Abstract
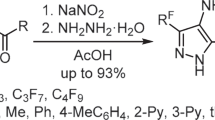
We studied different approaches to the synthesis of polyfluoroalkyl-containing 1-methyl-4-nitrosopyrazoles, which are based on the cyclization of 4,4,4-trifluoro-3,3-dihydroxy-2-hydroxyimino-1-R-butan-1-ones or 2-hydroxyimino-1,3-diketones with methylhydrazine and on a one-pot sequential treatment of 1,3-diketones with sodium nitrite and methylhydrazine. It was found that the regioselectivity of the formation of 1-methyl-4-nitrosopyrazoles is affected by the steric factors of 1,3-dicarbonyl reagents. The study of the mycostatic effect of 4-nitrosopyrazoles showed that the introduction of a bulky polyfluoroalkyl or tert-butyl substituent leads to a decrease in their activity.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
L. Pizzuti, A. Barschak, F. Stefanello, M. Farias, C. Lencina, M. Roesch-Ely, W. Cunico, S. Moura, C. Pereira, Curr. Org. Chem., 2014, 18, 115; DOI: https://doi.org/10.2174/13852728113179990029.
R. Pérez-Fernández, P. Goya, J. Elguero, Arkivoc, 2014, Part ii, 233; DOI: https://doi.org/10.3998/ark.5550190.p008.131.
Ş. G. Küçükgüzel, S. Şenkardeş, Eur. J. Med. Chem., 2015, 97, 786; DOI: https://doi.org/10.1016/j.ejmech.2014.11.059.
D. Havrylyuk, O. Roman, R. Lesyk, Eur. J. Med. Chem., 2016, 113, 145; DOI: https://doi.org/10.1016/j.ejmech.2016.02.030.
A. Ansari, A. Ali, M. Asif, S. Shamsuzzaman, New J. Chem., 2017, 41, 16; DOI: https://doi.org/10.1039/C6NJ03181A.
P. K. Mykhailiuk, Chem. Rev., 2021, 121, 3, 1670; DOI: https://doi.org/10.1021/acs.chemrev.0c01015.
K. Kaur, V. Kumar, G. K. Gupta, J. Fluorine Chem., 2015, 178, 306; DOI: https://doi.org/10.1016/j.jfluchem.2015.08.015.
J. C. Sloop, C. Holder, M. Henary, Eur. J. Org. Chem., 2015, 3405; DOI: https://doi.org/10.1002/ejoc.201500258.
D. O’Hagan, J. Fluorine Chem., 2010, 131, 1071; DOI: https://doi.org/10.1016/j.jfluchem.2010.03.003.
J. Wang, M. Sánchez-Roselló, J. L. Aceña, C. del Pozo, A. E. Sorochinsky, S. Fustero, V. A. Soloshonok, H. Liu, Chem. Rev., 2014, 114, 2432; DOI:https://doi.org/10.1021/cr4002879.
Y. Zafrani, D. Yeffet, G. Sod-Moriah, A. Berliner, D. Amir, D. Marciano, E. Gershonov, S. Saphier, J. Med. Chem., 2017, 60, 797; DOI: https://doi.org/10.1021/acs.jmedchem.6b01691.
L. V. Politanskaya, G. A. Selivanova, E. V. Panteleeva, E. V. Tretyakov, V. E. Platonov, P. V. Nikul’shin, A. S. Vinogradov, Y. V. Zonov, V. M. Karpov, T. V. Mezhenkova, A. V. Vasilyev, A. B. Koldobskii, O. S. Shilova, S. M. Morozova, Y. V. Burgart, E. V. Shchegolkov, V. I. Saloutin, V. B. Sokolov, A. Y. Aksinenko, V. G. Nenajdenko, M. Y. Moskalik, V. V. Astakhova, B. A. Shainyan, A. A. Tabolin, S. L. Ioffe, V. M. Muzalevskiy, E. S. Balenkova, A. V. Shastin, A. A. Tyutyunov, V. E. Boiko, S. M. Igumnov, A. D. Dilman, N. Y. Adonin, V. V. Bardin, S. M. Masoud, D. V. Vorobyeva, S. N. Osipov, E. V. Nosova, G. N. Lipunova, V. N. Charushin, D. O. Prima, A. G. Makarov, A. V. Zibarev, B. A. Trofimov, L. N. Sobenina, K. V. Belyaeva, V. Y. Sosnovskikh, D. L. Obydennov, S. A. Usachev, Russ. Chem. Rev., 2019, 88, 425; DOI: https://doi.org/10.1070/RCR4871.
Ya. V. Burgart, N. A. Agafonova, E. V. Shchegolkov, O. P. Krasnykh, S. O. Kushch, N. P. Evstigneeva, N. A. Gerasimova, V. V. Maslova, G. A. Triandafilova, S. Yu. Solodnikov, M. V. Ulitko, G. F. Makhaeva, E. V. Rudakova, S. S. Borisevich, N. V. Zilberberg, N. V. Kungurov, V. I. Saloutin, O. N. Chupakhin, Eur. J. Med. Chem., 2020, 208, 112768; DOI: https://doi.org/10.1016/j.ejmech.2020.112768.
Nitrofural, https://www.drugbank.ca/drugs/DB00336.
M. Vega-Teijido, I. Caracelli, J. Zukerman-Schpector, J. Mol. Graph. Model., 2006, 24, 349; DOI: https://doi.org/10.1016/j.jmgm.2005.09.008.
V. I. Saloutin, Y. V. Burgart, Z. E. Skryabina, O. G. Kuzueva, J. Fluorine Chem., 1997, 84, 107; DOI: https://doi.org/10.1016/S0022-1139(97)00054-7.
O. G. Khudina, Y. V. Burgart, V. I. Saloutin, M. A. Kravchenko, Russ. J. Org. Chem., 2011, 47, 904; DOI: https://doi.org/10.1134/s1070428011060121.
O. G. Khudina, Y. V. Burgart, V. I. Saloutin, M. A. Kravchenko, Russ. Chem. Bull., 2010, 59, 1967; DOI: https://doi.org/10.1007/s11172-010-0341-7.
D. N. Bazhin, Yu. S. Kudyakova, N. A. Nemytova, Ya. V. Burgart, V. I. Saloutin, J. Fluorine Chem., 2016, 186, 28; DOI: https://doi.org/10.1016/j.jfluchem.2016.04.009.
Ya. V. Burgart, N. A. Agafonova, E. V. Shchegolkov, V. V. Maslova, G. A. Triandafilova, S. Yu. Solodnikov, O. P. Krasnykh, V. I. Saloutin, Chem. Heterocycl. Compd., 2019, 55, 52; DOI: https://doi.org/10.1007/s10593-019-02418-4.
G. M. Sheldrick, Acta Crystallogr., Sect. A: Found. Crystallogr., 2008, 64, 112; DOI: https://doi.org/10.1107/S0108767307043930.
Author information
Authors and Affiliations
Corresponding author
Additional information
Dedicated to Academician of the Russian Academy of Sciences V. N. Charushin on the occasion of his 70th birthday.
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 6, pp. 1135–1140, June, 2021.
The work was carried out in the framework of the Russian state assignment AAAA-A19-119011790134-1, using the equipment of the Centre for Joint Use “Spectroscopy and Analysis of Organic Compounds” (CJU “SAOC”).
This work does not involve human participants and animal subjects.
The authors declare no competing interest.
Rights and permissions
About this article
Cite this article
Agafonova, N.A., Burgart, Y.V., Gerasimova, N.A. et al. Alternative approaches to the synthesis of polyfluoroalkyl-containing 1-methyl-4-nitrosopyrazoles. Russ Chem Bull 70, 1135–1140 (2021). https://doi.org/10.1007/s11172-021-3195-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-021-3195-2