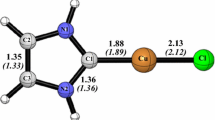
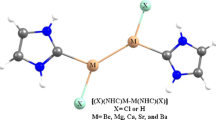
Abstract
A DFT study was carried out of how the nature of metal, the oxidation state of the metal (0 and +2), as well as the structures of N-heterocyclic carbene (NHC) and other ligands influence the heterolytic dissociation energies of the metal-ligand bond in the complexes M-NHC (M = Ni, Pd, Pt). It was shown that a change in the oxidation state of the metal can be followed by a considerable change in the M-NHC bond dissociation energy (up to nearly 21 kcal mol−1), which is also strongly influenced by the ligand in the trans-position to NHC.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
S. Díez-González, N. Marion, S. P. Nolan, Chem. Rev., 2009, 109, 3612.
M. N. Hopkinson, C. Richter, M. Schedler, F. Glorius, Nature, 2014, 510, 485.
R. D. J. Froese, C. Lombardi, M. Pompeo, R. P. Rucker, M. G. Organ, Acc. Chem. Res., 2017, 50, 2244.
N. Hazari, P. R. Melvin, M. M. Beromi, Nat. Rev. Chem., 2017, 1, 0025.
I. E. Chikunov, G. S. Ranny, A. V. Astakhov, V. A. Tafeenko, V. M. Chernyshev, Russ. Chem. Bull., 2018, 67, 2003.
S. B. Soliev, A. V. Astakhov, D. V. Pasyukov, V. M. Chernyshev, Russ. Chem. Bull., 2020, 69, 683.
R. Visbal, M. C. Gimeno, Chem. Soc. Rev., 2014, 43, 3551.
T. Fleetham, G. Li, J. Li, Adv. Mater., 2017, 29, 1601861.
M. Elie, J. L. Renaud, S. Gaillard, Polyhedron, 2018, 140, 158.
L. Oehninger, R. Rubbiani, I. Ott, Dalton Trans., 2013, 42, 3269.
W. Liu, R. Gust, Coord. Chem. Rev., 2016, 329, 191.
T. Zou, C.-N. Lok, P.-K. Wan, Z.-F. Zhang, S.-K. Fung, C.-M. Che, Curr. Opin. Chem. Biol., 2018, 43, 30.
W. A. Herrmann, Angew. Chem., Int. Ed., 2002, 41, 1290.
K. J. Cavell, A. T. Normand, in N-Heterocyclic Carbenes in Transition Metal Catalysis and Organocatalysis, Ed. C. S. J. Cazin, Springer Netherlands, Dordrecht, 2011, p. 299.
B. R. M. Lake, M. R. Chapman, C. E. Willans, in Organometallic Chemistry, Royal Society of Chemistry, 2016, vol. 40, p. 107.
D. J. Nelson, J. M. Praetorius, C. M. Crudden, in N-Heterocyclic Carbenes: From Laboratory Curiosities to Efficient Synthetic Tools, Royal Society of Chemistry, London, 2017, p. 46.
A. V. Astakhov, O. V. Khazipov, E. S. Degtyareva, V. N. Khrustalev, V. M. Chernyshev, V. P. Ananikov, Organometallics, 2015, 34, 5759.
E. G. Gordeev, D. B. Eremin, V. M. Chernyshev, V. P. Ananikov, Organometallics, 2018, 37, 787.
A. Astakhov, S. Soliev, E. Gordeev, V. Chernyshev, V. Ananikov, Dalton Trans., 2019, 48, 17052.
V. M. Chernyshev, A. V. Astakhov, I. E. Chikunov, R. V. Tyurin, D. B. Eremin, G. S. Ranny, V. N. Khrustalev, V. P. Ananikov, ACS Catal., 2019, 9, 2984.
A. Y. Chernenko, D. V. Pasyukov, A. V. Astakhov, V. A. Tafeenko, V. M. Chernyshev, Russ. Chem. Bull., 2018, 67, 1196.
V. M. Chernyshev, O. V. Khazipov, M. A. Shevchenko, A. Y. Chernenko, A. V. Astakhov, D. B. Eremin, D. V. Pasyukov, A. S. Kashin, V. P. Ananikov, Chem. Sci., 2018, 9, 5564.
O. V. Khazipov, M. A. Shevchenko, D. V. Pasyukov, A. Y. Chernenko, A. V. Astakhov, V. A. Tafeenko, V. M. Chernyshev, V. P. Ananikov, Catal. Sci. Technol., 2020, 10, 1228.
A. V. Astakhov, O. V. Khazipov, A. Y. Chernenko, D. V. Pasyukov, A. S. Kashin, E. G. Gordeev, V. N. Khrustalev, V. M. Chernyshev, V. P. Ananikov, Organometallics, 2017, 36, 1981.
O. V. Khazipov, M. A. Shevchenko, A. Y. Chernenko, A. V. Astakhov, D. V. Pasyukov, D. B. Eremin, Y. V. Zubavichus, V. N. Khrustalev, V. M. Chernyshev, V. P. Ananikov, Organometallics, 2018, 37, 1483.
A. Y. Chernenko, A. V. Astakhov, D. V. Pasyukov, P. V. Dorovatovskii, Y. V. Zubavichus, V. N. Khrustalev, V. M. Chernyshev, Russ. Chem. Bull., 2018, 67, 79.
D. B. Eremin, E. A. Denisova, A. Yu. Kostyukovich, J. Martens, G. Berden, J. Oomens, V. N. Khrustalev, V. M. Chernyshev, V. P. Ananikov, Chem.-Eur J., 2019, 25, 16564.
V. I. Minkin, Pure Appl. Chem., 1999, 71, 1919.
L. Cavallo, A. Correa, C. Costabile, H. Jacobsen, J. Organomet. Chem., 2005, 690, 5407.
H. Jacobsen, A. Correa, A. Poater, C. Costabile, L. Cavallo, Coord. Chem. Rev., 2009, 253, 687.
T. Dröge, F. Glorius, Angew. Chem., Int. Ed., 2010, 49, 6940.
R. Dorta, E. D. Stevens, C. D. Hoff, S. P. Nolan, J. Am. Chem. Soc., 2003, 125, 10490.
A. K. de K. Lewis, S. Caddick, F. G. N. Cloke, N. C. Billingham, P. B. Hitchcock, J. Leonard, J. Am. Chem. Soc., 2003, 125, 10066.
R. Dorta, E. D. Stevens, N. M. Scott, C. Costabile, L. Cavallo, C. D. Hoff, S. P. Nolan, J. Am. Chem. Soc., 2005, 127, 2485.
K. Matsubara, S. Miyazaki, Y. Koga, Y. Nibu, T. Hashimura, T. Matsumoto, Organometallics, 2008, 27, 6020.
E. P. A. Couzijn, E. Zocher, A. Bach, P. Chen, Chem.-Eur. J., 2010, 16, 5408.
C. Dash, M. M. Shaikh, R. J. Butcher, P. Ghosh, Dalton Trans., 2010, 39, 2515.
A. Comas-Vives, J. N. Harvey, Eur. J. Inorg. Chem., 2011, 2011, 5025.
R. Jagadeesan, G. Velmurugan, P. Venuvanalingam, RSC Advances, 2015, 5, 80661.
K. K. Pandey, Int. J. Quantum Chem., 2016, 116, 537.
R. H. Crabtree, Chem. Rev., 2015, 115, 127.
M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, B. Mennucci, G. A. Petersson, H. Nakatsuji, M. Caricato, X. Li, H. P. Hratchian, A. F. Izmaylov, J. Bloino, G. Zheng, J. L. Sonnenberg, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, J. A. Montgomery, Jr., J. E. Peralta, F. Ogliaro, M. J. Bearpark, J. Heyd, E. N. Brothers, K. N. Kudin, V. N. Staroverov, R. Kobayashi, J. Normand, K. Raghavachari, A. P. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, N. Rega, N. J. Millam, M. Klene, J. E. Knox, J. B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R. E. Stratmann, O. Yazyev, A. J. Austin, R. Cammi, C. Pomelli, J. W. Ochterski, R. L. Martin, K. Morokuma, V. G. Zakrzewski, G. A. Voth, P. Salvador, J. J. Dannenberg, S. Dapprich, A. D. Daniels, Ö. Farkas, J. B. Foresman, J. V. Ortiz, J. Cioslowski, D. J. Fox, Gaussian 09, Rev. D.01, Gaussian, Inc., Wallingford, 2009.
E. Lee, D. V. Yandulov, J. Organomet. Chem., 2011, 696, 4095.
L. Dobrzańska, E. Stander-Grobler, C. E. Strasser, S. Cronje, H. G. Raubenheimer, Helv. Chim. Acta, 2012, 95, 2528.
L. A. Curtiss, M. P. McGrath, J. P. Blaudeau, N. E. Davis, R. C. Binning, Jr., L. Radom, J. Chem. Phys., 1995, 103, 6104.
R. Krishnan, J. S. Binkley, R. Seeger, J. A. Pople, J. Chem. Phys, 1980, 72, 650.
A. D. McLean, G. S. Chandler, J. Chem. Phys., 1980, 72, 5639.
C. E. Check, T. O. Faust, J. M. Bailey, B. J. Wright, T. M. Gilbert, L. S. Sunderlin, J. Phys. Chem. A, 2001, 105, 8111.
P. J. Hay, W. R. Wadt, J. Chem. Phys., 1985, 82, 299.
C. Adamo, V. Barone, J. Chem. Phys., 1999, 110, 6158.
J. P. Perdew, K. Burke, M. Ernzerhof, Phys. Rev. Lett., 1996, 77, 3865.
S. M. Bachrach, in Computational Organic Chemistry, Ed. S. M. Bachrach, John Wiley & Sons, Inc., 2014, p. 1.
F. Lipparini, G. Scalmani, B. Mennucci, E. Cancès, M. Caricato, M. J. Frisch, J. Chem. Phys., 2010, 133, 014106.
G. Scalmani, M. J. Frisch, J. Chem. Phys., 2010, 132, 114110.
J. Tomasi, B. Mennucci, R. Cammi, Chem. Rev., 2005, 105, 2999.
A. E. Reed, L. A. Curtiss, F. Weinhold, Chem. Rev., 1988, 88, 899.
G. Frenking, N. Fröhlich, Chem. Rev., 2000, 100, 717.
A. J. Arduengo, S. F. Gamper, J. C. Calabrese, F. Davidson, J. Am. Chem. Soc., 1994, 116, 4391.
A. A. Danopoulos, D. Pugh, Dalton Trans., 2008, 30.
Y. Hoshimoto, Y. Hayashi, H. Suzuki, M. Ohashi, S. Ogoshi, Organometallics, 2014, 33, 1276.
A. F. Henwood, M. Lesieur, A. K. Bansal, V. Lemaur, D. Beljonne, D. G. Thompson, D. Graham, A. M. Z. Slawin, I. D. W. Samuel, C. S. J. Cazin, E. Zysman-Colman, Chem. Sci., 2015, 6, 3248.
F. Hering, J. Nitsch, U. Paul, A. Steffen, F. M. Bickelhaupt, U. Radius, Chem. Sci., 2015, 6, 1426.
E. A. B. Kantchev, C. J. O’Brien, M. G. Organ, Angew. Chem., Int. Ed., 2007, 46, 2768.
R. Jothibasu, K.-W. Huang, H. V. Huynh, Organometallics, 2010, 29, 3746.
D. S. McGuinness, K. J. Cavell, B. W. Skelton, A. H. White, Organometallics, 1999, 18, 1596.
S. Caddick, F. G. N. Cloke, P. B. Hitchcock, A. K. de K. Lewis, Angew. Chem., Int. Ed., 2004, 43, 5824.
H. V. Huynh, C. Holtgrewe, T. Pape, L. L. Koh, E. Hahn, Organometallics, 2006, 25, 245.
J. Berding, M. Lutz, A. L. Spek, E. Bouwman, Appl. Organomet. Chem., 2011, 25, 76.
C. H. Lee, D. A. Lutterman, D. G. Nocera, Dalton Trans., 2013, 42, 2355.
M. Heckenroth, E. Kluser, A. Neels, M. Albrecht, Dalton Trans., 2008, 6242.
D. M. Khramov, E. L. Rosen, J. A. V. Er, P. D. Vu, V. M. Lynch, C. W. Bielawski, Tetrahedron, 2008, 64, 6853.
A. Zanardi, J. A. Mata, E. Peris, Organometallics, 2009, 28, 4335.
P. L. Arnold, F. G. N. Cloke, T. Geldbach, P. B. Hitchcock, Organometallics, 1999, 18, 3228.
G. C. Fortman, N. M. Scott, A. Linden, E. D. Stevens, R. Dorta, S. P. Nolan, Chem. Commun., 2010, 46, 1050.
G. Dahm, C. Bailly, L. Karmazin, S. Bellemin-Laponnaz, J. Organomet. Chem., 2015, 794, 115.
Y. Zhang, C. R. P. Fulong, C. E. Hauke, M. R. Crawley, A. E. Friedman, T. R. Cook, Chem.-Eur. J., 2017, 23, 4532.
Author information
Authors and Affiliations
Corresponding author
Additional information
The authors express their gratitude to V. P. Ananikov for fruitful discussions and valuable comments.
This work was financially supported by the Russian Foundation for Basic Research (Project No. 19-73-10100).
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 11, pp. 2073—2081, November, 2020.
Rights and permissions
About this article
Cite this article
Astakhov, A.V., Soliev, S.B. & Chernyshev, V.M. Metal-ligand bond dissociation energies in the Ni, Pd, and Pt complexes with N-heterocyclic carbenes: effect of the oxidation state of the metal (0, +2). Russ Chem Bull 69, 2073–2081 (2020). https://doi.org/10.1007/s11172-020-3002-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-020-3002-5