Abstract
Upon mechanical wounding, plants locally induce necrosis, accumulate methyl jasmonate (MeJA) and acquire systemic resistance in nearby tissues. One-month-old in vitro grown Panax ginseng seedlings were treated with either 50 µM MeJA or mechanical wounding alone or a combination of both, to evaluate jasmonic acid (JA) signaling and terpene biosynthetic pathway genes along with terpenoid accumulation. After MeJA treatment, JA pathway genes, such as lipoxygenase (PgLOX), hydrogen peroxidase lyase (PgHPL), allene oxide synthase (PgAOS), and allene oxide cyclase (PgAOC1), and terpene pathway genes, such as isopentenyl diphosphate isomerase (PgIPP) and farnesyl diphosphate synthase (PgFPS), were highly expressed and resulted in the accumulation of mono- and sesquiterpenes. During mechanical wounding, PgLOX expression was induced relatively late after 72 h of treatment, however PgAOC1 was not induced. This resulted in decreased production of MeJA that in turn may have lowered terpenoid production. In contrast, wounding + MeJA treatment increased PgAOC1 and PgLOX gene expression earlier after 6 h and slowly promoted the production of mono- and sesquiterpenes. Furthermore, we monitored the effect of MeJA upon wounding in in vitro grown 1-month-old seedlings treated with MeJA + wounding. These result demonstrated that exogenous MeJA is able to promote recovery from the wounding effect by functioning as a long distance signal. Additionally, these results suggest that exogenous MeJA supplied at the time of mechanical wounding prevents necrosis in the ginseng leaves by increasing the production of terpenoids.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Mechanical wounding is one of the major threats to the survival of all living organisms. In plants, wounding responses have been widely studied. Plants in the wild seldom escape some degree of damage from environmental stresses, such as wind, hail, sand and rain (Graham et al. 1986). When plants get wounded, each wounded cell makes competent each other for the activation of defense responses, which largely depend on the activation of specific genes (León et al. 2001), by triggering specific signal transduction pathways. In Arabidopsis, mechanical wounding increases the accumulation of endogenous jasmonic acid (JA) and the release of cell wall derived oligogalacturonides (OGA) that tend to activate wound responsive (WR) and jasmonate responsive (JR) genes (Rojo et al. 1998). Local release of JA at the wound site systemically increases resistance in distal parts of the plant, indicating that JA plays a role as a long distance signal. JA biosynthesis via the octadeconoid pathway starts with the release of linolenic acid from membranes and induces the production of various secondary metabolites, including volatiles (Thines et al. 2007; Chini et al. 2007; Staswick 2008). Plant damage can also remarkably increase the accumulation of volatiles, including mono- and sesquiterpenes.
Terpenes are a group of volatile compounds present at a basal level in undamaged plants. Their accumulation increases in response to various biotic and abiotic stresses. In particular, JA acts as an intermediate signal transduction molecule and increases the production of volatiles in both maize and rice (Schmelz et al. 2003; Xu et al. 2003; Cheng et al. 2007). In Panax ginseng C.A. Meyer, mono- and sesquiterpenes are the most important wide spread groups of secondary metabolites. The biosynthesis of terpenes through activated 5-carbon isoprenoid units, isopentenyl diphosphate (IPP) and dimethylallyl diphosphate (DMAPP), is derived from the acetate/mevalonate or methylerythritol phosphate pathways (for review, see Chappell 1995; Gershenzon and Kreis 1999). The C5 units are condensed by prenyltransferases to form elongated linear prenyl diphosphates, such as geranyl diphosphate (GPP, a C10 compound) and farnesyl diphosphate (FPP, a C15 compound), which are the precursors of mono- and sesquiterpenes, respectively. The prenyl diphosphates in turn undergo a wide range of cyclizations and other modifications to produce the various structural types of mono- and sesquiterpenes.
Pests, including weeds, insects, diseases, and rodents, are the greatest concern of ginseng growers. It is highly probable that ginseng, as a perennial herb, is exposed to various biotic and abiotic conditions. Hence, avoiding stressful conditions while cultivating ginseng is a serious concern among ginseng farmers. In Arabidopsis, wounding-induced endogenous methyl jasmonate (MeJA) caused systemic resistance but failed to exhibit local resistance. There are large amounts of data showing that MeJA increases the production of terpenoids in various plants. For e.g. in Catharanthus roseus combined elicitor treatment is used for the enhancement of terpenoid indole alkaloids (Almagro et al. 2014). However, to date, no data exist for MeJA-dependent regulation of terpenoids in P. ginseng. Furthermore, no report has shown that exogenously supplied MeJA can prevent the physical local damage caused by wounding. Hence, the present study was designed to understand how plants resist wounding when exogenous MeJA is simultaneously supplied.
To this end, P. ginseng seedlings were treated with exogenously supplied MeJA and wounding, alone or in the combination, to investigate the role of exogenous MeJA on plant mechanical damage. To understand terpenoid regulation and the JA pathway at the molecular level, we first studied the expression profiles of various mono- and sesquiterpene biosynthetic pathway related genes, such as PgFPS and PgIPPI, and JA biosynthetic pathway related genes, such as PgLOX, PgHPL, PgAOS, PgAOC1, and PgAOC2. We also quantified mono- and sesquiterpene accumulation.
Materials and methods
Plant growth conditions
The P. ginseng seeds were obtained from the Korean Ginseng Resource Bank, South Korea. Ginseng seeds were surface sterilized in 70 % ethanol for 1 min, washed with 2 % sodium hypochlorite solution for 15 min with occasional shaking and then washed with sterile distilled water 3–5 times. After carefully dissecting the zygotic embryos, they were placed on ½ MS media (Duchefa Biochemie, The Netherlands), 3 % sucrose, and 0.7 % agar, pH 5.8 for 1 week. Later, germinated seedlings were transferred to solidified full MS media with 3 % sucrose and supplemented with gibberellic acid (30 mg/ml) at 25 °C under a 16 h photoperiod for 3 weeks.
Wounding and MeJA treatments
For wounding treatment, 1-month-old in vitro grown ginseng seedlings were wounded in all parts of the seedling using a sterile, sharp scalpel and placed in ½ MS liquid medium. For MeJA treatments, seedlings were just placed in ½ MS liquid medium containing 50 µM MeJA. For the combined treatment, seedlings were first wounded and then placed in ½ MS liquid medium containing 50 µM MeJA. Control group seedlings were placed in ½ MS medium without undergoing wounding treatment or supplied with any MeJA. All the treated and untreated seedlings were grown at 25 °C under a 16 h photoperiod and harvested after 24, 48, 72, 96 h, 8 and 15 days. The experiments were repeated twice with similar results. For gene expression studies and terpene analysis, plants were wounded and treated as described above, harvested at different time points, immediately frozen in liquid nitrogen and stored at −70 °C for future experiments.
Gene expression analysis by RT-PCR and qRT-PCR analyses
RNA was extracted from P. ginseng organs that had been subjected to all experimental treatments using the RNeasy kit (Qiagen, USA), according to the manufacturer’s instructions. The quality and concentration of RNA was measured using a spectrophotometer (GE Nanovalue, USA). To obtain first strand cDNA, 1 μg of total RNA was used, and cDNA was synthesized using a Power cDNA kit (Invitrogen, USA) following the supplied instructions. For semi-quantitative reverse transcription PCR (RT-PCR), the P. ginseng actin gene (PgActin) was used as a constitutively expressed control gene. The initial PCR was performed with PgActin primers, product levels were compared, and 100 ng of cDNA samples were used to ensure equal template concentrations. PCR was subsequently performed for 30 amplification cycles using primers specific for the genes of interest. PgActin gene specific primers were always included in each experiment.
We performed qRT-PCR using a real-time rotary analyzer (Rotor-Gene 6000; Corbett Life Science, Australia) using 100 ng of cDNA in a 10 μl reaction volume, using SYBR® Green SensiMix Plus Master Mix (Quantace, England), and the gene specific primers listed in Table 1. The housekeeping gene encoding the PgActin protein was used as a standard for all samples. For transcript analysis of stress treated samples, untreated samples were used as negative controls. A melting point analysis of PCR products was carried out, which resulted in a single peak, indicating the presence of a single PCR product. The melting temperatures varied from 78.6 to 80 °C with no clear differences between the treatments. The PCR conditions for each of the 40 cycles were 95 °C for 10 s, 60 °C for 10 s, and 72 °C for 20 s. Fluorescence was detected and measured in the real-time reverse transcriptase PCR thermocycler, and the geometric increase of the fluorescence corresponding to an exponential increase of the product was used to determine the threshold cycle (Ct) in each reaction using the formula 2−ΔΔCt. All real time reverse transcriptase PCR experiments were performed in triplicate.
Mono- and sesquiterpene analysis by GC–MS
1 g of fresh sample was pulverized in liquid nitrogen using a mortar and pestle, and then extracted sequentially with 5 ml of hexane:ethyl acetate (85:15) twice. The extracts were combined and purified by two rounds of silica column chromatography. The extract was applied to a silica column (230 mm in length) and washed with 6 ml of fresh hexane:ethyl acetate (85:15). The collected eluent was then applied to a second silica column and washed with 3 ml of hexane:ethyl acetate (85:15) to elute hydrocarbons and oxygenated compounds. The final eluent (approximately 20 ml) was concentrated to 1–2 ml under nitrogen before analysis by GC-MS. Mono- and sesquiterpenes in the samples were quantified using a Thermo Finnigan DSQ GC/MS system, as described by Lee and Chappell (2008).
Results
Morphological changes in wounded ginseng leaves under MeJA treatment
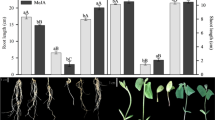
Morphological changes in in vitro grown ginseng seedlings were evaluated 24, 48, 72, 96 h, 8 and 15 days after wounding alone or in combination with exogenously supplied MeJA. The seedlings subjected to wounding showed damage effects as early as 6 h post-treatment (data not shown). Upon prolonged exposure, the lesion surface areas tended to show a gradual increase 24 h after wounding. The wounded surface areas increased over time and reached a peak at 15 days, resulting in a fully necrotized phenotype. Interestingly, exogenous MeJA supplied at the time of wounding decreased necrosis and did not induce necrosis even 15 days after treatment in ginseng seedling leaves (Fig. 1).
Recovery from mechanical wounding damage by exogenously supplied MeJA. Representative images of in vitro grown 1-month-old P. ginseng seedlings treated with 50 µM MeJA, wounding and a combination of wounding + 50 µM MeJA. Arrows indicate the wounding site in P. ginseng seedlings. Images were captured 24, 48, 72, 96 h, 8 and 15 days after treatment
Changes in the gene expression profiles of JA pathway and terpenoid biosynthetic genes
To determine whether terpenoid gene expression is mediated through the JA pathway, we monitored changes in terpenoid-related genes, such as PgIPPI and PgFPS, along with JA pathway genes, such as PgLOX, PgHPL, PgAOS, PgAOC1 and PgAOC2, in in vitro grown 1-month-old ginseng seedlings treated with MeJA, or wounding and the combination of both treatments by semi-quantitative PCR and qRT-PCR (Figs. 2, 3). qRT-PCR was carried out only for certain genes like PgLOX, PgIPPI and PgFPS (Fig. 2). Changes in the expression patterns of genes under various stresses also proved their roles in defense mechanisms by crosstalk between the JA and terpene pathways. Upon MeJA treatment, expression of PgIPPI was slightly upregulated, reached a maximum level at 48 h (13.9-fold) and declined gradually at 72 h post-treatment (Fig. 2a). After wounding, the PgIPPI transcripts were expressed at their highest level at 6 h (16.6-fold) and 24 h (15.6-fold), with decreased levels observed 12, 48 and 72 h post-treatment. The expression pattern of PgIPPI under MeJA + wounding showed a slight fluctuation compared to 0 h, and showed the highest levels of expression at 12 and 24 h. The PgFPS transcript increased gradually and reached a maximum 72 h (14.7-fold) after MeJA treatment (Fig. 2b). Due to the combination of wounding and MeJA, PgFPS transcripts started to show increased expression beginning at 6 h, reached a maximum level at 48 h and declined thereafter. The expression of PgFPS increased gradually, was induced 18.08-fold at 48 h and then gradually decreased in wounded leaves. MeJA increased PgLOX gene expression at 6 h and showed maximum accumulation with a 17.36-fold increase at 72 h (Fig. 2c). Upon wounding, PgLOX expression was highest at 72 h, with an average 16.78-fold increase. PgHPL and PgAOS were induced by wounding, MeJA and wounding + MeJA treatments. PgAOC2 seemed to be upregulated by MeJA treatment, but downregulated by wounding. Interestingly, expression of PgAOC2 seemed to be activated by MeJA treatment. However, AOC1 did not show any significant increase or decrease in gene expression (Fig. 3).
Differential expression levels of terpene pathway related genes and PgLOX in response to MeJA, wounding and a combination of wounding + MeJA treatments. RNA was isolated from treated seedlings at the indicated times, and the expression levels of the indicated mRNAs were determined by qRT-PCR. The Ct values for the tested genes were normalized to the Ct value for β-actin and calculated relative to the calibrator using the formula 2∆∆CT. Data represent the mean ± SD for three independent replicates
Altered pattern of jasmonic acid biosynthetic gene expression in P. ginseng seedlings treated with MeJA, wounding and a combination of wounding + MeJA. RNA was isolated from treated seedlings at the indicated times, and the expression levels of lipoxygenase (PgLOX), hydrogen peroxidase lyase (PgHPL), allene oxide synthase (PgAOS), allene oxide cyclase (PgAOC) were determined by RT-PCR
Production of mono-and sesquiterpenes
During the 6–48 h post-treatment period, the production of total mono and sesquiterpenes increased as a result of MeJA treatment, wounding or a combination of both treatments. Ginseng seedlings treated with MeJA alone showed maximum accumulation of terpenes at 48 h (levels were higher than those of wounding alone, or in combination with wounding) (Fig. 4a). In comparison with MeJA, wounding resulted in less accumulation (5.9 %; however, this was higher than untreated controls) of mono- and sesquiterpenes (Fig. 4b). Interestingly but not surprisingly, seedlings treated with MeJA simultaneously after wounding showed slightly higher accumulations of mono- and sesquiterpenes (11.8 %) but less accumulation than in response to MeJA treatment (Fig. 4c).
Percentage of total terpenoids in P. ginseng seedlings treated with MeJA, wounding and a combination of wounding + MeJA. Twenty-one-day-old in vitro grown seedlings treated with 50 µM MeJA, wounding and a combination of wounding + 50 µM MeJA were collected at different time intervals for mono and sesquiterpene analysis. Means of three independent replicates were statistically analyzed and compared with controls (*p < 0.05; **p < 0.01; ***p < 0.001) using a Student’s t test
Geraniolene, limonene, panasinsene, elemene, farnesane, alpha carophyllene, beta carophyllene, germacrene, bulsenol, citronellal, and falcrinol were all detectable in in vitro grown ginseng seedlings treated with MeJA. The wounding treatment resulted in less production of mono- and sesquiterpenes when compared to MeJA treatment alone or in combination. Only six different terpenes, including geraniolene, panasinsene, elemene, farnesane, alpha-carophyllene, benzoquin, were identified in wounded samples.
Discussion
The present study describes the influence of MeJA and mechanical wounding alone or in combination on volatile terpene biosynthesis. All three treatments individually resulted in an increase in terpenoid accumulation. However, each treatment accumulated different level of terpenes in the following order: MeJA > MeJA + wounding > wounding. From a biotechnological point of view, the relevant advantage with respect to damage recovery of using exogenous MeJA may contribute to plant wellbeing by preventing cell damage caused by mechanical wounding. The exposure of in vitro grown ginseng seedlings to wounding resulted in lower terpene levels compared to other treatments; the latter being prevalently released when ginseng seedlings were treated with MeJA in combination with wounding. However, seedlings treated with MeJA predominantly accumulated high levels of terpenes. These results demonstrate exogenous MeJA is able to help the plant recover from wounding stress by producing high levels of volatiles.
The effect of MeJA on terpene metabolite accumulation has been extensively studied in Norway spruce (Erbilgin et al. 2006; Zeneli et al. 2006). In our study, we determined the expression of terpene related genes, such as PgIPPI and PgFPS, terpene metabolite accumulation and JA mediated pathway genes, like PgLOX, PgHPL, PgAOS, PgAOC1 and PgAOC2, after treating seedlings with MeJA. Upon MeJA treatment, terpene-related gene transcripts and PgLOX (a key enzyme in the JA biosynthesis pathway) mRNA levels were upregulated, and resulted in the highest level of terpenoid accumulation, relative to untreated controls. The PgHPL, PgAOS, and PgAOC1 transcripts accumulated to their highest levels as a result of this treatment. With respect to the metabolite profile, seedlings treated with MeJA accumulated significant amounts of the farnesane and beta-carophyllene sesquiterpenes and others in trace amounts, compared to controls. The largest increase in both terpenoid and gland production was observed in plants treated with JA, a ubiquitous plant hormone known to mediate defense responses to biotic and abiotic stresses. Several hundred studies have demonstrated that exogenous application of JA induces defensive responses (Heil 2008; Wasternack 2007; Farmer et al. 2003), while no data has been published on the effect of MeJA on the biosynthesis of terpenoids in P. ginseng.
Mechanical wounding increases endogenous MeJA at the wounded site, which can act as a long distance signal and result in systemic resistance by inducing a broad spectrum of defense-related compounds, such as volatiles, alkaloid production, trichome formation and secretion of extrafloral nectar (Heil and Ton 2008; Chini et al. 2007). Terpenoids are the largest group of plant secondary metabolites and play vital roles in plant–microbe, plant-herbivore, plant–plant, and plant–environment interactions (Chappell 1995; Mahmoud and Croteau 2002; Pichersky and Gershenzon 2002; Aharoni et al. 2003). When plants are damaged by herbivores, volatiles released from the damaged portion of the plant vaporize and induce defensive responses in neighboring tissues or in other plants. Upon wounding stress in P. ginseng, terpene pathway-related gene transcripts seemed to be up-regulated at different time periods. When considering the JA pathway genes, PgLOX showed late but strong expression 72 h after wounding. Interestingly, PgAOC1, a downstream gene of the JA pathway, was expressed at lower levels after wounding, compared to after MeJA treatment. Therefore, the total production of mono- and sesquiterpenes was reduced compared to MeJA and wounding + MeJA treatments. The wounded seedlings also showed increased mono- and sesquiterpenes at all-time points, however, significant increases were observed only after 24 h of treatment. Among the different type of sesquiterpenes, only farnesene showed a significant increase compared to the level of untreated controls; the other terpenes, such as geraniolene, panasinsene, elemene, alpha-carophyllene, and benzoquin, accumulated but the levels were not significantly greater than those found in untreated controls. As a result, wounded plants showed local necrosis and failed to exhibit local resistance due to decreased MeJA production.
Panax ginseng seedlings exposed to MeJA at the time of wounding were able to recover from the damage caused by mechanical wounding, likely by inducing the production of volatiles. Furthermore, increases in PgLOX transcript levels after MeJA + wounding occurred much earlier than upon wounding stress and it is interesting to note that the PgAOC1 transcript also increased when wounded plants were supplied with MeJA. This result may be due to the fact that necrosis caused by wounding stress can be recovered by increased production of MeJA. Terpene-related gene transcripts, such as PgIPPI and PgFPS, were also up-regulated at the earlier time points, indicating that MeJA is a key factor in modulating terpene genes at early times and in the accumulation of higher levels of terpenes, compared to the levels induced by wounding stress (Fig. 5).
Model illustrating the possible mechanism of the response to MeJA, wounding and a combination of wounding + MeJA treatments in ginseng plants. Wounding induced local MeJA which can act as a long distance signal and increase the levels of volatiles in systemic tissues, resulting in resistance in distal parts of the plants but also in severe wounding damage at the local site. In contrast, exogenous MeJA mimicked endogenous JA and resulted in local resistance, thereby preventing wounding-induced necrosis by increasing volatile emissions
The present results in P. ginseng demonstrated that necrosis caused by mechanical wounding at damaged sites can be recovered by exogenously supplied MeJA, thereby proving that MeJA acts as the long distance signal and plays a defensive role by producing systemic resistance, as suggested in other plant systems.
Abbreviations
- JA:
-
Jasmonic acid
- MeJA:
-
Methyl jasmonate
- OGA:
-
Oligogalacturonides
- WR:
-
Wound responsive
- IPPI:
-
Isopentenyl diphosphate isomerase
- DMAPP:
-
Dimethylallyl diphosphate
- GPP:
-
Geranyl diphosphate
- FPS:
-
Farnesyl diphosphate synthase
- LOX:
-
Lipoxygenase
- HPL:
-
Hydrogen peroxidase lyase
- AOC:
-
Allene oxide cyclase
- AOS:
-
Allene oxide synthase
- GC–MS:
-
Gas chromatography mass spectrometry
- RT-PCR:
-
Reverse transcription PCR
- qRT-PCR:
-
Quantitative real time-reverse transcriptase polymerase chain reaction
References
Aharoni A, Giri AP, Deuerlein S, Griepink F, De Kogel WJ, Verstappen FW, Verhoeven HA, Jongsma MA, Schwab W, Bouwmeester HJ (2003) Terpenoid metabolism in wild-type and transgenic Arabidopsis plants. Plant Cell 15:2866–2884
Almagro L, Gutierrez J, Pedreno MA, Sottamayor M (2014) Synergistic and additive influence of cyclodextrins and methyl jasmonate on the expression of the terpenoid indole alkaloid pathway genes and metabolites in Catharanthus roseus. Plant Cell Tissue Organ Cult 119:543–551
Chappell J (1995) The biochemistry and molecular biology of isoprenoid metabolism. Plant Physiol 107:1–6
Cheng AX, Xiang CY, Li JX, Yang CQ, Hu WL, Wang LJ, Lou YG, Chen XY (2007) The rice (E)-beta-caryophyllene synthase (OsTPS3) accounts for the major inducible volatile sesquiterpenes. Phytochemistry 68:1632–1641
Chini A, Fonseca S, Fernandez G, Adie B, Chici JM, Lorenzo O et al (2007) The JAZ family of repressors is the missing link in jasmonate signaling. Nature 448:666–672
Erbilgin N, Christiansen E, Krokene P, Zeneli G, Gershenzon J (2006) Exogenous application of methyl jasmonate elicits defences in Norway spruce (Picea abies) and reduces host colonization by the bark beetle Ips typographus. Oecologia 148:426–436
Farmer EE, Alméras E, Krishnamurthy V (2003) Jasmonates and related oxylipins in plant responses to pathogenesis and herbivory. Curr Opin Plant Biol 6:372–378
Gershenzon J, Kreis JW (1999) Biochemistry of terpenoids: monoterpenes, sesquiterpenes, diterpenes, sterols, cardiac glycosides, and steroid saponins. In: Wink M (ed) Biochemistry of plant secondary metabolism: annual plant reviews, vol 21. Sheffield Academic Press, Sheffield, pp 222–299
Graham JS, Hall G, Pearce G, Ryan CA (1986) Regulation of synthesis of proteinase inhibitors I and II mRNAs in leaves of wounded tomato plants. Planta 169:399–405
Heil M (2008) Indirect defense via tritrophic interactions. New Phytol 178:41–61
Heil M, Ton J (2008) Long-distance signaling in plant defense. Trends Plant Sci 13:264–272
Lee S, Chappell J (2008) Biochemical and genomic characterization of terpene synthases in Magnolia grandiflora. Plant Physiol 147:1017–1033
León J, Rojo E, Sánchez-Serrano JJ (2001) Wounding signalling in plants. J Exp Bot 52(354):1–9
Mahmoud SS, Croteau RB (2002) Strategies for transgenic manipulation of monoterpene biosynthesis in plants. Trends Plant Sci 7:366–373
Pichersky E, Gershenzon J (2002) The formation and function of plant volatiles: perfumes for pollinator attraction and defense. Curr Opin Plant Biol 5:237–243
Rojo E, Titarenko E, Leon J, Berger S, Vancanneyt G, Sanchez-Serrano JJ (1998) Reversible protein phosphorylation regulates JA-dependent and -independent wound signal transduction pathways in Arabidopsis thaliana. Plant J 13:153–287
Schmelz EA, Alborn HT, Banchio E, Tumlinson JH (2003) Quantitative relationships between induced jasmonic acid levels and volatile emission in Zea mays during Spodoptera exigua herbivory. Planta 216:665–673
Staswick PE (2008) JAZing up jasmonate signaling. Trends Plant Sci 13:66–71
Thines B, Katsir L, Melotto M, Niu Y, Mandaokar A, Liu G et al (2007) JAZ repressor proteins are targets of the SCF COI1 complex during jasmonate signaling. Nature 448:661–666
Wasternack C (2007) Jasmonates: an update on biosynthesis, signal transduction and action in plant stress response, growth a development. Ann Bot (Lond) 100:681–697
Xu T, Zhou Q, Chen W, Zhang GR, He GF, Zhang WQ (2003) Involvement of jasmonate-signaling pathway in the herbivore-induced rice plant defense. Chin Sci Bull 48:1982–1987
Zeneli G, Krokene P, Christiansen E, Krekling T, Gershenzon J (2006) Methyl jasmonate treatment of mature Norway spruce (Picea abies) trees increases the accumulation of terpenoid resin components and protects against infection by Ceratocystis polonica, a bark beetle-associated fungus. Tree Physiol 26:977–988
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Balusamy, S.R.D., Rahimi, S., Sukweenadhi, J. et al. Exogenous methyl jasmonate prevents necrosis caused by mechanical wounding and increases terpenoid biosynthesis in Panax ginseng . Plant Cell Tiss Organ Cult 123, 341–348 (2015). https://doi.org/10.1007/s11240-015-0838-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-015-0838-8