Abstract
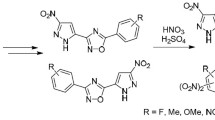
We developed an approach to the synthesis of 2,5-disubstituted pyrazole-containing 1,3,4-oxadiazoles by acylation of 5-(nitropyrazolyl)tetrazoles with alkyl, aryl, and hetaryl acyl chlorides with subsequent recyclization of the intermediate N-acyltetrazoles into 1,3,4-oxadiazoles and studied nitration of the obtained 2-aryl-5-(nitropyrazolyl)-1,3,4-oxadiazoles.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
S. Y. Kotian, C. D. Mohan, A. A. Merlo, S. Rangappa, S. C. Nayak, K. M. L. Rai, K. S. Rangappa, J. Mol. Liq., 2020, 297, 111686; DOI: https://doi.org/10.1016/j.molliq.2019.111686.
I. E. Mikhailov, Yu. M. Artyushkina, G. A. Dushenko, V. I. Minkin, Russ. Chem. Bull., 2020, 69, 2302; DOI: https://doi.org/10.1007/s11172-020-3039-5.
X. Xu, Z. Li, Z. Bi, T. Yu, W. Ma, K. Feng, Y. Li, Q. Peng, Adv. Mater., 2018, 30, No. 28, Art. ID 1800737; DOI: https://doi.org/10.1002/adma.201800737.
Y. Du, Z. Qu, H. Wang, H. Cui, X. Wang, Propellants, Explos., Pyrotech., 2021, 46, 860; DOI: https://doi.org/10.1002/prep.202000318.
I. Yu. Gudkova, I. N. Zyuzin D. B. Lempert, Russ. J. Phys. Chem. B, 2020, 14, 302; DOI: https://doi.org/10.1134/S1990793120020062.
Salahuddin, A. Mazumder, M. S. Yar, R. Mazumder, G. S. Chakraborthy, M. J. Ahsan, M. U. Rahman, Synth. Commun., 2017, 47, 1805; DOI: https://doi.org/10.1080/00397911.2017.1360911.
H. Z. Zhang, Z. L. Zhao, C. H. Zhou, Eur. J. Med. Chem., 2018, 144, 444; DOI: https://doi.org/10.1016/j.ejmech.2017.12.044.
A. Benassi, F. Doria, V. Pirota, Int. J. Mol. Sci., 2020, 21, 8692; DOI: https://doi.org/10.3390/ijms21228692.
A. Vaidya, D. Pathak, K. Shah, Chem. Biol. Drug Des., 2021, 97, 572; DOI: https://doi.org/10.1111/cbdd.13795.
M. Krasavin, A. Shetnev, T. Sharonova, S. Baykov, S. Kalinin, A. Nocentini, V. Sharoyko, G. Poli, T. Tuccinardi, S. Presnukhina, T. B. Tennikova, C. T. Supuran, Eur. J. Med. Chem., 2019, 164, 92; DOI: https://doi.org/10.1016/j.ejmech.2018.12.049.
V. C. Damalanka, Y. Kim, K. R. Alliston, P. M. Weerawarna, A. C. G. Kankanamalage, G. H. Lushington, N. Mehzabeen, K. P. Battaile, S. Lovell, K. O. Chang, W. C. Groutas, J. Med. Chem., 2016, 59, 1899; DOI: https://doi.org/10.1021/acs.jmedchem.5b01464.
F. Caputo, S. Corbetta, O. Piccolo, D. Vigo, Org. Process Res. Dev., 2020, 24, 1149; DOI: https://doi.org/10.1021/acs.oprd.0c00155.
G. Chawla, B. Naaz, A. A. Siddiqui, Mini Rev. Med. Chem., 2018, 18, 216; DOI: https://doi.org/10.2174/1389557517666170127121215.
V. B. Makane, V. S. Krishna, E. V. Krishna, M. Shukla, B. Mahizhaveni, S. Misra, S. Chopra, D. Sriram, V. N. A. Dusthackeer, H. B. Rode, Future Med. Chem., 2019, 11, 499; DOI: https://doi.org/10.4155/fmc-2018-0378.
P. Pitasse-Santos, V. Sueth-Santiago, M. E. F. Lima, J. Braz. Chem. Soc., 2018, 29, 435; DOI 10.21577/0103-5053.20170208.
M. Y. Wani, A. Ahmad, R. A. Shiekh, K. J. Al-Ghamdi, A. J. F. N. Sobral, Bioorg. Med. Chem., 2015, 23, 4172; DOI: https://doi.org/10.1016/j.bmc.2015.06.053.
J. V. Faria, P. F. Vegi, A. G. C. Miguita, M. S. dos Santos, N. Boechat, A. M. R. Bernardino, Bioorg. Med. Chem., 2017, 25, 5891; DOI: https://doi.org/10.1016/j.bmc.2017.09.035.
V. V. Utochnikova, I. A. Vatsadze, D. M. Tsymbarenko, A. S. Goloveshkin, S. Z. Vatsadze, Phys. Chem. Chem. Phys., 2021, 23, 25480; DOI: https://doi.org/10.1039/D1CP03924E.
S. Zhang, Z. Gao, D. Lan, Q. Jia, N. Liu, J. Zhang, K. Kou, Molecules, 2020, 25, 3475; DOI: https://doi.org/10.3390/molecules25153475.
S. G. Zlotin, I. L. Dalinger, N. N. Makhova, V. A. Tartakovsky, Russ. Chem. Rev., 2020, 89, 1; DOI: https://doi.org/10.1070/RCR4908.
N. V. Muravyev, K. A. Monogarov, I. L. Dalinger, N. Koga, A. N. Pivkina, Phys. Chem. Chem. Phys., 2021, 23, 11797; DOI: https://doi.org/10.1039/d1cp01530c.
M. S. Klenov, D. B. Lempert, A. A. Konnov, D. A. Gulyaev, I. A. Vatsadze, T. S. Kon’kova, Yu. N. Matyushin, E. A. Miroshnichenko, A. B. Vorob’ev, Ya. O. Inozemtsev, A. V. Inozemtsev, A. N. Pivkina, A. O. Dmitrienko, V. A. Tartakovsky, Russ. Chem. Bull., 2022, 71, 1123; DOI: https://doi.org/10.1007/s11172-022-3512-4.
T. E. Khoranyan, T. K. Shkineva, I. A. Vatsadze, A. Kh. Shakhnes, N. V. Muravyev, A. B. Sheremetev, I. L. Dalinger, Chem. Heterocycl. Compd., 2022, 58, 37; DOI: https://doi.org/10.1007/s10593-022-03054-1.
V. P. Sinditskii, A. D. Smirnova, V. V. Serushkin, N. V. Yudin, I. A. Vatsadze, I. L. Dalinger, V. G. Kiselev, A. B. Sheremetev, Thermochim. Acta, 2021, 698, 178876; DOI: https://doi.org/10.1016/j.tca.2021.178876.
I. L. Dalinger, T. K. Shkineva, I. A. Vatsadze, A. V. Kormanov, A. M. Kozeev, K. Yu. Suponitsky, A. N. Pivkina, A. B. Sheremetev, FirePhysChem, 2021, 1, 83; DOI: https://doi.org/10.1016/j.fpc.2021.04.005.
T. K. Shkineva, I. A. Vatsadze, T. E. Khoranyan, D. L. Lipilin, A. N. Pivkina, I. L. Dalinger, Chem. Heterocycl. Compd., 2021, 57, 828; DOI: https://doi.org/10.1007/s10593-021-02987-3.
A. I. Dalinger, A. V. Medved’ko, A. I. Balalaeva, I. A. Vatsadze, I. L. Dalinger, S. Z. Vatsadze, Chem. Heterocycl. Compd., 2020, 56, 180; DOI: https://doi.org/10.1007/s10593-020-02643-2.
S. Ningaiah, U. K. Bhadraiah, S. D. Doddaramappa, S. Keshavamurthy, C. Javarasetty, Bioorg. Med. Chem. Lett., 2014, 24, 245; DOI: https://doi.org/10.1016/j.bmcl.2013.11.029.
S. Bansal, M. Bala, S. K. Suthar, S. Choudhary, S. Bhattacharya, V. Bhardwaj, S. Singla, A. Joseph, Eur. J. Med. Chem., 2014, 80, 167; DOI: https://doi.org/10.1016/j.ejmech.2014.04.045.
T. Yan, G. Cheng, H. Yang, New J. Chem., 2020, 44, 6643; DOI: https://doi.org/10.1039/d0nj00518e.
X.-x. Zheng, T.-o. Yan, L. Qian, H.-w. Yang, G.-b. Cheng, Def. Technol., 2022, DOI: https://doi.org/10.1016/j.dt.2022.03.003.
H. Li, L. Zhang, N. Petrutik, K. Wang, Q. Ma, D. Shem-Tov, F. Zhao, M. Gozin, ASC Cent. Sci., 2020, 6, 54; DOI: https://doi.org/10.1021/acscentsci.9b01096.
R. Huisgen, J. Sauer, H. Sturm, Angew. Chem., 1958, 70, 272; DOI: https://doi.org/10.1002/ange.19580700918.
T. F. Osipova, G. I. Koldobsky, V. A. Ostrovsky, Zh. Org. Khim. [J. Org. Chem. USSR], 1984, 20, 2468 (in Russian).
V. A. Ostrovskii, G. I. Koldobskii, R. E. Trifonov, in Comprehensive Heterocyclic Chemistry III, Eds A. R. Katritzky, Ch. A. Ramsden, E. F. V. Scriven, R. J. K. Taylor, 2008, Vol. 6, Elsevier, Amsterdam, p. 259.
Q. Wang, K. C. Mgimpatsang, M. Konstantinidou, S. V. Shishkina, A. Dömling, Org. Lett. 2019, 21, 7320; DOI: https://doi.org/10.1021/acs.orglett.9b02614.
I. L. Dalinger, A. V. Kormanov, I. A. Vatsadze, O. V. Serushkina, T. K. Shkineva, K. Yu. Suponitsky, A. N. Pivkina, A. B. Sheremetev, Chem. Heterocycl. Compd., 2016, 52, 1025; DOI: https://doi.org/10.1007/s10593-017-2003-2.
A. S. Gavrilov, V. V. Kachala, N. E. Kuz’mina, E. L. Golod, Russ. J. Gen. Chem., 2004, 74, 752; DOI: https://doi.org/10.1023/B:RUGC.0000039090.05255.64.
K. Nepali, H.-Y. Lee, J.-P. Liou, J. Med. Chem., 2019, 62, 2851; DOI: https://doi.org/10.1021/acs.jmedchem.8b00147.
A. M. Starosotnikov, D. V. Shkaev, M. A. Bastrakov, I. V. Fedyanin, S. A. Shevelev, I. L. Dalinger, Mendeleev Commun., 2018, 28, 638; DOI: https://doi.org/10.1016/j.mencom.2018.11.025.
S. P. Korolev, M. A. Pustovarova, A. M. Starosotnikov, M. A. Bastrakov, Yu. Yu. Agapkina, S. A. Shevelev, M. B. Gottikh, Biochemistry, 2017, 11, 286; DOI: https://doi.org/10.1134/S1990750817030064.
M. Bastrakov, A. Starosotnikov, Pharmaceuticals, 2022, 15, 705; DOI: https://doi.org/10.3390/ph15060705.
P. Wardman, Curr. Med. Chem., 2001, 8, 739; DOI: https://doi.org/10.2174/0929867013372959.
I. L. Dalinger, A. V. Kormanov, T. K. Shkineva, A. B. Sheremetev, Asian J. Org. Chem., 2020, 9, 811; DOI: https://doi.org/10.1002/ajoc.201900757.
T. K. Shkineva, I. A. Vatsadze, I. L. Dalinger, Mendeleev Commun., 2019, 29, 429; DOI: https://doi.org/10.1016/j.mencom.2019.07.025.
I. L. Dalinger, I. A. Vatsadze, T. K. Shkineva, G. P. Popova, S. A. Shevelev, Yu. V. Nelyubina, J. Heterocycl. Chem., 2013, 59, 911; DOI: https://doi.org/10.1002/jhet.1026.
A. A. Zaitsev, I. L. Dalinger, S. A. Shevelev, Russ. Chem. Rev., 2009, 78, 589; DOI: https://doi.org/10.1070/RC2009v078n07ABEH004015.
V. M. Vinogradov, T. I. Cherkasova, I. L. Dalinger, S. A. Shevelev, Russ. Chem. Bull., 1993, 42, 1552; DOI: https://doi.org/10.1007/BF00699193.
J. A. Greenberg, T. Sammakia, J. Org. Chem., 2017, 82, 3245; DOI: https://doi.org/10.1021/acs.joc.6b02931.
Yu. A. Manaev, M. A. Andreeva, V. P. Perevalov, B. I. Stepanov, V. A. Dubrovskaya, V. I. Seraya, J. Gen. Chem. USSR, 1982, 52, 2291.
V. P. Perevalov, M. A. Andreeva, L. I. Baryshnenkova, Yu. A. Manaev, G. S. Yamburg, B. I. Stepanov, V. A. Dubrovskaya, Chem. Heterocycl. Compd., 1983, 19, 1326; DOI: https://doi.org/10.1007/BF00842843.
P. Rzepecki, M. Wehner, O. Molt, R. Zadmard, K. Harms, T. Schrader, Synthesis, 2003, 1815; DOI: https://doi.org/10.1055/s-2003-41031.
Author information
Authors and Affiliations
Corresponding author
Additional information
Dedicated to the Academician of the Russian Academy of Sciences V. A. Tartakovsky on the occasion of his 90th birthday.
No human or animal subjects were used in this research.
The authors declare no competing interests.
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 8, pp. 1737–1744, August, 2022.
Rights and permissions
About this article
Cite this article
Shkineva, T.K., Serushkina, O.V., Vatsadze, I.A. et al. Synthesis of 2,5-disubstituted pyrazolyl-1,3,4-oxadiazoles by the Huisgen reaction. Russ Chem Bull 71, 1737–1744 (2022). https://doi.org/10.1007/s11172-022-3584-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-022-3584-1