Abstract
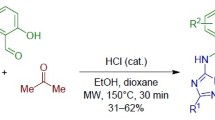
A method for producing substituted 4H-1,3-benzoxazines and their fused derivatives bearing N,N-dimethylamino moiety in the position 2 of 4H-1,3-oxazine ring from phenolic and naphtholic Mannich bases, their iodomethylates or salicylic alcohols, and 1,1,3,3-tetramethylguanidine has been developed. This reaction is supposed to proceed via the generation of o-quinone methides followed by [4+2]-cycloaddition and elimination of dimethylamine.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
S. K. Ihmaid, J. M. A. Al-Rawi, C. J. Bradley, M. J. Angove, M. N. Robertson, Eur. J. Med. Chem., 2012, 57, 85; DOI: https://doi.org/10.1016/j.ejmech.2012.08.035.
R. Morrison, Z. Zheng, I. G. Jennings, P. E. Thompson, J. M. A. Al-Rawi, Bioorg. Med. Chem. Lett., 2016, 26, 5534; DOI: https://doi.org/10.1016/j.bmcl.2016.10.003.
R. Morrison, J. M. A. Al-Rawi, J. Enzyme Inhib. Med. Chem., 2016, 31(S2), P. 86; DOI: https://doi.org/10.1080/14756366.2016.1190710.
R. B. Gammill, T. M. Judge, J. Morris, WO9006921A1, 1990; Chem. Abstrs., 1990, 114, 42797.
R. B. Gammill, T. M. Judge, J. Morris, WO9119707A2, 1991; Chem. Abstrs., 1991, 116, 235438.
R. Morrison, J. M. A. Al-Rawi, I. G. Jennings, P. E. Thompson, Eur. J. Med. Chem., 2016, 110, 326; DOI: https://doi.org/10.1016/j.ejmech.2016.01.042.
A. A. Schmalstig, K. M. Zorn, S. Murcia, A. Robinson, S. Savina, E. Komarova, V. Makarov, M. Braunstein, S. Ekins, Tuberculosis, 2022, 132, 102168; DOI: https://doi.org/10.1016/j.tube.2022.102168.
S. K. Ihmaid, C. Fitzgibbon, J. M. A. Al-Rawi, Med. Chem. Res., 2015, 24, 2825; DOI: https://doi.org/10.1007/s00044-015-1338-4.
W. S. I. Abou-Elmagd, A. I. Hashem, Med. Chem. Res., 2013, 22, 2005; DOI: https://doi.org/10.1007/s00044-012-0205-9.
S. W. Myers, H. K. Spencer, Pat. US 4164407, 1979; Chem. Abstrs., 1979, 91, 175367.
W. Schroth, E. Borgmann, K. Jasche, M. Klepel, S. Kühne, J. Müller, H.-D. Schädler, DD273571A1, 1989; Chem. Abstrs., 1989, 113, 2062.
J. F. Bereznak, E. A. Marshall, WO2000051992A1, 2000; Chem. Abstrs., 2000, 133, 207905.
R. R. Schmidt, Synthesis, 1972, 333; DOI: https://doi.org/10.1055/s-1972-21882.
E. V. Sazonova, A. N. Artemov, V. I. Faerman, N. A. Aksenova, A. A. Timofeeva, Yu. A. Zaytseva, N. V. Somov, N. Yu. Grishina, Russ. Chem. Bull., 2021, 70, 171; DOI: https://doi.org/10.1007/s11172-021-3073-y.
E. Grigat, R. Pütter, K. Schneider, K. F. Wedemeyer, Chem. Ber., 1964, 97, 3036; DOI: https://doi.org/10.1002/cber.19640971111.
B. Liu, M. Yin, H. Gao, W. Wu, H. Jiang, J. Org. Chem., 2013, 78, 3009; DOI: https://doi.org/10.1021/jo400002f.
S. Huang, Y. Pan, Y. Zhu, A. Wu, Org. Lett., 2005, 7, 3797; DOI: https://doi.org/10.1021/ol051458e.
J. Azizian, K. Yadollahzadeh, A. S. Delbari, M. M. Ghanbari, Monatsh. Chem., 2012, 143, 1417; DOI: https://doi.org/10.1007/s00706-011-0716-y.
Superbases for Organic Synthesis: Guanidines, Amidines, Phosphazenes and Related Organocatalysts, Ed. T. Ishikawa, John Wiley & Sons, Chichester, 2009, p. 24.
V. A. Osyanin, A. V. Lukashenko, D. V. Osipov, Russ. Chem. Rev., 2021, 90, 324; DOI: https://doi.org/10.1070/RCR4971.
D. V. Osipov, V. A. Osyanin, Yu. N. Klimochkin, Russ. Chem. Rev., 2017, 86, 625; DOI: https://doi.org/10.1070/RCR4679.
V. A. Osyanin, Dokt Diss., Samara, 2014, 458 p.
M. A. Zherebtsov, E. R. Zhiganshina, N. A. Lenshina, R. S. Kovylin, E. V. Baranov, N. Yu. Shushunova, M. P. Shurygina, M. V. Arsenyev, S. A. Chesnokov, V. K. Cherkasov, Russ. Chem. Bull., 2021, 70, 780; DOI: https://doi.org/10.1007/s11172-021-3151-1.
R. W. Van De Water, T. R. R. Pettus, Tetrahedron, 2002, 58, 5367; DOI: https://doi.org/10.1016/S0040-4020(02)00496-9.
I. Szatmári, F. Fülöp, Tetrahedron Lett., 2011, 52, 4440; DOI: https://doi.org/10.1016/j.tetlet.2011.06.074.
H. Sugimoto, S. Nakamura, T. Ohwada, Adv. Synth. Catal., 2007, 349, 669; DOI: https://doi.org/10.1002/adsc.200600508.
C. E. Augelli-Szafran, A. Arbor, T. M. Böhme, R. D. Schwarz, Pat. US 0220335, 2003; Chem. Abstrs., 2001, 135, 357936.
D. V. Osipov, V. A. Osyanin, G. D. Khaysanova, E. R. Masterova, P. E. Krasnikov, Yu. N. Klimochkin, J. Org. Chem., 2018, 83, 4775; DOI: https://doi.org/10.1021/acs.joc.8b00692.
V. A. Shiryaev, E. V. Radchenko, V. A. Palyulin, N. S. Zefirov, N. I. Bormotov, O. A. Serova, L. N. Shishkina, M. R. Baimuratov, K. M. Bormasheva, Y. A. Gruzd, E. A. Ivleva, M. V. Leonova, A. V. Lukashenko, D. V. Osipov, V. A. Osyanin, A. N. Reznikov, V. A. Shadrikova, A. E. Sibiryakova, I. M. Tkachenko, Yu. N. Klimochkin, Eur. J. Med. Chem., 2018, 158, 214; DOI: https://doi.org/10.1016/j.ejmech.2018.08.009.
D. V. Osipov, V. A. Osyanin, Yu. N. Klimochkin, Russ. J. Org. Chem., 2013, 49, 398; DOI: https://doi.org/10.1134/S1070428013030147.
B. Büyükkıdan, S. Bilgiç, O. Bilgiç, Synth. Commun., 2001, 31, 1263; DOI: https://doi.org/10.1081/SCC-100104015.
V. A. Osyanin, D. V. Osipov, M. R. Demidov, Yu. N. Klimochkin, J. Org. Chem., 2014, 79, 1192; DOI: https://doi.org/10.1021/jo402543s.
Author information
Authors and Affiliations
Corresponding author
Additional information
The study was financially supported by the the Russian Science Foundation (Project No. 22-13-00253) with the use of the equipment of the Multi-Access Center “Investigation of the Physicochemical Properties of Substances and Materials” of the SSTU.
No human or animal subjects were used in this research.
The authors declare no competing interests.
Based on the materials of the VI North Caucasus Symposium on Organic Chemistry NCOCS-2022 (April 18–22, 2022, Stavropol, Russia).
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 11, pp. 2451–2459, November, 2022.
Rights and permissions
About this article
Cite this article
Osyanin, V.A., Osipov, D.V., Krasnikov, P.E. et al. [4+2]-Cycloaddition of 1,1,3,3-tetramethylguanidine and o-quinone methides: synthesis of arene-fused 2-dimethylamino-4H-1,3-oxazines. Russ Chem Bull 71, 2451–2459 (2022). https://doi.org/10.1007/s11172-022-3673-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-022-3673-1