Abstract
This study aimed to evaluate the antioxidant effects of onion (Allium cepa L.) powder on the immunological variables, redox state, and semen quality of rabbit bucks exposed to severe heat stress. Thirty-six mature bucks (7 months old) were divided into three groups consisting of 12 bucks each, namely group I, control; group II, 400 mg onion powder/kg diet; and group III, 800 mg onion powder/kg diet. The quality of semen was evaluated for volume, pH, motility, concentration, total sperm output, viability, and packed sperm volume. Blood samples were collected in the 12th week for estimation of red blood cells (RBC), white blood cells (WBC), and erythrocytic indices. Serum proteins, glutamate oxaloacetate (GOT), glutamate pyruvate transaminase (GPT), urea, creatinine, testosterone, follicle-stimulating hormone (FSH), luteinizing hormone (LH), immunoglobulins, malondialdehyde (MDA), total antioxidant capacity (TAC), glutathione, superoxide dismutase (SOD), and catalase (CAT) were measured. The temperature-humidity index (THI) obtained was within the range of 28.85–33.08 indicating severe heat stress. The results show that mass and individual motility, concentration, total sperm output, sperm viability, and packed sperm volume were higher (P < 0.05) in groups II and group III, with group III having the highest (P < 0.05) levels compared to group I. Compared to group I, groups II and III had higher (P < 0.05) concentrations of RBC, MCV, MCH, FSH, LH, SOD, and catalase. The highest concentration (P < 0.05) of GPT was obtained in group III compared to other groups. The highest concentration of IgG (P < 0.05) was obtained in group II while the lowest was in group I. In conclusion, dietary supplementation with onion powder at 400 or 800 mg/kg diet improves semen quality, RBC, FSH, LH, SOD, catalase, and IgG while ameliorating the adverse effects of heat stress and improve the health and reproduction of rabbits.
Graphical abstract

Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Climatic conditions, which are exacerbated by global warming, have an impact on rabbit production (Oladimeji et al., 2022). The thermoneutral zone for rabbits is between 21–25 °C and 60–70% for ambient temperature and relative humidity, respectively (Cheeke 1987; National Research Council, 1996). Temperature above the thermoneutral zone induces heat stress in rabbits (Mutwedu et al., 2022). Rabbits have few or no sweat glands and thick insulating fur making it difficult to dissipate heat from the body easily thereby predisposing it to heat stress. Rabbits are extremely sensitive to temperature fluctuations and easily exhibit stress reactions (Marai et al., 2002; Oladimeji et al., 2022). Heat stress increases the formation of reactive oxygen species (ROS), which overwhelms the antioxidant system and results in oxidative stress. The activity of antioxidant enzymes (superoxide, catalase, and glutathione peroxidase) is outweighed by oxidative stress which may reduce the body’s antioxidant capacity (Chauhan et al., 2021). Increased ROS formation causes lipid peroxidation of cell membranes measured by the concentration of plasma malondialdehyde (MDA) (Sunil et al., 2011; Hosny et al., 2020). Heat stress adversely affects the reproductive ability of bucks; it decreases semen quality and quantity due to high polyunsaturated fatty acids in sperm membrane with low antioxidant levels within the cytoplasm (El-Gindy, 2022) that leads to the destruction of cells causing apoptosis of spermatozoa (El-Ratel et al., 2022; El-Sherbiny et al., 2022). Besides, heat stress weakens the immune functions of animals by shifting from cell-mediated to humoral immunity which makes them susceptible easily to diseases (Inbaraj et al., 2016; Chauhan et al., 2021).
Some natural plants contain bioactive components which may have positive impacts on animal reproduction (El-Gindy et al., 2022a) and combat the negative effects of thermal stress under conditions through various physiological processes (El-Gindy et al., 2023). Onion (Allium cepa) is a bulb vegetable that grows worldwide (Lawande, 2012). Onion has beneficial effects on health due to its contents of several antioxidants and sulfur-containing compounds (Srinivasan, 2014; Sidhu et al., 2019). Onions are rich sources of flavonoids, quercetin, glycosides, anthocyanins, allicin, and polyphenols. The antioxidant capacity of onion is associated with the content of phenol, flavonoids, and quercetin (Ye et al., 2013; Omar et al., 2020). The antioxidant activity is due to its ability to scavenge ROS by giving out electrons or hydrogen atoms (Nuutila et al., 2003). Its anti-inflammatory and antioxidant properties have been connected to enhanced bone health, lower blood sugar levels, and a decreased risk of cancer (Jafarpour-Sadegh et al., 2017; Zhao et al., 2021). Onion improves antioxidant enzyme activities, immune status, and performance of birds (Omar et al., 2020; ur Rahman et al., 2022). Onion extract improves ruminant performance, total antioxidant capacity, and leptin and ghrelin concentrations (Amiri et al., 2019). The purpose of the study was to assess the antioxidant activity of onion (Allium cepa L.) powder on the redox status, immunological characteristics, and semen quality of rabbit bucks that were subjected to severe heat stress.
Material and methods
Ethical approval
The Institutional Animal Care and Use Committee of Alexandria University gave its approval for the study with Reference Number: 14-20-09-20-3-13.
Study area
The research was conducted at Alexandria University in Alexandria, Egypt, during the hot-dry season (July–September 2020) in the Department of Fish and Animal Production, Saba Basha Faculty of Agriculture.
Source of onion powder
Onion powder was purchased from American Garden, NY, 11783, USA.
Animals, management, and diets
The experimental subjects consisted of 36 male V-Line rabbits aged 7 months, with an average body weight of 3.55 kg. The rabbits were kept in galvanized wire cages in the rabbitry of the Department of Fish and Animal Production. The rabbitry was well ventilated, and biosecurity measures were ensured. The rabbits were allowed to acclimatize to the environment for 3 weeks before commencing the study which lasted for 9 weeks. They were randomly allocated into three groups of twelve (12) bucks each, namely 1st group, control without any supplementation; 2nd group, onion powder at levels 400 mg/kg diet; 3rd group, onion powder at levels 800 mg/kg diet, according to (Saber et al., 2019). The composition of experimental diets is presented in Table 1.
Meteorological data
During the experiment, daily readings of the ambient temperature (°C) and relative humidity (%) were taken in the rabbitry. Temperature-humidity index (THI) was computed according to Marai et al. (2001) as follows:
where db, dry bulb temperature (°C), and RH, relative humidity (%).
Semen collection and evaluation
An artificial vagina containing warm water (approximately 45°C) was used to collect semen samples from 36 mature V-line rabbit bucks every 2 weeks. The period between the doe being placed in the buck’s cage and ejaculating was used to calculate libido. Libido was measured from when the doe was placed in the buck’s cage to the moment of ejaculation. The ejaculated volume was measured to the nearest 0.01 ml after the removal of any gel plug. With the aid of pH paper strips (range: 0–14, resolution: 1.0 pH unit; Sigma-Aldrich VR), the ejaculate’s hydrogen ion concentration (pH) was determined immediately. Sperm mass motility was determined on freshly obtained semen placed on a warm stage at 37°C. To quantify sperm individual motility, the semen samples were diluted with physiological saline solution, and observations were done at a magnification of ×400. The modified Newbuer hemocytometer slide reported by Smith and Mayer (1955) was used to measure the concentration of sperm using a mild eosin-formalin (10% formalin) solution. The total sperm output per ejaculate was calculated. A mixture of eosin and aniline blue stain was used to measure the sperm viability percentage (Shaffer and Almquist,1948). According to Yousef (2005), packed sperm volume (PSV) was determined using Micro-AIDVR microhematocrit tubes and a microhematocrit centrifuge for 5 min at 700 g.
Blood collection and evaluation
Six bucks from each group had exactly 7 mL of blood drawn from the marginal ear. A blood sample of 2 mL was placed in a vacuum tube with EDTA as an anticoagulant, and a sample of 5 mL was placed in a vacuum tube without an anticoagulant. The blood samples without anticoagulant were centrifugated at 700g for 10 min, serum was harvested and stored at −20°C pending biochemical analysis.
Hematological parameters
The concentration of hemoglobin (Hb) was measured using the cyanomethemoglobin method (Wintrobe, 1965), and the packed cell volume on the microhematocrit tubes was used to calculate the hematocrit percentage. Red blood cells (RBC) were counted using Natt and Herrick’s (1952) method. Hepler’s (1966) method was used to count the white blood cells (WBC). The Pappenheim method (Giemsa stain) was used to determine the WBC’s cell differentiation. According to Jain’s (1983) formulae, mean corpuscular volume (MCV), mean corpuscular hemoglobin (MCH), and mean corpuscular hemoglobin concentration (MCHC) were computed.
Blood biochemical parameters
In accordance with the manufacturer’s instructions, commercial kits (Biodiagnostic, Egypt) were used to spectrophotometrically measure total protein, albumin, GPT, GOT, urea, and creatinine. The albumin/globulin ratio and globulin were also calculated.
Redox status and immunity
The following parameters were measured by spectrophotometer using commercial kits (Biodiagnostic, Egypt) in accordance with the manufacturer’s instructions: total antioxidant capacity (TAC), malondialdehyde concentration (MDA), glutathione (GSH), superoxide dismutase (SOD), and catalase (CAT). An ELISA technique was used to measure the serum immunoglobulins (IgG and IgM).
Reproductive hormones
The serum levels of luteinizing hormone (LH), follicle-stimulating hormone (FSH), and testosterone in rabbits were measured using commercial enzyme immunoassay test kits (rabbit testosterone, free ELISA kit Category No. MBS012519; rabbit luteinizing hormone, LH ELISA Kit No. MBS912114; and rabbit Follicle-stimulating hormone (FSH) ELISA Kit No. MBS2600270).
Statistical analysis
Mean ± standard error of the mean (mean ± SEM) was used to present the study’s data. All data results were analyzed with a one-way analysis of variance (ANOVA), and Tukey’s post hoc test was used to assess how the means of the various treatment groups varied from one another. To analyze the data, Windows SPSS Version 16 was used. Values of P < 0.05 were regarded as significant.
Results
Thermal conditions in the rabbitry
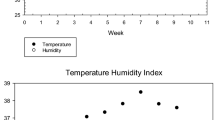
The air temperature, relative humidity, and THI obtained during the study period ranged between 30.66–34.66 °C, 64–74%, and 28.85–33.08, respectively. Natural daylight in the study ranged between 14 and 16 h per day (Table 2).
Semen quality
Significantly (P < 0.05) higher levels of semen mass, individual motility, concentration, total sperm output, sperm viability, and packed sperm volume were obtained in groups II and III, with group I, having the lowest levels in each parameter. Between groups, there was no significant difference (P > 0.05) in the pH of the semen. In comparison to the other groups, group III had significantly shorter (P < 0.05) libido (Table 3).
Hematology parameters
The RBC count was significantly (P < 0.05) higher in groups II and III than in group I. Group III had the highest (P < 0.05) WBC and monocyte counts compared to groups I and II. The hematocrit, neutrophil, and eosinophil counts did not differ (P > 0.05) between the groups. In comparison to group I, groups II and III had significantly (P < 0.05) higher MCV and significantly lower MCH values. The highest (P < 0.05) MCHC was obtained in group III than in other groups (Table 4).
Serum biochemical parameters
Total protein, albumin, globulin, and GOT concentrations did not differ (P > 0.05) between the groups. Group III had the highest (P < 0.05) GPT concentration when compared to the other groups. The concentrations of urea and creatinine were not significantly different (P > 0.05) across the groups (Table 5).
Reproductive hormones
The highest (P < 0.05) testosterone concentration was obtained in group II than in other groups. In comparison to group I, groups II and III had significantly (P < 0.05) higher FSH and LH concentrations (Table 6).
Semen immunoglobulins
Immunoglobulin G concentration was significantly (P < 0.05) increased in groups II (755.83mg/dL) and III (722.00 mg/dL) than in group I (582.80 mg/dL). The highest (P < 0.05) IgM concentration was obtained in group III while the lowest was in groups I and II (Fig. 1).
Serum redox status
The TAC and MDA concentrations did not differ significantly (P > 0.05) among groups. Glutathione concentration was highest (P < 0.05) in group I (1.88 nmol/mL) and lowest in group III (1.61 nmol/mL). The activities of SOD and catalase were significantly (P < 0.05) increased in group II (2.04 U/L and 436.00 U/L, respectively) and III (1.91 U/L and 500.20 U/L, respectively) than in group I (1.71 U/L and 333.20 U/L, respectively (Fig. 2).
Discussion
The meteorological condition of the study area shows that air temperature and relative humidity were above the thermoneutral zone (21–25 °C and 60–70%, respectively) meant for rabbits. The temperature-humidity index obtained in the study was between 28.85 and 33.08. The temperature-humidity index, which measures an animal’s heat burden, has been divided into four categories for rabbits: none (27.8), moderate (27.8–28.8), severe (28.9–29.9), and very severe (>30.0) (LPHSI, 1990). The result obtained from the study showed that the rabbits were under severe heat stress conditions. The finding is similar to that of Mutwedu et al. (2022) which demonstrated that temperature above the thermoneutral zone induced heat stress in rabbits. Heat stress has been reported to adversely affect the physiology of rabbits (Liang et al., 2022). Measures must be ensured at ameliorating the impact of the thermal conditions to mitigate the adverse effect of heat stress on the rabbits.
The increase in semen volume, motility, concentration, total sperm output, sperm viability, and packed sperm volume in groups administered onion powder than in non-supplemented rabbits shows that onion powder improved the semen quality of the rabbits. This finding is similar to the report of El-Gindy et al. (2022b) who showed increased semen quality and a decrease in the negative impact of heat stress on the semen quality of rabbits administered with an antioxidant (fresh onion juice). Onion as an antioxidant scavenges excess reactive oxygen species by increasing the antioxidant capacity of the body (Omar et al., 2020). The decrease in the generation of ROS decreases the effects of heat stress on semen (Hosny et al. 2020). According to Marai et al. (2002), heat stress reduces the volume of semen by lowering sperm concentration and the activity of accessory glands and testes. Due to negative effects on epididymal function, heat stress also reduces sperm motility due to lower sperm concentration and increases the percentage of dead spermatozoa (Naseer et al. 2018).
The increased RBC count in groups administered onion powder at 400 or 800 mg/kg diet shows that onion being an antioxidant was involved in increased erythropoiesis. This may be due to the antioxidant effect of onion in increasing the antioxidant capacity which decreases the heat stress effect on RBC. The decrease in red blood cell count in the non-supplemented group shows the effect of heat stress. Heat stress increases the production of ROS which causes lipid peroxidation of cell membrane leading to the destruction of RBC. This may lead to a reduction in the number of RBCs (Cheeseman, 1993; Abdelnour et al., 2020). The result is consistent with the findings of Abdelnour et al. (2020) on heat stress effects on RBC. The increased WBC in groups given onion powder, especially at 800mg/kg diet shows that onion powder may increase the immune response of the rabbits to heat stress. Onion powder at 400mg/kg nor 800 mg/kg did not affect the concentration of proteins, GOT, urea, and creatinine. The increased concentration of GPT in the group given onion at 800mg/kg diet shows that onion at the concentration induces hepato-cellular damage. The results of our study agree with Thomson et al. (1998), which found that high doses of onion (500 mg/kg) produced obvious histological changes in rat liver tissue.
The increased testosterone level in groups given onion powder, especially in group II, shows the antioxidant effect of onion on the heat-stressed bucks. The finding is due to the increased production of LH and FSH and the defense system of the animal. The enhanced defense system ameliorates the adverse effects of ROS on testosterone levels. The finding agrees with the report of Banihani (2019) who showed that onion enhances testosterone levels in male animals. The finding of decreased testosterone levels in non-supplemented heat-stressed rabbits agrees with the findings of Fadl et al. (2022) who showed decreased testosterone in rams subjected to heat stress. Heat stress adversely affects testicular cells which are involved in the production of testosterone (Shahat et al., 2020).
The increase in immunoglobulin G obtained in groups given onion powder especially group II shows that onion powder enhances the immunity of the rabbit under severe heat stress. Due to the antioxidant properties of onion, the production of ROS is suppressed, reducing the adverse effects of heat stress and boosting the immune response of the rabbits. The finding is similar to El-Kholy et al. (2019) who reported increased immunoglobulins in rabbits administered antioxidants.
The enhanced activities of SOD and CAT in groups given onion powder show the antioxidant property of onion. Onion powder increases the antioxidant capacity which enhances the defense system of the body. The body’s first line of defense against oxidative stress is comprised of the SOD and CAT enzymes. The enhanced antioxidant enzymes scavenge excess ROS generated, reducing the effects of heat stress on the rabbits. The decreased SOD and CAT obtained in the non-supplemented group shows that heat stress increases the generation of ROS which overwhelms the antioxidant capacity of the body. This supports the results of Oladimeji et al. (2022), who discovered that rabbits exposed to heat stress have lower antioxidant capacity.
Conclusion
It was concluded that onion powder improved the quality of the semen, hematological parameters (RBC and WBC), reproductive hormones (testosterone, LH, and FSH), immunoglobulin G and M, and redox status (SOD and CAT) in order to reduce the negative effects of severe heat stress on the rabbits. The high dose of onion powder in an 800 mg/kg diet could cause damage to liver tissue that is conformed by increased GPT. Onion powder at a 400 mg/kg diet may ameliorate the adverse effects of heat stress and improve the health and reproduction of rabbits.
Data availability
Not applicable.
References
Abdelnour, S. A., Swelum, A. A., Salama, A., Al-Ghadi, M. Q., Qattan, S. Y., Abd El-Hack, M. E. and El-Saadony, M. T. (2020). The beneficial impacts of dietary phycocyanin supplementation on growing rabbits under high ambient temperature. Italian Journal of Animal Science, 19(1), 1046-1056.
Amiri, M., Jelodar, G. A., Erjaee, H. and Nazifi, S. (2019). The effects of different doses of onion (Allium cepa. L) extract on leptin, ghrelin, total antioxidant capacity, and performance of suckling lambs. Comparative Clinical Pathology, 28(2), 391-396.
Banihani, S. A. (2019). Testosterone in males as enhanced by onion (Allium Cepa L.). Biomolecules, 9(2), 75.
Chauhan, S. S., Rashamol, V. P., Bagath, M., Sejian, V. and Dunshea, F. R. (2021). Impacts of heat stress on immune responses and oxidative stress in farm animals and nutritional strategies for amelioration. International Journal of Biometeorology, 65(7), 1231-1244.
Cheeseman, K. H. (1993). Mechanisms and effects of lipid peroxidation. Molecular aspects of medicine, 14(3), 191-197.
El-Gindy, Y. M., Zahran, S. M., Ahmed, M. H., Adegbeye, M. J., Salem, A. Z., & Salam, M. Y. (2022a). Enhancing semen quality, antioxidant status and sex hormones of V-line rabbit bucks fed on supplemented diets with dried moringa leaves. Animal Biotechnology, 1-10.
El-Gindy, Y. M., Zahran, S. M., Hassan, M. A. and Sabir, S. A. (2022b). Effect on physiological parameters and semen quality upon oral administration of fresh onion juice to V-line rabbit buck during severe heat stress. Animal Biotechnology, 1-9.
El-Gindy, Y. M. (2022). Improvement in quality and storage ability of rabbit semen by using black or thyme seed as dietary supplementation. Journal of Animal Physiology and Animal Nutrition, 106(3), 642-654.
El-Gindy, Y. M., Hafsa, S. H. A., & El-Deeb, N. M. (2023). The expression of liver TNF-α gene, liver and small intestine histology of thermal stressed growing rabbits affected by allicin and lycopene. Journal of Thermal Biology, 113, 103521.
El-Kholy, K. H., El-Deen, H. T., Abd-El-Lateif, A. I. and Mekaouy, A. I. (2019). Effects of dietary selenium sources on metabolic, enzymatic and immunoglobulin serum profiles in growing rabbits. Pakistan Journal of Nutrition, 2019-430.
El-Ratel, I., El-Moghazy, M., El-Gaml, A. and El-Naser, I. A. (2022). Effects of propolis ethanolic extract administration on quality of fresh and cryopreserved semen, redox status, and sperm flow cytometry parameters of heat-stressed rabbit bucks. Advances in Animal and Veterinary Sciences 10(12), 2578-2588.
El-Sherbiny, H. R., Abdelnaby, E. A., El-Shahat, K. H., Salem, N. Y., Ramadan, E. S., Yehia, S. G. and Fathi, M. (2022). Coenzyme Q10 Supplementation enhances testicular volume and hemodynamics, reproductive hormones, sperm quality, and seminal antioxidant capacity in goat bucks under summer hot humid conditions. Veterinary Research Communications, 46(4), 1245-1257.
Fadl, A. M., Abdelnaby, E. A. and El-Sherbiny, H. R. (2022). Supplemental dietary zinc sulphate and folic acid combination improves testicular volume and haemodynamics, testosterone levels and semen quality in rams under heat stress conditions. Reproduction in Domestic Animals
Hepler, O.E., 1966. Manual of Clinical Laboratory Methods. (4th ed). Charles C Thomas publisher, Illinois.
Hosny, N. S., Hashem, N. M., Morsy, A. S. and Abo-Elezz, Z. R. (2020). Effects of organic selenium on the physiological response, blood metabolites, redox status, semen quality, and fertility of rabbit bucks kept under natural heat stress conditions. Frontiers in Veterinary Science, 7, 290.
Inbaraj, S., Sejian, V., Bagath, M. and Bhatta, R. (2016). Impact of Heat Stress on Immune Responses of Livestock: A Review. Pertanika Journal of Tropical Agricultural Science, 39(4).
Jafarpour-Sadegh, F., Montazeri, V., Adili, A., Esfehani, A., Rashidi, M. R. and Pirouzpanah, S. (2017). Consumption of fresh yellow onion ameliorates hyperglycemia and insulin resistance in breast cancer patients during doxorubicin-based chemotherapy: a randomized controlled clinical trial. Integrative Cancer Therapies, 16(3), 276-289.
Lawande K.E. (2012). Onion Handbook of Herbs and Spices (Second edition) Woodhead Publishing Series in Food Science Technology and Nutrition, 417-429
Liang, Z. L., Chen, F., Park, S., Balasubramanian, B. and Liu, W. C. (2022). Impacts of heat stress on rabbit immune function, endocrine, blood biochemical changes, antioxidant capacity and production performance, and the potential mitigation strategies of nutritional intervention. Frontiers in Veterinary Science, 9.
LPHSI, (1990). Livestock and poultry heat stress indices. agriculture engineering technology guide. ClemsonUniversity, Clemson, SC 29634, US
Marai IFM, Ayyat MS, Abd El-Monem UM. (2001). Growth performance and reproductive traits at first parity of New Zealand White female rabbits as affected by heat stress and its alleviation under Egyptian conditions. Tropical Animal Health Production, 33(6):451–462.
Marai, I. F. M., Habeeb, A. A. M. and Gad, A. E. (2002). Rabbits’ productive, reproductive and physiological performance traits as affected by heat stress: a review. Livestock production Science, 78(2), 71-90.
Mutwedu, V. B., Nyongesa, A. W., Kitaa, J. M., Ayagirwe, R. B., Baharanyi, C. and Mbaria, J. M. (2022). Effects of Moringa oleifera aqueous seed extracts on reproductive traits of heat-stressed New Zealand white female rabbits. Frontiers in Veterinary Science, 9. https://doi.org/10.3389/fvets.2022.883976.
Naseer, Z., Ahmad, E., Şahiner, H. S., Epikmen, E. T., Fiaz, M., Yousuf, M. R. and Aksoy, M. (2018). Dietary quercetin maintains the semen quality in rabbits under summer heat stress. Theriogenology, 122, 88-93.
Natt, M. P. and Herrick, C. A. (1952). A new blood diluent for counting erythrocytes and leucocytes of the chicken. Poultry Science, 31, 735–738.
Nuutila, A. M., Puupponen-Pimiä, R., Aarni, M. and Oksman-Caldentey, K. M. (2003). Comparison of antioxidant activities of onion and garlic extracts by inhibition of lipid peroxidation and radical scavenging activity. Food chemistry, 81(4), 485-493.
Oladimeji, A. M., Johnson, T. G., Metwally, K., Farghly, M. and Mahrose, K. M. (2022). Environmental heat stress in rabbits: Implications and ameliorations. International Journal of Biometeorology, 66(1), 1-11.
Omar, A. E., Al-Khalaifah, H. S., Mohamed, W. A., Gharib, H. S., Osman, A., Al-Gabri, N. A., and Amer, S. A. (2020). Effects of phenolic-rich onion (Allium cepa L.) extract on the growth performance, behavior, intestinal histology, amino acid digestibility, antioxidant activity, and the immune status of broiler chickens. Frontiers in Veterinary Science, 7, 582612.
Rahman SU, Iftikhar F, Sajid Z, Khan S, Khan R, Rahman FU. (2022). Influence of onion (Allium cepa L.) supplementation on physio-chemical composition and lipid profile of broiler meat. Biomedical Letters, 8(2), 126-135.
Saber, S., El-Gindy, Y., Morshedy, S., Zahran, S., Ahmed, M., Zeweil, S. SEMEN QUALITY, SEX HORMONE AND ANTIOXIDANT STATUS OF MALE RABBITS AS INFLUENCED BY TWO FORMS OF ONION. Egyptian Poultry Science Journal, 2019; 39(1): 31-39. https://doi.org/10.21608/epsj.2019.28788
Shaffer, H.E., Almquist, J.O. (1948). Vital staining of bovine spermatozoa with an eosin–aniline blue staining mixture. Journal of Dairy Science 31, 677–678.
Shahat, A. M., Rizzoto, G. and Kastelic, J. P. (2020). Amelioration of heat stress-induced damage to testes and sperm quality. Theriogenology, 158, 84-96.
Sidhu, J. S., Ali, M., Al-Rashdan, A. Ahmed, N. (2019). Onion (Allium cepa L.) is potentially a good source of important antioxidants. Journal of Food Science and Technology, 56(4), 1811-1819.
Smith, J.T., Mayer, D.T. (1955). Evaluation of sperm concentration by the hemacytometer method. Comparison of four counting fluids. Fertility and Sterility 6, 271–275.
Srinivasan K. (2014). Antioxidant potential of spices and their active constituents. Critical Review in Food Science and Nutrition, 54:352–372. https://doi.org/10.1080/10408398.2011.585525.
Thomson, M., Alnaqeeb, M. A., Bordia, T., Al-Hassan, J. M., Afzal, M., & Ali, M. (1998). Effects of aqueous extract of onion on the liver and lung of rats. Journal of ethnopharmacology, 61(2), 91-99.
Wintrobe, M.M. (1965). Clinical Haematology. Lea and Febiger, Philadelphia. Jain N. 1983. Scanning electron micrograph of blood cells. Schalm’s Veterinary Haematology, 4:63–70.
Ye, C. L., Dai, D. H., Hu, W. L. (2013). Antimicrobial and antioxidant activities of the essential oil from onion (Allium cepa L.). Food Control 30: 48-53. https://doi.org/10.1016/j.foodcont.2012.07.033
Yousef, M. I. (2005). Reproductive performance, blood testosterone, lipid peroxidation and seminal plasma biochemistry of rabbits as affected by feeding Acacia saligna under subtropical conditions. Food and Chemistry Toxicology, 43(2):333–339.
Zhao, X. X., Lin, F. J., Li, H., Li, H. B., Wu, D. T., Geng, F. and Gan, R. Y. (2021). Recent advances in bioactive compounds, health functions, and safety concerns of onion (Allium cepa L.) Frontiers in Nutrition, 8.
Author information
Authors and Affiliations
Contributions
YMEG, SAS, SMZ, and MAH conceived and designed the experiment; YMEG, SAS, SMZ, and MAH prepared the data analysis, YMEG, SAS, SMZ, MAH, NEO, RCR, and AZMS prepared the manuscript. All authors approved the manuscript.
Corresponding author
Ethics declarations
Ethical approval
The Institutional Animal Care and Use Committee of Alexandria University gave its approval for the study with Reference Number: 14-20-09-20-3-13.
Competing interests
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
El-Gindy, Y.M., Sabir, S.A., Zahran, S.M. et al. Effect of dietary onion (Allium cepa L.) powder as an antioxidant on semen quality, blood biochemicals, and reproductive parameters, as well as immunological variables of rabbit bucks under severe heat stress. Trop Anim Health Prod 55, 380 (2023). https://doi.org/10.1007/s11250-023-03788-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11250-023-03788-w