Abstract
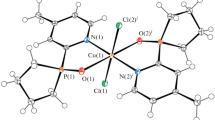
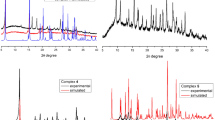
Copper(ii) and manganese(ii) chelate complexes were synthesized using the new N,O-hybrid ligand 1,3-dibenzhydryl-5-(pyridin-2-yl)-1,3,5-diazaphosphinane 5-oxide. The synthesized complexes were characterized by physicochemical methods. The molecular and crystal structures of the complexes were established by X-ray diffraction. All new compounds were evaluated for cytotoxicity against the cancer cell lines M-HeLa and HuTu80 and the normal cell line Chang liver.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
U. Ndagi, N. Mhlongo, M. Soliman, Drug Des. Dev. Ther., 2017, 11, 599; DOI: https://doi.org/10.2147/DDDT.S119488.
J.-X. Liang, H.-J. Zhong, G. Yang, K. Vellaisamy, D.-L. Ma, C.-H. Leung, J. Inorg. Biochem., 2017, 177, 276; DOI: https://doi.org/10.1016/j.jinorgbio.2017.06.002.
T. Johnstone, K. Suntharalingam, S. Lippard, Chem. Rev., 2016, 116, 3436; DOI: https://doi.org/10.1021/acs.chemrev.5b00597.
V. Brabec, O. Hrabina, J. Kasparkova, Coord. Chem. Rev., 2017, 351, 2; DOI: https://doi.org/10.1016/j.ccr.2017.04.013.
P. Starha, J. Vanc, Z. Travnicek, Coord. Chem. Rev., 2019, 380, 103; DOI: https://doi.org/10.1016/j.ccr.2018.09.017.
A. Matesanz, C. Hernandez, A. Rodriguez, P. Souza, Dalton Trans., 2011, 40, 5738; DOI: https://doi.org/10.1039/C1DT10212E.
A. Barve, A. Kumbhar, M. Bhat, B. Joshi, R. Butcher, U. Sonawane, R. Joshi, Inorg. Chem., 2009, 48, 9120; DOI: https://doi.org/10.1021/ic9004642.
C. Santini, M. Pellei, V. Gandin, M. Porchia, F. Tisato, C. Marzano, Chem. Rev., 2014, 114, 815; DOI: https://doi.org/10.1021/cr400135x.
M. Porchia, M. Pellei, F. Bello, C. Santini, Molecules, 2020, 25, 5814; DOI: https://doi.org/10.3390/molecules25245814.
G. Barone, A. Terenzi, A. Lauria, A.-M. Almerico, J. Leal, N. Busto, B. Garcia, Coord. Chem. Rev., 2013, 257, 2848; DOI: https://doi.org/10.1016/j.ccr.2013.02.023.
H. Wang, M. Sorolla II, X. Wang, A. Jacobson, H. Wang, A. Pillai, Transition Met. Chem., 2019, 44, 237; DOI: https://doi.org/10.1007/s11243-018-0288-3.
I. A. Lutsenko, M. A. Kiskin, K. A. Koshenskova, P. V. Primakov, A. V. Khoroshilov, O. B. Bekker, I. L. Eremenko, Russ. Chem. Bull., 2021, 70, 463; DOI: https://doi.org/10.1007/s11172-021-3109-3.
C. Freidline, R. Tobias, Inorg. Chem., 1966, 5, 354; DOI: https://doi.org/10.1021/ic50037a006
T. Iiyama, M. Chikira, T. Oyoshi, H. Sugiyama, J. Biol. Inorg. Chem., 2003, 8, 135; DOI: https://doi.org/10.1007/s00775-002-0398-3.
R. Chen, C.-S. Liu, H. Zhang, Y. Guo, X.-H. Bu, M. Yang, J. Inorg. Biochem., 2007, 101, 412; DOI: https://doi.org/10.1016/j.jinorgbio.2006.11.001.
Y.-L. Wang, Y.-C. Liu, Z.-S. Yang, G.-C. Zhao, Bioelectrochemistry, 2004, 65, 77; DOI: https://doi.org/10.1016/j.bioelechem.2004.07.002.
S. Tardito, O. Bussolati, F. Gaccioli, R. Gatti, S. Guizzardi, J. Uggeri, L. Marchiò, M. Lanfranchi, R. Franchi-Gazzola, Histochem. Cell Biol., 2006, 126, 473; DOI: https://doi.org/10.1007/s00418-006-0183-4.
N. S. Rukk, L. G. Kuzmina, G. A. Davydova, G. A. Buzanov, S. K. Belus, E. I. Kozhukhova, V. M. Retivov, T. V. Ivanova, V. N. Krasnoperova, B. M. Bolotin, Russ. Chem. Bull., 2020, 69, 1394; DOI: https://doi.org/10.1007/s11172-020-2914-4.
E.-J. Gao, Y. Zhang, L. Lin, R.-S. Wang, L. Dai, Q. Liang, M.-C. Zhu, M.-L. Wang, L. Liu, W.-X. He, Y.-J. Zhang, J. Coord. Chem., 2011, 64, 3992; DOI: https://doi.org/10.1080/00958972.2011.634910.
K. Trigulova, A. Shamsieva, R. Faizullin, P. Lonnecke, E. Hey-Hawkins, A. Voloshina, E. Musina, A. Karasik, Russ. J. Coord. Chem., 2020, 46, 600; DOI: https://doi.org/10.1134/S1070328420090055.
A. Karasik, E. Musina, A. Balueva, I. Strelnik, O. Sinyashin, Pure Appl. Chem., 2017, 89, 293; DOI: https://doi.org/10.1515/pac-2016-1022.
B. A. Arbuzov, O. A. Erastov, G. N. Nikonov, T. A. Zyablikova, D. S. Yufit, Yu. T. Struchkov, Bull. Acad. Sci. USSR, Div. Chem. Sci., 1981, 30, 1539.
A. V. Shamsieva, K. R. Trigulova, R. R. Fayzullin, V. V. Khrizanforova, Yu. G. Budnikova, E. I. Musina, A. A. Karasik, Russ. Chem. Bull., 2018, 67, 1206; DOI: https://doi.org/10.1007/s11172-018-2203-7.
A. Artem’ev, M. Davydova, A. Berezin, T. Sukhikh, D. Samsonenko, Inorg. Chem. Front., 2021, 8, 2261; DOI: https://doi.org/10.1039/d1qi00036e.
A. Artem’ev, M. Davydova, A. Berezin, V. Brel, V. Morgalyuk, I. Bagryanskaya, D. Samsonenko, Dalton Trans., 2019, 48, 16448; DOI: https://doi.org/10.1039/c9dt03283e.
A. Berezin, M. Davydova, I. Bagryanskaya, O. Artyushin, V. Brel, A. Artem’ev, Inorg. Chem. Commun., 2019, 107, 107473; DOI:https://doi.org/10.1016/j.inoche.2019.107473.
D. Montagner, B. Fresch, K. Browne, V. Gandin, A. Erxleben, Chem. Commun., 2017, 53, 134; DOI: https://doi.org/10.1039/C6CC08100B.
A. Voloshina, S. Gumerova, A. Sapunova, N. Kulik, A. Mirgorodskay, A. Kotenko, T. Prokopyeva, V. Mikhailov, L. Zakharova, O. Sinyashin, Biochi. Biophys. Acta, 2020, 1864, 129728; DOI: https://doi.org/10.1016/j.bbagen.2020.129728.
G. Sheldrick, Acta Crystallogr., Sect. A, 2015, 71, 3; DOI:https://doi.org/10.1107/S2053273314026370.
G. Sheldrick, Acta Crystallogr., Sect. C, 2015, 71, 3; DOI: https://doi.org/10.1107/S2053229614024218.
A. Spek, Acta Crystallogr., Sect. C, 2015, 71, 9; DOI: https://doi.org/10.1107/S2053229614024929.
A. Spek, Acta Crystallogr., Sect. D, 2009, 65, 148; DOI: https://doi.org/10.1107/S090744490804362X.
C. Macrae, I. Sovago, S. Cottrell, P. Galek, P. McCabe, E. Pidcock, M. Platings, G. Shield, J. Stevens, M. Towler, P. Wood, J. Appl. Crystallogr., 2020, 53, 226; DOI: https://doi.org/10.1107/S1600576719014092.
Author information
Authors and Affiliations
Corresponding author
Additional information
Dedicated to Academician of the Russian Academy of Sciences V. I. Ovcharenko on the occasion of his 70th birthday.
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 7, pp. 1410–1421, July, 2022.
The measurements were carried out using the equipment of the Assigned Spectral-Analytical Center of Shared Facilities for Study of Structure, Composition and Properties of Substances and Materials of the FRC Kazan Scientific Center of the Russian Academy of Sciences.
No human or animal subjects were used in this research.
The authors declare no competing interests.
Rights and permissions
About this article
Cite this article
Trigulova, K.R., Shamsieva, A.V., Kasimov, A.I. et al. Copper(ii) and manganese(ii) complexes based on a new N,O-chelating ligand bearing the 1,3,5-diazaphosphorinane moiety. Russ Chem Bull 71, 1410–1421 (2022). https://doi.org/10.1007/s11172-022-3547-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-022-3547-6