Abstract
Secondary metabolites (SMs) are biologically active compounds that have a very high commercial value due to their invaluable medicinal properties. However, the production level in in-vivo and in-vitro cultures remains low to meet the commercial demands. To overcome this problem, nano-elicitation has emerged as a novel and effective strategy which helps in enhancing the level of SMs production. The principle behind elicitation is that when external stress such as nanoparticles (NPs) is applied to the plant, the accumulation of SMs enhances in plant tissue cultures as a natural defense mechanism. In recent years, research on nano-elicitors using different types of NPs, especially metal oxide NPs (MONPs) has intensified due to enormous increase in medicinally important SMs. MONPs have gained special attention due to their unique physiochemical characteristics. In this review, we have explored the different routes of exposure of MONPs in plants as well as their role as novel elicitors of important classes of SMs (phenols, flavonoids, alkaloids, and terpenes). Moreover, the underlying mechanism of nano-elicitation and their uptake and translocation in plants have also been discussed. The review has been concluded with the knowledge gaps and future research direction, which if addressed will be helpful in solving the challenges of modern times.
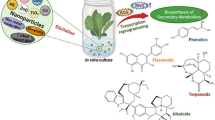
Graphical abstract

Key message
Metal oxide nanoparticles play a vital role in activation of the defense mechanism of plants which in turn activate the enhanced production of secondary metabolites in in-vitro cultures of medicinally important plants.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Secondary metabolites (SMs) are a class of chemical compounds synthesized by plants through metabolic routes that branch off from major primary metabolic pathways (Jones 1953). Some SMs are produced in response to the activation of morphological differentiation and emerge when cells differentiate and mature throughout the plant growth (Bhattacharya 2019). These metabolic intermediates or products are not necessary to the development and survival of the plant but are required for the plant to interact with its environment under stress conditions (Hatami et al. 2019). Thus, these compounds are produced to allow the plants to compete and increase their viability in their natural habitat by accurately controlling plant growth and development, acting as a reservoir for essential phytochemicals, and safeguarding plants from a variety of environmental barriers (Ashraf et al. 2018). Under stress conditions, they also serve as crucial mediators of interactions with biotic (living) or (abiotic) nonliving entities and have a variety of impacts on the plant and other living beings. The bioactive compounds including flavonoids, alkaloids, phenolics, and carotenoids have a high commercial value due to their potential applications in the pharmaceutical, medical, cosmetic, food, and agriculture industries (Rivero-Montejo et al. 2021).
Despite having huge commercial value, the in vitro and in vivo production of SMs remains low. There are several challenges associated with the large-scale synthesis of the majority of economically significant bioactive metabolites from farmed and wild plants, and various tissue cultures. In this regard, elicitation has emerged as an effective approach used to improve or enhance the production of SMs in plants (Hatami et al. 2019). Elicitation involves the exogenous addition of biotic or abiotic elicitors to the growth medium, which in turn induces a stress response and an increases the secondary metabolite production level in cultured cells, tissues or organs (Narayani and Srivastava 2017). Abiotic elicitors encompass non-biological substances and can be categorized into chemical, hormonal and physical factors. Conversely, biotic elicitors consist of biologically derived substances, such as polysaccharides from plant cell walls (e.g. pectin, cellulose, chitin) and microorganisms (Poornananda and Jameel 2016). Elicitors promote biochemical and physiological processes for activating the defensive system in targeted plants. They serve as signals, which are detected by elicitor-specific receptors present on the cell membrane of plants and induce defensive responses during elicitation, leading to an increase in the production and storage of SMs (Halder et al. 2019). Over the last few decades, the application of low concentrations of abiotic and biotic elicitors in differentiated or undifferentiated tissue cultures of various plant species has resulted in the induction of SMs accumulation in vitro or their release into the culture medium (Namdeo 2007).
Nanoparticles (NPs) are a class of abiotic elicitors applied to enhance the production of SMs in plant tissue cultures (Rivero-Montejo et al. 2021). While additional research is necessary to elucidate the precise mechanism, many researchers have proposed that NPs stimulate the production of secondary signaling molecules and reactive oxygen species (ROS), which play a role in the transcriptional control and regulation of plant secondary metabolism (Marslin et al. 2017). Metal oxide nanoparticles (MONPs), including zinc oxide NPs (ZnO NPs), copper oxide NPs (CuO NPs), and titanium dioxide NPs (TiO2 NPs), have been suggested as potential elicitors to enhance the production of SMs in in vitro plant cell cultures (Abbasi et al. 2019). The potential of MONPs to enhance plant SMs synthesis has been demonstrated in various plant species under different culture conditions (Anjum et al. 2019). Moreover, it has been found that MONPs play a crucial role in a plant’s response to abiotic stress and exhibit various regulatory functions. These particles have been shown to help plant cells resist oxidative stress and regulate ROS production (Khan et al. 2021a, b).
From NPs synthesis to their administration in plant research, the competency of these MONPs for boosting the production of major classes of medicinally and commercially important SMs in plants has been widely investigated. This review explores the current knowledge and gaps in this field and aims to further shed light on the possible mechanisms underlying this promising approach.
Uptake and translocation of MONPs in plants
Owing to their distinctive attributes, such as high surface area to volume ratio, minute size, high reactivity, and ability to facilitate electron exchange, MONPs can readily penetrate and interact with several plant cell and tissue components (Nair et al. 2010). A range of microscopic/spectroscopic methods, such as transmission electron microscopy, X-ray fluorescence microscopy, scanning electron microscopy, energy dispersive spectroscopy, confocal microscopy, atomic force microscopy, light microscopy, and two-photon excitation spectroscopy, have been employed to detect and verify the absorption of NPs by plants (Hatami et al. 2016) NPs may be absorbed by plants in three primary ways; by a foliar spray, through the soil and by means of artificially created nutritional medium as shown in Fig. 1.
In order to infiltrate plant tissue, MONPs need to overcome the initial obstacle of the primary barrier, namely the plant cell wall. Openings in the plant cell wall, with diameters ranging from 5 to 20 nm, may offer a direct entry route for NPs that are smaller than the pore size (Ma et al. 2015). Small NPs (diameters in range of 3–5 nm) have been shown to penetrate plant roots through capillary pressures, osmotic forces or directly by root epidermal cells (Lin and Xing 2008).
Several studies have reported the entry of NPs exceeding the size of plant cell wall pores, achieved either by altering the size of existing pores or by promoting the formation of larger pores. Subsequently, NPs traverse the plant cell wall to reach the cell membrane (Anjum et al. 2019). The internalization of the cell membrane into the cytosol or other organelles takes place through various mechanisms such as endocytosis, induction of new pores via ion carrier substances, or specific membrane-bound transporter proteins (Tripathi et al. 2017).
Once internalized, NPs have the potential to be transferred between cells through either the apoplastic or symplastic pathways (Fig. 2). Following penetration of the cell wall, NPs are transported through extracellular spaces in an apoplastic manner until they reach the central vascular cylinder, allowing them to move upward unidirectional through the xylem. In order to reach the innermost vascular cylinder, NPs must traverse the Casparian strip barrier via symplastic transport (Ali et al. 2021). After entry, NPs have the capacity to interact with diverse organelles and subcellular constituents of plant cells, which may disrupt the primary and specialized metabolism of plants. This disruption can be caused by the generation of ROS or other mechanisms, as elaborated in subsequent sections (Marchiol et al. 2014).
In a research, the modifications of MONPs were studied utilizing synchrotron X-ray absorption spectroscopy (XAS) during plant exposure to various plants (Du et al. 2011). When Zea mays plants were exposed to MONPs in hydroponic conditions, the maximum accumulation of ZnONPs was found in the roots and shoots. This is likely due to increased solubility in the plant uptake, rhizosphere, and translocation of Zn+2 ions (Lv et al. 2015). Currently, NPs absorption, accumulation, translocation, and association with plant cells or tissues are a fairly novel area, with limited published data on their destiny in plant cells. To fully comprehend the process of NP elicitation of specialized metabolites, it is critical to thoroughly define their penetration, transport, and interaction mechanisms inside plant cells and tissues (Anjum et al. 2019).
Mechanism of nano-elicitation by MONPs
There are numerous theories that have been postulated in regards to the signaling pathways associated with the metabolism of SMs however a definite one has not been identified. This is largely credited to the fact that the mechanism itself is elaborative involving a multitude of mediators and stimuli (Anjum et al. 2019). Furthermore, the cascade of events varies on the basis of 3 main factors: the physicochemical properties of the plant namely the plant species, the type of tissue involved, the stage of development it is at, the nutrition available to it and the mode of uptake of NPs, the nature of the elicitor, including its morphology and concentration, and the pertinent environmental conditions involved (Hatami et al. 2016).
The initiation of secondary metabolite production occurs on the interaction of the nano-elicitors with the specific receptors embedded within the plasma membrane of the plant cell (Ghorbanpour and Hadian 2015). Resultantly, Ca+2/H+ ions move into the cell through ion channels causing acidification of the cytosol while simultaneous efflux of Cl− and K+ ions alkalinize the extracellular environment (Khan et al. 2021a, b). The abrupt influx of Ca+2 is pertinent to the subsequent phases as it regulates the series of reactions that lead to the activation of genes for SMs metabolism (Zhang et al. 2012).
Oxidases of the plant cell particularly NADPH oxidase are speculated to be activated by the aforementioned Ca+2 flood that overproduce superoxide ions (O−2), ROS, hydroxyl radicals (OH−), reactive nitrogen species (RNS), and hydrogen peroxide species (H2O2) that culminate into an oxidative burst (Hatami et al. 2016). Consequently, rapid phosphorylation and de-phosphorylation of mitogen-activated protein kinases (MAPKs) via c-GMP protein kinase, causes transcriptional reprogramming and the expression of secondary metabolite production related genes (Anjum et al. 2019). Moreover, GTP binding proteins are also hypothesized to be involved in the transduction of signals as they activate phospholipases that can hydrolyze phospholipids into fatty acids and lysophospholipids (Garcia-Brugger et al. 2006). These are in turn in charge of the release of Ca+2 from intracellular pools of relevant organelles (Ramirez-Estrada et al. 2016) and the spike in Ca+2 concentration causes the induction of stress signaling molecules such as jasmonate through the octadecanoid pathway, ethylene and salicylic acid which are also involved in expressing genes responsible for defense response (Marslin et al. 2017). The complex network of the diverse parallel and cross-linking signaling pathways that target different responses in order to accomplish a common goal of inciting the production of SMs is shown in Fig. 3.
Schematic representation of the mechanism proposed for nano-elicitation in a plant cell (complete arrows depict direct reactions while dashed arrows represent indirect reactions; NP nanoparticles PL phospholipase; RNS reactive nitrogen species; ROS reactive oxygen species; MAPKs mitogen activated protein kinases; TF transcription factor)
MONPs as elicitors of different class of secondary metabolites
Studies have reported the effect of MONPs on the production of various classes of SMs. In relation to the effective induction of secondary metabolism and ease of implementation, the use of MONPs as elicitors is considered as an attractive strategy to enhance pharmaceutically significant metabolites as shown in Fig. 4 (Anjum et al. 2019). The studies discussed ahead highlight the outcomes of elicitation of a variety of plant species by different MONPs to enhance the production of the major classes of SMs as discussed below.
Nano-elicitation of phenols
Phenolic compounds are one of the preeminent types of SMs that are distinguished by their structural appearance with one benzene ring and single or multiple hydroxyl substitutes (Velderrain-Rodríguez et al. 2014). They are naturally produced in plants through the metabolization of phenylpropanoid (Randhir et al. 2004). With respect to human physiology, phenol rich foods have been reported to reduce the risk of cardiovascular diseases, diabetes associated hyperglycemia, cancerous activities and inflammation (Lin et al. 2016). Commercially, their use is made in the food industry as preservatives and colorants, in the cosmetic industry as UV protectors and anti-aging products, in the packaging industry for their anti-microbial and anti-oxidant properties and in various other niches (Albuquerque et al. 2021). A myriad of plant species and cultures have been utilized to test the viability of MONPs in stimulating the production of phenolic as listed in Table 1.
An experiment comparing the effects of zinc oxide NPs (ZnO NPs) and titanium dioxide (TiO2 NPs) as potential pathogen controllers for beetroot concluded that ZnO NPs not only had a definite impact on the growth of the plant but also reduced disease indices more efficiently than TiO2 NPs .This was caused by the increase in phenylalanine ammonia lyase (PAL) activity that catalyzed the overproduction of coumarin, cinnamic acid, chlorogenic acid and lignin (Siddiqui et al. 2019). Similarly, another study reported the enhanced gallic acid synthesis in ZnO NPs elicited Hyoscyamus reticulatus hairy root culture in accordance to which 1.67 mg gallic acid/g fresh weight was acquired after treatment with 100 mg/L of ZnO NPs for 72 h (Asl et al. 2019). In an attempt to study the influence of various metallic NPs on the biological activity of Lavandula viridis and Thymus lotocephalus in vitro shoot cultures, it was revealed that ZnO NPs were accountable for the induction of rosmarinic acid in high concentrations in T. lotocephalus however they negatively affected the total phenolic content of L. viridis indicating that the results of nano-elicitation with the same type of nanoparticle can vary with plant species (Gonçalves et al. 2021). While most studies employ chemically synthesized ZnO NPs, for example a experiment was conducted using Phoenix dactylifera mediated ZnO NPs in order to analyze their effect on the total phenolic content (TPC) of Juniperus procera. Corresponding to the results obtained with chemically prepared NPs, these biological ones at 160 mg/L also exhibited a distinguishable increase in the TPC and bioactive compound production of J. procera imparting extraordinary antioxidant capabilities to the plant (Salih et al. 2021). Yet another study highlighted the enhanced antioxidative properties of the potato plant as they synthesized 99.1 mg/g fresh weight of phenolics when subjected to ZnO NPs at 300 ppm concentration. The toxicity promoted by the oxidative stress of the NPs gave rise to the secondary metabolite outpour (Raigond et al. 2017).
Copper oxide nanoparticles (CuO NPs) are also largely employed for the stimulation of secondary metabolite production. An experiment conducted on Withania somnifera demonstrated a significant increase in the polyphenol content of the shoots and roots when elicited by CuO NPs. 27.215 ± 0.73 mg/g DW phenolic amount was recorded in shoots while 26.455 ± 0.365 mg/g DW was found in the roots after a treatment period of 20 days (Singh et al. 2018). Yet in another study where ZnO and CuO NPs synthesized by the extracts of Nigella sativa were utilized for elicitation of phytochemicals, three varieties of Vigna radiata were tested. At 0.5 mg/L of each type of nanoparticle, all three classes of mung beans showed enhanced phenolic content i.e. 26%, 25.6%, and 22.7%. Moreover, 50%, 37.5%, and 25% increase in glycoside production was also observed in the three varieties of beans respectively (Iqbal et al. 2022). Much like any other stress applied to a plant system, imposing a concentration higher than the bearable threshold amount can deem toxic. This was analyzed in an experiment using Stevia rebaudiana which was subjected to the formulations of ZnO and CuO NPs. It was concluded that ZnO was a more potent elicitor of steviol glycosides, phenolics and flavonoids at 100 mg/L than CuO at 10 mg/L. The reason was attributed to the toxicity imposed by CuO NPs on S. rebaudiana above 10 mg/L (Javed et al. 2018). Nonetheless, this threshold value varies with plant species. Another study reported optimum phytochemical generation in Chinese cabbage at 100 mg/L of CuO NPs. It included hydroxybenzoic acid (889.23 µg/g), flavonols (1293.09 µg/g), and hydroxycinnamic acid (922.9 µg/g) (Chung et al. 2018). The efficiency of CuO NPs for the stimulation of SMs in Gymnema sylvestre was assessed by subjecting it to three different concentrations of the elicitor (1, 3 and 5 mg/L). Highest yields of gymnemic acid II, phenolics, and flavonoids were obtained at 3 mg/L of CuO NP after 48 h of interaction. A total of 2 fold increase was reported in the TPC and TFC and around 2.3 times enhancement in gymnemic acid II (Chung et al. 2019).
Titanium dioxide nanoparticles (TiO2 NPs) are considered to be the most common components of environmental pollutants as a result of which most plant systems are accustomed to interacting with them and prone to taking up them with soil solutions. Various researches pertaining to the ability of TiO2 NPs to instigate the production of metabolites in a range of plant systems have been done. An experiment involving Ziziphora clinopodioides was conducted which reported the production of various phenolic compounds (ortho-diphenols, phenolic acids, flavonoids, and proanthocyanidin). 60 mg/L of TiO2 NP was not only revealed to be optimal for the growth of the plant root and shoot but the highest yield of phenol and flavonoid (1.261 ± 0.51 mg/g and 1.04 ± 0.13 mg/g fresh weight respectively) was obtained at this concentration too. Moreover a positive correlation was discovered between the antioxidative characteristics of the plant elicited by NPs and the increase in the TPC (Asadi and Cheniany 2022). In a recent study focused on enhancing the phenolic content, rosmarinic acid and caffeic acid produced by Mentha piperita. 150 mg/L of TiO2 NPs proved to not only enhance the dry weight of shoot, length of the shoot and the membrane stability index but it also improved the release of phenolic content by the plant system by 76%. Further, 87% increase in rosmarinic acid and 78% in caffeic acid was noted (Shenavaie Zare et al. 2022). Similarly, researchers aiming to enhance the induction of a callus through callogenesis and the production of certain SMs in leaf explants and apical meristems of Salvia tebesana, employed 6-Benzylaminopurine (BAP) and 2,4-D1 TiO2 NPs respectively to achieve it. The production of rosmarinic acid, an essential phenol, various flavonoids and anti-oxidants were recorded against the elicitation of TiO2 NPs at 10, 60, and 120 mg/L concentration. Results demonstrated promising outputs at 60 mg/L for leaf explants and at 10 mg/L for apical meristems by which high values for phenolic acid, TPC, O-diphenols, flavonoids, flavanols and proanthocyanidins were obtained. In addition, direct relations were observed between the rosmarinic acid derivative and the anti-oxidant activity of the treated calli (Shoja et al. 2022).
The ability to initiate the release of SMs in plants has not only been studied in chemically synthesized NPs but plant based TiO2 NPs as well with comparable outcomes.
A study reporting the use of Moringa oleifera reduced TiO2 NPs to trigger the synthesis of proline, phenols and flavonoids in Triticum aestivum L. revealed that the TPC and TFC values increased at 4 mg/L, however they displayed a marked reduction in comparison to plants elicited under Puccini striiformis stress (Satti et al. 2022). Yet another study gave prominence to the need to revolutionize the pharmaceutical industry by optimizing extraction from alternative cultivation system. In order to fulfill this objective, TiO2 NPs ranging from 10 to 50 mg/L were used on hairy root cultures of Saponaria officinalis L. to observe the production of polyphenolic compounds. Maximum hairy roots with a fresh weight of 3.09 g were obtained in cultures treated with 10 mg/L for 24 h. Moreover, the highest rate of phenolic content, 9.79 mg /g fresh weight was measured at 50 mg/L after 24 h. The production of various other polyphenols such as cinnamic acid, rutin and rosmarinic acid were also induced with the same treatment (Hedayati et al. 2022).
Nano-elicitation of flavonoids
Flavonoids impart pigments to plants and belongs to a subclass of polyphenols. They contain over 10,000 compounds and are classified by their aromatic rings, namely two phenyl rings (Teoh 2015). Flavonoids have been reported to be eminent bearers of medicinal benefits that include anti-cancerous, anti-viral, anti-inflammatory and antioxidant properties as a result they are one of the most desired class of SMs (Ullah et al. 2020). Several types of MONPs have been tested as nano-elicitors of flavonoids as shown in Table 2.
The seedlings of Glycyrrhiza glabara L. also known as Licorice, were subjected to ZnO NPs of ranging sizes i.e., nano and bulk dimensions in order to determine if it had an effect on the production of flavonoids, anthocyanins, glycyrrhizin and phenols. The results concluded that ZnO NPs caused a greater increase in all the compounds that were studied in comparison to their bulk counterparts (Oloumi et al. 2015) and the rational reasoning suggested that the smaller size not only allows for better adsorption inside the plant cells but confers better compatibility with the organelles of the cell as well (Nair et al. 2010). In the course of such studies, the toxicity at specific concentrations of ZnO NPs was uncovered and a solution was derived. Research on the combination of ZnO NPs and humic acid for elicitation was done on Lilium ledebourii and the results showed that maximum production of flavonoids (25 mg/L) was achieved at 25 mg/L of ZnO NPs while highest concentration of anthocyanins (100 mg/L) was acquired at 75 mg/L. In addition, humic acid deemed beneficial for the growth parameters of the plant suggesting that it may be helpful in mitigating and compensating for the damage caused by ZnO NPs elicitation (Chamani et al. 2015). Similarly, Momordica charantia L. was treated with three types of abiotic elicitors i.e., jasmonate, ZnO NPs and chitosan in a dose-dependent manner. While ZnO NPs did not have any notable impact on the weight of the shoot, it progressively amplified the amount of anthocyanin and flavonoid produced. At 100 ppm of ZnO NPs, 228 mg/g fresh weight and 14 mg/g fresh weight anthocyanin and flavonoid were reported respectively (Sharifi-Rad et al. 2020). S. rebaudiana was treated with ZnO NPs with the aim of bio-fortification and an 87.8% increase of TFC was noticed at 75 mg/L of the NPs while the growth parameters of the plant were not disturbed (Velázquez-Gamboa et al. 2021). Nevertheless, above a certain threshold limit of elicitation, the flavonoid content may begin to decrease as was reported in the experiment conducted on Echinacea purpurea. A callus culture was exposed to a wide range of Zn ONPs concentrations, out of which optimum outcomes were represented at 75 mg/L with 23.25 mg/g dry weight of flavonoid. However, above this value, the phytotoxicity of ZnO NPs sets in, inhibiting further synthesis (Karimi et al. 2018).
Similar to the promising outcomes reported with phenolic content elicitation, CuO NPs were also claimed to be effective in increasing flavonoids in plants. Lens culinaris, an edible legume, was treated with CuO NPs, biosynthesized using the extract of Adiantum lunulatum. On evaluation of the TPC of the plant by elicitation with the NPs, a 2.33 fold increase was reported at 0.025 mg/mL (Sarkar et al. 2020). Nano-CuO stress on Lavandula stoechas exhibited remarkable outcomes with an increase in phytochemicals at all four concentrations i.e. 0, 25, 50, and 100 mg/L relative to the control. However the highest amount of TFC obtained was at the 50 mg/L with an exposure period of 5 days with 0.68 mg/g FW. Moreover, 36.51 mg/g FW anthocyanins and 0.29 mg/gFW flavonols were also reported (Asadollahei et al. 2023). Medicinal plants such as Ocimum bascilicum have also been subjected to CuO and MnO NPs to gain insight on their effect on the biochemical profile of the plants. The comparative analysis showed that CuO NPs at 10 mg/L resulted in the highest accumulation of flavonoids (9.1 mg/g DW) and instigated peroxidase and superoxide dismutase more effectively than MnO NPs (Nazir et al. 2021). G. sylvestre subjected to three concentrations of CuO NPs 1, 3, 5 mg/L caused a 2 fold increase in both the TPC and the TFC at the optimal concentration of 3 mg/L (Chung et al. 2019).
The dominant influence of TiO2 NPs as elicitors has also been discussed before however, experiments pertaining to enhancement in flavonoid content have also been conducted. S. rebaudiana when treated with a range of TiO2 concentrations over a time period of 3 weeks exhibited a 40% increase in flavonoid content as compared to the controls while 51% increase in phenolic content and 53% improvement in chlorophyll content was also reported. This occurred at the highest concentration that was tested i.e. 400 µL/L (Rezaizad et al. 2021). A comparative analysis of Methyl Jasmonate and TiO2 NPs as elicitors of Teucrium polium revealed a 1.5 fold increment in the flavonoid, flavones, rosmarinic acid and phenolic content of the plant in relation with the non-elicited controls. While 10 mg/L of TiO2 NPs was concluded to be the most preferable concentration, 50 µM methyl jasmonate produced even better results (Tabarifard et al. 2023). Yet another parallel study conducted by Modarresi et al. (2020) tested TiO2, Al2O3 and NiO NPs as elicitors of Nigella arvensis and reported a 2.2 times increment in flavonoid content at 1000 mg/L of TiO2 relative to the control. Nevertheless, NiO NPs proved to be even more effective by raising the flavonoid content by 2.5 folds at a lower concentration of 100 mg/L. In another study, a remarkable elevation of 118% in the flavonoid content of Oryza sativa was reported on exposure to 400 mg/L of TiO2 NPs. Moreover free radicals present in the metabolites were depleted by 84% in contrast to the untreated control (Chutipaijit and Sutjaritvorakul 2020). In accordance with a previously mentioned study, S. officinalis was subjected to TiO2 to determine its effect on the plants phytochemical profile resulting in 1.06 mg QE/g FW of TFC which was the highest amount obtained by treatment with 30 mg/L of the nano-elicitor for 48 h (Hedayati et al. 2022).
Nano-elicitation of alkaloids
Alkaloids are bioactive components that contain a distinctive heterocyclic ring with one or more nitrogen atoms attached to it (Teoh 2015). The principles of nano-elicitation are availed to reap alkaloids for commercial use as the quantities extracted through their natural resource are often insufficient (Anjum et al. 2021). Although, alkaloids are commercially important metabolites, only a limited number of studies are available in literature in which nano-elicitation has been performed (Table 3).
ZnONPs have proven to be promising in the enhanced metabolism of alkaloids as well, as seen in the elicitation of Trigonella foenum-graecum L commonly known as Fenugreek. This plant contains a precious compound called trigonelline that holds significant medicinal importance. A hairy root culture of the plant was prepared and subjected to 2× ZnONPs in conjugation with an Agrobacterium rhizogenes strain and resulted in double the trigonelline produced in comparison to the control i.e. hairy roots not transformed with the NPs (Tariverdizadeh et al. 2021). As mentioned earlier, in order to obtain an optimum yield of alkaloids, specific concentrations of ZnONPs treatments must be given otherwise the results may disappoint. This was tested in a study wherein ZnONPs and a Chitosan-ZnO bioformulation were used administered to Nicotiana benthamiana callus. Nicotine constitutes the majority of the alkaloids present in N. benthamiana and its overproduction was maximally stimulated at 400 ppm of ZnONPs. At this concentration, the callus produced 125% more nicotine than the control but once the concentration was raised to 800 ppm, significant reduction (25%) in nicotine production was recorded (Patel et al. 2020). Yet another plant that houses valuable tropane alkaloids, for instance, hyoscyamine and scopolamine is Hyoscyamus reticulatus L. The alkaloids derived from these plants prove to be pertinent compounds to treat various neurological diseases like schizophrenia, Parkinson’s and epilepsy. Investigations at three different concentrations (50, 100, 200 mg/L) of ZnONPs with three intervals of time (24 h, 48 h, 72 h) were done and the ideal results were obtained at 100 mg/L of ZnONPs at which 37% hyoscyamine and 37.63% scopolamine was produced after 48 and 72 h of exposure respectively (Asl et al. 2019). ZnO has been developed with different formulations and tested as an alkaloid elicitor. A comparative analysis was performed between ZnONPs and a chitosan/ZnO nanocomposite on Capsicum annuum and a number of physiological patterns were studied. Chitosan/ZnO nanocomposites seemed to excel in all aspects with a 60% increase in alkaloid production, 40% more phenol accumulation and improved organogenesis of the plant in vitro (Asgari-Targhi et al. 2021).
The enhanced production of alkaloids in plants in response to other MONPs is evident from the published literature reports. Al-Oubaidi and Jawad (2021) published a study in which CuO NPs were applied to Physalis peruiviana for the enhanced production of atropine and scopolamine. The experiments were performed at different concentrations of CuO NPs (0.5, 1.0, 1.5, and 2.0 mg/L). Highest concentration of the before-mentioned alkaloids was obtained at 2.0 mg/L CuO NPs concentration. Likewise, hyoscyamine and scopalamine production was enhanced in Hyoscyamus niger L. by the application of TiO2 NPs. Highest content of hyoscyamine was obtained at 80 mg/L TiO2 NPs concentration whereas 20 mg/L concentration was found optimum for the enhanced production of scopolamine (Ghourbanpour et al. 2015). Similarly, silicon dioxide NPs (SiO2 NPs) were also found to have elicitation potential for alkaloids as evident from the study performed by (Hedayati et al. 2020). Two species of Hyoscyamus were selected to study the effect of SiO2 NPs for the production scopolamine and hyoscyamine. The highest content of both metabolites was obtained at 100 mg/L and 10 mg/L respectively (15.56 µg/g FW and 7.42 µg/g FW) indicating the use of SiO2 NPs as efficient elicitors. Similarly, Fe3O4 NPs has also been employed for tropane alkaloids production in genetically transformed H. reticulatus root cultures by Moharrami et al. (2017) At 450 mg/L Fe3O4 NPs concentration, maximum increase (20.3%) was observed in case of scopolamine as compared to hyoscyamine, of which maximum content was observed at 900 mg/L concentration (43.82%) after the treatment of 48 h and 24 h respectively. As mentioned before, Modarresi et al. (2020) further reported the impact of TiO2 NPs, NiO NPs and Al2O3 NPs on the production of glaucine, a commercially important alkaloid, in N. arvensis. The highest concentration of glaucine was observed when 100 mg/L of Al2O3 was applied to the plant. In case of NiO and TiO2 NPs, the alkaloid content was reduced. The increase was more than 50% as compared to the control groups.
Nano-elicitation of terpenes
Terpenes are a broad class of natural organic compounds primarily existing in plants. They are derived from a 5-carbon compound called isoprene; the polymers of isoprene are called as terpenes. More than 60,000 terpenoids have been identified up to date, making terpenes a major class of SMs (Jahangeer et al. 2021). The terms terpenes and terpenoids are used interchangeably, however they differ in the addition of a functional group in terpenoids with general structure same as terpenes. Being SMs, terpenes and its derivatives provide plants with defensive strategies against the environment (Toffolatti et al. 2021). They have long been used as natural flavoring compounds, insecticides, pharmaceutical compounds, fragrances and industrial compounds. With the growth of pharmaceutical uses of plant based drugs and the uses of terpenes as alternative fuels, scientists are looking for ways to enhance the production of terpenes (Tholl 2015). The potential of MONPs has been exploited for eliciting of terpenoids in several studies as shown in Table 4.
ZnO NPs have extensively been used to produce SMs evident from a variety of reported studies. Expression Analysis of ADS, DBR2, ALDH1and SQS genes was done in the hairy root cultures of A. vulgaris by Yarizade and Hosseini (2015). These are some of the key genes that regulate the artemisinin biosynthesis pathway in plants. The up-regulation of these genes result in the enhanced synthesis of artemisinin as well (Wani et al. 2021). Elicitation of the expressed gene products i.e., terpenes was mediated by cobalt nanoparticles (Co NPs) and ZnO NPs. After the treatment of culture with NPs, the expression level was examined after 8, 24, 48 and 42 h. It was reported that 1.0 mg/L ZnO NPs resulted in the maximum expression of all the genes, whereas 0.25 mg/L of Co NPs resulted in the maximum gene expression. According to the study, ZnO NPs were established as the nano elicitor, however, Co NPs had an advantage that in addition to up regulating the ADS gene, it down regulated the SQS gene, its antagonist for the maximum elicitation of artemisinin. Bhardwaj et al. (2018) evaluated the nano-elicitation potential of ZnO NPs by applying them to cell suspension cultures of Baccopa monnieri. HPLC analysis showed that varying concentrations of ZnO NPs showed varying bacoside yield. A two fold increase in bacoside A production was observed in the suspension cultures supplemented with 1ppm ZnO NPs as compared to control. Another study was conducted by Mosavat et al. (2019) in order to evaluate the elicitation effect of ZnO NPs on thymol and carvacrol production in callus cultures of three different thymus species. The concentration of the two mentioned terpenoids was measured by HPLC which indicated that 150 mg/L ZnO NPs resulted in the highest content of thymol and carvacol (28.8 mg/L and 0.68 mg/L respectively) making them potential nano-elicitors of terpenoids in T. kotschyanus and T. daenasis respectively (Mosavat et al. 2019). The effect of ZnO NPs, jasmonate and chitosan as non-biological elicitors were studied on the M. charantia shoot cultures as mentioned previously. The results showed an overall increase in the secondary metabolite content. At 20 ppm ZnO NPs concentration, the carotenoid content increased to 2.767 mg/g FW as compared to control, when carotenoid content was 1.086 mg/g FW (Sharifi-Rad et al. 2020). In addition to evaluating their potential of nano-elicitation of phenolic compounds, ZnO NPs have also established themselves as the potent nano-elicitor of glycyrrhizin, which is structurally a saponin, in licorice seedlings. The content of glycyrrhizin was increased by supplementation of 1mM of ZnO NPs in seedlings (Oloumi et al. 2015). Nekoukhou et al. (2022) studied the effect of ZnO NPs on the synthesis of medicinally important terpenes, geranial, geraniol and neral by applying ZnO NPs formulation in dragonhead (Dracocephalum moldavica L.). The result showed the gradual increase in the biomass, total chlorophyll and total secondary metabolite content in dose-dependent manner. The dose of 160 mg/L of ZnO NPs was found suitable for the increased concentration of terpenes.
Although limited evidence is available on the exploitation of CuO NPs for the nano-elicitation. However, existing studies have shown promising results regarding the production of terpene and their derivatives. In a study performed by Ahmad et al. (2020) tissue culture regenerates of S. rebaudiana were comparatively studied after the application of ZnO NPs and CuO NPs. Different concentrations of both NPs were applied to the MS media in which tissue culture were grown and their effects on the morpho-physiological responses viz. plant biomass, shooting and rooting and the number of leaves, roots and nodes were studied as well as the synthesis of SMs namely seviol glycosides (SGs). The results obtained by HPLC showed that greatest content of SGs was obtained at 2 mg/l of ZnO NPs and 20 mg/l of CuO NPs. Both type of NPs were reported to have adverse effects on the plant biomass and antioxidant activities of plants and elicited the SMs response i.e., synthesis of SGs. As SGs have significant anti-diabetic and anti-cancerous properties and are also used pharmaceutically for obesity relief (Dey et al. 2013; Ashwell 2015). CuO NPs and ZnO NPs has proved as potential nano-elicitors and can be applied at industrial level to extract SGs from tissue cultured SGs-rich plants. It is supported by another study performed by same authors who studied the comparative effect of ZnO NPs and CuO NPs as mentioned previously. In this study, micro-propagated shoots of S. Rubaudiana were investigated after the application of NPs. The result showed a significant increase in SGs content, however a decrease in TPC and TFC was observed (Javed et al. 2017). Chung et al. (2019) reported the increased yield of gymnemic acid II in the cell suspension cultures of G. sylvestre as mentioned in prior heading. The highest yield was observed after the treatment of cultures with 3 mg/L of CuO NPs for 48 h, where the amount of gymnemic acid II increased nine-fold as compared to untreated cultures. Gymnemic acid is a triterpene saponin with potential anti-diabetic applications (Li et al. 2019). Nekoukhou et al. (2023) studied the impact of different concentrations of CuO NPs and chelated-CuO NPs on the production and yield of SMs such as geraniol, neral, geranial and geranyl acetate in dragonhead (D. moldavica L.). The study showed promising results at 80 mg/L of CuO NPs as compared to 160 mg/L of conventionally chelated-CuO NPs. A recent research reported by Asadollahei et al. (2023) stated the potential effect of CuO NPs as elicitors on the essential oils and terpenoid components in Lavandula stoechas L. The results showed a significant increase in the essential components such as lavandulol (21.68%), germacrene D (9.33%) 1, 8-cineole (17.21%), and E-nerlidol (8.11%). The study proved the significant potential of CuO NPs for its application in SMs production for industrial and medicinal use.
A number of research reports have provided promising results regarding the use of TiO2 NPs for SMs production. Gohari et al. (2020) reported the effect of TiO2 NPs on the agronomic traits of D. moldavica grown under saline stress. The results showed a considerable increase in all the plant traits under study. The essential oil content consisting of geranial, geranyl acetate and z-citral increased significantly (1.19%) at 100 mg/L TiO2 NPs. Moreover, this concentration sharply ameliorated the salt stress condition in the plant. The content and yield of menthol increased to 9.6% in control and 124.1% in TiO2 NPs-treated M. piperita L. as reported by Ahmad et al. (2018). 150 mg/L concentration of TiO2 NPs was found suitable for the significant increase in the production of essential oil and menthol content. The study investigated the effect of TiO2 NPs on the enzymatic activities and photosynthetic rate as well. Another study reported the increase in menthol production when TiO2 NPs were applied exogenously combined with methyl jasmonate to M. piperita L. The study provided astonishing results where the highest amount (44.51%) of menthol was produced when the subject plant was treated simultaneously with 150 mg/L TiO2 NPs and 0.1mM methyl jasmonate (Shenavaie Zare et al. 2022). Sheikhalipour et al. (2021) reported the stress alleviating effect of TiO2 NPs and Selenium NPs (SE NPs) functionalized with chitosan in Stevia rebaudiana Bertoni under salt stress. TiO2 NPs proved potentially successful in countering the adverse effects of salt stress in the plant under study. The results showed a significant increase in the concentration of stevioside (17.81%) and rebaudioside A (18.18%), tetracyclic triterpenoid compounds, and carotenoids (21.99%) in S. rebaudiana by the application of TiO2 NPs. Fazeli-Nasab et al. (2018) reported the increase in the concentration of thymol and carvacol when TiO NPs were applied to Thymus vulgaris under drought stress. The result showed that the yield of thymol and carvacol was enhanced by 82.302% and 12.33% in plants under drought stress of 70% field capacity upon TiO2 NPs application.
Concluding remarks
With the growing population, there is a growing need to find innovative solutions to the problems related to the pharmaceutical sector such as a need for higher production level of medicinally important SMs to combat diseases. To counter this, MONPs are widely explored in plant biotechnology research as novel elicitors of plant SMs. Numerous research found MONPs as an effective elicitor of SMs in terms of productivity and specificity. Despite immense research on MONPs as novel elicitors of SMs, there are still some knowledge gaps which need to be filled. Firstly, a deep understanding is required on dose dependent nano-elicitation of SMs for its successful industrial adoption. This can be achieved by targeted delivery and surface functionalization of NPs with various bio-molecules which can enhances the efficacy of elicitation using lower concentration of NPs and also prevents phyto-toxicity. Moreover, an understanding of plant response to the nanomaterials applied as elicitors either individually or in combination with other classes of NPs or with other types of elicitors is essential for developing strategies for enhanced SMs production.
Furthermore, the knowledge gap exists on the exact mechanism of nano-elicitation due to the involvement of complex events and variation from plant to plant, parts of the plant and growing conditions, depending on other factors as well such as, size, structure, morphology and exposure time of NPs (Dey and Das 2013; Anjum et al. 2019). However, it is clear from our review that MONPs can trigger specialized pathways for SMs production in in-vitro cultures. Various studies have been performed on cultured cells, tissues and organs of medicinally important plants. Therefore, case to case analysis on the effect of MONPs on the production of SMs, metabolic modeling, omics-based studies, and computer assisted simulations can be a future research direction in the area of nano-elicitation. This knowledge will be helpful in targeting specific metabolic pathway and adapt innovative strategies for nano-elicitation to gain commercial benefits. Further research on nano-elicitation will also be helpful in the production of novel SMs to combat emerging diseases as well. Once these research gaps are addressed, MONPs can be applied for important biotechnological applications and to meet the emerging demands of SMs.
Data availability
All data generated or analysed during this study are included in this published article.
References
Abbasi BH, Zahir A, Ahmad W, Nadeem M, Giglioli-Guivarc’h N, Hano C (2019) Biogenic zinc oxide nanoparticles-enhanced biosynthesis of lignans and neolignans in cell suspension cultures of Linum usitatissimum L. Artif Cells Nanomed Biotechnol 47:1367–1373. https://doi.org/10.1080/21691401.2019.1596942
Ahmad B, Shabbir A, Jaleel H, Khan MMA, Sadiq Y (2018) Efficacy of titanium dioxide nanoparticles in modulating photosynthesis, peltate glandular trichomes and essential oil production and quality in Mentha piperita L. Curr Plant Biol 13:6–15. https://doi.org/10.1016/j.cpb.2018.04.002
Ahmad MA, Javed R, Adeel M, Rizwan M, Ao Q, Yang Y (2020) Engineered ZnO and CuO Nanoparticles ameliorate morphological and biochemical response in tissue culture regenerants of Candyleaf (Stevia rebaudiana). Molecules 25:1356. https://doi.org/10.3390/molecules25061356
Al-Oubaidi MH, Jawad L (2021) Increasing some medical alkaloid compound for gooseberry physalis peruiviana using nano-copper oxide in vitro. Biochem Cell Archives https://connectjournals.com/03896.2021.21.4043
Albuquerque BR, Heleno SA, Oliveira MBPP, Barros L, Ferreira ICFR (2021) Phenolic compounds: current industrial applications, limitations and future challenges. Food Funct 12:14–29. https://doi.org/10.1039/D0FO02324H
Ali S, Mehmood A, Khan N (2021) Uptake, translocation, and consequences of nanomaterials on plant growth and stress adaptation. J Nanomater 2021:6677616. https://doi.org/10.1155/2021/6677616
Anjum S, Anjum I, Hano C, Kousar S (2019) Advances in nanomaterials as novel elicitors of pharmacologically active plant specialized metabolites: current status and future outlooks. RSC Adv 9:40404–40423. https://doi.org/10.1039/C9RA08457F
Anjum S, Komal A, Abbasi BH, Hano C (2021) Nanoparticles as Elicitors of biologically active ingredients in plants. Nanatechnol Plant Growth Promot Prot Recent Adv Impacts. https://doi.org/10.1002/9781119745884.ch9
Asadi A, Cheniany M (2022) Enhancing effect of titanium dioxide nanoparticles on growth, phenolic metabolites production and antioxidant potential of Ziziphora clinopodioides Lam. Russ J Plant Physiol 69:74. https://doi.org/10.1134/S1021443722040021
Asadollahei MV, Tabatabaeian J, Yousefifard M, Mahdavi SME, Nekonam MS (2023) Impact of elicitors on essential oil compositions and phytochemical constituents in Lavandula stoechas L. Plant Physiol Biochem 194:722–730. https://doi.org/10.1016/j.plaphy.2022.12.019
Asgari-Targhi G, Iranbakhsh A, Oraghi Ardebili Z, Hatami Tooski A (2021) Synthesis and characterization of chitosan encapsulated zinc oxide (ZnO) nanocomposite and its biological assessment in pepper (Capsicum annuum) as an elicitor for in vitro tissue culture applications. Int J Biol Macromol 189:170–182. https://doi.org/10.1016/j.ijbiomac.2021.08.117
Ashraf MA, Iqbal M, Rasheed R, Hussain I, Riaz M, Arif MS, Ahmad P, Ahanger MA, Singh VP et al (2018) Plant Metabolites and Regulation Under Environmental Stress. Academic Press, Cambridge, pp 153–167
Ashwell M (2015) Stevia, nature’s zero-calorie sustainable sweetener: a new player in the fight against obesity. J Nutr today 50:129. https://doi.org/10.1097/NT.0000000000000094
Asl KR, Hosseini B, Sharafi A, Palazon J (2019) Influence of nano-zinc oxide on tropane alkaloid production, h6h gene transcription and antioxidant enzyme activity in Hyoscyamus reticulatus L. hairy roots. Eng Life Sci 19:73–89. https://doi.org/10.1002/elsc.201800087
Bhardwaj P, Goswami N, Narula P, Jain CK, Mathur A (2018) Zinc oxide nanoparticles (ZnO NP) mediated regulation of bacosides biosynthesis and transcriptional correlation of HMG-CoA reductase gene in suspension culture of Bacopa monnieri. Plant Physiol Biochem 130:148–156. https://doi.org/10.1016/j.plaphy.2018.07.001
Bhattacharya A (2019) Chap. 5-High-Temperature Stress and Metabolism of Secondary Metabolites in Plants. In: Bhattacharya A (ed) Effect of High Temperature on Crop Productivity and Metabolism of Macro Molecules. Academic Press, Cambridge, pp 391–484
Chamani E, Karimi Ghalehtaki S, Mohebodini M, Ghanbari A (2015) The effect of zinc oxide nano particles and humic acid on morphological characters and secondary metabolite production in Lilium ledebourii Bioss. Iran J Genet Plant Breed 4:11–19
Chung I-M, Rekha K, Rajakumar G, Thiruvengadam M (2018) Production of bioactive compounds and gene expression alterations in hairy root cultures of chinese cabbage elicited by copper oxide nanoparticles. Plant Cell Tissue Organ Culture (PCTOC) 134:95–106. https://doi.org/10.1007/s11240-018-1402-0
Chung I-M, Rajakumar G, Subramanian U, Venkidasamy B, Thiruvengadam M (2019) Impact of copper oxide nanoparticles on enhancement of bioactive compounds using cell suspension cultures of Gymnema sylvestre (Retz.) R. Br. Appl Sci 9:2165. https://doi.org/10.3390/app9102165
Chutipaijit S, Sutjaritvorakul T (2020) Enhancements of growth and metabolites of indica rice callus (Oryza sativa L. cv. pathumthani1) using TiO2 nanoparticles (NANO-TiO2). Digest J Nanomaterials Biostructures 15:483–489. https://doi.org/10.1088/2043-6254/aaf1af
Dey P, Das N (2013) Carbon Nanotubes: it’s role in modern health care. Int J Pharm Pharm Sci 5:9–13
Dey A, Kundu S, Bandyopadhyay A, Bhattacharjee A (2013) Efficient micropropagation and chlorocholine chloride induced stevioside production of Stevia rebaudiana Bertoni. CR Biol 336:17–28. https://doi.org/10.1016/j.crvi.2012.11.007s
Du W, Sun Y, Ji R, Zhu J, Wu J, Guo H (2011) TiO2 and ZnO nanoparticles negatively affect wheat growth and soil enzyme activities in agricultural soil. J Environ Monit 13:822–828. https://doi.org/10.1039/C0EM00611D
Fazeli-Nasab B, Sirousmehr A-R, Azad H (2018) Effect of titanium dioxide nanoparticles on essential oil quantity and quality in Thymus vulgaris under water deficit. J Med Plants By-prod 7:125–133. https://doi.org/10.22092/jmpb.2018.118140
Garcia-Brugger A, Lamotte O, Vandelle E, Bourque S, Lecourieux D, Poinssot B, Wendehenne D, Pugin A (2006) Early signaling events induced by elicitors of plant defenses. Mol Plant Microbe Interact 19:711–724. https://doi.org/10.1094/MPMI-19-0711
Ghorbanpour M, Hadian J (2015) Multi-walled carbon nanotubes stimulate callus induction, secondary metabolites biosynthesis and antioxidant capacity in medicinal plant Satureja khuzestanica grown in vitro. Carbon 94:749–759. https://doi.org/10.1016/j.carbon.2015.07.056
Ghourbanpour M, Hatami M, Hatami M (2015) Activating antioxidant enzymes, hyoscyamine and scopolamine biosynthesis of Hyoscyamus niger L. plants with nano-sized titanium dioxide and bulk application. Acta Agric Slov 105:23–32. https://doi.org/10.14720/aas.2015.105.1.03
Gohari G, Mohammadi A, Akbari A, Panahirad S, Dadpour MR, Fotopoulos V, Kimura S (2020) Titanium dioxide nanoparticles (TiO2 NPs) promote growth and ameliorate salinity stress effects on essential oil profile and biochemical attributes of Dracocephalum moldavica. Sci Rep 10:1–14. https://doi.org/10.1038/s41598-020-57794-1
Gonçalves S, Mansinhos I, Rodríguez-Solana R, Pereira-Caro G, Moreno-Rojas JM, Romano A (2021) Impact of metallic nanoparticles on in vitro culture, phenolic profile and biological activity of two Mediterranean Lamiaceae species: Lavandula viridis L’Hér and Thymus lotocephalus G. López and R. Morales. Molecules 26:6427. https://doi.org/10.3390/molecules26216427
Halder M, Sarkar S, Jha S (2019) Elicitation: a biotechnological tool for enhanced production of secondary metabolites in hairy root cultures. Eng Life Sci 19:880–895. https://doi.org/10.1002/elsc.201900058
Hatami M, Kariman K, Ghorbanpour M (2016) Engineered nanomaterial-mediated changes in the metabolism of terrestrial plants. Sci Total Environ 571:275–291. https://doi.org/10.1016/j.scitotenv.2016.07.184
Hatami M, Naghdi Badi H, Ghorbanpour M (2019) Nano-elicitation of secondary pharmaceutical metabolites in plant cells: a review. J Med Plants 18:6–36. https://doi.org/10.29252/jmp.3.71.6
Hedayati A, Hosseini B, Palazon J, Maleki R (2020) Improved tropane alkaloid production and changes in gene expression in hairy root cultures of two Hyoscyamus species elicited by silicon dioxide nanoparticles. Plant Physiol Biochem 155:416–428. https://doi.org/10.1016/j.plaphy.2020.07.029
Hedayati A, Naseri F, Nourozi E, Hosseini B, Honari H, Hemmaty S (2022) Response of Saponaria officinalis L. hairy roots to the application of TiO2 nanoparticles in terms of production of valuable polyphenolic compounds and SO6 protein. Plant Physiol Biochem 178:80–92. https://doi.org/10.1016/j.plaphy.2022.03.001
Iqbal Z, Javad S, Naz S, Shah AA, Shah AN, Paray BA, Gulnaz A, Abdelsalam NR (2022) Elicitation of the in vitro cultures of selected varieties of Vigna radiata L. with zinc oxide and copper oxide nanoparticles for enhanced phytochemicals production. Front Plant Sci. https://doi.org/10.3389/fpls.2022.908532
Jahangeer M, Fatima R, Ashiq M, Basharat A, Qamar SA, Bilal M, Iqbal H (2021) Therapeutic and biomedical potentialities of terpenoids—a review. J Pure Appl Microbiol 15:471–483. https://doi.org/10.22207/JPAM.15.2.04
Javed R, Usman M, Yücesan B, Zia M, Gürel E (2017) Effect of zinc oxide (ZnO) nanoparticles on physiology and steviol glycosides production in micropropagated shoots of Stevia rebaudiana Bertoni. Plant Physiol Biochem 110:94–99. https://doi.org/10.1016/j.plaphy.2016.05.032
Javed R, Yucesan B, Zia M, Gurel E (2018) Elicitation of secondary metabolites in callus cultures of Stevia rebaudiana Bertoni grown under ZnO and CuO nanoparticles stress. Sugar Tech 20:194–201. https://doi.org/10.1007/s12355-017-0539-1
Jones ME (1953) Albrecht Kossel, a biographical sketch. Yale J biology Med 26:80–97
Karimi N, Behbahani M, Dini G, Razmjou A (2018) Enhancing the secondary metabolite and anticancer activity of Echinacea purpurea callus extracts bytreatment with biosynthesized ZnO nanoparticles. Adv Nat Sci: Nanosci Nanotechnol 9:045009
Khan AK, Kousar S, Tungmunnithum D, Hano C, Abbasi BH, Anjum S (2021a) Nano-elicitation as an effective and emerging strategy for in vitro production of industrially important flavonoids. Appl Sci 11:1694. https://doi.org/10.3390/app11041694
Khan AU, Khan T, Khan MA, Nadhman A, Aasim M, Khan NZ, Ali W, Nazir N, Zahoor M (2021b) Iron-doped zinc oxide nanoparticles-triggered elicitation of important phenolic compounds in cell cultures of Fagonia indica. Plant Cell Tissue and Organ Culture (PCTOC) 147:287–296. https://doi.org/10.1007/s11240-021-02123-1
Li Y, Sun M, Liu Y, Liang J, Wang T, Zhang Z (2019) Gymnemic Acid alleviates type 2 diabetes Mellitus and suppresses endoplasmic reticulum stress in vivo and in Vitro. J Agric Food Chem 67:3662–3669. https://doi.org/10.1021/acs.jafc.9b00431
Lin D, Xing B (2008) Root uptake and phytotoxicity of ZnO nanoparticles. Environ Sci Technol 42:5580–5585. https://doi.org/10.1021/es800422x
Lin D, Xiao M, Zhao J, Li Z, Xing B, Li X, Kong M, Li L, Zhang Q, Liu Y, Chen H, Qin W, Wu H, Chen S (2016) An overview of plant phenolic compounds and their importance in human nutrition and management of type 2 diabetes. Molecules. https://doi.org/10.3390/molecules21101374
Lv J, Zhang S, Luo L, Zhang J, Yang K, Christie P (2015) Accumulation, speciation and uptake pathway of ZnO nanoparticles in maize. Environ Science: Nano 2:68–77. https://doi.org/10.1039/C4EN00064A
Ma C, White JC, Dhankher OP, Xing B (2015) Metal-based nanotoxicity and detoxification pathways in higher plants. Environ Sci Technol 49:7109–7122. https://doi.org/10.1021/acs.est.5b00685
Marchiol L, Mattiello A, Pošćić F, Giordano C, Musetti R (2014) In vivo synthesis of nanomaterials in plants: location of silver nanoparticles and plant metabolism. Nanoscale Res Lett 9:101. https://doi.org/10.1186/1556-276X-9-101
Marslin G, Sheeba CJ, Franklin G (2017) Nanoparticles alter secondary metabolism in plants via ROS Burst. Front Plant Sci. https://doi.org/10.3389/fpls.2017.00832
Modarresi M, Chahardoli A, Karimi N, Chahardoli S (2020) Variations of glaucine, quercetin and kaempferol contents in Nigella arvensis against Al2O3, NiO, and TiO2 nanoparticles. Heliyon 6:e04265. https://doi.org/10.1016/j.heliyon.2020.e04265
Moharrami F, Hosseini B, Sharafi A, Farjaminezhad M (2017) Enhanced production of hyoscyamine and scopolamine from genetically transformed root culture of Hyoscyamus reticulatus L. elicited by iron oxide nanoparticles. Vitro Cell Dev Biol-Plant 53:104–111. https://doi.org/10.1007/s11627-017-9802-0
Mosavat N, Golkar P, Yousefifard M, Javed R (2019) Modulation of callus growth and secondary metabolites in different Thymus species and Zataria multiflora micropropagated under ZnO nanoparticles stress. Biotechnol Appl Chem 66:316–322. https://doi.org/10.1002/bab.1727
Nair R, Varghese SH, Nair BG, Maekawa T, Yoshida Y, Kumar DS (2010) Nanoparticulate material delivery to plants. Plant Sci 179:154–163. https://doi.org/10.1016/j.plantsci.2010.04.012
Namdeo AG (2007) Plant cell elicitation for production of secondary metabolites: a review. Pharmacogn Rev 1:69–79
Narayani M, Srivastava S (2017) Elicitation: a stimulation of stress in in vitro plant cell/tissue cultures for enhancement of secondary metabolite production. Phytochem Rev 16:1227–1252. https://doi.org/10.1007/s11101-017-9534-0
Nazir S, Jan H, Zaman G, Khan T, Ashraf H, Meer B, Zia M, Drouet S, Hano C, Abbasi BH (2021) Copper oxide (CuO) and manganese oxide (MnO) nanoparticles induced biomass accumulation, antioxidants biosynthesis and abiotic elicitation of bioactive compounds in callus cultures of Ocimum basilicum. Artif Cells Nanomed Biotechnol 49:625–633. Thai basilhttps://doi.org/10.1080/21691401.2021.1984935
Nekoukhou M, Fallah S, Abbasi-Surki A, Pokhrel LR, Rostamnejadi A (2022) Improved efficacy of foliar application of zinc oxide nanoparticles on zinc biofortification, primary productivity and secondary metabolite production in dragonhead. J Clean Prod 379:134803. https://doi.org/10.1016/j.jclepro.2022.134803
Nekoukhou M, Fallah S, Pokhrel LR, Abbasi-Surki A, Rostamnejadi A (2023) Foliar enrichment of copper oxide nanoparticles promotes biomass, photosynthetic pigments, and commercially valuable secondary metabolites and essential oils in dragonhead (Dracocephalum moldavica L.) under semi-arid conditions. Sci Total Environ 863:160920. https://doi.org/10.1016/j.scitotenv.2022.160920
Oloumi H, Soltaninejad R, Baghizadeh A (2015) The comparative effects of nano and bulk size particles of CuO and ZnO on glycyrrhizin and phenolic compounds contents in Glycyrrhiza glabra L. seedlings. Indian J Plant Physiol 20:157–161. https://doi.org/10.1007/s40502-015-0143-x
Patel KV, Nath M, Bhatt MD, Dobriyal AK, Bhatt D (2020) Nanofomulation of zinc oxide and chitosan zinc sustain oxidative stress and alter secondary metabolite profile in tobacco. 3 Biotech 10:477. https://doi.org/10.1007/s13205-020-02469-x
Poornananda MN, Jameel MAK (2016) Abiotic and biotic elicitors-role in secondary metabolites production through in vitro culture of medicinal plants. In: Arun KS, Chitra S (eds) Abiotic and Biotic Stress in Plants. IntechOpen, Rijeka
Raigond P, Raigond B, Kaundal B, Singh B, Joshi A, Dutt S (2017) Effect of zinc nanoparticles on antioxidative system of potato plants. J Environ Biol 38:435. https://doi.org/10.22438/jeb/38/3/MS-209
Ramirez-Estrada K, Vidal-Limon H, Hidalgo D, Moyano E, Golenioswki M, Cusidó RM, Palazon J (2016) Elicitation, an effective strategy for the Biotechnological production of bioactive high-added value compounds in plant cell factories. Molecules 21:182. https://doi.org/10.3390/molecules21020182
Randhir R, Lin Y-T, Shetty K (2004) Stimulation of phenolics, antioxidant and antimicrobial activities in dark germinated mung bean sprouts in response to peptide and phytochemical elicitors. Process Biochem 39:637–646. https://doi.org/10.1016/S0032-9592(03)00197-3
Rezaizad MS, Abbaspour H, Hashemi-Moghaddam H, Gerami M, Ramezani M (2021) Photocatalytic activity of Titanium Dioxide Nanoparticles (TiO2) on the physiological and Phytochemical Properties of Stevia [Stevia rebaudiana (Bertoni). Bertoni] J Med plants By-products 10:169–177. https://doi.org/10.22092/jmpb.2020.342676.1205
Rivero-Montejo SdJ, Vargas-Hernandez M, Torres-Pacheco I (2021) Nanoparticles as novel elicitors to improve bioactive compounds in plants. Agriculture 11:134. https://doi.org/10.3390/agriculture11020134
Salih AM, Al-Qurainy F, Khan S, Tarroum M, Nadeem M, Shaikhaldein HO, Gaafar A-RZ, Alfarraj NS (2021) Biosynthesis of zinc oxide nanoparticles using Phoenix dactylifera and their effect on biomass and phytochemical compounds in Juniperus procera. Sci Rep 11:19136. https://doi.org/10.1038/s41598-021-98607-3
Sarkar J, Chakraborty N, Chatterjee A, Bhattacharjee A, Dasgupta D, Acharya K (2020) Green synthesized copper oxide nanoparticles ameliorate defence and antioxidant enzymes in Lens culinaris. Nanomaterials 10:312. https://doi.org/10.3390/nano10020312
Satti SH, Raja NI, Ikram M, Oraby HF, Mashwani Z-U-R, Mohamed AH, Singh A, Omar AA (2022) Plant-based titanium dioxide nanoparticles trigger biochemical and proteome modifications in Triticum aestivum L. under biotic stress of Puccinia striiformis. Molecules 27:4274. https://doi.org/10.3390/molecules27134274
Sharifi-Rad R, Esmaeilzadeh Bahabadi S, Samzadeh-Kermani A, Gholami M (2020) The effect of non-biological elicitors on physiological and biochemical properties of medicinal plant Momordica charantia L. Iran J Sci Technol Trans A: Sci 44:1315–1326. https://doi.org/10.1007/s40995-020-00939-8
Sheikhalipour M, Esmaielpour B, Gohari G, Haghighi M, Jafari H, Farhadi H, Kulak M, Kalisz A (2021) Salt stress mitigation via the foliar application of chitosan-functionalized selenium and anatase titanium dioxide nanoparticles in Stevia (Stevia rebaudiana Bertoni). Molecules 26:4090. https://doi.org/10.3390/molecules26134090
Shenavaie Zare A, Ganjeali A, Vaezi Kakhki MR, Cheniany M, Mashreghi M (2022) Plant elicitation and TiO2 nanoparticles application as an effective strategy for improving the growth, biochemical properties, and essential oil of peppermint. Physiol Mol Biol Plants 28:1391–1406. https://doi.org/10.1007/s12298-022-01215-2
Shoja AA, Çirak C, Ganjeali A, Cheniany M (2022) Stimulation of phenolic compounds accumulation and antioxidant activity in in vitro culture of Salvia tebesana Bunge in response to nano-TiO2 and methyl jasmonate elicitors. Plant Cell Tissue Organ Cult (PCTOC) 149:423–440. https://doi.org/10.1007/s11240-022-02251-2
Siddiqui ZA, Khan MR, AbdAllah EF, Parveen A (2019) Titanium dioxide and zinc oxide nanoparticles affect some bacterial diseases, and growth and physiological changes of beetroot. Int J Veg Sci 25:409–430. https://doi.org/10.1080/19315260.2018.1523267
Singh OS, Pant NC, Laishram L, Tewari M, Dhoundiyal R, Joshi K, Pandey C (2018) Effect of CuO nanoparticles on polyphenols content and antioxidant activity in Ashwagandha (Withania somnifera L. Dunal). J Pharmacogn Phytochem 7:3433–3439
Tabarifard M, Cheniany M, Khalilian-movahhed M (2023) Artificial neural network prediction and comparative evaluation of pharmaceutical important flavones and antioxidant compositions in Teucrium polium callus culture elicited with methyl jasmonate and TiO2 nanoparticles. Res Square. https://doi.org/10.21203/rs.3.rs-2396091/v1
Tariverdizadeh N, Mohebodini M, Chamani E, Ebadi A (2021) Iron and zinc oxide nanoparticles: an efficient elicitor to enhance trigonelline alkaloid production in hairy roots of fenugreek. Ind Crops Prod 162:113240. https://doi.org/10.1016/j.indcrop.2021.113240
Teoh ES (2015) Secondary metabolites of plants. Med Orchids Asia. https://doi.org/10.1007/978-3-319-24274-3_5
Tholl D (2015) Biosynthesis and biological functions of terpenoids in plants. Biotechnol isoprenoids. https://doi.org/10.1007/10_2014_295
Toffolatti SL, Maddalena G, Passera A, Casati P, Bianco PA, Quaglino F (2021) 16 - Role of terpenes in plant defense to biotic stress. In: Jogaiah S (ed) Biocontrol Agents and Secondary Metabolites. Woodhead Publishing, Sawston, pp 401–417
Tripathi DK, Singh S, Singh VP, Prasad SM, Dubey NK, Chauhan DK (2017) Silicon nanoparticles more effectively alleviated UV-B stress than silicon in wheat (Triticum aestivum) seedlings. Plant Physiol Biochem 110:70–81. https://doi.org/10.1016/j.plaphy.2016.06.026
Ullah A, Munir S, Badshah SL, Khan N, Ghani L, Poulson BG, Emwas A-H, Jaremko M (2020) Important flavonoids and their role as a therapeutic agent. Molecules 25:5243. https://doi.org/10.3390/molecules25225243
Velázquez-Gamboa MC, Rodríguez-Hernández L, Abud-Archila M, Gutiérrez-Miceli FA, González-Mendoza D, Valdez-Salas B, González-Terreros E, Luján-Hidalgo MC (2021) Agronomic biofortification of Stevia rebaudiana with zinc oxide (ZnO) phytonanoparticles and antioxidant compounds. Sugar Tech 23:453–460. https://doi.org/10.1007/s12355-020-00897-w
Velderrain-Rodríguez GR, Palafox-Carlos H, Wall-Medrano A, Ayala-Zavala JF, Chen CYO, Robles-Sánchez M, Astiazaran-García H, Alvarez-Parrilla E, González-Aguilar GA (2014) Phenolic compounds: their journey after intake. Food Funct 5:189–197. https://doi.org/10.1039/C3FO60361J
Wani KI, Choudhary S, Zehra A, Naeem M, Weathers P, Aftab T (2021) Enhancing artemisinin content in and delivery from Artemisia annua: a review of alternative, classical, and transgenic approaches. Planta 254:29. https://doi.org/10.1007/s00425-021-03676-3
Yarizade K, Hosseini R (2015) Expression analysis of ADS, DBR2, ALDH1 and SQS genes Artemisia vulgaris hairy root culture under nano cobalt and nano zinc elicitation. Ext J App Sci 3:69
Zhang B, Zheng LP, Wang JW (2012) Nitric oxide elicitation for secondary metabolite production in cultured plant cells. Appl Microbiol Biotechnol 93:455–466. https://doi.org/10.1007/s00253-011-3658-8
Funding
The authors did not receive support from any organization for the submitted work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Authors declare there is no conflict of interest.
Additional information
Communicated by Jericó Jabín Bello-Bello.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Inam, M., Attique, I., Zahra, M. et al. Metal oxide nanoparticles and plant secondary metabolism: unraveling the game-changer nano-elicitors. Plant Cell Tiss Organ Cult 155, 327–344 (2023). https://doi.org/10.1007/s11240-023-02587-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-023-02587-3