Abstract
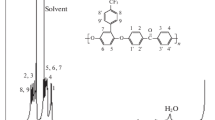
Monoesters of tri- and tetraglycol and 1-pyrenecarboxylic acid were characterized for the first time by NMR spectroscopy and elemental analysis. The photophysical properties of the compounds obtained were studied. A selective sensory response of the esters to nitro-containing explosives in an aqueous medium was revealed. The maximum value of the Stern—Volmer fluorescence quenching constant for 2,4,6-trinitrotoluene was found to be 1.13•104 L mol−1.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
J. Yinon, Trends Anal. Chem., 2002, 21, 292; DOI: https://doi.org/10.1016/S0165-9936(02)00408-9.
J. Yinon, Counterterrorist Detection Techniques of Explosives, Elsevier, 2007, 454 p.
F. Akhgari, H. Fattahi, Y. M. Oskoei, Sensors and Actuators, B: Chem., 2015, 221, 867; DOI: https://doi.org/10.1016/B978-0-444-52204-7.X5017-2.
D. Rembelski, J. Bordet, Q. Brouard, B. Minot, C. Barthet, C. Frénois, Procedia Engineering, 2015, 120, 1011; DOI: https://doi.org/10.1016/j.proeng.2015.08.680.
X. Chang, G. Wang, C. Yu, Y. Wang, M. He, J. Fan, Y. Fang, J. Photochem. Photobiol. A Chem., 2015, 298, 9; DOI: https://doi.org/10.1016/j.jphotochem.2014.10.008.
N. N. Dioubankova, A. D. Malakhov, D. A. Stetsenko, V. A. Korshun, Russ. Chem. Bull., 2004, 53, 463; DOI: https://doi.org/10.1023/B:RUCB.0000030825.05631.40.
I. S. Kovalev, N. V. Slovesnova, D. S. Kopchuk, G. V. Zyryanov, O. S. Taniya, V. L. Rusinov, O. N. Chupakhin, Russ. Chem. Bull., 2014, 63, 1312; DOI: https://doi.org/10.1007/s11172-014-0596-5.
E. S. Barskaya, V. V. Shorokhov, A. V. Rzheutsky, A. D. Khudyakov, I. V. Yudin, V. A. Tafeenko, N. V. Zyk, E. K. Beloglazkina, Russ. Chem. Bull., 2019, 68, 638; DOI: https://doi.org/10.1007/s11172-019-2468-5.
N. M. Zadymova, Ph. D. Thesis (Chem.), Lomonosov Moscow State University, Moscow, 2014, 273 pp. (in Russian).
J. Hu, P. Wang, X. Zhao, L. Lv, S. Yang, B. Song, Q. Wang, Chem. Commun., 2014, 50, 14125; DOI: https://doi.org/10.1039/c4cc05195e.
Y. Niko, Y. Hiroshige, S. Kawauchi, G. I. Konishi, Tetrahedron, 2012, 68, 6177; DOI: https://doi.org/10.1016/j.tet.2012.05.072.
P. Šoustek, M. Michl, N. Almonasy, O. Machalický, M. Dvořák, A. Lyčka, Dyes Pigment., 2008, 78, 139; DOI: https://doi.org/10.1016/j.dyepig.2007.11.003.
Y. Suzuki, T. Morozumi, H. Nakamura, M. Shimomura, T. Hayashita, R. A. Bartsh, J. Phys. Chem. B, 1998, 102, 7910; DOI: https://doi.org/10.1021/jp981567t.
P. Hrdlovič, L. Horinová, Š. Chmela, Can. J. Chem., 1995, 73, 1948; DOI: https://doi.org/10.1139/v95-240.
D. Daems, M. Van den Zegel, N. Boens, F. C. De Schryver, Eur. Biophys. J., 1985, 12, 97; DOI: https://doi.org/10.1007/BF00260432.
J. J. Kim, R. A. Beardslee, D. T. Phillips, H. W. Offen, J. Chem. Phys., 1969, 51, 2761; DOI: https://doi.org/10.1063/1.1672406.
A. Ruiu, M. Vonlanthen, S. M. Rojas-Montoya, I. González-Méndez, E. Rivera, Molecules, 2019, 24, 4083, DOI: https://doi.org/10.3390/molecules24224083.
T. I. Lin, Biophys. Chem., 1982, 15, 277; DOI: https://doi.org/10.1016/0301-4622(82)80011-2.
X. Wang, L. Liu, S. Zhu, J. Peng, L. Li, RSC Adv., 2017, 7, 40842; DOI: https://doi.org/10.1039/c7ra08142a.
S. Kanagalingam, J. Spartalis, T. M. Cao, J. Duhamel, Macromolecules, 2002, 35, 8571; DOI: https://doi.org/10.1021/ma020784w.
S. G. Bertolotti, C. M. Previtali, J. Macromol. Sci., Part A, 1994, 31, 439; DOI: https://doi.org/10.1080/10601329409351530.
I. S. Kovalev, O. S. Taniya, D. S. Kopchuk, K. Giri, A. Mukherjee, S. Santra, A. Majee, M. Rahman, G. V. Zyryanov, V. A. Bakulev, O. N. Chupakhin, New J. Chem., 2018, 42, 19864; DOI: https://doi.org/10.1039/c8nj03807d.
A. D. Hughes, I. C. Glenn, A. D. Patrick, A. Ellington, E. V. Anslyn, Chem. — A Eur. J., 2008, 14, 1822; DOI: https://doi.org/10.1002/chem.200701546.
J. H. Hong, J. H. Choi, D. G. Cho, Bull. Korean Chem. Soc., 2014, 35, 3158; DOI: https://doi.org/10.5012/bkcs.2014.35.11.3158.
A. Shrivastava, V. Gupta, Chronicles Young Sci., 2011, 2, 21, DOI: https://doi.org/10.4103/2229-5186.79345.
Author information
Authors and Affiliations
Corresponding author
Additional information
Dedicated to Academician of the Russian Academy of Sciences V. N. Charushin on the occasion of his 70th birthday.
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 6, pp. 1174–1179, June, 2021.
This work was financially supported by the Russian Foundation for Basic Research (Project No. 19-33-90155).
This paper does not contain descriptions of studies on animals or humans.
The authors declare no competing interests.
Rights and permissions
About this article
Cite this article
Sadieva, L.K., Taniya, O.S., Kovalev, I.S. et al. Pyrene-1-carboxylic acid polyethylene glycol esters: synthesis and photophysical studies. Russ Chem Bull 70, 1174–1179 (2021). https://doi.org/10.1007/s11172-021-3201-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-021-3201-8