Abstract
Palm oil is a natural energy source ingredient in poultry diets that offers a broad range of beneficial effects on the performance of broiler chickens. This review was conducted to highlight the impact of palm oil as a feed ingredient on growth performance and carcass quality, as well as the biochemical, antioxidant activity and tissue fatty acids (FA) composition of broiler chickens. Palm oil inclusion in broiler chickens’ rations contributes significantly to the high metabolisable energy (ME) of feed formulation, increases feed palatability and decreases digesta passage rate in the intestine. The reviewed literature indicated that dietary palm oil has a beneficial effect on broiler chickens’ overall growth performance traits. The addition of palm oil can also improve the heat tolerance of chickens reared in high ambient temperature conditions. Regardless of breed and breeding conditions, palm oil exhibits good oxidative stability in broiler chickens due to the presence of prevalent phytonutrient elements in this oil. The inclusion of palm oil increased palmitic (C16:0) and oleic (C18:1) acids in tissue deposits, which improves meat stability and quality. Moreover, molecular studies have revealed that higher mRNA expression of several lipid-related hepatic genes in broiler chickens fed palm oil. Nonetheless, dietary palm oil can influence FA deposition in tissues, modulate lipoprotein and triglycerides (TG) levels, and cytokine contents in the blood serum of broiler chickens.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Chicken meat is a widely available and affordable source of high-quality protein for the majority of the world’s human population. Malaysia is one of the highest chicken meat consumers in Asia, with estimated per-capita consumption of 49.3 kg in 2020 (DVS. 2019). Global broiler chickens production is projected to continue to grow as the demand for chicken meat is driven by the increasing population growth rates. Diversifying the feeding strategies is one of the pivotal aspects of broiler chicken production due to the increasing feed ingredient prices. However, meeting the recommended metabolism energy supply in poultry feeds is essential in optimizing the cost of feeding (Long et al. 2019). The use of oils or fats is a common economic practice in modern poultry production because they provide 2.25 times more energy per gram than carbohydrates (Oladunjoye 2018).
Oils contribute significantly to high-energy feed formulation in poultry diets and improve the growth performance of broiler chickens (Khatun et al. 2018). Oils have commonly been used as metabolisable energy (ME) sources in diets to enhance energy density that can be easily stored in adenosine triphosphate (ATP) by non-ruminants animals (Jalali et al. 2015). Enhancement of nutrient utilisation and performance of broiler chickens via optimal inclusion of fats or oils has been extensively investigated (Wu et al. 2011; Khatun et al. 2018; Attia et al. 2020; Villanueva-Lopez et al. 2020). Most of the studies mainly focused on the effects of oil inclusion on growth performance, feed efficiency, carcass quality, fat deposition in muscle and synthesis of fatty acid (FA) deposits in adipose tissues of broiler chickens. Besides supplying energy, dietary oil also could increase the diet palatability, diminish the pulverulence and increase hydrolysis of lipoproteins, contributing to essential FA (Chwen et al. 2013; Nobakht et al. 2011). However, the effects of lipid sources on overall performance in broiler chickens mainly depend on the types, structure and functions of the lipids, the breeder type of broiler chicken and the calorie-protein ratio of the diets (Ahiwe et al. 2018).
Oil palm (Elaeis quineensis Jacq.) fruits are made of almost 80% mesocarp (by weight of the fruit), and approximately 40–45% of this mesocarp presents itself as high-quality edible oil. The oil from mesocarp extracted mechanically is recognised as crude palm oil (CPO) which is a deep orange-coloured liquid due to the high carotenoid content (Kua et al. 2018). The CPO contains several substances considered impurities (Table 1) that must be removed using several processes. CPO is usually refined via a chemical or physical process where it turns into a fine golden-yellow refined oil for further end-use applications. Figure 1 shows the typical flowchart of palm oil processing.
Like other natural oils, palm oil comprises mainly triacylglycerols (TAG) and minimal free fatty acid (FFA) content. Palm oil consists of both saturated fatty acids (SFA) and unsaturated fatty acids (UFA), which might influence poultry production parameters depending on inclusion levels in the diet. SFA of palm oil comprises mainly of palmitic acid (C16:0, 44%), with 4.5% stearic acid (C18:0) and small amounts of myristic acid (C14:0). The SFA contributes to palm oil stability and has potential as natural free radical scavengers (Sundram et al. 2003). The UFA of palm oil mainly consists of monounsaturated fatty acids (MUFA), namely oleic acid (C18:1, 39.3%) and polyunsaturated acid (PUFA), with 10% linoleic acid (C18:2) and a minimal amount of linolenic acid (C18:3) (Sundram et al. 2003). In addition, palm oil also consists of phytonutrients, such as vitamin E (600–1000 ppm), carotenoids (500–700 ppm), phytosterols (300–620 ppm), squalene (250–540 ppm), co-enzyme Q10 (10–80 ppm), polyphenolics (40–70 ppm) and phospholipids (20–100 ppm), which are the main elements though which it exhibits its nutritional and antioxidant properties (Loganathan et al. 2010).
Palm oil is pervasively used in broiler and layer chickens’ feeds in Malaysia for decades (Saminathan et al. 2020). Palm oil is relatively cheap with high ME content that can substitute high-cost conventional feed ingredients to produce cost-effective poultry feed. Most studies revealed that palm oil shows pronounced efficiency in promoting the growth of broiler chickens (Zulkifli et al. 2003; Rahman et al. 2010; Tancharoenrat et al. 2013; Akter et al. 2014; Nooraida and Abidah 2020). Moreover, dietary palm oil could impact the meat cholesterol and serum lipid profile in broiler chickens (Khatun et al. 2017). Palm oil also exhibits oxidative stability due to the presence of prevalent phytonutrient elements (May and Nesaretnam 2014). There is extensive information on the nutritional value, biochemical properties, feeding responses and energy metabolisms in broiler chickens fed with diets containing palm oil, and the prospect for palm oil utilisation in broiler chicken production is enormous. This paper reviews the use of palm oil to improve broiler chicken growth performance, carcass characteristics, meat quality, FA profile and deposition, antioxidant activity and blood and serum profiles.
Effect of dietary palm oil on growth performance of broiler chickens
Broiler chickens fed with low-fat rations showed poor growth performance and feathering and high mortality rates, affecting overall meat production (Nooraida and Abidah 2020). Nevertheless, it has been extensively documented that broiler chickens fed on high-energy rations had improved feed and energy consumption, daily weight gain (DWG) and feed conversion ratio (FCR) (Abdulla et al. 2016; Long et al. 2018; Nooraida and Abidah 2020). To the best of our knowledge, oils have also been extensively used in the diets of broiler chickens reared at high ambient temperature to increase feed energy concentration and promote bird’s growth performance (Mujahid et al. 2009). Furthermore, palm oil could be less expensive in some areas of the world relative to other oils, and its widespread use in broiler chicken feeding could reduce feed costs and improve performance and production (Table 2).
A study conducted by Panja et al. (1995) found that the inclusion of 60 g/kg of palm oil in the broiler diet improved DWG by 12.4% and energy consumption under warm, humid conditions and comparable to broiler chickens fed with soybean oil. Likewise, feeding a diet consisting of 60 g/kg palm oil resulted in significantly higher average daily gain (ADG, 1.87 g/day) and greater duodenal villi height (101 μm) in male broiler chickens (strain Cobb 500) than that fed linseed oil (Abdulla et al. 2016). A recent study by Nooraida and Abidah (2020) showed that supplementing 40 g/kg CPO into the Cobb 500 broiler chickens diet significantly increased weight gain and carcass weight by 0.72 and 0.61 kg, respectively than the control. Another study by Long et al. (2019) reported that 40 g/kg or 60 g/kg palm oil inclusion did not significantly affect ADG but had a lower average daily FI (P = 0.019) and feed-to-gain ratio (P = 0.031) compared to birds fed with a basal diet from day 1 to 42. Previously, Long et al. (2018) showed that Arbor Acres-strain chicks fed 30 g/kg mixed plant oils containing 20% palm oil showed greater ADG (59.5 g/day) than that fed soybean oil alone (54.9 g/day) in an overall trial period (1 to 42 days). The improvement in weight gain could be due to the favourable effects of the counter-balance between SFAs (from 20% palm oil) and PUFAs (from 15% linseed oil) in mixed oils. On the other hand, Nwoche et al. (2003) reported that partial replacement of maize with optimal palm oil inclusion (40 g/kg) resulted in a significant increase in body weight by 8.4% and least cost per unit weight gain when compared to birds fed with that of the control (diet without palm oil addition). In contrast, Khatun et al. (2018) found that the addition of palm oil or soybean oil in broiler chicken rations did not significantly influence birds’ growth performance. Comparable energy efficacy between palm oil and soybean oil rations due to a similar SFA to UFA ratio content (2.05 and 2.15, respectively) could result in similar growth performance effects.
The definite mechanism(s) underlying the growth-promoting effects of palm oil is unclear, but a plausible explanation for significant improvement in broilers’ growth performance is the improvement in palatability of diets, decreased digesta passage rate and presence of high concentrations of palmitic and oleic acid in palm oil that improved oil utilisation and, eventually, enhanced weight gain (Jung et al. 2010). Moreover, it has been well documented that broiler diets with palm oil addition also had high-energy efficacy than diets without the oil inclusion. Therefore, palm oil can be used to partially substitute corn grain as a high-energy grain source and a cost-effective ingredient in the broiler diet without compromising the overall growth performance traits.
Several studies have found that FI of broiler chickens was not affected by palm oil (Jung et al. 2010; Abdulla et al. 2016; Long et al. 2018). As a matter of fact, broiler chickens fed a diet containing palm oil improved the efficiency of feed utilisation (resulting in lower FCR) compared to chickens fed diets with the same caloric amount of linseed oil or soybean oil (Abdulla et al. 2016). Panja et al. (1995) found that broiler chickens fed with 60 g/kg palm oil had a better FCR than birds fed the basal diet on day 28 and thereafter. For chickens, the dietary factor that influences the efficiency of feed utilisation is the passage rate of feed through the digestive system. Abdulla et al. (2016) suggested that a broiler diet supplemented with palm oil could reduce the transit time of digesta in a chicken’s alimentary canal and thus help increase nutrient utilisation and absorption from the diet, which would eventually reduce the FI and result in lower FCR.
Effect of dietary palm oil on heat tolerance in broiler chickens
Commonly, broiler chickens raised in a tropical region have a heat intolerance ability and are more susceptible to heat stress due to increased metabolic heat generated concerning rapid growth rates when heat dissipation capacity does not grow likewise (Rafei-Tari et al. 2021). It is widely accepted that the utilisation of dietary oils as supplementation in a hot weather feeding programme is favourable for broiler chicken production strategy (Zulkifli et al. 2007; Daghir 2009).
The beneficial effects of palm oil in a broiler ration on alleviating the consequences of heat stress have been shown to have a marked reduction in mortality rates (Zulkifli et al. 2003). Based on previous studies (Zulkifli et al. 2003; Htin et al. 2007; Wang et al. 2013), the mortality data suggest that feeding broiler chickens with a diet containing palm oil could enhance the survival rate in extremely high ambient temperature conditions compared to other edible oils. This is because palm oil contains almost 50% SFA, which has marginal effects against high environmental temperature stress resistance. In accordance with this, Wang et al. (2013) found that the growth performance of Arbor Acres broiler chickens under heat stress conditions was enhanced by palm oil addition (40 g/kg in starter diet and 50 g/kg in finisher diet) rather than linseed oil. Htin et al. (2007) found that adding 80 g/kg palm oil to broiler chickens grower diet (21–52 days) improved their growth performance in a heat stress challenged environment (36 ± 1 °C for 2 h/day) by reducing the impact of heat stress when compared to that of the coconut oil and soybean oil. Zulkifli et al. (2003) revealed that increased levels of palm oil in poultry diets could improve the survivability of Cobb 500 chickens exposed to high environmental temperatures. The study showed that broiler rations containing 91.1 g/kg of palm oil reduced mortality of heat stress challenged broiler chickens by as much as 75%. Broiler chickens’ energy metabolisms and body temperatures are closely associated with heat-related stress survivability. Therefore, feeding palm oil-based rations to birds could restrain body temperature rises in response to heat stress that might lead to hyperthermia (Rafei-Tari et al. 2021).
The higher inclusion level of palm oil (80–90 g/kg) in broiler chickens’ diet could resist against high environmental temperatures, but it also led to the meat of an undesirable composition of fatty acid and reduced the palatability of the feed (Zulkifli et al. 2007). On the contrary, edible oil is rich in UFA, which produces the meat of more favourable balanced fatty acid, but UFA aggravates heat stress by inducing higher body temperature and reducing the performance of chickens (Ferrini et al. 2008). It has been suggested that the combination of palm oil and other edible oil rich in UFA such as linseed oil and soybean oil could have a synergistic effect in mitigating heat stress in broiler chickens as well as improving the performance and meat quality. Therefore, further study is needed to determine the combination effects as a better alternative than single oil for broilers exposed to high environmental temperature.
Effect of dietary palm oil on carcass characteristics and meat quality of broiler chickens
Palm oil, CPO or a mixture of palm oil and palm fatty acids distillate (PFAD) are important sources of vegetable oils with differing FA compositions that could be used in broiler chicken feed as an alternative to animal fats without any negative impacts that compromise carcass quality (Rodriguéz et al. 2002). The utilisation of dietary palm oil in chicken diets is fascinating because of its SFA content, which can positively impact meat juiciness and firmness (Smink et al. 2008). However, the high content of SFA and MUFA in palm oil tends to increase the abdominal fat deposit in broiler chickens more than other commercial vegetable oils, mainly due to the low oxidation rate of FA (Ferrini et al. 2008).
Das et al. (2014) reported that carcass yield, breast meat and thigh meat of Hubbard Classic broiler chickens had an increased level from palm oil inclusion up to 30 g/kg. On the other hand, CPO inclusion in broiler diet, regardless of level, gave significantly higher (P < 0.05) carcass weight compared to other oil palm-based fats (PFAD and Malaysian Palm Oil Board-High Energy, MPOB-HIE, a fat-based on PFAD but with emulsifier inclusion), basal and commercial feed (Nooraida and Abidah 2020). Specifically, the highest thigh and drumstick weights were observed from broiler chicken fed diets with 40 g/kg and 60 g/kg CPO, compared with other fats. Meanwhile, a diet containing 80 g/kg CPO corresponds to the highest weight for the breast part. According to the previous research, rapid development in protein accretion in chicken muscle when the level of oil supplementation increases in broiler ration could increase breast and thigh weight of broiler chickens (Das et al. 2014). Moreover, higher dietary protein and energy levels particularly from oil supplementation had little influence on carcass yield or the yield and composition of edible meat (Skřivan et al. 2018).
Valencia et al. (1993) discovered that dressing percentage, breast, drumsticks and abdominal fat weight were not significantly affected by different levels of palm oil addition in male chickens of commercial broiler strain diet; however, an undesirable composition of FAs was observed in adipose tissue by the inclusion of palm oil from day 21 to 42 of the trial. Smink et al. (2010) found that palmitic acid concentration in breast muscle increased by 11.4% in broilers fed with palm oil compared to those fed with sunflower oil. Similarly, Skřivan et al. (2018) found that the concentration of palmitic acid was significantly high in the meat (605 mg/100 g) and abdominal fat (21,544 mg/100 g) of Ross 308 broiler chickens when fed a diet with palm oil. The increasing level of palmitic acid is more pronounced because this acid is highly dense in palm oil and readily absorbed and deposited within the chicken’s muscle. Ura et al. (2008) also found significant improvement in vitamin E concentration in the liver, breast and thigh meat of broiler chickens supplemented with increasing levels of palm oil up to 80 g/kg for 1 to 42 days when compared to the basal diet (without palm oil). Vitamin E has an essential function as a biological antioxidant in reducing meat deterioration and helps to maintain immune function and animal health.
Meat colour is one of the important factors when consumers assess the meat quality trait since they correlate colour to freshness. Ayed et al. (2015) showed that chickens receiving palm oil supplemented diet traduce a slight loss of the red colour (a*) and an increase in yellow colour (b*) of muscle after 6 days of storage when compared chickens fed with basal diet (without palm oil). The increase in yellowness is might due to the deposition of carotene from palm oil into the chicken muscle. However, Khatun et al. (2018) and Zaki et al. (2018) found that meat colour did not change with 6% palm oil or soybean oil dietary supplementation. Effect of feeding broiler chicken on different types of vegetable oils showed significant differences in pH values of chicken meat. Several studies revealed that chickens fed with palm oil produced breast meat with a pH ranging from 5.78 to 5.99 (Khatun et al. 2018; Long et al. 2018; Zaki et al. 2018). These values are within the ranges of normal pH reported for chicken breast meat.
Although there are some reports describing the effects of palm oil on meat quality (Smink et al. 2010; Skřivan et al. 2018), little research has been done to study the impact of palm oil on drip and cooking loss in chicken meat and meat products. Data of cooking loss (%) and drip loss (%) of broiler chicken meat fed on different types of oils showed that no significant differences were found between broiler fed on palm oil and soybean oil (Zaki et al. 2018; Khatun et al. 2018). However, Panja et al. (1995) found that dietary palm oil had a significant effect on the proximate analysis of broiler chicken meat. Accordingly, crude fat in the carcass was increased from 32.03 to 38.28% with increasing palm oil (0 to 8%) in broiler diets, but the opposite trends were found for crude protein and ash content. The decline in crude protein and ash is likely due to the substantially higher rate of fat gain during the feeding period. Fat, protein and ash are the basic body constituents making up most of the carcass dry matter content. Similarly, Ayed et al. (2015) observed that broiler carcass fat content was higher in the palm oil treatment group compared to the basal diet. The increase in fat content also indicated that the amount of energy consumed by the chicken was excessive and concurrently produced higher abdominal fat deposits in birds. Fat deposition is crucial in modern broiler chicken production schemes because it is associated with selection for increased body weight in broilers.
Effect of dietary palm oil on gene expression in broiler chickens
Few studies have addressed the effects of palm oil on gene expression profiling in broiler chickens related to mRNA expression, oxidation activity, lipid metabolism and heat stress modulation. Basically, modulation in the gene expression is strongly associated with mediatory FA metabolism effects in the cell, either by directly or indirectly altering signalling pathways via a nuclear hormone receptor, such as peroxisome proliferator-activated receptors (PPARs) genes (Sato et al. 2004). A wide range of FA derivatives triggers these PPARs’ gene expression activities at the cellular level, and therefore, they are called lipid sensors. Furthermore, the effects of various dietary oil on the regulation of transcription and gene expression have been defined in poultry. Nonetheless, underlying alterations to the anti-inflammatory and immune response that might contribute to changes in downstream performance are not as well described.
Scientific evaluation of the relationship between mRNA regulation level and biological parameters can assess the functionality of gene expression data obtained from a study. Royan et al. (2011) discovered signs of a strong relationship between the adipose PPARγ gene upregulation that occurred in the abdominal fat deposition and adipose tissue of broiler chickens fed with a diet supplemented with palm oil compared to that of the soybean oil. The finding revealed that palm oil could be safely incorporated into the diets of broiler chickens to enrich chicken meat with potential health-beneficial FA. Skřivan et al. (2018) demonstrated that the higher mRNA expression of hepatic FA synthase and desaturases (Δ5-, Δ6- and Δ9-) and mitochondrial enzyme carnitine palmitoyltransferase (CPT) were detected in Ross 308 strain chickens fed with palm oil compared to that of the chickens fed with rapeseed oil. However, the expression of HMG-CoA (rate-limiting enzyme of cholesterol synthesis) reductase was increased in broiler chickens fed palm oil compared to those fed diets containing rapeseed oil or lard (Skřivan et al. 2018). Smink et al. (2010) found that mRNA expression of CPT, which is a key gene in control mitochondrial, peroxisomal fatty acid oxidation and beta-oxidation of fatty acids, as well as the enzyme activity of CPT hepatic activities, was comparable in broiler chickens fed diets supplemented with palm oil or sunflower oil.
Recently, Khatun et al. (2020) found that the combination of palm oil and sunflower oil with l-arginine and vitamin E altered the expression of cytokine genes by reducing the expression of NF-α, whereas TGF-ß1 was upregulated, which could positively affect the immune response of the chickens. Although several studies indicated that the inclusion of palm oil in broiler chicken rations could influence cytokines expression and modulate immunological function, further research is required to better understand the impact of palm oil on gene expression under stress conditions and specific disease challenges.
Effect of dietary palm oil on blood and serum profiles in broiler chickens
The FA composition of depot fat or edible poultry tissues reflects that of dietary supplementation of the fats or oils in complete rations (Rymer and Givens 2005; Khatun et al. 2017). Likewise, oil supplementation in the poultry diet can modulate the lipoprotein and triglycerides (TG) levels in the serum (Wongsuthavas et al. 2007; Zhu et al. 2014). The altering effect of various types of oil supplementation on serum lipoproteins and TG concentrations has been widely discussed (Viveros et al. 2009; Velasco et al. 2010; Aghaei et al. 2012). However, the actual influencing mechanisms behind the reported effect of dietary oil supplementation on serum lipid alteration have not been completely understood. Moreover, edible oil in the poultry diet can influence serum lipid profile and meat cholesterol level, which corresponds to the type of oil consumed.
Generally, dietary oils containing a significantly higher level of SFA tend to increase blood TG concentration and decrease the unsaturated FA/SFA ratio of deposition fat in broiler chickens’ meat (Milićević et al. 2014). Palm oil is high in SFAs, and feeding broiler chickens a diet high in palm oil may reduce ß-oxidation rate by concurrently decreasing TG absorption from the blood to adipose tissues, which increases blood TG. In accordance with this statement, Velasco et al. (2010) demonstrated that feeding broiler chickens a diet supplemented with palm oil had a higher blood TG concentration compared to those fed a diet supplemented with sunflower oil. In addition, chickens fed with palm oil had significantly (P < 0.05) higher serum TG levels and significantly (P < 0.05) decrease low-density lipoprotein concentrations than chickens that consumed soybean oil or a mixture of dietary palm oil and soybean oil (Gheisari et al. 2017). Triglycerides are important for health, but high levels can raise blood cholesterol levels and increase the risk of heart disease in broiler chickens.
Other advantages of oil addition into the broiler diet are improved absorption of fat-soluble vitamins and calcium (Leeson and Atteh 1995). Palm oil is rich in vitamin E, with the majority present as tocotrienols with a minor tocopherols component that can be easily absorbed into the animal bloodstream (Nagendran et al. 2000). The combination effects of tocotrienols with other palm oil phytonutrients, such as tocopherols, polyphenols and carotenes, have been shown to have a synergistic effect on protection against serum peroxidation and reducing oxidative stress (Ali and Woodman 2015). Long et al. (2019) reported that dietary inclusion of 20–40 g/kg palm oil treatments significantly increased glutathione peroxide activity and reduced serum lipid peroxidation (malondialdehyde (MDA) concentration) of broiler chickens when compared to the diet without palm oil. Furthermore, supplementation of 60 g/kg palm oil in broiler diet significantly decreased total antioxidant capacity (T-AOC) (p = 0.027) and catalase activity (P = 0.018) in serum samples as compared to chickens fed with basal diet, which prevent tissue or cells damage (Long et al. 2019). However, the authors concluded that no significant differences (P > 0.05) were observed in broiler serum MDA and T-AOC levels among the dietary treatments supplemented with palm oil (0–60 g/kg) at 42 days of age, which could suggest that the inclusion of palm oil had less effect on the antioxidant status of serum in female Sanhuang chickens.
Sudharsan et al. (2020) demonstrated that a combination of rapeseed oil and palm oil in the broiler chicken diet significantly decreased the serum lipid concentration, whereas Khatun et al. (2018) revealed that feeding a ration containing palm oil could alter the blood lipid profiles without compromising the meat quality in broiler chickens. However, detrimental effects of increases in total plasma creatine kinase activity have been addressed in poultry production in response to exposure and transportation conditions (Mitchell and Sandercock 1995). In earlier work, Zulkifli et al. (2007) revealed that broiler chickens of the Cobb strain fed a diet consisting of 80 g/kg palm oil under heat stress conditions resulted in a significant (P < 0.05) reduction in serum creatine kinase concentration, which showed mild damage of individual muscle that causes minimal membrane permeability and consequent leakage of enzymes and other markers into the bloodstream. However, we did not find any information in the literature on the effect of palm oil supplementation on serum glutathione peroxidase GPxA and lipid peroxidation activity, which play an essential biological role in protecting chickens from oxidative damage.
Modulation effects of palm oil supplement in broiler diet on the anti-inflammatory cytokines interleukin-10 (IL-10) and transforming growth factor-β (TGF-β), as well as on pro-inflammatory cytokine IL-1β, IL-6 and tumour necrosis factor-α (TNF-α), in the serum have been documented. A study by Long et al. (2019) revealed that the inclusion of palm oil (40 g/kg and 60 g/kg) in the Sanhuang broiler chicken diet modulates the balance between pro-and anti-inflammatory cytokines in serum by significantly increasing IL-1β content but improves TGF-β and IL-10 contents in both starter and grower periods when compared to basal diet treatment. Moreover, the addition of 60 g/kg palm oil in the broiler diet significantly reduced TNF-α content on days 21 and 42 compared with the basal diet (without palm oil). These results suggest that dietary inclusion of palm oil could modulate anti-inflammatory cytokines (a series of immunoregulatory molecules) and shows the limited effect on antioxidative status in broiler chickens.
Effect of dietary palm oil on fatty acids profile and fat deposition in broiler chickens
Nutritional benefits of chicken meat are mainly correlated to its fat content and FA composition (Rodriguez-Sanchez et al. 2019). The FA composition in monogastric animal muscle or fat deposition may be significantly influenced by the diet administered (Abdulla et al. 2015). Fatty acid digestibility in the gastrointestinal tract and lipid composition in chicken meat is influenced by the type of dietary fat or lipid source in broiler rations and the age of the chickens (Peña-Saldarriaga et al. 2020). The stored fats in meat are mainly SFA and MUFA. The FA properties of broiler chickens’ abdominal fat could also be modulated by various sources and levels of oil or fat supplementations. For instance, SFAs are often readily tolerant of being absorbed and deposited in abdominal fat or body tissues, while PUFAs are susceptible to oxidation to generate energy (Baião and Lara 2005). Moreover, lack of enzyme activity Δ12 and Δ15 desaturase can cause chickens to be unable to synthesize essential FA (Head et al. 2019). Thus, dietary intake of oils is needed as a precursor for important FA.
Extreme amounts of n-6 PUFA concentration and a concurrently high level of n-6/n-3 ratio in a diet can prompt the pathogenesis of many diseases in humans, including inflammatory diseases, autoimmune diseases and cardiovascular diseases; in contrast, a lower n-6/n-3 ratio (increased level of n-3 PUFA) exerts suppressive effects. Zaki et al. (2018) demonstrated that broiler chickens fed with palm oil had an increased total n-3 and lower n-6/n-3 ratio when compared to broiler chickens fed with soybean oil. In another study, broiler chickens fed starter or grower diets supplemented with palm oil produced healthy meat enriched with n-3 PUFA concentration and a lower n-6/n-3 FA ratio (López-Ferrer et al. 2001). Therefore, palm oil could be used as a potential energy source in broiler chicken feeding with beneficial effects on meat quality and human health. In another study, increasing levels of palm oil in broiler rations resulted in improving the SFA deposition efficacy and a reduced level of PUFA, which could promote modulatory effects on chicken adipose tissue hardness, thus proving to be beneficial in a marketing strategy (Valencia et al. 1993). Consistent with other studies, Nyquist et al. (2013) reported that substitution of animal fat with palm oil showed positive effects on the nutritional value of broiler chicken muscle with regard to FA proportions.
Nonetheless, several studies showed that the inclusion of dietary oil rich in SFA in broiler chickens’ rations resulted in efficient FA deposition in broiler chickens when compared to that of those fed rations rich in PUFA (Crespo and Esteve-Garcia 2001, 2002; Long et al. 2018). Abdulla et al. (2015) reported that palmitic acid (C16:0) concentration increased in meat from broiler-fed palm oil compared to those fed diets supplemented with linseed oil and soybean oil. The higher content of palmitic acid (C16:0) in broiler chickens fed on palm oil may be due to palm oil’s high palmitic acid content. Furthermore, Zhong et al. (2014) reported that palm oil is more stable than linseed oil, increasing the fats’ oxidative stability and maintaining an apposite SFA balance in chicken meat. However, Wang et al. (2013) revealed that the combination of palm oil and linseed oil in broiler rations could maintain the balance between the SFA and PUFA proportion and enhance the n-3 PUFA content in the meat of Arbor Acres broiler chickens. These findings suggest that high levels of α-linolenic acid (C18:3n-3) in mixed oils (palm oil and linseed oil) may act as a precursor for long-chain n-3 PUFA such as C20:5n-3 and C22:6n-3 so that more deposition of n-3 PUFA occurs in the muscle of broiler chickens (Viveros et al. 2009).
Effect of dietary palm oil on antioxidant activity in broiler chickens
Antioxidant properties of palm oil are another interesting subject in poultry production, as they can prevent lipid peroxidation in the muscles of broiler chicken meat (Rama Rao et al. 2011). Previous studies have attempted to systematise edible vegetable oil antioxidant compounds’ characteristics and their effects, as summarised in Table 3. Even though phytonutrients are present in a minimal amount (< 1%) in palm oil, they nonetheless have acted as a natural antioxidant agent, protecting the poultry and its products from oxidative damage (May and Nesaretnam 2014; Dian et al. 2017). Moreover, several studies have reported the oxidative stability effect of carotenes and α-tocopherol in palm oil on meat quality (Ura et al. 2008; Nyquist et al. 2013).
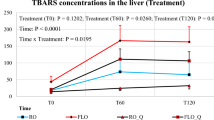
Lipid peroxidation, a dynamic chain reaction of free radicals, includes oxidative stress triggered by thermal processing in the presence of oxygen. The high content of PUFAs in broiler diets can improve the quality of meat, but they are susceptible to lipid damage that can cause oxidative damage, which later can reduce the quality of meat and harm the consumer (Attia et al. 2020). This lipid oxidation reaction rate is associated with the duration and intensity of thermal processing. Contrarily, SFA is stable against lipid oxidation. Adeyemi et al. (2015) reported that the dietary oil blend of a balanced SFA and UFA, such as palm oil, in broiler chickens could be an effective strategy to enhance the product quality and minimise oxidative spoilage. A study by Akter (2015) also found that supplementation of 30–40 g/kg CPO in the broiler chickens’ rations could be effective for enhancing oxidative stability and meat quality and reduced (P < 0.05) the lipid peroxidation (TBARS) values in the liver sample.
The beneficial effects of oil palm tocotrienols have been widely studied for their nutritional and health properties, including antioxidant activities, anti-cancer effects and cholesterol-lowering mechanisms (Shahidi and de Camargo 2016). Several studies have shown that ingestion of palm tocotrienols resulted in enhanced antioxidant activity by inhibiting chicken protein oxidation and lipid peroxidation compared with α-tocopherols (other isomers of vitamin E in palm oil) (Kamat et al. 1997; Seppanen et al. 2010). Thus, the incorporation of palm oil into poultry diets provides a natural dietary source of vitamin E that is absorbed in the chicken’s intestine to perform its functions, improving the body’s total antioxidative status.
The inclusion of palm oil in poultry diets leads to increased tocotrienol and tocopherol contents in meat, liver and adipose tissues (Kang et al. 1998). It has been suggested that feeding chickens with a diet that contains palm oil resulted in a more excellent oxidative stability in broiler meat, which is caused by the higher levels of palm vitamin E (potential antioxidative agents) rather than by the lower PUFA content of palm oil (Rebolé et al. 2006). Long et al. (2019) found that incorporation of 20 g/kg or 40 g/kg palm oil in chicken diets decreased MDA, an oxidative damage indicator, and catalase activity, which is the antioxidant indicator, and increased total antioxidant capacity (T-AOC) in chicken meat when compared to the diet without palm oil inclusion at 21 days of age.
Conclusions and future recommendations
The rising price of energy-rich feedstuff has prompted a search for alternative energy sources in the poultry diet. Replacement of conventional feed ingredients such as grain corn in the broiler chickens diet with palm oil was economically beneficial in contributing significantly to high ME and improving growth performance and biochemical properties of tissues and blood. There is strong evidence that palm oil has a positive effect on the production performance of broiler chickens, which is reflected in enhanced BWG and improved FCR and meat quality. Palm oil also exhibits oxidative stability in chickens due to the presence of prevalent phytonutrient elements. Moreover, palm oil in broiler diets was found to have a stronger impact on mRNA expression involved in lipid metabolism and modulate the balance between pro- and anti-inflammatory cytokines. Therefore, palm oil has great potential for use as an ingredient in broiler chicken production in oil palm-producing countries due to its contribution to developing a cost-effective feeding system and, ultimately, reducing the production cost without affecting their growth performance.
The utilisation of palm oil in the broiler chickens’ diet should be strengthened to expand its role in increasing the productivity of high-quality meat and improving other potential health benefits on a commercial scale. Although palm oil can enhance the survival rate of broiler chickens in extremely high ambient temperature conditions, it is recommended to consider analysing changes in different environments keeping in mind the expected environmental changes (in future), including increased temperatures. In addition, the authors concluded that there is a lack of studies on the impact of palm oil on the gastrointestinal tract microbiota for better growth and improved health of poultry. Therefore, future studies should be conducted to evaluate the efficacy of dietary palm oil in mitigating the colonisation of pathogenic microorganisms in poultry gut systems using a molecular approach.
Data availability
Not applicable.
Code availability
Not applicable.
References
Abdulla, N.R., Loh, T.C., Akit, H., Sazili, A.Q., and Foo, H.L., 2016. Effects of dietary oil sources and calcium: phosphorus levels on growth performance, gut morphology and apparent digestibility of broiler chickens. South African Journal of Animal Science, 46, 42-53.
Abdulla, N.R., Loh, T.C., Akit, H., Sazili, AQ., Foo, HL., Mohamad, R., Rahim, R.A., Ebrahimi, M., and Sabow, A.B., 2015. Fatty acid profile, cholesterol and oxidative status in broiler chicken breast muscle fed different dietary oil sources and calcium levels. South African Journal of Animal Science, 45, 153-163.
Adeyemi, K.D., Sabow, A.B., Shittu, R.M., Karim, R., and Sazili, A.Q., 2015. Influence of dietary canola oil and palm oil blend and refrigerated storage on fatty acids, myofibrillar proteins, chemical composition, antioxidant profile and quality attributes of semimembranosus muscle in goats. Journal of Animal Science and Biotechnology, 6, 1-13.
Aghaei, N., Safamehr, A., Mehmannavaz, Y., and Chekaniazar, S., 2012. Blood and tissue fatty acid compositions, lipoprotein levels, performance and meat flavor of broilers fed fish oil: changes in the pre-and post-withdrawal design. Animal, 6, 2031-2040.
Ahiwe, E.U., Omede, A.A., Abdallh, M.B., and Iji, P.A., 2018. Managing dietary energy intake by broiler chickens to reduce production costs and improve product quality. In: Yucel B, Taskin T, editors. Animal husbandry and nutrition. London: IntechOpen; p. 115–145.
Akter, Y., 2015. Effects of carotenes and alpha-tocopherol in crude palm oil layer and broiler performance. Ph.D thesis, Universiti Putra Malaysia.
Akter, Y., Kasim, A., Omar, H., and Sazili, A.Q., 2014. Effect of storage time and temperature on the quality characteristics of chicken eggs. Journal of Food, Agriculture and Environment, 12, 87-92.
Ali, S.F., and Woodman, O.L., 2015. Tocotrienol rich palm oil extract is more effective than pure tocotrienols at improving endothelium-dependent relaxation in the presence of oxidative stress. Oxidative Medicine and Cellular Longevity, 2015, Article 150829.
Anigbogu, N.M., 2001. Performance efficiency of broiler chicks fed diets with different levels of palm oil and tannia tuber meal. Journal of Applied Chemistry and Agricultural Research, 7, 1-16.
Attia, Y.A., Al-Harthi, M.A., and El-Maaty, H.M.A., 2020. The effects of different oil sources on performance, digestive enzymes, carcass traits, biochemical, immunological, antioxidant, and morphometric responses of broiler chicks. Frontiers in Veterinary Science, 7, 181.
Ayed, H.B., Attia, H., and Ennouri, M., 2015. Effect of oil supplemented diet on growth performance and meat quality of broiler chickens. Advanced Techniques in Biology & Medicine, 4, 1–4.
Baião, N.C., and Lara, L.J.C., 2005. Oil and fat in broiler nutrition. Brazilian Journal of Poultry Science, 7, 129-141.
Chwen, L.T., Foo, H.L., Thanh, N.T., and Choe, D., 2013. Growth performance, plasma fatty acids, villous height and crypt depth of preweaning piglets fed with medium chain triacylglycerol. Asian-Australasian Journal of Animal Sciences, 26, 700–704.
Crespo, N., and Esteve-Garcia, E., 2001. Dietary fatty acid profile modifies abdominal fat deposition in broiler chickens. Poultry Science, 80, 71-78.
Daghir, N.J., 2009. Nutritional strategies to reduce heat stress in broilers and broiler breeders. Lohmann Information, 44, 6-15.
Das, G.B., Hossain, M.E., and Akbar, M.A., 2014. Performance of broiler with feeding different levels of palm oil. Bangladesh Journal of Animal Science, 43, 45-51.
DVS., 2019. Department of Veterinary Services: Ministry of Agriculture, Malaysia. End of Year Statistics and Report. Kuala Lumpur, Malaysia, 2020. http://www.dvs.gov.my/index.php/pages/view/3338?mid=42, accessed on 13 Feb 2021.
Ferrini, G., Baucells, M.D., Esteve-Garcia, E., and Barroeta, A.C., 2008. Dietary polyunsaturated fat reduces skin fat as well as abdominal fat in broiler chickens. Poultry Science, 87, 528-535.
Gheisari, A., Fosoul, S.A.S., Pourali, S., Esfahani, E.N., and Mohammadrezaei, M., 2017. Blood lipid metabolites and meat lipid peroxidation responses of broiler chickens to dietary lecithinized palm oil. South African Journal of Animal Science, 47, 526-534.
Goh, S.H., Khor, H.T., and Gee, P.T., 1982. Phospholipids of palm oil (Elaeis guineensis). Journal of the American Oil Chemists’ Society, 59, 296-299.
Goh, S.H., Tong, S.L., and Gee, P.T., 1984. Total phospholipids in crude palm oil: quantitative analysis and correlations with oil quality parameters. Journal of the American Oil Chemists' Society, 61, 1597-1600.
Gupta, A., Sharma, R., Sharma, S., and Singh, B., 2019. Oilseed as potential functional food Ingredient. Trends & Prospects in Food Technology, Processing and Preservation, 1st ed.; Prodyut Kumar, P., Mahawar, MK, Abobatta, W., Panja, P., Eds, 191–215.
Head, B., Bionaz, M., and Cherian, G., 2019. Flaxseed and carbohydrase enzyme supplementation alters hepatic n-3 polyunsaturated fatty acid molecular species and expression of genes associated with lipid metabolism in broiler chickens. Veterinary Science, 6, 25.
Htin, N.N., Zulkifli, I., Alimon, A.R., Loh, T.C., and Hair-Bejo, M., 2007. Effects of sources of dietary fat on broiler chickens exposed to transient high temperature stress. Archiv fur Geflugelkunde, 71, 74-80.
Jacobsberg, B., 1974. Palm oil characteristics and quality. In Proceedings of the 1st Mardi Workshop on Oil Palm Technology (pp. 48–68). Kuala Lumpur: Malaysian Agriculture Research and Development Institute (MARDI).
Jalali, S.M.A., Rabiei, R., and Kheiri, F., 2015. Effects of dietary soybean and sunflower oils with and without L-carnitine supplementation on growth performance and blood biochemical parameters of broiler chicks. Archives Animal Breeding, 58, 387-394.
Jung, S., Choe, J.H., Kim, B., Yun, H., Kruk, Z.A., and Jo, C., 2010. Effect of dietary mixture of gallic acid and linoleic acid on antioxidative potential and quality of breast meat from broilers. Meat Science, 86, 520-526.
Kamat, J.P., Sarma, H.D., Devasagayam, T.P., Nesaretnam, K., and Basiron, Y., 1997. Tocotrienols from palm oil as effective inhibitors of protein oxidation and lipid peroxidation in rat liver microsomes. Molecular and Cellular Biochemistry, 170, 131–137.
Kang, K.R., Cherian, G., and Sim, J.S., 1998. Tocopherols, retinol and carotenes in chicken egg and tissues as influenced by dietary palm oil. Journal of Food Science, 63, 592-596.
Khatun, J., Loh, T.C., Akit, H., Foo, H.L., and Mohamad, R., 2018. Influence of different sources of oil on performance, meat quality, gut morphology, ileal digestibility and serum lipid profile in broilers. Journal of Applied Animal Research, 46, 479-485.
Khatun, J., Loh, T.C., Akit, H., Foo, H.L., and Mohamad, R., 2017. Fatty acid composition, fat deposition, lipogenic gene expression and performance of broiler fed diet supplemented with different sources of oil. Animal Science Journal, 88, 1406-1413.
Khatun, J., Loh, T.C., Foo, H.L., Akit, H., and Khan, K.I., 2020. Growth performance, cytokine expression, and immune responses of broiler chickens fed a dietary palm oil and sunflower oil blend supplemented with L-Arginine and varying concentrations of vitamin E. Frontiers in Veterinary Science, 7, 1-13.
Kua, Y.L., Gan, S., Morris, A., and Ng, H.K., 2018. Simultaneous recovery of carotenes and tocols from crude palm olein using ethyl lactate and ethanol. In Journal of Physics: Conference Series (Vol. 989, No. 1, p. 012005). IOP Publishing.
Leeson, S., and Atteh J. O., 1995. Utilization of fats and fatty acids by turkey poults. Poultry Science, 74, 2003-2010.
Lin, S.W., 2011. 2 Palm Oil. Vegetable oils in food technology: composition, properties and uses, 25.
Loganathan, R., Selvaduray, K.R., Nesaretnam, K., and Radhakrishnan, A.K., 2010. Health promoting effects of phytonutrients found in palm oil. The Malaysian Journal of Nutrition, 16, 309-322.
Long, G.L., Hao, W.X., Bao, L.F., Li, J.H., Zhang, Y., and Li, G.H., 2019. Effects of dietary inclusion levels of palm oil on growth performance, antioxidative status and serum cytokines of broiler chickens. Journal of Animal Physiology and Animal Nutrition, 103, 1116-1124.
Long, S., Xu, Y., Wang, C., Li, C., Liu, D., and Piao, X., 2018. Effects of dietary supplementation with a combination of plant oils on performance, meat quality and fatty acid deposition of broilers. Asian-Australasian Journal of Animal Sciences, 31, 1773-1780.
López-Ferrer, S., Baucells, M.D., Barroeta, A.C., Galobart, J., and Grashorn, M., 2001. n-3 enrichment of chicken meat. 2. Use of precursors of long-chain polyunsaturated fatty acids: linseed oil. Poultry Science, 80, 753-761.
May, C.Y., and Nesaretnam, K., 2014. Research advancements in palm oil nutrition. The European Journal of Lipid Science and Technology, 116, 1301-1315.
Milićević, D., Vranić, D., Mašić, Z., Parunović, N., Trbović, D., Nedeljković-Trailović, J., and Petrović, Z., 2014. The role of total fats, saturated/unsaturated fatty acids and cholesterol content in chicken meat as cardiovascular risk factors. Lipids in Health and Disease, 13, 1-12.
Mitchell, M.A., and Sandercock, D.A., 1995. Creatine kinase isoenzyme profiles in the plasma of the domestic fowl (Gallus domesticus): Effects of acute heat stress. Research in Veterinary Science, 59, 30–34.
Mujahid, A., Akiba, Y., and Toyomizu, M., 2009. Progressive changes in the physiological responses of heat-stressed broiler chickens. The Journal of Poultry Science, 46, 163-167.
Nagendran, B., Unnithan, U.R., Choo, Y.M., and Sundram, K., 2000. Characteristics of red palm oil, a carotene-and vitamin E–rich refined oil for food uses. Food and Nutrition Bulletin, 21, 189-194.
Nooraida, W.W., and Abidah, M.N., 2020. Effects of pellet supplemented with different percentages of oil palm lipid sources on broiler performance, carcass trait and feed quality. Journal of Oil Palm Research, 32, 313-325.
Noor Lida H.M.D, Hamid, R.A., Kanagaratnam, S., Isa, W.A., Hassim, N.A.M., Ismail, N.H., Omar, Z., and Sahri, M.M., 2017. Palm oil and palm kernel oil: Versatile ingredients for food applications. Journal of Oil Palm Research, 29, 487-511.
Nwoche, G.N., Ndubuisi, E.C., and Iheukwumere, F.C., 2003. Effects of dietary palm oil on the performance of broiler chicks. International Journal of Agriculture and Rural Development, 4, 81-86.
Nyquist, N.F., Rødbotten, R., Thomassen, M., and Haug, A., 2013. Chicken meat nutritional value when feeding red palm oil, palm oil or rendered animal fat in combinations with linseed oil, rapeseed oil and two levels of selenium. Lipids in Health and Disease, 12, 1-13.
Panja, P., Kassim, H., and Jalaludin, S., 1995. Effects of palm oil and soybean oil as fat sources in isonitrogenous and isocaloric diets on the performance of broilers. Asian-Australasian Journal of Animal Sciences, 8, 223-229.
Peña-Saldarriaga, L M., Fernández-López, J., and Pérez-Alvarez, J A., 2020. Quality of chicken fat by-products: Lipid profile and colour properties. Foods, 9, 1046.
Rafei-Tari, A., Sadeghi, A.A., and Mousavi, S.N., 2021. Inclusion of vegetable oils in diets of broiler chicken raised in hot weather and effects on antioxidant capacity, lipid components in the blood and immune responses. Acta Scientiarum. Animal Sciences, 43, e50587-e50587.
Rahman, M.S., Akbar, M.A., Islam, K.M.S., Iqbal, A., and Assaduzzaman, M., 2010. Effect of dietary inclusion of palm oil on feed consumption, growth performance and profitability of broiler. Bangladesh Journal of Animal Science, 39, 176-182.
Rama Rao, S.V., Raju, M.V.L.N., Panda, A.K., Poonam, N.S., and Shyam Sunder, G., 2011. Effect of dietary α-tocopherol concentration on performance and some immune responses in broiler chickens fed on diets containing oils from different sources. British Poultry Sciences, 52, 97-105.
Rebolé, A., Rodriguez, M.L., Ortiz, L.T., Alzueta, C., Centeno, C., Viveros, A., Brenes, A., and Arija, I., 2006. Effect of dietary high-oleic acid sunflower seed, palm oil and vitamin E supplementation on broiler performance, fatty acid composition and oxidation susceptibility of meat. British Poultry Science, 47, 581-591.
Rodriguéz, M.A., Crespo, N.P., Medel, P., Creus, E., and Cortés, M., 2002. Efecto del tipo de grasa de la dieta en la alimentacion del broiler, con enfasis en los productos derivados del aceite de palma. Selecciones avícolas, 44, 693-702.
Rodriguez-Sanchez, R., Tres, A., Sala, R., Garcés-Narro, C., Guardiola, F., Gasa, J., and Barroeta, A.C., 2019. Effects of dietary free fatty-acid content and saturation degree on lipid-class composition and fatty-acid digestibility along the gastrointestinal tract in broiler starter chickens. Poultry Science, 98, 4929-4941.
Royan, M., Meng, G.Y., Othman, F., Sazili, A.Q., and Navidshad, B., 2011. Effects of conjugated linoleic acid, fish oil and soybean oil on PPARs (α & γ) mRNA expression in broiler chickens and their relation to body fat deposits. International Journal of Molecular Sciences, 12, 8581-8595.
Rymer, C., and Givens, D.J., 2005. n-3 fatty acid enrichment of edible tissue of poultry: A review. Lipids, 40, 121-130.
Saminathan, M., Mohamed, W.N.W., Noh, A., Ibrahim, N.A., Fuat, M.A., Noor Lida, H.M.D, and Ramiah, S.K., 2020. Potential of feeding crude palm oil and co-products of palm oil miling on laying hen’s performance and egg quality: A review. Journal of Oil Palm Research, 32, 547-558.
Sato, K., Fukao, K., Seki, Y., and Akiba, Y., 2004. Expression of the chicken peroxisome proliferator activated receptor-γgene is influenced by aging, nutrition, and agonist administration. Poultry Science, 83, 1342-1347.
Seppanen, C.M., Song, Q., and Saari Csallany, A., 2010. The antioxidant functions of tocopherol and tocotrienol homologues in oils, fats, and food systems. Journal of the American Oil Chemists’ Society, 87, 469-481.
Shahidi, F., and De Camargo, A.C., 2016. Tocopherols and tocotrienols in common and emerging dietary sources: Occurrence, applications, and health benefits. International Journal of Molecular Sciences, 17, 1745.
Skřivan, M., Marounek, M., Englmaierová, M., Čermák, L., Vlčková, J., and Skřivanová, E., 2018. Effect of dietary fat type on intestinal digestibility of fatty acids, fatty acid profiles of breast meat and abdominal fat, and mRNA expression of lipid-related genes in broiler chickens. PLoS ONE, 13, e0196035.
Smink, W., Gerrits, W.J.J., Hovenier, R., Geelen, M.J.H., Lobee, H.W.J., Verstegen, M.W.A., and Beynen, A.C., 2008. Fatty acid digestion and deposition in broiler chickens fed diets containing either native or randomized palm oil. Poultry Science, 87, 506-513.
Smink, W., Gerrits, W.J.J., Hovenier, R., Geelen, M.J.H., Verstegen, M.W.A., and Beynen, A.C., 2010. Effect of dietary fat sources on fatty acid deposition and lipid metabolism in broiler chickens. Poultry Science, 89, 2432-2440.
Sudharsan, C., Murugan, S.S., Chacko, B., and Juliet, S., 2020. Influence of dietary substitution of palm oil by rapeseed oil at different levels on growth performance and economics of broilers. Indian Journal of Animal Research, 55, 445-450.
Sundram, K., Sambanthamurthi, R., and Tan, Y.A., 2003. Palm fruit chemistry and nutrition. Asia Pacific Journal of Clinical Nutrition, 12, 355-362.
Tancharoenrat, P., Ravindran, V., Zaefarian, F., and Ravindran, G., 2013. Influence of age on the apparent metabolisable energy and total tract apparent fat digestibility of different fat sources for broiler chickens. Animal Feed Science and Technology, 186, 186-192.
Ura, B., Taharnklaew, R., and Kijparkorn, S., 2008. The effects of Vitamin E in crude palm oil on growth performance, lipid peroxidation and tissue vitamin e concentration in broilers. In : Proceedings of the 7th Chulalongkorn University Veterinary Annual Conference; May 1, 2008.
Valencia, M.E., Watkins, S.E., Waldroup, A.L., and Waldroup, P.W., 1993. Utilization of crude and refined palm and palm kernel oils in broiler diets. Poultry Science, 72, 2200-2215.
Velasco, S., Ortiz, L.T., Alzueta, C., Rebole, A., Trevino, J., and Rodriguez, M.L., 2010. Effect of inulin supplementation and dietary fat source on performance, blood serum metabolites, liver lipids, abdominal fat deposition, and tissue fatty acid composition in broiler chickens. Poultry Science, 89, 1651-1662z
Villanueva-Lopez, D.A., Infante-Rodríguez, F., Nájera-Pedraza, O.G., Barrios-García, H.B and Salinas-Chavira, J., 2020. Effect of dietary frying fat, vegetable oil and calcium soaps of palm oil on the productive behavior and carcass yield of broiler chickens. Brazilian Journal of Poultry Science, 22, 1-8.
Viveros, A., Ortiz, L.T., Rodríguez, M.L., Rebolé, A., Alzueta, C., Arija, I., Centeno, C., and Brenes, A., 2009. Interaction of dietary high-oleic-acid sunflower hulls and different fat sources in broiler chickens. Poultry Science, 88, 141-151.
Wang, J., Zhu, Q., Hussain, A., Zhang, X., and Wang, T., 2013. Combination of linseed and palm oils is a better alternative than single oil for broilers exposed to high environmental temperature. Journal of Poultry Science, 50, 332-339.
Wongsuthavas, S., Yuangklang, C., Vasupen, K., Mitchaothai, J., Srenanual, P., Wittayakun, S., and Beynen, A.C., 2007. Assessment of de-novo fatty acid synthesis in broiler chickens fed diets containing different mixtures of beef tallow and soybean oil. International Journal of Poultry Science, 6, 800-806.
Wu, H., Gong, L.M., Guo, L., Zhang, L.Y., and Li, J.T., 2011. Effects of the free fatty acid content in yellow grease on performance, carcass characteristics, and serum lipids in broilers. Poultry Science, 90, 1992-1998.
Zaki, E.F., El Faham, A.I., and Mohmed, N.G., 2018. Fatty acids profile and quality characteristics of broiler chicken meat fed different dietary oil sources with some additives. International Journal of Health, Animal Science and Food Safety’s, 5, 40-50.
Zhong, X., Gao, S., Wang, J.J., Dong, L., Huang, J., Zhang, L.L., and Wang, T., 2014. Effects of linseed oil and palm oil on growth performance, tibia fatty acid and biomarkers of bone metabolism in broilers. British Poultry Science, 55, 335-342.
Zhu, X., Liu, W., Yuan, S., and Chen, H., 2014. The effect of different dietary levels of thyme essential oil on serum biochemical indices in Mahua broiler chickens. Italian Journal of Animal Science, 13, 576-581.
Zulkifli, I., Htin, N.N., Alimon, A.R., Loh, T.C., and Hair-Bejo, M., 2007. Dietary selection of fat by heat-stressed broiler chickens. Asian-Australasian Journal of Animal Sciences, 20, 245-251.
Zulkifli, I., Liew, P.K., Israf, D.A., Omar, A.R., and Hair-Bejo, M., 2003. Effects of early age feed restriction and heat conditioning on heterophil/lymphocyte ratios, heat shock protein 70 expression and body temperature of heat-stressed broiler chickens. Journal of thermal Biology, 28, 217-222.
Acknowledgements
The authors are particularly grateful to the Director-General of MPOB for permission to publish this article.
Author information
Authors and Affiliations
Contributions
Drafting the manuscript: M.S.; contribution to the concept or design of the manuscript: M.S.; interpreting the relevant literature: M.S., W.N.W.M., N.A.I., A.M.N., M.A.F.; critical revision for the important intellectual content of the manuscript: M.S., S.K.R.; final editing and improving the version to be published: M.S., W.N.W.M., N.A.I., A.M.N., M.A.F., S.K.R. All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
All authors have checked and approved the manuscript for publication.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Saminathan, M., Mohamed, W.N.W., Noh, ‘.M. et al. Effects of dietary palm oil on broiler chicken productive performance and carcass characteristics: a comprehensive review. Trop Anim Health Prod 54, 64 (2022). https://doi.org/10.1007/s11250-022-03046-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11250-022-03046-5