rates and abundance give similar results when modelling specific responses to environmental predictors (Torre et al. 2014), we used the site-occupancy rate rather than the mean abundance as a proxy for bat box occupancy. The occupancy rate was defined as the number of occasions on which a bat species was detected at a locality divided by the total number of box inspections per locality × 100.
Landscape composition
The influence of land cover on the occupancy rate for both Nyctalus leisleri and Pipistrellus sp. (both Kuhl’s pipistrelle P. kuhlii and soprano pipistrelle P. pygmaeus have been reported for this area) was analysed within a 5-km buffer zone (Boughey et al. 2011; Flaquer and Puig-Montserrat 2012) around the bat box stations. This buffer size was chosen following Boughey et al. (2011), who assessed the effect of the configuration of broad-leaved woodland at a landscape scale on roost selection in other European bat species. Buffers of this size cover similar bat species’ home ranges and so these potential zones of influence could greatly affect bat box occupancy success (Boughey et al. 2011). Using a dataset with nine different habitat categories (Table 1), all buffer zones were characterized according to the habitat cover derived from the information available in the 1:50,000-scale Catalan habitat cartography (Departament de Medi Ambient i Habitatge, 2005). Within each buffer the proportions of each habitat landscape cover were considered as predictor variables. A measure of habitat diversity (Shannon Index) and the distance to the nearest urban area were also added to the models to assess how habitat composition at landscape scale (within the buffers) might affect bat box occupancy rates (Table 1).
Statistical analysis
A matrix with the frequencies of occurrence of all the bat species found in the boxes (N. leisleri, P. kuhlii and P. pygmaeus.) was built and associated with environmental variables (Braak and Smilauer 2002; Leps and Smilauer 2003) using a constrained correspondence analysis (DCA) and CANOCO 4.5 software. Given that gradient lengths were less than four, linear ordination methods were used (Leps and Smilauer 2003).
Redundancy analysis (RDA) was performed to assess whether differences in bat box occupancy rates between sites could be explained by environmental variables. Spatial autocorrelation was previously examined through a Mantel test including a Monte Carlo permutation test, computed with the ‘ade4’ package (Dray and Dufour 2007). Spatial autocorrelation was found to affect bat box occupancy, indicating higher similarity on occupation rates among bat boxes that are close together than among those spatially distant (r2 = 0.149, nrep = 9999, p = 0.0372). In order to control for spatial autocorrelation in the occupancy rates, we included a set of spatial predictors – a third-degree polynomial function of the geographical coordinates – as suggested by Bocard et al. (1992). This use of partial constrained ordination analyses allowed us to determine separately the influence of the environment and the spatial situation of bat boxes on the bat box occupancy rates. This ordination splits the total variance into a) the environmental variation (non-spatial), b) the spatial variance (non-environmental), c) the spatial-environmental fraction (shared variance) and (d) the unexplained fraction of variance (see Bocard et al. 1992 for details). SpaceMaker2 software (Borcard et al. 2004) was used to obtain the polynomial terms from the geographical coordinates of the sampled sites.
Next, we modelled and compared the effect of landscape composition on bat box occupancy success (using logistic generalized linear models) for forest-dwelling (N. leisleri) and house-dwelling (all Pipistrellus sp.) bats. Due to the limited number of bat box stations, only three categories of land cover were considered as predictors in this analysis: urban, forest and grasslands/crops, plus the mean distance to the nearest urban area. To avoid multicollinearity, the autocorrelation between the predictors in the models was calculated using the ‘corrplot’ package (Wei and Simko 2016) and all predictors with r > 0.6 were excluded. Then, following Burnham and Anderson (2004), the most parsimonious models were selected using Akaike‘s Information Criterion corrected for small sample sizes (AICc). Model-averaging was used to obtain parameter estimates for the models with an AICc difference from the best model (Δi) < 2 (Burnham and Anderson 2004) using the R package ‘bestglm’ v. 0.34 (McLeod and Xu 2014). The results of these models are presented using the corresponding odds ratio (OR) and their confidence intervals. Models were built and tested using R software, version 3.2.0 (R Foundation for Statistical Computing).
Results
A total of 47.5 % of the stations (19 stations) were occupied by bats at least once during the study period. In all, 81 of 200 bat boxes (40.5 %) were occupied at least once during the nine-year survey period; 1404 out of 1659 bat box inspections were negative, that is, only 15 % were found to host bats during inspections. Considering only positive inspections, 6 % of bat boxes were found to host more than one bat (up to 50 bats), although mixed colonies with more than one species were never found in the area. The mean occupancy rate and mean bat abundance in bat boxes were strongly correlated in all 40 bat box stations (r = 0.67, P < 0.0001).
Leisler’s bat (N. leisleri) was only found roosting at seven stations (17.5 %), corresponding to 29 of the 200 bat boxes (14.5 %), in groups ranging from 1 to 11 individuals. Leisler’s females usually appear during mid-August and remain in the bat boxes until mid-May. On the other hand, males were found throughout the whole year but only gathered in harems with females from late August to November.
Individuals of the genus Pipistrellus were detected at all bat box stations that were also occupied by N. leisleri (in different boxes) but were also found at stations not occupied by this forest-dwelling bat species. Both maternity and mating colonies were reported for the genus Pipistrellus. Thirteen bat boxes (6.5 % of the 200 boxes) were occupied at least once by P. kuhlii. This species was found to be breeding just once and mating four times. The Pipistrellus sp. group, which includes the soprano pipistrelle (P. pygmaeus) and the Kuhl’s pipistrelle (P. kuhlii), was found in 39 of the 200 boxes at least once (19.5 %), either mating or alone (range 1–15).
The first redundancy analysis of box occupancy rates constrained only by environmental variables showed that these variables explained 56 % of the variance, and that both the first axis (F ratio = 19.1, P = 0.004) and the four axes together (F ratio = 2.8, P = 0.01; Table 1) were significant. Grassland cover (30 %), habitat diversity (8 %) and rocky outcrops (5 %) were selected as environmental variables by the model. Bat box occupancy rates for both N. leisleri and Pipistrellus sp. increased from stations in urban and open habitats (grasslands, shrublands and croplands) to more heterogeneous forest habitats far from human settlements, as shown in Fig. 3. The first axis can be interpreted as a gradient from open and human-made habitats to forests. While forest cover and distance from human settlements both had positive scores on this axis, open and human-made habitats had a negative weight. All bat species showed significant responses in occupancy rates to the environmental gradient described by the first axis extracted in the RDA (Fig. 4). Areas with high occupancy rates (> 66.6 %) had land-use profiles that were largely dominated by forest cover (79.78 % ± 0.98 SE), whereas areas with low occupancy rates (<33.3 %) had more evenly distributed land-use profiles: forest cover (38.20 % ± 3.68), urban (21.83 ± 3.13), crops (22.05 ± 4.29) and shrubland (17.32 ± 2.93) (Fig. 5).
Results of the first redundancy analysis (RDA) showing total occupancy rates of bat boxes in the environmental space delimited by 5-km buffer areas around bat box stations. Shaded lines are frequencies of occurrence of bats in bat boxes. Abbreviations of the variables are given in Table 1
Land-cover composition within 5-km radius buffer areas surrounding the prospected stations, categorized according to the occupancy rates of the bat boxes. The median is represented by a thicker horizontal line, the box limits are given at their respective lower (Q1) and upper (Q3) quartiles, and the vertical extending lines are standard deviations. Outliers are plotted as individual red dots
In the second redundancy analysis with occupancy rates constrained by the spatial situation of bat boxes, environmental variables explained 42 % of the variance, and both the first axis (F ratio = 19.1, P = 0.004) and the four axes together (F ratio = 2.8, P = 0.01; Table 1) were significant. The final model yielded greater influence for environmental variables (30 %) followed by space (16 %), but with an important fraction of shared variance (26 %) and 28 % of unexplained variance. Together, environmental and spatial variables in the model explained 72 % of the variance in bat box occupancy rates.
Landscape-cover effects modelled using a generalized linear model confirmed a positive effect for forest cover in forest-dwelling (Table 2, Fig. 6). For house-dwelling bats, the forest cover effect was only slightly significant, together with the distance to the near human settlement (Table 3, Fig. 6). The negative influence of urban cover was only detected for N. leisleri (a forest-dwelling bat), no relation being found for Pipistrellus sp. (Fig. 6).
Discussion
The placing of bat boxes is one of the most commonly used bat conservation techniques in Europe; bat lovers, technical staff, town councils, forest managers, researchers and educational institutions all use these boxes as alternative roost sites for bats near urban areas (Rueegger 2016). Nonetheless, there is a remarkable lack of specific information regarding the influence of the surrounding landscape composition on box occupancy, a factor that would seem to be important for forest-dwelling bats (Boughey et al. 2011; Rueegger 2016). This consideration should be borne in mind when designing box placement strategies that will guarantee project efficiency, especially when funding for biological conservation tasks is tight and closely controlled.
We provide here strong evidence to suggest that in the study area bat box occupancy was influenced by landscape composition around bat boxes and by the spatial distribution of bat boxes. This pattern was similar for both N. leisleri and Pipistrellus sp., which have different roosting strategies (tree-dwelling and house-dwelling, respectively). Occupancy rates decreased along the gradient of anthropic influence, from areas covered by natural forests to areas covered by human landscapes (urban areas and croplands). Thus, our data shows that boxes placed in non-native forests with a high percentage of nearby urban land cover will have lower-than-expected occupation rates. Thus, if landscape composition in surrounding areas is not taken into account, bat population trends cannot be properly measured in forests simply by monitoring bat boxes, contrary to what is pointed out in the Guidelines for Surveillance and Monitoring of European Bats (Battersby 2010, and references therein). However, placing bat boxes in forested areas far from highly urbanized areas is a more reliable technique for surveying certain bat species than other available methods (Jones and McLeish 2004). The fact that our models also indicated the same patterns as the RDA analysis strongly supports our hypothesis that forest cover has a positive effect on bat box occupancy success and that urban cover has the opposite effect.
The effect that bat box spatial distribution within the landscape has on occupancy rates could reflect the territorial behaviour of the studied species, which usually return to the same roost site every year (specially N. leisleri as a migratory forest-dwelling bats). This also suggests that bats switch between nearby roosts within the same natural area, thereby increasing the probability of finding occupied bat boxes near to known colonies (as opposed to far from known colonies) (Bartonička and Řehák 2007; Ruczyński and Bogdanowicz 2008; Dondini and Vergari 2009). Phylopatry could explain why nearby bat boxes (between stations or within the same region) tend to be more often occupied by bats than isolated boxes (Boston 2008; Boston et al. 2012). Areas with greater insect availability or that are better protected from adverse weather conditions could also possess regionally higher abundances of bats (mostly house-dwelling bats), which could provoke these spatial contagious occupation effects between stations or natural parks (Bartonička and Řehák 2007). In wetlands with rice paddies where there are no suitable buildings or trees for roosting, bat box occupancy rates are extremely high (>95 %, Flaquer et al. 2006).
In our study the occupation rate of bat boxes by P. kuhlii and P. pygmaeus was roughly similar (and reduced in comparison with the occupation rates in swamps or other areas) to those of N. leisleri. However, abundance of Pipistrellus sp. in the study areas is much higher than N. leisleri (Flaquer et al. 2007, 2010; Flaquer and Puig-Montserrat 2012). Such low occupancy rates could be the consequence of unfavourable habitat suitability around bat boxes or due to the presence of alternative roosts. The fact that urban land cover around bat boxes reduces the occupation rates of house-dwelling bats could be attributable to the exceptionally high number of available roosts that this type of landscape provides (Russo et al. 2004). Buildings often offer optimum roosting sites for house-dwelling bats and it is possible that only large bat boxes will tempt these bats to move away from buildings (Tuttle et al. 2004; Flaquer et al. 2014). Due to the low rates of occupied bat boxes in urban environments, we do not recommend to place bat boxes on a large scale in non-native forests near cities or in highly urbanized landscapes.
The fact that bat box occupancy by the tree-dwelling bat N. leisleri strongly decreases with urban cover (Fig. 6) highlights this species’ need for a minimum size of forest for roosting during the mating season and when wintering in the Mediterranean region (no evidence of breeding was found in the study area). This is probably due to the fact that this species of bat usually positively selects sites such as hollow trees, natural crevices and woodpecker nests in which to roost and breed (Ruczynski and Bogdanowicz 2005). In general, bat boxes are able to provide relevant data for less abundant species that would be otherwise almost impossible to obtain. For instance, N. leisleri (a tree-dwelling species) was first recorded in the study area in bat boxes (Flaquer et al. 2010). This species is a long-distant migrant in Europe and is difficult to capture. It forms small male-only colonies throughout the year, in which in the northern part of the Iberian Peninsula females are only present during the mating season (Ibáñez et al. 2009). Currently, other noctule species are being monitored using bat boxes in urban areas (Ibáñez et al. 2009; Alcalde et al. 2013).
Conclusions
As noted by Boughey et al. (2011), landscape composition has a strong effect on roost occupancy in tree-dwelling bats and must be taken into consideration when setting up bat box stations for bat conservation. Small groups of boxes located in continuous extensions of non-native forests, far from urbanized landscapes, could provide alternative roost sites for woodland species and have relatively high monitoring efficiency (>30 %). However, the placing of bat boxes in highly fragmented secondary forests surrounded by urban areas is not effective as a strategy for bat conservation and does not provide alternative roost sites.
References
Alcalde JT, Campion J, Fabo J, Marín F, Artázco A, Martínez I, Antón I (2013) Ocupación de cajas-refugio por murciélagos en Navarra. Barb 6(1):34–43. doi:10.14709/BarbJ.6.1.2013.05
Bartonička T, Řehák Z (2007) Influence of the microclimate of bat boxes on their occupation by the soprano pipistrelle Pipistrellus pygmaeus: possible cause of roost switching. Acta Chiropterol 9(2):517–526. doi:10.3161/1733-5329(2007)9[517:IOTMOB]2.0.CO;2
Battersby J (2010) Guidelines for Surveillance and Monitoring of European Bats. EUROBATS Publication Series. No. 5. UNEP / EUROBATS Secretariat, Bonn, Germany. ISBN 978–92–95058–27–9
Benzal J (1991) Population dynamics of the brown long-eared bat (Plecotus auritus) occupying bird boxes in a pine forest in central Spain. Neth J Zool 41:241–249. doi:10.1163/156854291X00171
Bocard D, Legendre P, Drapeau P (1992) Partialling out the space component of ecological variation. Ecol 73:1045–1055
Borcard D, Legendre P, Avois-Jacquet C, Tuomisto H (2004) Dissecting the spatial structure of ecological data at multiple scales. Ecol 85:1826–1832. doi:10.1890/03-3111
Boston ESM (2008) Molecular ecology and conservation genetics of the Leisler's bat (Nyctalus leisleri) in Ireland. Dissertation, Queen's University of Belfast
Boston ES, Roué SG, Montgomery WI, Prodöhl PA (2012) Kinship, parentage, and temporal stability in nursery colonies of Leisler’s bat (Nyctalus leisleri). Behav Ecol 23(5):1015–1021. doi:10.1093/beheco/ars065
Boughey KL, Lake IR, Haysom KA, Dolman PM (2011) Effects of landscape-scale broadleaved woodland configuration and extent on roost location for six bat species across the UK. Biol Conserv 144(9):2300–2310. doi:10.1016/j.biocon.2011.06.008
Boyd IL, Stebbings RE (1989) Population changes of brown long-eared bats (Plecotus auritus) in bat boxes in Thetford Forest. J Appl Ecol 26:101–112
Braak CJF, Smilauer P (2002) CANOCO reference manual and CanoDraw for Windows user's guide: software for canonical community ordination (version 4.5)
Brigham RM, Vonhof MJ, Barclay RMR, Gwilliam JC (1997) Roosting behaviour and roost-site preferences of forest-dwelling California bats (Myotis californicus). J Mammal 78:1231–1239
Burnham KP, Anderson DR (2004) Multimodel inference understanding AIC and BIC in model selection. Sociol meth res 33(2):261–304. doi:10.1177/0049124104268644
Chambers CL, Alm V, Siders MS, Rabe MJ (2002) Use of artificial roosts by forest-dwelling bats in northern Arizona. Wildlife Soc B 30(4):1085–1091
Dondini G, Vergari S (2009) Harem size and male mating tactics in Nyctalus leisleri (Kuhl, 1817) (Chiroptera, Vespertilionidae). Hystrix 20(2):147–154. doi:10.4404/hystrix-20.2-4445
Dray S, Dufour AB (2007) The ade4 package: implementing the duality diagram for ecologists. J Stat Softw 22(4):1–20
Flaquer C, Puig-Montserrat X (2012) Els Ratpenats de Catalunya. Guia de Camp. Edicions Brau, Barcelona
Flaquer C, Ruiz-Jarillo R, Torre I, Arrizabalaga A (2005) First resident population of Pipistrellus nathusii (Keyserling and Blasius, 1839) in the Iberian Peninsula. Acta Chiropterol 7:183–188. doi:10.3161/1733-5329(2005)7[183:FRPOPN]2.0.CO;2
Flaquer C, Torre I, Jarillo R (2006) The value of bat boxes in the conservation of Pipistrellus pygmaeus in wetland rice paddies. Biol Conserv 128:223–230. doi:10.1016/j.biocon.2005.09.030
Flaquer C, Torre I, Arrizabalaga A (2007) Selección de refugios, gestión forestal y conservación de los quirópteros forestales. In: Camprodon J, Plana E. (ed) Conservación de la biodiversidad y gestión forestal: su aplicación en la fauna vertebrada, Edicions de la Universitat de Barcelona pp: 469–488.
Flaquer C, Puig X, Fàbregas E, Guixé D, Torre I, Ràfols RG, Páramo F, Camprodon J, Cumplido JM, Jarillo RR, López-Baucells A, Freixas L, Arrizabalaga A (2010) Revisión y aportación de datos sobre quirópteros de Catalunya: propuesta de lista roja. Galemys 22(1):21–54
Flaquer C, Puig X, López-Baucells A, Torre I, Freixas L, Mas M, Porres X, Arrizabalaga A (2014) Could overheating turn bat boxes into death traps? Barb 7(1):46–53. doi:10.14709/BarbJ.7.1.2014.08
Geoffrey C, Agnew SG (2002) The value of bat boxes for attracting hollow-dependent fauna to farm forestry plantations in southeast Queensland. Ecol. Manage. Restor. 3(1):37–46. doi:10.1046/j.1442-8903.2002.00088.x
Ibáñez C, Guillén A, Agirre-Mendi PT, Juste J, Schreur G, Cordero AI, Popa-Lisseanu AG (2009) Sexual Segregation in Iberian Noctule Bats. J Mammal 90(1):235–224. doi:10.1644/08-MAMM-A-037.1
Jones AJM, McLeish AP (2004) The bat worker's manual. 3rd Edition. Joint nature conservation committee: UK populations 172
Kerth G, Weissmann K, König B (2001) Day roost selection in female Bechstein’s bats (Myotis bechsteinii): a field experiment to determine the influence of roost temperature. Oecologia 126:1–9. doi:10.1007/s004420000489
Kunz TH (1982) In: Kunz TH (ed) Roosting ecology of bats. In: “Ecology of Bats”. Plenum Press, New York
Kunz TH, Lumsden LF (2003) In: Kunz TH, Fenton MB (eds) Ecology of cavity and foliage bats. In “Bat ecology”. University of Chicago Press, Chicago
Kunz TH, Parsons S (2009) Ecological and behavioral methods for the study of bats. Johns Hopkins University Press, Baltimore
Kunz TH, de Braun TE, Bauer D, Lobova T, Fleming TH (2011) Ecosystem services provided by bats. Ann N Y Acad Sci 1223:1–38. doi:10.1111/j.1749-6632.2011.06004.x.
Leps J, Smilauer P (2003) Multivariate analysis of ecological data using CANOCO. Cambridge University Press, Cambridge
Lourenço SI, Palmeirim JM (2004) Influence of temperature in roost selection by Pipistrellus pygmaeus (Chiroptera): relevance for design of bat boxes. Biol Conserv 119:237–243. doi:10.1016/j.biocon.2003.11.006
Mcleod AI, Xu CJ (2014) Best subset glm using AIC, BIC, EBIC, BICq or cross-validation. R package version 0.34. https://cran.r-project.org/web/packages/bestglm/. Accessed 3 Mar 2014
Patriquin KJ, Barclay RMR (2003) Foraging by bats in cleared, tinned and unharvested boreal forest. J Appl Ecol 40:646–657. doi:10.1046/j.1365-2664.2003.00831.x
Pierson ED (1998) In: Kunz TH, Racey PA (eds) Tall trees, deep holes and scarred landscapes: conservation biology of North American bats. In “Bat Biology and Conservation”. Smithsonian Institution Press, Washington, D. C.
Puig-Montserrat X, Torre I, López-Baucells A, Guerrieri E, Monti MM, Ràfols-García R, Ferrer X, Gisbert D, Flaquer C (2015) Pest control service provided by bats in Mediterranean rice paddies: linking agroecosystems structure to ecological functions. Mamm Biol. 80(3):237–245. doi:10.1016/j.mambio.2015.03.008
Racey PA (1998) In: Kunz TH, Racey PA (eds) Ecology of European bats in relation to their conservation. In “Bat biology and Conservation”. Smithsonian Institution Press, Washington, D.C
Ruczynski I, Bogdanowicz W (2005) Roost cavity selection by Nyctalus noctula and N. leisleri (Vespertilionidae, Chiroptera) in Bialowieza primeval forest, eastern Poland. J Mammal 86(5):921–930. doi:10.1644/1545-1542(2005)86[921:RCSBNN]2.0.CO;2
Ruczyński I, Bogdanowicz W (2008) Summer roost selection by tree-dwelling bats Nyctalus noctula and N. leisleri: A multiscale analysis. J Mammal 89(4):942–951. doi:10.1644/07-MAMM-A-134.1
Rueegger N (2016) Bat boxes - a review of their use and application, past, present and future. Acta Chiropt 18(1):279–299. doi:10.3161/15081109ACC2016.18.1.017
Russo D, Ancillotto L (2015) Sensitivity of bats to urbanization: a review. Mamm Biol - Zeitschrift für Säugetierkunde, 80(3):205–212. doi:10.1016/j.mambio.2014.10.003
Russo D, Cistrone L, Jones G, Mazzoleni S (2004) Roost selection by barbastelle bats (Barbastella barbastellus, chiroptera: vespertilionidae) in beech woodlands of central Italy: consequences for conservation. Biol Conserv 117:73–81. doi:10.1016/S0006-3207(03)00266-0
Russo D, Cistrone L, Jones G (2005) Spatial and temporal patterns of roost use by tree-dwelling barbastelle bats Barbastella barbastellus. Ecography 28(6):769–776
Russo D, Cistrone L, Jones G (2007) Emergence time in forest bats: the influence of canopy closure. Acta Oecol 31(1):119–126
Torre I, Díaz M, Arrizabalaga A (2014) Additive effects of climate and vegetation structure on the altitudinal distribution of greater white-toothed shrew Crocidura russula in a Mediterranean mountain range. Acta Theriol 59:139–147. doi:10.1007/s13364-013-0128-y
Tuttle M, Kiser M, Kiser S (2004) The Bat House Builder’s Handbook. Bat Conservation International Inc, Austin
Wei T, Simko V (2016) Package ‘corrplot’. R package version 0.77. https://cran.r-project.org/web/packages/corrplot/. Accessed 21 Apr 2016
Acknowledgments
This project received financial support from the Barcelona Provincial Council (Diputació de Barcelona). We are especially thankful to Parc de Collserola, Parc del Foix, Parc Natural del Montnegre i el Corredor, Parc de la Serralada de Marina and the Parc de la Serralada Litoral. The Comissionat per a Universitats i Recerca de la Generalitat de Catalunya provided the material necessary for this research. We are also greatly indebted for their support to the following people: A. Cabrerizo, C. Perez, D. Guinart, F. Llimona, G. Llimós, I. Cabrera, I. Pac, J. Capdevila, J. Torrentó, L. C. Cabañeros, M. Vila, P. Mundó, P. Panon, R. Loireand and T. Bombí. Finally, we would like to make special mention of Dr. Danilo Russo, who has acted as our mentor during this formative period of our research (which is still underway!).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
López-Baucells, A., Puig-Montserrat, X., Torre, I. et al. Bat boxes in urban non-native forests: a popular practice that should be reconsidered. Urban Ecosyst 20, 217–225 (2017). https://doi.org/10.1007/s11252-016-0582-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11252-016-0582-9