Abstract
As part of a survey of the parasites infecting the fishes of Moreton Bay, Queensland, Australia, the gill lamellae of two species, the common silver-biddy Gerres oyena (Forsskål) and the common silver belly Gerres subfasciatus Cuvier (both Gerreidae), were found infected with monogenoids as follows: Metahaliotrema filamentosum Venkatanarasaiah, 1981 (Dactylogyridae) was redescribed from G. oyena and recorded from G. subfasciatus (both new host records); Metahaliotrema imparilis n. sp., Protogyrodactylus monacanthus n. sp., Protogyrodactylus vulgaris n. sp. (all Dactylogyridae), Gyrodactylus xynus n. sp. (Gyrodactylidae), and Polylabris gerres (Sanders, 1944) Mamaev & Parukin, 1976 (Microcotylidae) were reported from G. subfasciatus; and Protogyrodactylus scalmophorus n. sp., Protogyrodactylus ichthyocercus n. sp., and Protogyrodactylus similis n. sp. were recorded from G. oyena. The new species are described, and the finding of M. filamentosum and P. gerres from Moreton Bay represent new geographic records. Haliotrema digyroides Zhang, 2001 and Haliotrema subancistroides Zhang, 2001 are transferred to Metahaliotrema Yamaguti, 1953 as Metahaliotrema digyroides (Zhang, 2001) n. comb. and Metahaliotrema subancistroides (Zhang, 2001) n. comb., respectively.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The Gerreidae includes 53 species of primarily marine fishes occurring in coastal waters of warm seas. Commonly known as mojarras or silver-biddies, the family currently contains eight genera: Gerres (with 28 species), Eucinostomus (10 species), Eugerres (6 species), Diapterus (4 species), Parequula (2 species), Pentapion (monotypic), Deckertichthys (monotypic), and Ulaema (monotypic) (Froese & Pauly, 2017; Eschmeyer & Fong, 2018). Although some gerreids are economically significant and represent important food sources in some regions of the world, only 20 gerreid species have been examined and reported as hosts for species of Monogenoidea (Table 1).
During a survey initiated by Drs Thomas Cribb and Scott Cutmore, University of Queensland, Australia, during 2016 to determine the parasite diversity in the fishes of Moreton Bay, Queensland, Australia, two species of gerreids, the common silver-biddy Gerres oyena (Forsskål) and the common silver belly Gerres subfasciatus Cuvier, were collected and examined for monogenoids. The present paper, representing the second installment recording the monogenoidean diversity in Moreton Bay, includes the report or description of two previously described and seven new species representing three monogenoidean families, the Dactylogyridae Bychowsky, 1933, Gyrodactylidae van Beneden & Hess, 1863, and Microcotylidae Taschenberg, 1879. In the first installment (Kritsky, 2018), Hareocephalus thiasae Young, 1969 and six new species of Hemirhamphiculus Bychowsky & Nagibina, 1969 (all Dactylogyridae) were recorded from three species of beloniform fishes in Moreton Bay.
Materials and methods
Specimens of the common silver-biddy and the common silver belly were collected by various methods during January 2016 and examined for monogenoids from Moreton Bay, Queensland, Australia. The fishes were transported alive to the Moreton Bay Research Station located in Dunwich, North Stradbroke Island, Queensland, where they were euthanized and identified; gill baskets were immediately removed and placed in separate vials containing hot (60 °C) sea water to relax and kill the attached helminths. A volume of 10% formalin, equal to that of the sea water in each container, was then added for fixation in 5% formalin; vials containing the preserved gills and sediments were later shipped to Idaho State University for study. Monogenoids were subsequently removed from the gills or sediment using a small probe and dissecting microscope. Some specimens were mounted unstained in Gray & Wess medium for study of sclerotized structures; other specimens were stained with Gomori’s trichrome or VanCleave’s hematoxylin (Kritsky et al., 1978; Pritchard & Kruse, 1982) and mounted in Canada balsam for observing internal anatomy. Illustrations were prepared with the aid of a camera lucida or microprojector. Measurements, all in micrometres, represented straight-line distances between extreme points and were expressed as the range followed by the mean and number (n) of structures measured in parentheses; body length included that of the haptor. Direction of the coil of the male copulatory organ (MCO), clockwise vs counterclockwise, was determined using the method of Kritsky et al. (1985). Scientific and common names of hosts were those presented by Johnson (2010); each name was verified in Froese & Pauly (2017) and Eschmeyer & Fong (2018). Type- and voucher specimens of helminths were deposited in the Queensland Museum, Brisbane, Australia (QM); the Invertebrate Zoology Collection, National Museum of Natural History, Smithsonian Institution, Washington, D. C. (USNM); and the University of Nebraska State Museum, Harold W. Manter Laboratory, Lincoln, Nebraska (HWML) as indicated in the following species accounts.
Results
The gill lamellae of seven specimens of each of two gerreid species, G. oyena and G. subfasciatus, were examined for monogenoids. All specimens of both species were infected with one or more helminth species assigned to either Protogyrodactylus Johnston & Tiegs, 1922 (five species) and Metahaliotrema Yamaguti, 1953 (two species) (both Dactylogyidae), Gyrodactylus von Nordmann, 1832 (one species) (Gyrodactylidae) or Polylabris Euzet & Cauwet, 1967 (one species) (Microcotylidae). The minimum prevalence (see Kritsky et al., 2013) for each helminth species encountered during the present study was recorded in the following species accounts.
Subclass Polyonchoinea Bychowsky, 1937
Order Dactylogyridea Bychowsky, 1937
Dactylogyridae Bychowsky, 1933
Metahaliotrema filamentosum Venkatanarasaiah, 1981
Type-host: Gerres filamentosus Cuvier (Gerreidae), whipfin silver-biddy.
Type-locality: Bay of Bengal, Kakinada, Andhra Pradesh, India.
Current records: Gerres oyena (Forsskål) (Gerreidae), common silver-biddy: Moreton Bay off Dunwich, North Stradbroke Island, Queensland, Australia (27°50′S, 153°40′E), 18.i.2016; Moreton Bay off Green Island, Queensland, Australia (27°26′S, 153°20′E), 12 and 21.i.2016. Gerres subfasciatus Cuvier (Gerreidae), common silver belly: Moreton Bay off Dunwich, North Stradbroke Island, Queensland, Australia (27°50′S, 153°40′E), 18.i.2016; Moreton Bay off Green Island, Queensland, Australia (27°26′S, 153°20′E), 12 and 21.i.2016; Moreton Bay off Amity Point, North Stradbroke Island, Queensland, Australia (27°24′S, 153°20′E), 11.i.2016.
Previous record: There have been no previous records for the species other than that of the original description of the species by Venkatanarasaiah (1981).
Site in host: Gill lamellae.
Minimum prevalence: 100% (7 or 7 G. oyena and 7 of 7 G. subfasciatus infected).
Specimens studied: 61 voucher specimens (from G. oyena), QM G237376-G237385), USNM 1484996-1485042, HWML 139452; 17 voucher specimens (from G. subfasciatus), QM G237386-G237389, USNM 1485043-1485055.
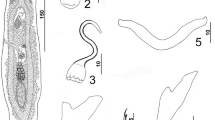
Redescription (Figs. 1–11)
Based on specimens from G. oyena and verified with specimens from G. subfasciatus. Body proper fusiform, flattened dorsoventrally. Tegument smooth. Cephalic region moderately broad, with 2 poorly developed bilateral cephalic lobes and rounded terminal lobe. Head organs poorly defined; bilateral groups of cephalic-gland cells posterolateral to pharynx. Two pairs of eyespots; members of posterior pair slightly closer together, somewhat larger than those of anterior pair, each with small inconspicuous lens often obscured by chromatic granules; members of anterior pair lacking lenses; accessory granules few or absent in cephalic region, infrequently observed in anterior trunk. Pharynx subspherical; oesophagus short; intestinal caeca uniting posterior to testis. Peduncle broad, gently tapered posteriorly; haptor subhexagonal, with 2 well-developed bilateral lobes. Ventral anchor with broad slightly depressed superficial root, knob-like deep root, gently curved shaft, elongate point extending slightly past level of tip of superficial root; ventral-anchor sclerite small, globose. Dorsal anchor with elongate slightly depressed superficial root, poorly developed deep root, slightly arcing shaft, point extending to near level of tip of superficial root. Ventral bar broadly U-shaped when observed in dorsoventral view, with slightly expanded ends directed laterally (Fig. 10); appearing rod-shaped with medial expansion in anteroposterior view (Fig. 9). Dorsal bar rod-shaped, with slight medial expansion. Hook with short upright thumb, delicate point, uniform shank; filamentous hook (FH) loop about 3/4 shank length. Gonads intercaecal, tandem, germarium pretesticular. Testis ovate. Proximal vas deferens, seminal vesicle not observed; small prostatic reservoir lying near base of MCO. Copulatory complex comprising basally articulated MCO and accessory piece. MCO a coiled tube having about 1¼ counterclockwise ring and slightly expanded base originating from variable fleshy-appearing flap. Accessory piece variable depending on orientation, comprising proximal articulation process and distal expanded portion having an elongate arm usually looping dorsally around distal shaft of MCO (Fig. 4) and a distal hook-like projection (Figs. 2, 4). Secondary copulatory sclerite usually absent, a short rod having longitudinal groove extending its length when present. Germarium elongate ovate. Uterus usually empty, delicate, proximally expanded; oötype and Mehlis’ gland not observed. Vagina, seminal receptacle not observed (absent). Vitellarium dense, coextensive with intestinal caeca, absent in regions of other reproductive organs; transverse vitelline duct at level of anterior end of germarium.
Metahaliotrema filamentosum Venkatanarasaiah, 1981. 1, Whole mount (composite, ventral view); 2, Copulatory complex (ventral view); 3, Accessory copulatory sclerite; 4, Copulatory complex (dorsal view); 5, Dorsal anchor; 6, 7, Dorsal bars; 8, Hook; 9, Ventral bar (anteroposterior view); 10, Ventral bar (dorsoventral view); 11, Ventral anchor
Measurements (measurements of specimens collected from G. subfasciatus follow in brackets those from G. oyena): Body 218–322 (275; n = 17) [325–326 (n = 1)] long; width at level of testis 41–61 (54; n = 16) [56–59 (58; n = 3)]. Haptor 55–90 (72; n = 17) [79–83 (81; n = 2)] wide. Ventral anchor 29–40 (35; n = 17) [34–42 (38; n = 12)] long; dorsal anchor 30–42 (36; n = 17) [35–46 (39; n = 12)] long. Ventral bar 26–37 (31; n = 16) [28–39 (33; n = 11)] long; dorsal bar 25–36 (31; n = 16) [30–39 (34; n = 12)] long. Hook 10–12 (11; n = 37) [10–12 (11; n = 31)] long. Pharynx 15–22 (18; n = 17) [17–18 (n = 2)] wide. Copulatory complex 26–34 (29; n = 13) [33–47 (39; n = 8)] long; ring diameter (MCO) 14–23 (19; n = 14) [14–20 (18; n = 7)]. Testis 27–52 (44; n = 14) [56–64 (60; n = 2)] long, 16–27 (21; n = 14) [19–26 (22; n = 2) wide; germarium 23–33 (27; n = 12) [29–39 (34; n = 2)] long, 14–22 (18; n = 12) [17–21 (19; n = 2)] wide.
Remarks
In the original description of M. filamentosum, Venkatanarasaiah (1981) did not show or describe a distal hook-like projection on the accessory piece of the copulatory complex. As a result, the specimens identified herein as M. filamentosum from G. oyena and G. subfasciatus from Australia were initially considered to be an undescribed species of Metahaliotrema. However, subsequent examination of the dactylogyrids infecting G. filamentosus in Vietnam (type-host of M. filamentosum) revealed that the hook-like projection was present in that species, although it was often obscured by other elements of the accessory piece (Nguyen & Kritsky, unpublished).
Examination of the original description of Haliotrema digyroides Zhang, 2001 from the gills of three species of Gerres in China, suggests that this species may be a synonym of M. filamentosum. Zhang (2001) did not indicate a hook-like projection on the accessory piece, but other features of the haptoral sclerites and the copulatory complex of H. digyroides, including the presence of a rod-shaped secondary copulatory sclerite, are similar to those originally described for M. filamentosum. However, the type-specimens of H. digyroides were not available for comparison (prior attempts to obtain and review types of Dr. Zhang’s species had been unsuccessful), and as a result, H. digyroides is not formally proposed as a junior synonym of M. filamentosum; a proposal of synonymy is withheld pending examination of the dactylogyrid species infecting gerreid hosts in the South China Sea. However, H. digyroides is transferred to Metahaliotrema as Metahaliotrema digyroides (Zhang, 2001) n. comb., based on the comparative morphology of the haptoral and copulatory sclerites and the apparent absence of a vagina; in the original description of H. digyroides, Zhang (2001) stated that the vagina was not observed.
Metahaliotrema filamentosum was the most common species encountered on the two hosts, G. oyena and G. subfasciatus, from Australia. Its occurrence on these hosts in Moreton Bay represents new host and locality records for the species.
Metahaliotrema imparilis n. sp.
Type-host: Gerres subfasciatus Cuvier (Gerreidae), common silver belly.
Type-locality: Moreton Bay off Dunwich, North Stradbroke Island, Queensland, Australia (27°50′S, 153°40′E), 18.i.2016.
Other localities: Moreton Bay off Green Island, Queensland, Australia (27°26′S, 153°20′E), 21.i.2016; Moreton Bay off Amity Point, North Stradbroke Island, Queensland, Australia (27°24′S, 153°20′E), 11.i.2016.
Type-material: Holotype, QM G237340; 4 paratypes, QM G237341, USNM 1485064, 1485065, HWML 139446.
Site in host: Gill lamellae.
Minimum prevalence: 71% (5 of 7 G. subfasciatus infected).
ZooBank registration: To comply with the regulations set out in article 8.5 of the amended 2012 version of the International Code of Zoological Nomenclature (ICZN, 2012), details of the new species have been submitted to ZooBank. The Life Science Identifier (LSID) for Metahaliotrema imparilis n. sp. is urn:lsid:zoobank.org:act:891DD1AB-F8B4-49F9-A544-CDB87766B1BB.
Etymology: The specific name, an adjective, is from Latin (imparilis = different, unequal) and refers to the morphological differences that differentiate the species from the morphologically similar Metahaliotrema subancistroides Zhang, 2001.
Description (Figs. 12–19)
Body proper fusiform, flattened dorsoventrally. Tegument smooth. Cephalic region broad, with 2 poorly developed bilateral cephalic lobes and terminal lobe usually with slight distal indentation. Three bilateral pairs of head organs poorly defined; cephalic glands not observed. Two pairs of eyespots; members of posterior pair larger, closer together than those of anterior pair; each posterior member with inconspicuous lens often obscured by chromatic granules; chromatic granules small, irregular to ovate; accessory granules few or absent in cephalic region. Pharynx subspherical; remaining digestive system unclear. Peduncle broad, gently tapered posteriorly; haptor subhexagonal, with 2 well-developed bilateral lobes containing hook pairs 2–4, 6, 7. Ventral anchor with elongate depressed superficial root, knob-like deep root, gently curved shaft, elongate point extending past level of tip of superficial root; ventral-anchor sclerite small, globose. Dorsal anchor with elongate tapered superficial root, poorly developed deep root, slightly arcing to straight shaft, point extending past level of tip of superficial root. Ventral bar broadly U-shaped, with slightly expanded ends. Dorsal bar rod-shaped, with subterminal constrictions, 2 anteriorly directed thorn-like processes at each end. Hook with short upright thumb, delicate point, heavy uniform shank; FH loop about 3/4 shank length. Gonads tandem, germarium pretesticular. Testis ovate; remaining unsclerotized structures of male reproductive system unclear. Copulatory complex comprising basally articulated MCO and accessory piece. MCO a coiled tube having about 1¼ counterclockwise rings and slightly expanded base. Accessory piece variable depending on orientation, comprising proximal articulation process and distal portion with complex of branches; hook-like termination absent. Secondary copulatory sclerite absent. Germarium irregular in outline; remaining structures of female reproductive system unclear. Vitellarium consisting of 2 bilateral bands confluent posterior to testis; transverse vitelline duct lying immediately anterior to germarium.
Measurements: Body 337–352 (345; n = 4) long; width at level of testis 75–79 (77; n = 3). Haptor 90–110 (98; n = 4) wide. Ventral anchor 47–53 (49; n = 5) long; dorsal anchor 50–57 (52; n = 5) long. Ventral bar 36–44 (41; n = 5) long; dorsal bar 42–49 (45; n = 5) long. Hook 10–12 (11; n = 15) long. Copulatory complex 35–40 (37; n = 3) long; ring diameter (MCO) 12–16 (14; n = 5). Pharynx 22–26 (23; n = 4) wide. Testis 37–47 (42; n = 4) long, 25–28 (26; n = 4) wide; germarium 30–35 (32; n = 3) long, 20–24 (22; n = 3) wide.
Remarks
Only five specimens of M. imparilis n. sp., one on each of five common silver bellies from Moreton Bay, were found. The species occurred concomitantly with the more abundant M. filamentosum. Because the two species could not be differentiated under low-power microscopy used to collect helminths from the gills and sediment, all five specimens were unknowingly mounted unstained in Gray & Wess medium, which precluded full determination of the soft anatomy of the new species. Nonetheless, M. imparilis is easily differentiated from M. filamentosum by having slightly larger haptoral sclerites and a dorsal bar with terminal anteriorly directed thorn-like projections (thorn-like projections absent in the dorsal bar of M. filamentosum).
Zhang (2001) described Haliotrema subancistroides Zhang, 2001 from the gills of five species of Gerres collected from the South China Sea (see Table 1). This species possesses a morphologically similar haptoral armament to that of M. imparilis. Although minimal information is available concerning the internal anatomy of H. subancistroides, Zhang (2001) did report that the vagina was not observed in his specimens. The apparent absence of a vagina and the presence of articulated dorsal and ventral bars, both features defining Metahaliotrema (see Kritsky et al., 2016), along with the similarity of the haptoral sclerites in H. subancistroides and M. imparilis, supports the transfer herein of H. subancistroides to Metahaliotrema as Metahaliotrema subancistroides (Zhang, 2001) n. comb. Metahaliotrema imparilis and M. subancistroides are the only species of Metahaliotrema currently known to possess bilateral thorn-like projections on the dorsal bar.
Methaliotrema imparilis is easily differentiated from M. subancistroides in the comparative morphology of the copulatory complex. In M. subancistroides, the MCO is noticeably larger with a greater diameter of the lumen than that of M. imparilis. In addition, the accessory piece extends well anterior of the tip of the MCO as an elongate flattened projection in M. subancistroides (accessory piece ending slightly anterior of the tip of MCO in M. imparilis).
Protogyrodactylus scalmophorus n. sp.
Type-host: Gerres oyena (Forsskål) (Gerreidae), common silver-biddy.
Type-locality: Moreton Bay off Green Island, Queensland, Australia (27°26′S, 153°20′E), 12 and 21.i.2016.
Other locality: Moreton Bay off Dunwich, North Stradbroke Island, Queensland, Australia (27°50′S, 153°40′E), 18.i.2016.
Type-material: Holotype, QM G237333; 19 paratypes (2 lost during re-mounting), QM G237334-G237339, USNM 148056-1485063, HWML 139445.
Site in host: Gill lamellae.
Minimum prevalence: 57% (4 of 7 G. oyena infected).
ZooBank registration: To comply with the regulations set out in article 8.5 of the amended 2012 version of the International Code of Zoological Nomenclature (ICZN, 2012), details of the new species have been submitted to ZooBank. The Life Science Identifier (LSID) for Protogyrodactylus scalmophorus n. sp. is urn:lsid:zoobank.org:act:A362382E-59E5-4DCE-A945-129A34253A71.
Etymology: The specific name, an adjective, is derived from Greek (scalme = a knife, blade or sword + -phor = to bear or carry) and refers to the blade-like medial terminations of the dorsal haptoral bars.
Description (Figs. 20–28)
Body proper fusiform, slightly flattened dorsoventrally. Tegument with closely applied spines extending from peduncle to the germarium in trunk. Cephalic region broad, with 2 poorly developed bilateral cephalic lobes and rounded terminal lobe. Head organs poorly defined; cephalic glands not observed. One pair of eyespots; each member with small lens; chromatic granules very small; accessory granules few or absent in cephalic region. Pharynx subspherical; oesophagus short; intestinal caeca uniting well posterior to testis. Peduncle broad, gently tapered posteriorly; haptor globose. Ventral anchor with short broad superficial root, knob-like deep root, gently curved short shaft, point extending slightly past level of tip of superficial root; ventral-anchor sclerite small, with well-defined lateral margins having delicate web between them. Dorsal anchor with elongate superficial root lacking terminal hook, short deep root, slightly arcing shaft, point extending to near level of tip of superficial root. Ventral bar rod-shaped, with 2 pairs of short projections arising from anterior margin near midlength. Dorsal bar rod-shaped, with acute medial blade-like termination extending posteriorly at approximately 45° angle from proximal portion of bar. Hook delicate, with short upright thumb, fine point, uniform shank; FH loop about 3/4 shank length. Gonads intercaecal, tandem, germarium pretesticular. Testis subspherical to ovate. Vas deferens, seminal vesicle not observed; well-defined prostatic vesicle lying near base of copulatory complex. Copulatory complex comprising basally articulated MCO and accessory piece; MCO a minimally tapered tube slightly undulating proximally, with distal clockwise coil of about one ring; accessory piece variable, lightly sclerotized (often not visible), extending along undulating portion of MCO. Germarium irregular in outline. Uterus, oötype and Mehlis’ gland not observed. Vaginal aperture dextroventral; distal vaginal canal a coil of about one ring and giving rise to undulating proximal section of canal extending to seminal receptacle; seminal receptacle lying on body midline anterior to germarium. Vitellarium dense, coextensive with intestinal caeca; transverse vitelline duct at level of seminal receptacle.
Measurements: Body 418–583 (488; n = 10) long; greatest width 66–122 (85; n = 11). Haptor 53–82 (62; n = 11) wide. Ventral anchor 18–21 (19; n = 10) long, ventral-anchor sclerite 11–14 (12; n = 8) long; dorsal anchor 21–24 (23; n = 10) long. Ventral bar 28–31 (30; n = 9) long; dorsal bar 17–21 (18; n = 9) long. Hook 9–11 (10; n = 9) long. Pharynx 25–32 (29; n = 8) wide. Copulatory complex 26–36 (31; n = 8) long. Testis 32–62 (44; n = 7) long, 16–30 (25; n = 7) wide; germarium 29–44 (36; n = 7) long, 20–35 (25; n = 7) wide.
Remarks
Only Protogyrodactylus delicatus Bychowsky & Nagibina, 1974 possesses blade-like medial terminations of the dorsal bars similar to those of P. scalmophorus n. sp. These species are easily differentiated, however, by P. scalmophorus having smaller ventral anchors with short shafts (ventral anchor shafts more than twice as long in P. delicatus) and by lacking transverse fracture-lines in the bases of the dorsal anchors (transverse fracture-lines present in the dorsal anchors of P. delicatus).
Based on the comparative morphology of the respective copulatory complexes and ventral anchor/bar complexes, P. scalmophorus n. sp. most closely resembles Protogyrodactylus youngi Bychowsky & Nagibina, 1974, parasitic on the gills of the slender silver-biddy Gerres oblongus Cuvier in the South China Sea. Both species have a comparatively straight proximal segment and a coiled distal segment of the MCO, ventral anchors with robust bases and short shafts, and ventral bars with anteromedial projections. Protogyrodactylus scalmophorus is easily differentiated from P. youngi by having a blade-like medial end of the dorsal bar (medial end of dorsal bar club-shaped in P. youngi), dorsal anchors larger than the ventral anchors (dorsal and ventral anchors subequal in length in P. youngi), and a distal coil of the vaginal canal (vaginal canal an inverted U in P. youngi) (see Bychowsky & Nagibina, 1974).
Protogyrodactylus ichthyocercus n. sp.
Type-host: Gerres oyena (Forsskål) (Gerreidae), common silver-biddy.
Type-locality: Moreton Bay off Dunwich, North Stradbroke Island, Queensland, Australia (27°50′S, 153°40′E), 18.i.2016.
Other locality: Moreton Bay off Green Island, Queensland, Australia (27°26′S, 153°20′E), 12 and 21.i.2016.
Type-material: Holotype, QM G237358; 19 paratypes, QM G237359-G237366, USNM 1485072-1485078, HWML 139450.
Site in host: Gill lamellae.
Minimum prevalence: 71% (5 of 7 G. oyena infected).
ZooBank registration: To comply with the regulations set out in article 8.5 of the amended 2012 version of the International Code of Zoological Nomenclature (ICZN, 2012), details of the new species have been submitted to ZooBank. The Life Science Identifier (LSID) for Protogyrodactylus ichthyocercus n. sp. is urn:lsid:zoobank.org:act:90FF6758-0F23-4D0F-A147-F2352F246952.
Etymology: The specific name, treated as an adjective, is from Greek (ichthyos = fish + kerkos = tail) and refers to distal end of the ventral-anchor sclerite that resembles a heterocercal fish tail.
Description (Figs. 29–35)
Body proper fusiform, slightly flattened dorsoventrally, often robust with broad posterior trunk. Tegument with closely applied spines extending from peduncle to pharynx. Cephalic region broad, with 2 moderately developed bilateral cephalic lobes and rounded terminal lobe. Head organs poorly defined; cephalic glands not observed. One pair of eyespots; each member with small lens; chromatic granules small, irregular; accessory granules irregular, often ovate, few or infrequently absent in cephalic region. Pharynx subspherical; oesophagus short; intestinal caeca uniting well posterior to testis. Peduncle broad, gently tapered posteriorly; haptor globose, with 2 bilateral lobes containing hook pairs 2–4, 6, 7. Ventral anchor with robust base, short depressed superficial root, small knob-like deep root, evenly curved short shaft and point; point extending slightly past level of ventral margin of anchor base; ventral-anchor sclerite distally shaped as an heterocercal fish tail. Dorsal anchor with elongate superficial root lacking terminal spine, short deep root, slightly arcing shaft, sharply recurved point extending just short of level of tip of superficial root; dorsal anchor base with transverse fracture-line. Ventral bar shaped as an inverted oxen yoke. Dorsal bar club- shaped, distally flared. Hook delicate, with short upright thumb, fine point, uniform shank; FH loop about 3/4 shank length. Gonads intercaecal, tandem, germarium pretesticular. Testis ovate. Proximal vas deferens dorsoventrally looping left intestinal caecum; distal vas deferens, seminal vesicle not observed; small prostatic vesicle lying near base of copulatory complex. Copulatory complex comprising basally articulated MCO and accessory piece; MCO a minimally tapered tube undulating proximally, with distal clockwise coil of about one ring; accessory piece variable, lightly sclerotized (often not visible), extending along MCO. Germarium irregular in outline, often lying diagonally in trunk in contracted specimens. Uterus, oötype and Mehlis’ gland not observed. Vagina aperture dextroventral; vaginal canal short, leading to small seminal receptacle lying dextral to anterior end of germarium. Vitellarium dense, coextensive with intestinal caeca; transverse vitelline duct immediately anterior to germarium.
Measurements: Body 314–463 (355; n = 8) long; greatest width 55–112 (76; n = 9). Haptor 65–105 (83; n = 9) wide. Ventral anchor 26–32 (29; n = 9) long, ventral-anchor sclerite 16–24 (19; n = 10) long; dorsal anchor 21–30 (24; n = 8) long. Ventral bar 31–47 (37; n = 10) long; dorsal bar 21–32 (25; n = 10) long. Hook 10–12 (11; n = 21) long. Pharynx 24–30 (28; n = 10) wide. Copulatory complex 17–25 (21; n = 4) long. Testis 34–52 (42; n = 6) long, 20–40 (28; n = 6) wide; germarium 28–35 (31; n = 5) long, 17–26 (22; n = 5) wide.
Remarks
The copulatory and haptoral sclerites of P. ichthyocercus n. sp. resemble those of P. similis n. sp. from G. oyena in Moreton Bay and Protogyrodactylus fissilis Bychowsky & Nagibina, 1974, parasitic on G.oblongus in the South China Sea. It differs from both species by its smaller size (314–463 vs 516–828 µm for P. similis and 790–1,000 µm for P. fissilis), and by having ventral anchors with comparatively straight shafts (ventral anchor shaft and point forming an evenly curved arc in P. fissilis) (see Bychowsky & Nagibina, 1974); in P. similis, the ventral anchor shaft is comparatively longer than that in P. ichthyocercus.
Bychowsky & Nagibina (1974) did not provide any details of the soft internal anatomy of P. fissilis and did not provide a drawing of a whole mount of the species. As a result, it was not possible to utilize any internal features to differentiate this species from P. ichthyocercus. However, the presence of two posterior diverticula of the intestinal caeca in P. similis further differentiates it from P. ichthyocercus (intestinal diverticula lacking in P. ichthyocercus).
Protogyrodactylus similis n. sp.
Type-host: Gerres oyena (Forsskål) (Gerreidae), common silver-biddy.
Type-locality: Moreton Bay off Green Island, Queensland, Australia (27°26′S, 153°20′E), 12 and 21.i.2016.
Other locality: Moreton Bay off Dunwich, North Stradbroke Island, Queensland, Australia (27°50′S, 153°40′E), 18.i.2016.
Type-material: Holotype, QM G237373; 17 paratypes, QM G237367-G237372, G237374, USNM 1485079-1485085, HWML 139451.
Site in host: Gill lamellae.
Minimum prevalence: 71% (5 of 7 G. oyena infected).
ZooBank registration: To comply with the regulations set out in article 8.5 of the amended 2012 version of the International Code of Zoological Nomenclature (ICZN, 2012), details of the new species have been submitted to ZooBank. The Life Science Identifier (LSID) for Protogyrodactylus similis n. sp. is urn:lsid:zoobank.org:act:6718AE2A-519C-425E-8A75-ED40D0F28270.
Etymology: The specific name, an adjective, is from Latin (similis = resembling) and refers to the morphological similarity of the species to two other congenerics (P. ichthyocercus and P. fissilis) occurring on silver-biddies in the western Pacific.
Description (Figs. 36–42)
Body proper foliform, flattened dorsoventrally. Tegument smooth. Cephalic region tapered anteriorly, with 2 poorly developed bilateral lobes and rounded terminal lobe. Head organs poorly defined; bilateral groups of cephalic-gland cells lying lateral to pharynx. One pair of eyespots; each member with small lens; chromatic granules small, irregular in outline; accessory granules few or absent in cephalic region. Pharynx large, ovate, often with constriction near midlength; oesophagus short; intestinal caeca with two posteriorly directed diverticula originating from posterior margin of caeca near their confluence posterior to testis. Peduncle short to non-existent; haptor globose, with bilateral lobes containing hook pairs 2–4, 6, 7. Ventral anchor with robust base having knob-like superficial root, short anteriorly directed deep root, comparatively long evenly curved shaft and point; point extending past level of ventral margin of anchor base; ventral-anchor sclerite with bifurcated medial end. Dorsal anchor with elongate superficial root lacking terminal spine, short deep root, slightly arcing shaft, sharply recurved point extending to near level of tip of superficial root; dorsal anchor base with transverse fracture-line. Ventral bar rod-shaped, bowed, with slight medial expansion. Dorsal bar club-shaped, distally expanded. Hook delicate, with short upright thumb, fine point, uniform shank; FH loop about 3/4 shank length. Gonads intercaecal, tandem; germarium pretesticular. Testis elongate ovate to bacilliform. Vas deferens not observed; seminal vesicle a dilation of distal vas deferens, lying posterior to small prostatic vesicle; prostatic vesicle near base of copulatory complex. Copulatory complex comprising basally articulated MCO and accessory piece. MCO with expanded base, slightly tapered tube; tube loosely coiled; coil clockwise, with about 1½ rings. Accessory piece variable, lightly sclerotized (often not visible), extending along MCO. Germarium ovate, lying diagonally in trunk. Uterus poorly defined; Mehlis’ gland represented by several glandular structures lying immediately anterior to germarium and immediately left of body midline. Vagina aperture dextroventral; vaginal canal short, coiled, leading to small seminal receptacle lying to right of anterior end of germarium. Vitellarium dense throughout trunk; transverse vitelline duct just anterior to germarium.
Measurements: Body 516–828 (711; n = 8) long; greatest width 122–203 (172; n = 9). Haptor 91–130 (117; n = 5) wide. Ventral anchor 29–33 (31; n = 4) long, ventral-anchor sclerite 17–21 (19; n = 8) long; dorsal anchor 23–29 (26; n = 5) long. Ventral bar 31–44 (38; n = 9) long; dorsal bar 24–36 (29; n = 9) long. Hook 11–13 (12; n = 14) long. Pharynx 46–64 (50; n = 9) wide. Copulatory complex 22–29 (25; n = 6) long. Testis 91–181 (145; n = 8) long, 35–75 (53; n = 7) wide; germarium 57–86 (68; n = 8) long, 41–63 (51; n = 8) wide.
Remarks
Protogyrodactylus similis n. sp. is most similar to P. fissilis, described by Bychowsky & Nagibina (1974) from G. macrosoma Bleeker (now G. oblongus) in the South China Sea. Both species are comparatively large when compared with the other congeners described herein, and both species possess very similar copulatory and haptoral sclerites. The two species may indeed by synonyms, but differences primarily in the morphology of the ventral and dorsal anchors appear to differentiate the two species at this time. In P. similis, the ventral anchor shafts are longer than those of P. fissilis, and the dorsal anchors are somewhat smaller than the ventral anchors (dorsal and ventral anchors subequal in P. fissilis). The type-specimens of P. fissilis, currently in the Zoological Institute of the Russian Academy of Sciences in St. Petersburg, Russia, were not available for examination; apparently international loans of specimens deposited in the Zoological Institute are no longer made.
With exception of Protogyrodactylus vulgaris n. sp., the presence of intestinal diverticula in P. similis differentiates it from all other congeners in which internal anatomy has been reported; the nature of the intestinal caeca in P. fissilis was not reported by Bychowsky & Nagibina (1974). Protogyrodactylus similis is easily differentiated from P. vulgaris by having larger ventral anchors with noticeably longer anchor shafts and in the comparative morphology of the ventral-anchor sclerite, ventral bar, and copulatory complex (compare Figs. 36–42 and 49–54).
Protogyrodactylus monacanthus n. sp.
Type-host: Gerres subfasciatus Cuvier (Gerreidae), common silver belly.
Type-locality: Moreton Bay off Amity Point, North Stradbroke Island, Queensland, Australia (27°24′S, 153°20′E), 11.i.2016.
Other locality: Moreton Bay off Dunwich, North Stradbroke Island, Queensland, Australia (27°50′S, 153°40′E), 18.i.2016.
Type-material: Holotype, QM G237342; 12 paratypes (one lost during re-mounting), QM G237343-G237347, USNM 1485066-1485069, HWML 139447, 139448.
Site in host: Gill lamellae.
Minimum prevalence: 71% (5 of 7 G. subfasciatus infected).
ZooBank registration: To comply with the regulations set out in article 8.5 of the amended 2012 version of the International Code of Zoological Nomenclature (ICZN, 2012), details of the new species have been submitted to ZooBank. The Life Science Identifier (LSID) for Protogyrodactylus monacanthus n. sp. is urn:lsid:zoobank.org:act:313D51CB-4E83-44B6-AE1A-66CF580EA347.
Etymology: The specific name, treated as a noun, is from Greek (mon/o = one + acanth/o = spine or thorn) and refers to the single anchor/bar complex in the species.
Description (Figs. 43–48)
Body proper fusiform, slightly flattened dorsoventrally. Tegument with parallel transverse ridges from peduncle to level of gonads. Cephalic region broad, with 2 poorly developed bilateral cephalic lobes and rounded to truncate terminal lobe. Three pairs of head organs; bilateral groups of cephalic-gland cells lying lateral to pharynx. One pair of eyespots; each member with lens often obscured by chromatic granules; chromatic granules small, irregular or ovate; accessory granules few in cephalic region. Pharynx subspherical; oesophagus short; intestinal caeca confluent posterior to testis. Peduncle broad, gently tapering posteriorly; haptor globose, with posteromedial indentation. Ventral anchor with slightly depressed superficial root, short knob-like deep root, comparatively long evenly curved shaft and point; point extending past level of ventral margin of anchor base; ventral-anchor sclerite simple, tapered distally. Dorsal anchor/bar complex absent; dorsal anchor apparently replaced by delicate bifurcated remnant (Fig. 47). Ventral bar rod-shaped, straight, with slightly enlarged ends. Hook delicate, with short upright thumb, fine point, uniform shank; FH loop approaching shank length. Gonads intercaecal, tandem; germarium pretesticular. Testis elongate ovate. Vas deferens not observed; seminal vesicle an apparent dilation of distal vas deferens, lying posterior to small prostatic vesicle; prostatic vesicle near base of copulatory complex. Copulatory complex comprising basally articulated MCO and compound accessory piece; MCO a straight broad tube having elongate curved distal hook-like projection of accessory piece overlying the tip; hook-like projection terminally acute or often blunt (rounded); second accessory piece variable, extending along MCO. Germarium ovate. Uterus and Mehlis’ gland not observed. Wide open vaginal aperture ventral, immediately to left of body midline just anterior to germarium; seminal receptacle not observed. Vitellarium dense, coextensive with intestinal caeca; transverse vitelline duct immediately anterior to germarium.
Measurements: Body 221–353 (279; n = 6) long; greatest width 50–62 (54; n = 5). Haptor 46–79 (62; n = 6) wide. Ventral anchor 25–29 (27; n = 7) long, ventral-anchor sclerite 10–12 (11; n = 7) long. Dorsal-anchor remnant 12–14 (13; n = 5) long. Ventral bar 31–38 (35; n = 7) long. Hook 10–12 (11; n = 14) long. Pharynx 16–22 (19; n = 6) wide. Copulatory complex 23–31 (28; n = 6) long. Testis 34–55 (41; n = 5) long, 16–24 (20; n = 5) wide; germarium 20–34 (26; n = 5) long, 14–26 (19; n = 5) wide.
Remarks
Protogyrodactylus monacanthus n. sp. is distinguished from all congeners by lacking the dorsal anchor/bar complex, which ostensively represents a secondary loss among members of the genus. In addition, P. monacanthus possesses a sinistroventral vaginal aperture. In its congeners for which internal anatomy has been recorded, including the remaining species from Moreton Bay, the ventral vaginal aperture lies just to the right of the body midline immediately anterior to the germarium.
Monogenoidean species possessing a single anchor/bar complex are not uncommon among members of the Dactylogyridae (see Kritsky et al., 2010 for a partial list of dactylogyrid genera containing species that lack either the ventral or dorsal anchor/bar complex). In all of these species, the absence of the complex is currently understood to represent secondary evolutionary losses within the family (see Kritsky & Boeger, 1989), and in some cases, secondary losses within individual genera, i.e. Euryhaliotrema Kritsky & Boeger, 2002 (see Kritsky & Boeger, 2002). Whereas absence of one or more of the anchor/bar complexes in a dactylogyrid may often justify a separate genus to accommodate the species, proposal of a new genus for P. monacanthus is not justified at this time, because to do so would undoubtedly result in Protogyrodactylus becoming paraphyletic.
Protogyrodactylus vulgaris n. sp.
Type-host: Gerres subfasciatus Cuvier (Gerreidae), common silver belly.
Type-locality: Moreton Bay off Amity Point, North Stradbroke Island, Queensland, Australia (27°24′S, 153°20′E), 11.i.2016.
Other locality: Moreton Bay off Dunwich, North Stradbroke Island, Queensland, Australia (27°50′S, 153°40′E), 18.i.2016.
Type-material: Holotype, QM G237348; 3 paratypes, QM G237349, USNM 1485070, 1485071.
Site in host: Gill lamellae.
Minimum prevalence: 43% (3 of 7 G. subfasciatus infected).
ZooBank registration: To comply with the regulations set out in article 8.5 of the amended 2012 version of the International Code of Zoological Nomenclature (ICZN, 2012), details of the new species have been submitted to ZooBank. The Life Science Identifier (LSID) for Protogyrodactylus vulgaris n. sp. is urn:lsid:zoobank.org:act:6A063A9C-7A19-4CCA-A283-F3B7916406F2.
Etymology: The specific name, an adjective, is from Latin (vulgaris = common) and refers to the recurrent basic morphology of the species with that of its congeners.
Description (Figs. 49–54)
Body proper fusiform, slightly flattened dorsoventrally. Tegument with closely applied spines extending from peduncle to pharynx. Cephalic region broad, with 2 poorly developed bilateral cephalic lobes and rounded terminal lobe. Three pairs of head organs; posterolateral head organ often divided into two; bilateral groups of cephalic-gland cells lateral to pharynx. One pair of eyespots; each member with lens; chromatic granules small, irregular in outline; accessory granules often ovate, few in cephalic region. Pharynx subspherical; oesophagus short; intestinal caeca uniting posterior to testis, with 2 bilateral diverticula extending posteriorly near caecal confluence. Peduncle broad, tapered posteriorly; haptor globose. Ventral anchor with enlarged base having protruding superficial root with distal cap, small knob-like deep root, evenly curved short shaft, point extending to near level of tip of superficial root; ventral-anchor sclerite bifid, with delicate web between branches. Dorsal anchor with elongate superficial root lacking terminal spine, short to non-existent deep root, slightly arcing shaft, sharply recurved elongate point extending to near level of tip of superficial root; anchor base with transverse fracture-line. Ventral bar generally straight, rod-shaped, with 2 short acute projections directed anteriorly near midlength of bar. Dorsal bar spatulate. Hook delicate, with short upright thumb, fine point, uniform shank; FH loop about 3/4 shank length. Gonads intercaecal, tandem; germarium pretesticular. Testis ovate. Vas deferens not observed; large spherical vesicle with tapered internal duct apparently representing seminal vesicle; small prostatic vesicle lying near base of copulatory complex. Copulatory complex comprising basally articulated MCO and accessory piece. MCO a tapered tube with expanded base, proximal recurve, and distal clockwise coil comprised of single ring. Accessory piece variable, lightly sclerotized, extending along MCO. Germarium irregular to subovate in outline. Uterus delicate; oötype and Mehlis’ gland not observed. Vaginal aperture dextroventral near body midline; vaginal canal short, coiled, leading to small seminal receptacle lying just to right of anterior end of germarium. Vitellarium dense, coextensive with intestinal caeca; transverse vitelline duct near level of vaginal aperture, anterior to germarium.
Measurements: Body 474–495 (486; n = 3) long; greatest width 93–104 (98; n = 3). Haptor 76–89 (82; n = 3) wide. Ventral anchor 23–24 (n = 1) long, ventral-anchor sclerite 13–14 (n = 1) long; dorsal anchor 29–30 (n = 1) long. Ventral bar 45–46 (n = 1) long; dorsal bar 17–18 (n = 1) long. Hook 10–12 (11; n = 3) long. Pharynx 30–35 (32; n = 3) wide. Copulatory complex 15–16 (n = 1) long. Testis 34–52 (43; n = 3) long, 25–31 (28; n = 3) wide; germarium 46–61 (53; n = 3) long, 32–40 (36; n = 3) wide.
Remarks
The copulatory complex of P. vulgaris n. sp. most closely resembles that of Protogyrodactylus leptocirrus Zhang & Lin, 2001, a parasite of the gills of Terapon jarbua (Forsskål) (Terapontidae) from off Guangdong Province, China. The new species is easily differentiated from P. leptocirrus by the comparative morphology of their haptoral sclerites. In P. vulgaris, the dorsal anchor has an elongate superficial root and a short to non-existent deep root (superficial roots short and deep roots well developed in the dorsal anchors of P. leptocirrus), the dorsal anchor has a transverse fracture-line (transverse fracture-line absent in P. leptocirrus), and the ventral-anchor sclerites are bifid with a delicate web between the two branches (ventral-anchor sclerite with terminal projection in P. leptocirrus).
Order Gyrodactylidea Bychowsky, 1937
Gyrodactylidae Van Beneden & Hesse, 1863
Gyrodactylus xynus n. sp.
Type-host: Gerres subfasciatus Cuvier (Gerreidae), common silver belly.
Type-locality: Moreton Bay off Amity Point, North Stradbroke Island, Queensland, Australia (27°24′S, 153°20′E), 11.i.2016.
Type-material: Holotype, QM G237350; 21 paratypes, QM G237351-G237357, USNM 1485086-1485092, HWML 139449.
Site in host: Gills.
Minimum prevalence: 29% (2 of 7 G. subfasciatus infected).
ZooBank registration: To comply with the regulations set out in article 8.5 of the amended 2012 version of the International Code of Zoological Nomenclature (ICZN, 2012), details of the new species have been submitted to ZooBank. The Life Science Identifier (LSID) for Gyrodactylus xynus n. sp. is urn:lsid:zoobank.org:act:BC409763-8D29-4B5A-80C7-AC567525154F.
Etymology: The specific name, an adjective, is from Greek (xynos = common) and refers to the comparatively ordinary morphology of the copulatory and haptoral sclerites of the species.
Description (Figs. 55–59)
Body proper fusiform; greatest width usually at level of uterus when containing embryo; peduncle short, tapering toward haptor. Tegument smooth. Cephalic lobes poorly to well developed, each containing large head organ; spike sensilla not observed. Cephalic glands comprising bilateral groups of unicellular glands posterolateral to pharynx; prepharyngeal cephalic glands not observed. Pharynx having 2 tandem bulbs, pharyngeal papillae inconspicuous; oesophagus short; intestinal caeca nonconfluent, extending posteriorly to level of testis. Testis with flattened anterior margin abutting posterior margin of germarium. Vas deferens, seminal vesicle, prostates not observed. MCO usually absent, lying slightly to left of body midline posterior to pharynx when present, armed with large spine and about four spinelets. Germarium subspherical, containing large oocyte surrounded by thin peripheral layer of germinal cells; oviduct not observed; uterus with one or two generations of embryos; uterine pore not observed. Vitellarium absent. Haptor subcircular. Hooks similar, extrahamular (see Kritsky & Mizelle, 1968); each with uniform shank; hooklet with fine evenly curved point, slightly arcing shaft, small globose heel, tapered toe lacking well-defined shelf; filamentous hooklet loop about ½ shank length. Anchor delicate; anchor base with small fold and conspicuous knob; superficial anchor root moderately long, tapered; anchor shaft slightly arced; point recurved, elongate. Superficial bar with short anterolateral rami inserted beneath anchor folds; superficial-bar shield short, tapered posteriorly, extending posteriorly about 1/3 the length of anchor shaft. Deep bar variably bent, a simple rod with attenuated ends inserted into anchor knobs.
Measurements: Body 225–244 (236; n = 9) long; greatest width 44–63 (54; n = 9). Haptor 51–74 (61; n = 10) long; 49–65 (58; n = 9) wide. Anchor 49–53 (51; n = 12) long, superfical bar 19–24 (21; n = 12) long, deep bar 12–17 (15; n = 8) long. Hook 26–29 (28; n = 20) long; hooklet 5–7 (6; n = 22) long. Anterior pharyngeal bulb 20–24 (22; n = 8) wide; posterior pharyngeal bulb 24–28 (26; n = 8) wide. MCO 8–12 (10; n = 3) in diameter. Testis 5–16 (8; n = 5) long, 4–13 (7; n = 5) wide; germarium 5–14 (9; n = 8) long, 6–15 (9; n = 8) wide.
Remarks
Specimens of Gyrodactylus xynus n. sp. were collected from the gills of its gerreid host. It is unknown if the species also occurs on the skin and fins, as body washes of G. subfasciatus were not conducted during the present study.
Assignment of a species of Gyrodactylus that most closely resembles G. xynus is difficult. As its specific name suggests, G. xynus lacks notable morphological features that would clearly differentiate it from the more than 400 previously described species of Gyrodactylus. In addition, comparison of G. xynus with congeners infecting fishes of related host groups was not possible. Gyrodactylus xynus is the only species of the genus recorded from a gerreid host, and phylogenetic studies have not been successful in determining the sister group of the Gerreidae (see Vari, 1978; Chen et al., 2007).
That Protogyrodactylus spp. are known to infect fishes assigned to the Gerreidae and Terapontidae (see Galli & Kritsky, 2008), suggests that species of Gyrodactylus similar to G. xynus might be found on terapontids. Although a search of the literature did not reveal any described species of Gyrodactylus infecting a terapontid, Ernst et al. (2001) reported an unidentified species of Gyrodactylus (their Gyrodactylus sp. 5) from the skin and fins of Pelates sexlineatus (Quoy & Gaimard) [now Helotes sexlineatus (Quoy & Gaimard)] (Terapontidae) from Moreton Bay, Australia. In Table 2 of their paper, Ernst et al. (2001) provided accession numbers from the Queensland Museum for their gyrodactylid from H. sexlineatus, but the helminth specimens were apparently never received by museum staff (M. Bryant, personal communication). As a result, a comparison of G. xynus with a gyrodactylid species from these associated hosts was not possible.
Subclass Heteronchoinea Boeger & Kritsky, 2001
Infrasubclass Oligonchoinea Bychowsky, 1937
Order Mazocraeidea Bychowsky, 1937
Microcotylidae Taschenberg, 1879
Polylabris gerres (Sanders, 1944) Mamaev & Parukin, 1976
Syns Microcotyle gerres Sandars, 1944; Nudimasculus (Nudimasculus) gerres (Sanders, 1944) Unnithan, 1971; Prostatomicrocotyla gerres Machida, 1973
Type-host: Gerres subfasciatus Cuvier (Gerreidae), common silver belly.
Type-locality: Mandurah, Western Australia.
Current record: G. subfasciatus: Moreton Bay off Amity Point, North Stradbroke Island, Australia (27°24′S, 153°20′E), 11.i.2016.
Previous records: Gerres ovatus Günther (now G. subfasciatus): Mandurah, Western Australia (Sanders, 1944); Swan River Estuary and Cockburn Sound, Perth, Western Australia [Williams (1991), this locality recorded as Rockingham, Western Australia, by Hayward (1996)]. G. oyena; Southern Bungo Channel, Ehime Prefecture, Shikoku Island, Japan (Machida, 1973).
Site in host: Gill lamellae.
Minimum prevalence: 14% (1 of 7 G. subfasciatus infected).
Specimen studied: Voucher specimen, QM G237375.
Remarks
Sanders (1944) originally described this species as Microcotyle gerres from G. ovatus (now G. subfasciatus) collected from Mandurah, Western Australia. The species was subsequently transferred to Nudimasculus as Nudimasculus (Nudimasculus) gerres (Sanders, 1944) Unnithan, 1971 by Unnithan (1971). Nudimasculus Unnithan, 1971 was considered a subjective synonym of Polylabris Euzet & Cauwet, 1967 by Mamaev & Parukin (1976), who transferred the species to the latter genus. The species was subsequently recorded from G. subfasciatus by Williams (1991) from additional localities in Western Australia. Finally, Machida (1973) described Prostatomicrocotyla gerres Machida, 1973 as a new species from G. oyena in Japan; Mamaev & Parukin (1976) considered Machida’s species to be a junior subjective synonym and homonym of Polylabris gerres.
Polylabris gerres was encountered only once, as a single specimen on the gills of G. subfasciatus during the present study. Although previously recorded from G. oyena by Machida (1973), none of the seven specimens of the latter host examined during the present study was found infected by this species. The finding of P. gerres in Morton Bay represents a new locality record for the helminth.
Change history
02 January 2020
During registration of specimens of Protogyrodactylus scalmophorus and Protogyrodactylus ichthyocercus for the Queensland Museum, it was noticed that the collection sites of the holotypes of the two species provided on the slide labels did not correspond to the published type-localities. While both species were collected from two localities within Moreton Bay as recorded in the paper, the type-locality for each is here corrected to that reported as the ���Other locality��� in the respective taxonomic summaries.
02 January 2020
During registration of specimens of Protogyrodactylus scalmophorus and Protogyrodactylus ichthyocercus for the Queensland Museum, it was noticed that the collection sites of the holotypes of the two species provided on the slide labels did not correspond to the published type-localities. While both species were collected from two localities within Moreton Bay as recorded in the paper, the type-locality for each is here corrected to that reported as the ���Other locality��� in the respective taxonomic summaries.
References
Aguirre-Macedo, M. L., Vidal-Martínez, V., González-Solis, D., & Caballero, P. I. (2007). Helminth communities of four commercially important fish species from Chetumal Bay, Mexico. Journal of Helminthology, 81, 19–31.
Bayoumy, E. M., & El-Monem, S. A. (2011). Surface topography and spermiogenesis of Lamellodiscus Diplodicus (Monogenea, Diplectanidae), a parasite of Gerres Oyena (teleostei, Gerridae) from the Suez Gulf, Egypt (sic). Life Science Journal, 8, 467–476.
Boeger, W. A., Diamanka, A., Pariselle, A., & Patella, L. (2012). Two new species of Protogyrodactylus (Monogenoidea: Dactylogyridae) from the gills of Gerres nigri (Teleostei: Gerreidae) from Senegal. Folia Parasitologica, 59, 59–63.
Bravo-Hollis, M., & Kohn, A. (1990). Una especies nueva del genero Microcotyle (Monogenea: Microcotylidae) de las branquias de Eucinostomus argenteus (Baird and Girard, 1854) de Rio de Janeiro, Brasil. Anales del Instituto de Biología da Universidad Nacional Autonóma de México, Serie Zoología, 61, 191–196.
Bunkley-Williams, L., & Williams, E. H., Jr. (1994). Parasites of Puerto Rican freshwater sport fishes. San Juan: Puerto Rico Department of Natural and Environmental Resources, Puerto Rico, 164 pp.
Bunkley-Williams, L., & Williams, E. H., Jr. (1995). Parásitos de peces de valor recreativo en aqua dulce de Puerto Rico. Mayagüez: Departamento de Recursos Naturales y Ambientales de Puerto Rico y el Departamento de Ciencias Marinas, Universidad de Puerto Rico, 190 pp.
Bychowsky, B. E., & Nagibina, L. F. (1974). New species of Protogyrodactylus (Dactylogyridae, Ancyrocephalinae). Parazitologiya, 8, 473–483 (In Russian).
Caballero, Y. C. E., & Bravo-Hollis, M. (1972). Monogenea (van Beneden, 1858) Carus, 1863 de peces marinos del litoral mexicano del Golfo de México y de Mar Caribe. V. Revista de Biología Tropical, 20, 151–165.
Chen, W.-J., Ruiz-Carus, R., & Orti, G. (2007). Relationships among four genera of mojarras (Teleostei: Perciformes: Gerreidae) from the western Atlantic and their tentative placement among peromorph fishes. Journal of Fish Biology, 70, 201–218.
Cohen, S. C., Cárdenas, M. Q., Fernandes, B. M. M., & Justo, M. C. N. (2017). Monogenoidea from Eucinostomus argenteus and Eucinostomus gula (Perciformes, Gerreidae) from the littoral of the State of Rio de Janeiro, Brazil. Pan-American Journal of Aquatic Sciences, 12, 80–84.
Domingues, M. V., Diamanka, A., & Pariselle, A. (2011). Monogenoids (Diplectanidae, Polyonchoinea) from the gills of mojarras (Perciformes, Gerreidae) with the resurrection of Neodiplectanum Mizelle & Blatz, 1941 and the proposal of Darwinoplectanum n. gen. Zootaxa, 3010, 1–19.
Dyer, W. G., Williams, E. H., Jr., & Bunkley Williams, L. (1989). Monogeneans from marine fishes of Okinawa, Japan. Proceedings of the Helminthological Society of Washington, 56, 64–68.
Ernst, I., Whittington, I. D., & Jones, M. K. (2001). Diversity of gyrodactylids from some marine fishes in tropical and subtropical Queensland, Australia. Folia Parasitologica, 48, 165–168.
Eschmeyer, W. N., & Fong J. D. (2018). Catalog of Fishes; Species by family/subfamily. Retrieved March 2, 2018, from http://researcharchive.calacademy.org/research/ichthyology/catalog/SpeciesByFamily.asp.
Froese, R., & Pauly, D. (Eds). (2017). FishBase. Retrieved March 2, 2018, from www.fishbase.org (version (10/2017).
Fuentes Zambrano, J. L. (1997). Dos especies nuevas de monogéneos (Diplectanidae) parásitos de peces marinos de la Laguna de la Restinga, Venezuela. Anales del Instituto de Biología da Universidad Nacional Autonóma de México, Serie Zoología, 68, 225–236.
Galli, P., & Kritsky, D. C. (2008). Three new species of Protogyrodactylus Johnston & Tiegs, 1922 (Monogenoidea: Dactylogyridae) from the gills of the longtail silverbiddy Gerres longirostris (Teleostei: Gerreidae) in the Red Sea. Systematic Parasitology, 69, 221–231.
González-Solís, D., & Sánchez-Ceballos. (2012). Metazoarios parásitos de la chihua, Eugerres plumieri (Perciformes, Gerreidae) en la Bahía de Chetumal y lagunas adyacentes, Quintana Roo, México. In: Del Moral-Flores, L. F., Martínez-Perez, J. A., Franco-López, J., Ramírez-Villalobos, A. J. & Tello-Musi, J. L. (Eds), Investigación Ictiológica en México, Temas Selectos en honor al Dr. José Luis Castro Aguirre. Mexico City: Universidad Nacional Autónoma de México, Facultad de Estudios Superiores Iztacala y Sociedad Ictiológica Mexicana, pp. 75−87.
Hayward, C. J. (1996). Revision of the monogenean genus Polylabris (Microcotylidae). Invertebrate Taxonomy, 10, 995–1039.
ICZN (2012). International Commission on Zoological Nomenclature: Amendment of articles 8, 9, 10, 21 and 78 of the International Code of Zoological Nomenclature to expand and refine methods of publication. Bulletin of Zoological Nomenclature, 69, 161–169.
Johnson, J. W. (2010). Fishes of the Moreton Bay Marine Park and adjacent continental shelf waters, Queensland, Australia. Memoirs of the Queensland Museum - Nature, 54, 299–353.
Kritsky, D. C. (2018). Dactylogyrids (Monogenoidea) infecting the gill lamellae of some beloniform fishes from Moreton Bay, Queensland, Australia, with a redescription of Hareocephalus thaisae Young, 1969 and descriptions of six new species of Hemirhamphiculus Bychowsky & Nagibina, 1969. Systematic Parasitology, 95, 33–54.
Kritsky, D. C., Ali, A. H., & Khamees, N. R. (2013). Gyrodactylus aff. mugili Zhukov, 1970 (Monogenoidea: Gyrodactylidae) from the gills of mullets (Mugiliformes: Mugilidae) collected from the inland waters of southern Iraq, with an evaluation of previous records of Gyrodactylus spp. on mullets in Iraq. Folia Parasitologica, 60, 441–447.
Kritsky, D. C., Aquaro, G., & Galli, P. (2010). Microncocotyle bicoccae n. gen., n. sp. (Monogenoidea: Dactylogyridae) from the gills of the longtail silverbiddy, Gerres longirostris (Teleostei: Gerreidae), in the Red Sea, Egypt. Comparative Parasitology, 77, 137–144.
Kritsky, D. C., & Boeger, W. A. (1989). The phylogenetic status of the Ancyrocephalidae Bychowsky, 1937 (Monogenea: Dactylogyroidea). Journal of Parasitology, 75, 207–211.
Kritsky, D. C., & Boeger, W. A. (2002). Neotropical Monogenoidea. 41: New and previously described species of Dactylogyridae (Platyhelminthes) from the gills of marine and freshwater perciform fishes (Teleostei) with proposal of a new genus and a hypothesis on phylogeny. Zoosystema, 24, 7–40.
Kritsky, D. C., Boeger, W. A., & Thatcher, V. E. (1985). Neotropical Monogenea. 7. Parasites of the pirarucu, Arapaima gigas (Cuvier), with descriptions of two new species and redescription of Dawestrema cycloancistrium Price and Nowlin, 1967 (Dactylogyridae, Ancyrocephalinae). Proceedings of the Biological Society of Washington, 98, 321–331.
Kritsky, D. C., & Galli, P. (2007). Dactylogyrids (Monogenoidea) parasitizing the gills of spinefoots (Teleostei: Siganidae): Revision of Pseudohaliotrema, with redescriptions of P. sphincteroporus and P. molnari from the Great Barrier Reef, Australia. Comparative Parasitology, 74, 9–22.
Kritsky, D. C., Leiby, P. D., & Kayton, R. J. (1978). A rapid stain technique for the haptoral bars of Gyrodactylus species (Monogenea). Journal of Parasitology, 64, 172–174.
Kritsky, D. C., & Mendoza-Franco, E. F. (2008). Revision of Aristocleidus (Monogenoidea: Dactylogyridae), rediscovery of Aristocleidus hastatus, and description of Aristocleidus lamothei n. sp. from the Peruvian mojarra Diapterus peruvianus (Teleostei: Gerreidae) in Mexico. Revista Mexicana de Biodiversidad, 79, 75S–82S.
Kritsky, D. C., & Mizelle, J. D. (1968). Studies on monogenetic trematodes. XXXV. Some new and previously described North American species of Gyrodactylus. American Midland Naturalist, 79, 205–215.
Kritsky, D. C., Nguyen, H. V., Ha, N. D., & Heckmann, R. A. (2016). Revision of Metahaliotrema Yamaguti, 1953 (Monogenoidea: Dactylogyridae), with new and previously described species from the spotted scat Scatophagus argus (Linnaeus) (Perciformes: Scatophagidae) in Vietnam. Systematic Parasitology, 93, 321–335.
Lamothe-Argumedo, R., García-Prieto, L., Osorio-Sarabia, E., & Pérez-Ponce de León, G. (1997). Catálogo de la Colección Nacional de Helmintos. Mexico City: Instituto de Biología, Universidad Nacional Autónoma de México y Comisión Nacional para el Conocimiento y Uso de la Biodiversidad, 211 pp.
Machida, M. (1973). Two new trematodes from the gerrid (sic) fish of Bungo Channel, Japan. Bulletin of the National Science Museum, 16, 429–435.
Mamaev, Yu. L. (1970). [Helminths of food fishes of the Tonkin Gulf]. In: Oshmarin, P. G., Mamaev, Yu. L. & Lebedev, G. I. (Eds), Helminths of Animals of Southeast Asia. Moscow: Nauka, pp. 127–190 (In Russian).
Mamaev, Yu. L, & Parukhin, A. M. (1976). On the genus Polylabris Euzet et Cauwet, 1967 and some closely allied species of microcotylids (Monogenoidea: Microcotylidae). Parazitologiya, 10, 245–254 (In Russian).
Mendoza-Franco, E. F., Osorio, M. T., & Caspeta-Mandujano, J. M. (2015). Two new species of Aristocleidus (Monogenea) from the gills of the Mexican mojarra Eugerres mexicanus (Percifomres, Gerreidae) from southwestern Mexico. Parasite, 22, 33.
Mendoza-Franco, E. F., Roche, D. G., & Torchin, M. E. (2008). New species of Diplectanum (Monogenoidea: Diplectanidae), and proposal of a new genus of the Dactylogyridae from the gills of gerreid fishes (Teleostei) from Mexico and Panama. Folia Parasitologica, 55, 171–179.
Mendoza-Franco, E. F., Violante-González, J., & Roche, D. G. (2009). Interoceanic occurrence of species of Aristocleidus Mueller, 1936 (Monogenoidea: Dactylogyridae) parasitizing the gills of gerreid fishes in the Neotropics. Parasitology Research, 105, 703–708.
Mizelle, J. D., & Blatz, V. (1941). Studies on monogenetic trematodes. VI. Two new dactylogyrid genera from Florida fishes. American Midland Naturalist, 26, 105–109.
Paperna, I. (1972). Monogenea from Red Sea Fishes. II. Monogenea of Mullidae. Proceedings of the Helminthological Society of Washington, 39, 39–45.
Pritchard, M. H., & Kruse, G. O. W. (1982). The collection and preservation of animal parasites. Lincoln, Nebraska: University of Nebraska Press, 141 pp.
Sanders, D. F. (1944). A contribution to the knowledge of the Microcotylidae of Western Australia. Transactions of the Royal Society of South Australia, 68, 67–81.
Skinner, R. H. (1982). The interrelation of water quality, gill parasites, and gill pathology of some fishes from South Biscayne Bay, Florida. Fisheries Bulletin, 80, 269–280.
Unnithan, R. V. (1971). On the functional morphology of a new fauna of Monogenoidea on fishes from Trivandrum and environs. Part IV. Microcotylidae sensu stricto and its repartition into subsidiary taxa. American Midland Naturalist, 85, 366–398.
Vari, R. P. (1978). The terapon perches (Percoidei, Teraponidae). A cladistics analysis and taxonomic revision. Bulletin of the American Museum of Natural History, 159, 177–340.
Venkatanarasaiah, J. (1981). On three new species of Metahaliotrema Yamaguti, 1953 (Monogenea, Dactylogyridae) from the gills of marine fishes from Andhra Pradesh, India. Helminthologia, 18, 247–256.
Venkatanarasaiah, J. (1982). Two new species of the genus Protogyrodactylus Johnston et Tiegs, 1922 from marine fishes from Bay of Bengal, Andhra Pradesh, India. Rivista di Parassitologia, 43, 433–438.
Violante-González, J., & Aguirre-Macedo, M. L. (2007). Metazoan parasites of fishes from Coyuca Lagoon, Guerrero, Mexico. Zootaxa, 1531, 39–48.
Violante-González, J., Aguirre-Macedo, M. L., & Mendoza-Franco, E. F. (2007). A checklist of metazoan parasites of fish from Tres Palos Lagoon, Guerrero, Mexico. Parasitology Research, 102, 151–161.
Violante-González, J., Pulido-Flores, G., Monks, S., Rojas-Herrera, A. A., Gil-Guerrero, J. S., Garcia-Ibáñez, S., & Carbajal-Violante, J. (2015). Parasitofauna de peces de lagunas costeras del estado de Guerrero, Mexico. In: Pulido-Flores, G., Monks, S. & López-Herrera (Eds), Estudios en Biodiversidad, Volume I. Lincoln: Zea Books, pp. 78–91.
Williams, A. (1991). Monogeneans of the families Microcotylidae Taschenberg, 1879 and Heteraxinidae Price, 1962 from Western Australia, including the description of Polylabris sandarsae n. sp. (Microcotylidae). Systematic Parasitology, 18, 17–43.
Wu, X.-Y., Zhu, X.-Q., Xie, M.-Q., & Li, A.-X. (2006). The radiation of Haliotrema (Monogenea: Dactylogyridae: Ancyrocephalinae): molecular evidence and explanation inferred from LSU rDNA sequences. Parasitology, 132, 659–668.
Wu, X.-Y., Zhu, X.-Q., Xie, M.-Q., & Li, A.-X. (2007). The evaluation for generic-level monophyly of Ancyrocephalinae (Monogenea, Dactylogyridae) using ribosomal DNA sequence data. Molecular Phylogenetics and Evolution, 44, 530–544.
Zhang J.-Y. (2001). [Ancyrocephalidae Bychowsky & Nagibina, 1978], Chapter 10. In: Zhang J.-Y., Yang T., Liu L. et al. (Eds), Monogeneans of Chinese Marine Fishes. Beijing, China: Agriculture Press, pp. 79–175 (In Chinese, English descriptions of new species).
Zhang J.-Y., & Lin L. (2001). [Protogyrodactylidae Johnston & Tiegs, 1922], Chapter 11. In: Zhang J.-Y., Yang T., Liu L. et al. (Eds), Monogeneans of Chinese Marine Fishes. Beijing, China: Agriculture Press, pp. 176–183 (In Chinese, English descriptions of new species).
Acknowledgements
This study was supported by an Australian Biological Resources Study Grant No. RF215-40 to Drs Thomas Cribb and Scott Cutmore, University of Queensland, for the exploration of the parasites of fishes in Moreton Bay. Mr John Page, Mr David Thompson and the many students are gratefully acknowledged for assistance during the collection of the fishes. The support provided by the staff of the Moreton Bay Research Station was instrumental during the field operations.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All applicable institutional, national, and international guidelines for the care and use of animals were followed.
Additional information
This article was registered in the Official Register of Zoological Nomenclature (ZooBank) as urn:lsid:zoobank.org:pub:499B6C3F-2AFA-4632-816B-F3297DD84776. This article was published as an Online First article on the online publication date shown on this page. The article should be cited by using the doi number. This is the Version of Record.
This article is part of the Topical Collection Monogenea.
Rights and permissions
About this article
Cite this article
Kritsky, D.C. Species of Monogenoidea infecting the gill lamellae of the common silver-biddy Gerres oyena (Forsskål) and the common silver belly Gerres subfasciatus Cuvier (Perciformes: Gerreidae) in Moreton Bay, Queensland, Australia. Syst Parasitol 95, 499–525 (2018). https://doi.org/10.1007/s11230-018-9800-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11230-018-9800-6