Abstract
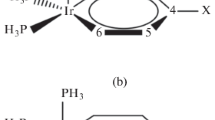
Features of the electronic structure of adducts of transition metal hydride complexes (Cp*M(dppe)H, dppe is the 1,2-(diphenylphosphino)ethane, M = Fe, Ru, Os; CpM(CO)3H, M = Mo, W) with acids and bases were analyzed with the ADF2014 program using energy decomposition analysis (EDA) by the Ziegler-Rauk method combined with the natural orbitals for chemical valence theory (ETS-NOCV). The nature of orbital interactions in the complex determines the reaction pathway: σMH → σ*OH interaction leads to the proton transfer to hydride ligand, nM → σ*OH leads to the metal atom protonation, nN → σ*MH implies the metal hydride deprotonation, and σMH → n*B corresponds to the hydride transfer to Lewis acid. It was shown that M-H bond polarization change has the similar character upon the formation of complexes with Brønsted and Lewis acids. The ease of polarization of M-H bonds in complexes CpM(CO)3H determines their reactivity as proton and hydride ion donors.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
R. G. Pearson, Chem. Rev., 1985, 85, 41.
R. H. Crabtree, in The Organometallic Chemistry of the Transition Metals, John Wiley and Sons, Inc., Hoboken, New Jersey, USA, 2014, Ch. 3, p. 69.
R. L. Sweany, J. Halpern, J. Am. Chem. Soc., 1977, 99, 8335.
D. P. Estes, A. K. Vannucci, A. R. Hall, D. L. Lichtenberger, J. R. Norton, Organometallics, 2011, 30, 3444.
S. S. Kristjansdottir, J. R. Norton, in Transition Metal Hydrides, Ed. A. Dedieu, VCH, New York, 1991, p. 309.
T. Y. Cheng, R. M. Bullock, Organometallics, 2002, 21, 2325.
J. Choi, L. Tang, J. R. Norton, J. Am. Chem. Soc., 2007, 129, 234.
R. M. Bullock, J. S. Song, J. Am. Chem. Soc., 1994, 116, 8602.
J. S. Song, D. J. Szalda, R. M. Bullock, C. J. C. Lawrie, M. A. Rodkin, J. R. Norton, Angew. Chem., Int. Ed., 1992, 31, 1233.
R. M. Bullock, Chem. Eur. J., 2004, 10, 2366.
H. Guan, M. Iimura, M. P. Magee, J. R. Norton, G. Zhu, J. Am. Chem. Soc., 2005, 127, 7805.
H. R. Guan, S. A. Saddoughi, A. P. Shaw, J. R. Norton, Organometallics, 2005, 24, 6358.
H. R. Guan, M. Iimura, M. P. Magee, J. R. Norton, K. E. Janak, Organometallics, 2003, 22, 4084.
R. F. Jordan, J. R. Norton, J. Am. Chem. Soc., 1982, 104, 1255.
K. Abdur-Rashid, T. P. Fong, B. Greaves, D. G. Gusev, J. G. Hinman, S. E. Landau, A. J. Lough, R. H. Morris, J. Am. Chem. Soc., 2000, 122, 9155.
E. S. Shubina, N. V. Belkova, E. V. Bakhmutova, L. N. Saitkulova, A. V. Ionidis, L. M. Epstein, Russ. Chem. Bull. (Int. Ed.), 1998, 47, 846. [Izv. Akad. Nauk, Ser. Khim., 1998, 817].
E. S. Shubina, N. V. Belkova, L. M. Epstein, J. Organomet. Chem., 1997, 536, 17.
V. A. Levina, O. A. Filippov, E. I. Gutsul, N. V. Belkova, L. M. Epstein, A. Lleds, E. S. Shubina, J. Am. Chem. Soc., 2010, 132, 1123.
N. V. Belkova, E. I. Gutsul, O. A. Filippov, V. A. Levina, D. A. Valyaev, L. M. Epstein, A. Lleds, E. S. Shubina, J. Am. Chem. Soc., 2006, 128, 3486.
S. Namorado, M. A. Antunes, L. F. Veiros, J. R. Ascenso, M. T. Duarte, A. M. Martins, Organometallics, 2008, 27, 4589.
M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, B. Mennucci, G. A. Petersson, H. Nakatsuji, M. Caricato, X. Li, H. P. Hratchian, A. F. Izmaylov, J. Bloino, G. Zheng, J. L. Sonnenberg, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, J. A. Montgomery, Jr., J. E. Peralta, F. Ogliaro, M. J. Bearpark, J. Heyd, E. N. Brothers, K. N. Kudin, V. N. Staroverov, R. Kobayashi, J. Normand, K. Raghavachari, A. P. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, N. Rega, N. J. Millam, M. Klene, J. E. Knox, J. B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R. E. Stratmann, O. Yazyev, A. J. Austin, R. Cammi, C. Pomelli, J. W. Ochterski, R. L. Martin, K. Morokuma, V. G. Zakrzewski, G. A. Voth, P. Salvador, J. J. Dannenberg, S. Dapprich, A. D. Daniels, Ö. Farkas, J. B. Foresman, J. V. Ortiz, J. Cioslowski, D. J. Fox, Gaussian 09, Revision C.01, Gaussian, Inc., Wallingford CT, 2009.
ADF2014, SCM, Theoretical Chemistry, Vrije Universiteit, Amsterdam, The Netherlands; http://www.scm.com.
T. Ziegler, A. Rauk, Inorg. Chem., 1979, 18, 1558.
T. Ziegler, A. Rauk, Inorg. Chem., 1979, 18, 1755.
F. M. Bickelhaupt, E. J. Baerends, in Rev. Comput. Chem., Eds K. B. Lipkowitz, D. B. Boyd, John Wiley and Sons, Inc., New York, USA, 2007, Vol. 15, p. 1.
M. P. Mitoraj, A. Michalak, T. Ziegler, J. Chem. Theory Comput., 2009, 5, 962.
O. A. Filippov, N. V. Belkova, L. M. Epstein, A. Lledos, E. S. Shubina, ChemPhysChem, 2012, 13, 2677.
Y. Zhao, N. E. Schultz, D. G. Truhlar, J. Chem. Phys., 2005, 123, 1611. 3.
L. E. Roy, P. J. Hay and R. L. Martin, J. Chem. Theory Comput., 2008, 4, 1029.
W. R. Wadt, P. J. Hay, J. Chem. Phys., 1985, 82, 284.
P. J. Hay, W. R. Wadt, J. Chem. Phys., 1985, 82, 270.
E. D. Glendening, J, K. Badenhoop, A. E. Reed, J. E. Carpenter, J. A. Bohmann, C. M. Morales, F. Weinhold, NBO 5.0, Theoretical Chemistry Institute, University of Wisconsin, Madison, 2001; http://www.chem.wisc.edu/~nbo5/.
A. E. Reed, L. A. Curtiss, F. Weinhold, Chem. Rev., 1988, 88, 899.
E. Van Lenthe, E. J. Baerends, J. Comput. Chem., 2003, 24, 1142.
M. P. Mitoraj, A. Michalak, T. Ziegler, Organometallics, 2009, 28, 3727.
I. V. Alabugin, M. Manoharan, S. Peabody, F. Weinhold, J. Am. Chem. Soc., 2003, 125, 5973.
A. Rauk, Orbital Interaction Theory of Organic Chemistry, 2nd ed., John Wiley and Sons, Inc., New York, USA, 2001.
N. V. Belkova, E. S. Shubina, L. M. Epstein, Acc. Chem. Res., 2005, 38, 624.
T.-Y. Cheng, B. S. Brunschwig, R. M. Bullock, J. Am. Chem. Soc., 1998, 120, 1312.
Author information
Authors and Affiliations
Corresponding author
Additional information
Dedicated to the 60th anniversary of foundation of the A. N. Nesmeyanov Institute of Organoelement Compounds of the Russian Academy of Sciences.
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 11, pp. 2428—2433, November, 2014.
Rights and permissions
About this article
Cite this article
Filippov, O.A., Golub, I.E., Osipova, E.S. et al. Activation of M—H bond upon the complexation of transition metal hydrides with acids and bases. Russ Chem Bull 63, 2428–2433 (2014). https://doi.org/10.1007/s11172-014-0758-5
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-014-0758-5