Abstract
Fisheries resources in Hong Kong have been overexploited since the 1970s due to intensive bottom trawling and other fishing activities that have depleted stocks and destroyed marine habitat. To rehabilitate depleted fisheries resources, a permanent ban on trawling in Hong Kong territorial waters came into force on December 31, 2012. In order to determine whether the trawl facilitated recovery of fish communities, trawl surveys were conducted at two sites in each of the eastern, southern and western (estuarine) coastal waters of Hong Kong before and three years after the trawl ban. A total of 315 species and 86 families of fishes in nine feeding groups were encountered during the surveys. Mean trophic level of the fish community, abundance and biomass of total fishes and of predatory fishes increased in eastern and western waters after the ban, but no changes or declines in these metrics were observed in southern waters. Although initial recovery in fish community were observed in eastern and western waters, anthropogenic disturbances might hinder the recovery process, including a large-scale reclamation for construction of coastal infrastructures in the west, illegal trawling, and expansion of non-trawling fishing efforts in the southern and eastern waters. Longer term monitoring is needed to evaluate the effects of the trawl ban, and determine whether recovery in the southern waters will continue to be constrained by the anthropogenic disturbances.
Graphic abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Overexploitation of fisheries resources has been documented in many regions globally (Pauly et al. 1998; Worm et al. 2009). Overfishing has substantially reduced the abundance of large, long-lived, predatory species over the past 60 years, while small, short-lived species at lower trophic levels have become dominant (Cheung et al. 2007; Christensen et al. 2014; Pauly et al. 1998). Bottom trawling is one of the most destructive and widespread anthropogenic disturbances of the seafloor and fishery resources (Hiddink et al. 2017; Sciberras et al. 2018). It captures fishes and macroinvertebrates regardless of their economic or ecological value (Jennings and Kaiser 1998; Jones 1992), altering the community composition and size structure of demersal and benthic communities (de Juan et al. 2007; Jennings et al. 2001; Mangano et al. 2014), while bottom trawling disturbs benthic habitat by sediment resuspension and removal of sessile organisms such as sponges, soft corals and bivalves (Kaiser et al. 2006; Thrush and Dayton 2002). To rebuild the depleted fisheries resources and facilitate benthic recovery, bans on trawling have been implemented in the Mediterranean (Pipitone et al. 2000), Scandinavia (Svedäng 2010), Kenya (Munga et al. 2012), the Philippines (Ronquillo and Llana 1987), Indonesia (Bailey 1997) and elsewhere, and have been effective in some instances.
Fisheries resources in Hong Kong have been overexploited since the 1970s as a result of an increase in the number of mechanized fishing vessels (particularly bottom trawlers) after World War II (Cheung and Sadovy 2004; Leung 2001). Fishery landings in absolute and in catch per unit effort terms from local waters decreased greatly during the late 1970s (Cheung 2015; Cheung and Sadovy 2004), and since then, communities in coastal waters that formerly included large predatory fishes shifted to dominance by short-lived, small-bodied fishes and crustaceans that feed at low trophic levels (Cheung and Sadovy 2004; ERM, 1998; Sadovy 1998). Excessive trawling, which formerly accounted for 50% of the total fisheries landings from Hong Kong waters, with an annual catch of around 12,000 tonnes in 2006 (AFCD, 2013), was seen as the major cause of this community shift (Cheung and Sadovy 2004; Morton 2011). To mitigate overfishing and destruction of benthic habitat caused by trawling, the Government of the Hong Kong Special Administrative Region (HKSAR) introduced a permanent territory wide trawl ban on pair, stern, shrimp and hang trawling in Hong Kong on December 31, 2012 (AFCD, 2011). The purpose of the ban is also intended to rebuild depleted fisheries resources and facilitate marine ecosystem recovery.
Previous studies have demonstrated that trawl bans allow fish communities to recover, leading to larger size, higher biomass and greater diversity (Bergman et al. 2015; Hoskin et al. 2011; Murawski et al. 2000). In the Bay of Biscay (western coast of France), for example, predatory fishes at high trophic levels benefitted from a four-year trawling ban (Serrano et al. 2011) and, more generally, the mean trophic level (MTL) of fish communities was elevated under lower fishing pressure after the ban (Pauly et al. 1998; Shannon et al. 2014). Parallel studies in Hong Kong waters have also demonstrated that increases in body size and mean trophic position of predatory crustaceans and a higher diversity of benthic crustacean assemblages were detected after the trawl ban (Tao et al. 2020a, 2020b, 2018). However, studies on the potential recovery of fish communities after the trawl ban in Hong Kong are limited.
In this study, we hypothesised that the ban would be associated with increases in: (1) total abundance and biomass of demersal fishes; (2) mean body size; (3) species richness; (4) predator abundance and biomass (in absolute and percentage terms), and (5) the MTL of the fish community. Hong Kong lacks any possible control site (i.e., an area without trawling in the past), so for this study, we conducted a before-and-after comparison without the control to determine whether demersal fish communities in Hong Kong waters showed signs of recovery three years after the trawl ban.
Materials and methods
Field sampling and sample processing
Hong Kong lies within the northern tropics (latitude 22°09′N to 22°31′N, longitude 113°51′E to 114°23′E) on the southeast coast of China, bordered by the South China Sea. The territorial coast waters of Hong Kong cover 1,650 km2 (SMO, 2021) and are strongly influenced by its monsoonal climate. The wet season is from May to September accompanied by southwest monsoon winds, while the dry season is from November to March with northeast monsoon winds; the months in between are transitional (Lee et al. 2006; HKO, 2021). During the wet season, temperatures are warm and rainfall is high with the consequence that salinities in the Pearl River Estuary are down to 15 psu (Goodkin et al. 2011; Lee et al. 2006). The western coastal waters of Hong Kong receive a continuous inflow of freshwater, nutrients, sediments and pollutants from the Pearl River, especially during the wet season associated with the southwest monsoon. The eastern waters are influenced by oceanic currents, while southern waters occupy a transitional zone between the estuarine west and the oceanic east (Morton and Morton 1983; Ng et al. 2016).
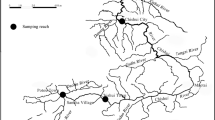
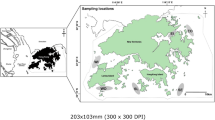
Standardised bottom trawl surveys were conducted at two sites in each of the east, south and west of Hong Kong: i.e., inner (EI) and outer (EO) Tolo Channel; southeastern waters (SE) and southern Lamma Island (SL); and inner (WI) and outer (WO) Pearl River estuary (Fig. 1). At each site, two transects were trawled per survey during daytime. Twelve sets of monthly samples were collected using commercial shrimp trawlers during January–December 2004 (2004; before the trawl ban), June 2013–May 2014 (2013–14; immediately after the trawl ban), and June 2015–May 2016 (2015–16; three years after the trawl ban) at the four sites in eastern and western waters, and at the additional two sites in the south during 2013–14 and 2015–16. The 2004 samples were assumed to represent conditions before the trawl ban, while 2013–14 and 2015–16 were representative of post-ban conditions.
Sampling locations of benthic fishes in Hong Kong waters: inner (EI) and outer (EO) Tolo Channel, southeastern waters (SE), southern Lamma Island (SL), outer (WO) and inner (WI) Pearl River estuary. Red arrows represent sampling locations in eastern waters, black arrows represent sampling locations in southern water, while blue arrows represent sampling locations in western waters. The dotted line is Hong Kong’s jurisdictional boundary within which all trawling has been banned since 31 December 2012
Along each transect, the trawler (beam size: 2 m; stretched mesh size: 2 cm), which had a 15-m outrigger and 10 nets, towed for 30 min at a speed of 5 to 7 km/h, and trawled a total area of 0.0375 − 0.0525 km2 per transect. Catches from the ten replicate nets in each transect were pooled and taken to the laboratory, where the vast majority of fishes were identified to species level or, where this was not possible, were combined into higher taxa (genus or family level; e.g., Cynoglossus spp., Muraenidae). Each taxon was classified into a feeding mode functional group (Table S1) based on the dietary information available on FishBase (Froese and Pauly 2019) and the Fish Database of Taiwan (Shao 2019). Nine feeding modes were defined: herbivore/detritivore (HV/DV), herbivore/omnivore (HV/OV), omnivore (OV), miscellaneous/opportunist (OP), zooplanktivore (ZP), zooplanktivore/zoobenthivore (ZP/ZB), zoobenthivore (ZB), zoobenthivore/piscivore (ZB/PV) and piscivore (PV). The abundance and biomass of fishes in each transect from each survey were standardized as individuals/km2 and kg/km2, respectively, according to Eq. 1:
where the beam width was 2 m for each net, the number of nets was 10, and trawl distance (km) was recorded during each trawl survey using a hand-held GPS device (GPSMAP-62S, Garmin) or estimated from the product of the average trawl speed and tow time.
Stable isotope analysis (SIA)
Apart from fish samples, samples of bivalves and turritellid gastropods were also collected during the trawl surveys and subjected to SIA. Samples from two transects in each site were pooled to include as many species as possible for SIA. For each species, one individual represented one replicate; 3 replicates were adopted whenever samples were available. Dorsal muscles from the fishes, foot and adductor muscle tissues from the bivalves, and foot muscle from gastropods were dissected, rinsed with Milli-Q water, freeze-dried for at least 72 h, then homogenised for the SIA. Samples (1.0 mg) were weighed into tin capsules and analysed for stable nitrogen isotopes (δ15N (‰)) using a Nu Perspective Isotope Ratio Mass Spectrometer (Nu instruments Ltd, Wrexham, United Kingdom) coupled with a Eurovector EA3028 Elemental Analyser (Isomass Scientific Inc., Alberta, Canada) at The University of Hong Kong Stable Isotope Laboratory. The SIA followed those described in Tao et. al (2020a).
Based on the δ15N value of each species using SIA, samples from the dry and wet seasons of 2012 (before the trawl ban) and the dry season of 2015 (after the trawl ban) were used to measure the trophic level (TL) of each fish species at each site with the following equation:
where TLijk and δ15Nijk are the trophic level and the mean δ15Ni, respectively, of fish species i in season k of year j. δ15Nref jk is the mean value of baseline species in season k of year j. This study used 2.9‰ as a trophic fractionation factor, which was suggested by McCutchan (2003) to account for δ15N enrichment per trophic transfer; 2 is the TL of the baseline (Post 2002). To estimate the TL of each fish species, δ15N values of long-lived filter feeders (i.e., bivalves from the families Anomiidae, Arcidae, Cardiidae, Corbulidae, Mytilidae, Ostreidae, Pectinidae, Pinnidae, Pteriidae or Veneridae, as well as turritellid gastropods) collected from the same trawl samples were used as baseline references (Post 2002).
Statistical analyses
Since there are significant spatial variations in the fish community in eastern, southern and western waters in Hong Kong (ERM, 1998), inter-year differences in the fish community at each site were investigated separately to determine the effectiveness of the trawl ban.
Univariate analyses
Wilcoxon signed-rank tests were applied to the following community metrics: total abundance, total biomass, species richness, and mean individual weight. The mean weight of fishes in each of the two transects taken each month at the sampling sites was obtained by dividing total fish biomass by the total number of individuals captured (Dulvy et al. 2004; Nicholson and Jennings 2004). At each site, we investigated the yearly differences among the three surveyed years in eastern (EI and EO) and western waters (WI and WO) and between the two surveyed years (post-trawl ban) in southern waters (SE and SL), using each of the 12 months in each sampled year as the paired sample (e.g. data from January in 2004 and January in 2014 were paired). Bonferroni corrections were performed to adjust statistical significance to account for multiple testing (Rice 1989).
Predatory fishes were defined as those that were piscivorous or fed on macroinvertebrates larger than macrozooplankton category (i.e., > 2 cm; (Shin et al. 2010)), and therefore fishes with feeding modes ZB, ZB/PV and PV were regarded as predators (Table S1). Wilcoxon signed-rank tests and Bonferroni corrections were also applied to inter-year tests of differences in absolute and relative abundance and biomass of predatory fishes at each site.
We quantified the mean trophic level (MTL) of fish communities in 2004, 2013–14 and 2015–16 at the eastern and western sites, and in 2013–14 and 2015–16 at the southern sites, according to Pauly (1998):
where TLijk and Bijk are the trophic level and biomass of species i in year j in site k, respectively. The TL of each species at each site in the 2012 dry season was used to calculate the MTL of fish community in 2004, while the TLs from the 2015 dry season at each site were used to calculate the MTL of fish communities in 2013–14 and 2015–16. At least 87.1% of the total abundance and 83.2% of the total biomass of the fishes collected could be assigned TLs using SIA data from the dry seasons of 2012 and 2015. For the minority of species that had not been subject to SIA in 2012 and 2015, data on their TL was derived from wet season data of 2012 at the same site, or neighbouring sites in the same area (i.e., EI and EO, SE and SL, WI and WO) or, in a very few cases, from the relevant trophic information available in FishBase (Froese and Pauly 2019).
To investigate inter-year changes in the biomass of TLs at each site, fish species were grouped into three trophic categories (TL < 3.25; 3.25 ≤ TL ≤ 3.75, and TL > 3.75) at each site in each year. The value of 3.25 has been widely used as a standard cut-off TL in assessing the status of fish stocks (Pauly and Watson 2005; Shannon et al. 2014), and biomass of predatory fishes at or above TL 3.75 has been declining substantially in the North Atlantic during recent decades (Christensen et al. 2003). The relative biomass of each trophic category at each site in each year was calculated by using the following equation:
where Bijk is the biomass of species k in the particular feeding mode i in year j, Bj is the total biomass of the fish community in the year j. Wilcoxon signed-rank tests with Bonferroni corrections were performed to determine whether the MTL, and the absolute and relative biomass of each trophic category differed among years at each site, using months as replicates.
Multivariate analyses
Species were combined to family level for multivariate analyses as the abundance and biomass for most individual species was small. Rare families (i.e. families that contributed to < 5% of the total catch) were excluded from the analyses (Clarke and Gorley 2006). Fish abundance and biomass data were Log10(X + 1) transformed before calculation of Bray–Curtis similarity index to down-weight the contribution of abundant families to dissimilarity patterns (Anderson et al. 2008). Permutational multivariate analysis of variance (PERMANOVA) was applied to abundance and biomass data to test for inter-year changes in community composition. Prior to running PERMANOVA, permutational tests of the homogeneity of multivariate dispersions (PERMDISP), were performed. If significant differences in fish community among years were observed, pairwise multiple comparison tests were conducted to determine which pairs of years differed, whereupon one-way similarity percentage (SIMPER) analyses were applied to identify the families responsible for most of the inter-year differences. The relative biomass change of major fish families was calculated as follows:
where Bij% is the relative biomass change in the family i in year j (i.e., 2013–2014 and 2015–2016). Given that the biomass of Gymnuridae was zero in EI and EO in 2004, the relative biomass change in Gymnuridae in 2015–2016 was calculated by using the equation:
Similarly, the relative biomass changes in major fish families in SE and SL were only compared between 2013–14 and 2015–16. All multivariate analyses were performed using PRIMER v6 with PERMANOVA (Anderson et al. 2008; Clarke and Gorley 2006).
Results
Abundance, biomass, size and species richness
Overall, we collected a total of 328,936 individuals (4,910 kg) of fish representing 315 species from 86 families during the three years of surveys; including 226 species (72 families) from the eastern waters, 181 species (63 families) from the southern waters, and 206 species (61 families) from the western waters of Hong Kong.
Yearly variations in the total fish abundance, biomass, mean individual weight and species richness were detected in each of the six sites (Table 1 and Fig. 2). From 2004 to 2016, total abundance, biomass and species richness of demersal fishes increased by 271%, 308% and 46% in EI and by 356%, 316% and 73% in WO after the trawl ban. In EO, fish abundance increased by 156% in 2015–16 and species richness increased by 27% in 2013–14, respectively. Similar findings were observed in WI, where higher biomass and species richness were found in 2015–16 compared to that of before the ban, with 65% and 33% increases in both metrics, respectively. However, the abundance, biomass, mean weight and species richness of the fishes in SL decreased by 44%, 69%, 37% and 27% in 2015–16, respectively, when compared to 2013–14. No significant temporal changes in abundance and biomass were found at SE.
Mean fish abundance (a; Mean + SEM; individuals/km2/year), biomass (b; Mean + SEM; kg/km2/year), mean weight (c; g/individual) and species richness (d) at six sites in Hong Kong coastal waters before (2012) and after (2015) the trawl-ban. In (c) and (d), the vertical line shows the third quantile (Q) + 1.5* interquartile range (IQR) to Q1-1.5*IQR of the data. The black horizontal bars and red diamonds in the boxes indicate the medians and the means, respectively. Significant differences between years are indicated by a and b, while n.s. means no significant difference. Site codes are given in Fig. 1
Changes in predatory fishes
Absolute biomass of predatory fish increased by 293% in EI in 2015–16 compared to before the ban (Table 1 and Fig. 3). Similarly, their absolute abundance increased by 590%, 368% and 172% and absolute biomass increased by 699%, 156% and 216% in EO, WI and WO after the ban, respectively; their absolute abundance and biomass remained unchanged in SE but decreased at SL. Moreover, higher relative abundance and biomass of predatory fishes was detected in WI after the ban, and the same increase was evident at WO in 2015–16 compared to 2013–14.
Mean predatory fish abundance (a; Mean + SEM; individuals/km2/year), biomass (b; Mean + SEM; kg/km2/year), relative abundance (c) and relative biomass (d) at six sites in Hong Kong coastal waters before (2004; 2013–14) and after (2015–16) the trawl-ban. For other explanation, see Fig. 2
Community trophic metrics
MTL of the demersal fish community increased by 7%, 6%, 11% and 14% after the ban in EI, EO, WI and WO, respectively, and did not differ between 2013–14 and 2015–16 at SE and SL (Table 1 and Fig. 4). The results were consistent with the findings that the relative and absolute biomass of fishes in the highest (TL > 3.75) and intermediate (3.25 ≤ TL ≤ 3.75) trophic categories increased at WI and WO after the ban, while that of the lower trophic category (TL < 3.25) decreased (Table 1 and Figs. 5 and 6). Such changes suggest a release of fishing pressure on the highest and intermediate TL fishes, and their increased biomass is likely to impose high predator pressure on the lower TL fishes and juveniles. Similarly, higher relative and absolute biomass of fishes in the intermediate trophic category were also detected in EI and EO after the ban, while the trophic categories of fishes at SE were generally similar between 2013–14 and 2015–16. However, the relative and absolute biomass of fishes with TL > 3.75 decreased in SL, while the relative abundance of fishes of intermediate trophic categories (3.25 ≤ TL ≤ 3.75) increased in 2015–16 at SL.
Mean trophic level of fish community (MTL) at six sites in Hong Kong coastal waters before (2004; 2013–14) and after (2015–16) the trawl-ban. Violin plots display (i) the density of data estimated by Kernel method and (ii) the median value (the line in the middle of the boxplot). For other explanation, see Fig. 2
Relative biomass of fish (%) in three trophic categories (category 1: trophic level (TL) < 3.25; category 2: 3.25 ≤ TL ≤ 3.75; category 3: TL > 3.75) at six sites in Hong Kong coastal waters before (2004; 2013–14) and after (2015–16) the trawl-ban. For site codes, see Fig. 2
Absolute biomass of fish (%) in three trophic categories (category 1: trophic level (TL) < 3.25; category 2: 3.25 ≤ TL ≤ 3.75; category 3: TL > 3.75) at six sites in Hong Kong coastal waters before (2004; 2013–14) and after (2015–16) the trawl-ban. For other explanation, see Fig. 2
The overall results of the present study are summarized in Fig. 7, and reveal initial signs of community recovery (e.g., larger fishes occupying higher trophic positions) in eastern and western waters. These changes were particularly prominent in EI and WO areas with increased fish abundance, biomass and MTL after the ban. However, no change or decreases in most metrics were observed in southern waters (SE and SL) in 2015–16 compared to 2013–14.
Summary of the indicators for detecting biodiversity change in the fish community by univariate statistical analysis. ‘↑’ represents an increase after the trawl-ban, whereas ‘↓’ represents a decrease after the ban. For site codes, see Fig. 2
Community composition
The PERMANOVA revealed that the structure of demersal fish communities differed significantly among the surveyed years in all six sites (Tables S2–3). Pairwise comparisons also confirmed that the fish community structure differed among all years in all six sites with only a single exception (2013–14 vs. 2015–16 in WI; Tables S2–3).
In EI, the biomass of most fish families increased after the ban (Fig. 8a). Specifically, the biomass of Siganidae (herbivore/omnivore) and Leiognathidae (omnivore), greatly increased immediately after the trawl ban (2013–14) but dropped in 2015–16 (Fig. 8a and Fig. S1a). The same pattern was seen at EO, although the decline in siganids in 2015–16 was offset by increases in leiognathids (Fig. 8b and Fig. S1c). Such changes might be ascribed to increased predatory pressures from fishes in higher trophic levels that had increased biomass after the trawl ban, including Gerreidae, Gymnuridae, Paralichthyidae, Platycephalidae and Sparidae at EI (Fig. 8a and Fig. S1b) and Apogonidae, Gymnuridae, Platycephalidae and Sparidae at EO (Fig. 8b and Fig. S1d).
Relative biomass change (%) of the major fish families (those contributing 5% to between-year dissimilarity in SIMPER analyses) caught in different years (i.e. 2004, 2013 and 2015) at six sites in Hong Kong coastal waters; for site codes, see Fig. 2. Abundance is not shown as the pattern was very similar to biomass
In SE, two families in higher trophic levels (i.e., Sillaginidae and Sciaenidae) had much lower biomass in 2015–16 compared to 2013–14 (Fig. 8c and Fig. S1e), but other families at higher trophic levels (e.g., Paralichthyidae, Triglidae, Rajidae and Synodontidae) increased their biomass in 2015–16. In contrast, at SL, the biomass of all families in higher trophic levels was reduced in 2015–16 compared to 2013–14 (Fig. 8d and Fig. S1f).
Pairwise PERMANOVA tests revealed that community structure in WI in 2004 was distinctly different from that in 2013–14 and 2015–16, but there was no difference between 2013–14 and 2015–16 (Table S2–3). Siganids dominated before the trawl ban but their biomass greatly decreased subsequently (Fig. 8e and Fig. S1g). In contrast, the biomass of families in high trophic levels increased after the ban, including Muraenesocidae, Synanceiidae, Polynemidae, Dasyatidae and Sciaenidae (all zoobenthivores/piscivores) which were greater in 2013–14 and 2015–16 than prior to the ban.
The biomass of Siganidae decreased while that of Leiognathidae greatly increased from 2004 to 2015–16 at WO (Fig. 8f and Fig. S1h-i). The biomass of many families at higher trophic levels tended to increase from 2004 to 2015–16, including Synanceiidae, Sillaginidae, Polynemidae, Platycephalidae and Sciaenidae.
Discussion
This study detected initial recovery of fish community in eastern and western coastal waters of Hong Kong, as indicated by increases in several community metrics after the trawl ban. All fishes in EI and WO, and predatory fishes in EO, WI and WO had higher absolute abundance and biomass, while all total fishes in EO and WI, and predatory fishes in EI showed an increasing trend after the ban between 2013–14 and 2015–16. Moreover, increases in MTL were also observed in all four sites in the eastern and western waters (i.e., EI, EO, WI and WO), suggesting a lower fishing pressure after prohibiting trawling activities (Colléter et al. 2014; Pauly et al. 1998). These changes were likely due to increases in the relative and absolute biomass of fishes in the highest and intermediate trophic categories at these sites after the ban, in accordance with the observed increases in predatory fishes that could be attributable to an initial recovery of upper trophic levels after the ban. Nonetheless, no consistent patterns (and even some decreases) in community metrics were observed in southern waters after the ban.
The results of our concurrent study on the benthic infaunal biodiversity in Hong Kong also indicated that the trawl ban resulted in increases of organic matter in sediment, leading to widespread increases in diversity, abundance and biomass of benthic infauna such as polychaete worms and bivalves (Wang et al. 2021). Such positive changes would have provided more food resources for demersal fishes and facilitated their recovery.
The beneficial effect of the trawl ban observed on fish communities in eastern and western waters of Hong Kong mirrors findings reported elsewhere (Dimarchopoulou et al. 2018; Pipitone et al. 2000). However, the effects in Hong Kong seem relatively small; for instance, an eight-fold increase in total catch was found after a four-year trawl ban in the Gulf of Castellammare of Mediterranean Sea (Pipitone et al. 2000). In contrast, in Malindi-Ungwana Bay, Kenya, a trawl ban in 2006, was associated with increasing exploitation of penaeid shrimps during 2008–2010 (Swaleh et al. 2015), indicating increased fishing pressure. Such variable responses to a trawl ban were likely attributable to the distinctiveness of each ecosystem, including such factors as the extent to which trawling had depleted stocks (and hence the potential for recovery), the life-cycle duration and reproductive rates of species of interest, the effectiveness of enforcement, the period since the ban (= time available for recovery) and the incidence of redistributed, intensified non-trawling fishing activities (Bailey 1997; Chong et al. 1987; Mullowney et al. 2012). The potential impacts of other non-fishing related disturbances such as habitat destruction, pollution or climate change may also account for differences in the magnitude of recovery after trawl bans (Lotze et al. 2006, 2011).
Given the important economic and ecological roles of predatory fishes, restoration of depleted stock is a major objective of fishery management (Baum and Worm 2009; Fung et al. 2013; Myers and Worm 2005). Indeed, Russ and Alcala (1996, 2004) reported that the biomass of large predatory fish increased greatly after establishment of a marine reserve in the Philippines, which they regarded this as demonstrating the success of the intervention. In Georges Bank, Southern New England, stock of several predatory fishes increased after bottom fishing had been closed for four years (Murawski et al. 2000). These findings parallel our observations that abundance and biomass of predatory fishes in EO, WI and WO had increased after the ban, implying (at least) a partial reconstruction of the trophic structure of the demersal fish community, and a general improvement in the status of the marine ecosystem in eastern and western waters following the ban.
The Mean Trophic Level (MTL) was higher under reduced fishing pressure, which has been reported for demersal fisheries in Beibu Gulf, South China Sea (Su et al. 2021) and reservoir fisheries in Mali (Kantoussan et al. 2010) and, along with a greater proportion of predatory fishes, was also detected after establishment of a protected area in a Senegalese estuary (Ecoutin et al. 2014). Consistent with these observations, we observed increases in MTL at four sites in eastern and western waters in Hong Kong, accompanied by increases in the relative and absolute biomass of fishes in the intermediate trophic category at the two eastern sites, and in the highest trophic category in the west after the trawl ban. The same pattern was seen in benthic crustacean assemblages with increases in MTL after the trawl ban (Tao et al. 2020a). Such improved situations might be attributable to rehabilitation of disturbed habitat, greater functional diversity of prey, and/or associated changed patterns of energy flow (Hooper et al. 2005; McHugh et al. 2010; Post and Takimoto 2007).
Although initial signs of recovery were detected in the eastern and western waters of Hong Kong three years after the trawl ban, the extent of that recovery, and the lack of any recovery in southern waters, could have been influenced by anthropogenic activities. Large scale construction (e.g. the Hong Kong-Zhuhai-Macao Bridge; East Sha Chau contaminated mud pit facility) and reclamation (extension of the Hong Kong International Airport) have been conducted in the western waters of Hong Kong, especially around Lantau Island. Reclamation, sediment dumping and dredging during the construction have generated huge large sediment plumes (Ip 2015) that could confound the effects of any trawling ban. The eastern waters are affected by eutrophication and associated algal blooms and hypoxia events that periodically occur in Tolo Harbour and Channel (EPD, 2018; Xu et al. 2010). Eutrophication degrades marine ecosystems and results in substantial changes of their structure and function (Diaz and Rosenberg 2008; Rabalais et al. 2002; Vaquer-Sunyer and Duarte 2008). The anthropogenic factors are likely to hinder the full recovery of benthic communities of fishes and crustaceans in western and eastern waters of Hong Kong—at least in the short term (Tao et al. 2020b, 2018).
The lack of any recovery of fish communities in the southern waters was consistent with the lack of signs of recovery of commercial crustacean resources in this area (Tao et al. 2020b). This was likely linked to various biotic and abiotic factors. Benthic ecosystem in the southern waters have been perturbed and overexploited by intensive dredging activities to collect sand for constructions in the early 1990s (Leung 1995; Leung and Leung 2000) and intensive fishing activities before the trawl ban (AFCD port survey in 2006). The expansion of non-trawling fishing effort (especially, purse-seine) and illegal trawling in Hong Kong waters might also have impeded the recovery process. Despite the implementation of the trawl ban in Hong Kong waters, the estimated total number of fishing vessels greatly increased from 3,988 in 2013 to 5,160 in 2016 (Table S4). The increase was largely attributable to a rise in miscellaneous craft, including small vessels such as sampans that employ a wide variety of fishing gears including lines, nets and traps (Sumaila et al. 2007), which are commonly operated inshore within Hong Kong territorial waters due to their small sizes. The ban may force fishers to use non-trawling fishing gear (e.g., gill nets and cages). Such practices were frequently observed in our field work after the trawl ban. Total landings from such fishing increased to account for 38% of landings in 2016–17 (AFCD, 2018), and a rise in fishing effort by vessels using purse-seines in 2013–14 was confirmed by preliminary results of night light satellite image analysis (Tao et al., unpubl. data).
Furthermore, illegal trawling was evident after the trawl ban, especially in southern waters, as evinced by the rise in the number of successfully prosecuted cases in this area (at least 30 cases from 2013 to 2016; AFCD, 2021). Anecdotes from fishermen also suggested that illegal trawling activities were quite common after the trawl ban (especially during night time). Therefore, demersal fish communities in the southern waters might have been subject to continual ecosystem degradation from the intensive fishing after the ban. Similarly, decreases in catches was detected in Indonesia after two years of a trawl ban as a result of dramatic expansion of small-scale demersal fisheries and illegal trawling, imposing high fishing pressure (Bailey 1997; Chong et al. 1987).
Conclusions
By analysing a suite of community and size-based indicators, we found initial signs of recovery in eastern and western waters three years after a permanent trawling ban came into force in Hong Kong. However, changes or deterioration situation were marked in the southern waters. To facilitate recovery of the demersal fish communities, enhancement of enforcement and patrolling to eliminate illegal trawling, regulation of fishing effort of other non-trawling based gears, and proper planning of coastal development will be essential. As a complete ecosystem recovery often occurs at decadal time-scales after management intervention (Russ and Alcala 2004; Babcock et al. 2010; Lotze et al. 2011; Sheehan et al. 2013), a full recovery of the demersal fish communities in Hong Kong may require 5–10 more years, or even longer. Thus, the long-term monitoring on the demersal fish communities in Hong Kong waters will be needed to confirm the effectiveness of the trawl-ban policy and measure the full benefits that it will bring in terms of recovery. It is also hoped that fisheries in the surrounding non-protected waters with depleted resources (i.e. South China Sea) will ultimately benefit from the trawl ban in Hong Kong, as local waters will become a source of recruits and enhance fish stocks in these waters.
References
AFCD Agriculture Fisheries and Conservation Department (2011) Legislative Proposals to Take Forward the Trawl Ban and Other Fisheries Management Measures. https://www.afcd.gov.hk/english/fisheries/fish_cap/fish_cap_con/files/LEGOEng.pdf. Accessed 26 November 2016
AFCD Agriculture Fisheries and Conservation Department (2013) report of the Committee on Sustainable Fisheries. https://www.afcd.gov.hk/english/fisheries/fish_cap/fish_cap_con/files/common/CSF_WP_10_01_2013Eng.pdf. Accessed 26 November 2016
AFCD Agriculture Fisheries and Conservation Department (2018) Port Survey 2016/17. Agriculture Fisheries and Conservation Department. The Government of the Hong Kong Special Administrative Region, Hong Kong, China
AFCD Agriculture Fisheries and Conservation Department (2021) Departmental annual report. https://www.afcd.gov.hk/english/publications/publications_dep/publications_dep.html. Accessed 15 Sept 2021
Anderson M, Gorley RN, Clarke RK (2008) Permanova+ for primer: guide to software and statisticl methods. PRIMER-E Ltd., Plymouth
Babcock RC, Shears NT, Alcala AC, Barrett NS, Edgar GJ, Lafferty K, Mcclanahan TR, Russ GR (2010) Decadal trends in marine reserves reveal differential rates of change in direct and indirect effects. Proc Natl Acad Sci 107:18256–18261
Bailey C (1997) Lessons from Indonesia’s 1980 trawler ban. Mar Policy 21:225–235
Baum JK, Worm B (2009) Cascading top-down effects of changing oceanic predator abundances. J Anim Ecol 78:699–714
Bergman MJN, Ubels SM, Duineveld GCA, Meesters EWG (2015) Effects of a 5-year trawling ban on the local benthic community in a wind farm in the Dutch coastal zone. ICES J Mar Sci 72:962–972
Cheung WWL, Sadovy Y (2004) Retrospective evaluation of data-limited fisheries: a case from Hong Kong. Rev Fish Biol Fish 14:181–206
Cheung WWL, Watson R, Morato T, Pitcher TJ, Pauly D (2007) Intrinsic vulnerability in the global fish catch. Mar Ecol Prog Ser 333:1–12
Cheung WWL (2015) Reconstructed catches in waters administrated by the Hong Kong Special Administrative Region. Fisheries Centre Working Paper #2015–93, University of British Columbia, Vancouver, Canada. 16p
Chong KC, Dwiponggo A, Ilyas S, Martosubroto P (1987) Some experiences and highlights of the Indonesian trawl ban: bioeconomics and socioeconomics. InIndo Pacific Fisheries Commission, Papers presented at the Symposium on Exploitation and Management of Marine Fishery Resources in Southeast Asia held in conjunction with the Twenty-Second Session of the Indo-Pacific Fishery Commission, Darwin, Australia. pp16–26
Christensen V, Guenette S, Heymans JJ, Walters CJ, Watson R, Zeller D, Pauly D (2003) Hundred-year decline of North Atlantic predatory fishes. Fish Fish 4:1–24
Christensen V, Coll M, Piroddi C, Steenbeek J, Buszowski J, Pauly D (2014) A century of fish biomass decline in the ocean. Mar Ecol Prog Ser 512:155–166
Clarke KR, Gorley RN (2006) Primer v6: user Manual/Tutorial. PRIMER-E Ltd., Plymouth
Colléter M, Gascuel D, Albouy C, Francour P, de Morais LT, Valls A, Le Loc’h F (2014) Fishing inside or outside? a case studies analysis of potential spillover effect from marine protected areas, using food web models. J Mar Syst 139:383–395
de Juan S, Thrush SF, Demestre M (2007) Functional changes as indicators of trawling disturbance on a benthic community located in a fishing ground (NW Mediterranean Sea). Mar Ecol Prog Ser 334:117–129
Diaz RJ, Rosenberg R (2008) Spreading dead zones and consequences for marine ecosystems. Science 321:926–929
Dimarchopoulou D, Dogrammatzi A, Karachle PK, Tsikliras AC (2018) Spatial fishing restrictions benefit demersal stocks in the northeastern Mediterranean Sea. Sci Rep 8:5967
Dulvy NK, Polunin NVC, Mill AC, Graham NAJ (2004) Size structural change in lightly exploited coral reef fish communities: evidence for weak indirect effects. Can J Fish Aquat Sci 61:466–475
Ecoutin JM, Simier M, Albaret JJ, Laë R, Raffray J, Sadio O, de Morais LT (2014) Ecological field experiment of short-term effects of fishing ban on fish assemblages in a tropical estuarine MPA. Ocean Coast Manag 100:74–85
EPD Environmental Protection Department (2018) Marine water quality data. http://epic.epd.gov.hk/EPICRIVER/marine/?lang=en Accessed 26 November 2018
ERM Environmental Resource Management (1998) Fisheries Resources and Fishing Operations in Hong Kong Waters, Final report. Prepared for Agriculture and Fisheries Department, Hong Kong Special Administrative Region Government, Hong Kong
Essington TE, Beaudreau AH, Wiedenmann J (2006) Fishing through marine food webs. Proc Natl Acad Sci 103:3171–3175
Froese R, Pauly D (2019) FishBase. World Wide Web electronic publication. www.fishbase.org
Fung T, Farnsworth KD, Shephard S, Reid DG, Rossberg AG (2013) Why the size structure of marine communities can require decades to recover from fishing. Mar Ecol Prog Ser 484:155–171
Goodkin NF, Switzer AD, McCorry D, DeVantier L, True JD, Hughen KA, Angeline N, Yang TT (2011) Coral communities of Hong Kong: long-lived corals in a marginal reef environment. Mar Ecol Prog Ser 426:185–196
Hiddink JG, Jennings S, Sciberras M, Szostek CL, Hughes KM, Ellis N, Rijnsdorp AD, McConnaughey RA, Mazor T, Hilborn R, Collie JS, Pitcher CR, Amoroso RO, Parma AM, Suuronen P, Kaiser MJ (2017) Global analysis of depletion and recovery of seabed biota after bottom trawling disturbance. Proc Natl Acad Sci 114:8301–8306
HKO Hong Kong Observatory (2021) Climate of Hong Kong. https://www.hko.gov.hk/en/cis/climahk.htm#:~:text=About%2080%20percent%20of%20the,are%20usually%20the%20driest%20months Accessed 6 August 2021
Hooper DU, Chapin FS, Ewel JJ, Hector A, Inchausti P, Lavorel S, Lawto JH, Lodge DM, Loreau M, Naeem S, Schmid B, Setälä H, Symstad AJ, Vandermeer J, Wardle DA (2005) Effects of biodiversity on ecosystem functioning: a consensus of current knowledge. Ecol Monogr 75:3–35
Hoskin MG, Coleman RA, Von Carlshausen E, Davis CM (2011) Variable population responses by large decapod crustaceans to the establishment of a temperate marine no-take zone. Can J Fish Aquat Sci 68:185–200
Ip TY (2015) Cumulative water quality impact study on coastal developments in western waters of Hong Kong. Master dissertation, The University of Hong Kong
Jennings S, Kaiser MJ (1998) The effects of fishing on marine ecosystems. Adv Mar Biol 34:201–352
Jennings S, Dinmore TA, Duplisea DE, Warr KJ, Lancaster JE (2001) Trawling disturbance can modify benthic production processes. J Anim Ecol 70:459–475
Jones JB (1992) Environmental impact of trawling on the seabed: a review. N Z J Mar Freshwat Res 26:59–67
Kaiser MJ, Clarke KR, Hinz H, Austen MCV, Somerfield PJ, Karakassis I (2006) Global analysis of response and recovery of benthic biota to fishing. Mar Ecol Prog Ser 311:1–14
Kantoussan J, Ecoutin JM, Fontenelle G, de Morais LT, Laë R (2010) Ecological indicators based on trophic spectrum as a tool to assess ecosystems fishing impacts. Ecol Indicators 10:927–934
Lee JH, Harrison PJ, Kuang C, Yin KD (2006) Eutrophication dynamics in Hong Kong coastal waters: physical and biological interactions. In: Wolanski E (ed) The Environment in Asia Pacific Harbours. Springer, Netherlands, pp 187–206
Leung SF, Leung KF (2000) The prawn resources of the southeastern waters of Hong Kong: A comparison of the 1992, 1995 and 1998 trawl surveys. In: Morton, B (ed) The Marine Flora and Fauna of Hong Kong and Southern China V Proceedings of the Tenth International Marine Biological Workshop Hong Kong University Press, Hong Kong pp 619–649
Leung SF (1995) The species composition and distribution of penaeid prawns in the southern waters of Hong Kong. In: Morton B (ed) The marine biology of the South China Sea II: proceedings of the Second International Conference held in Guangzhou, China World Publishing Corporation, Beijing, pp 59–69
Leung AWY (2001) Overfishing and changes to the fishing industry in Hong Kong. In: Proceedings of an International Workshop Reunion Conference, Hong Kong, pp 719–740
Lotze HK, Lenihan HS, Bourque BJ, Bradbury RH, Cooke RG, Kay MC, Kidwell SM, Kirby MX, Peterson CH, Jackson JBC (2006) Depletion, degradation, and recovery potential of estuaries and coastal seas. Science 312:1806–1809
Lotze HK, Coll M, Magera AM, Ward-Paige C, Airoldi L (2011) Recovery of marine animal populations and ecosystems. Trends Ecol Evol 26:595–605
Mangano MC, Kaiser MJ, Porporato EM, Lambert GI, Rinelli P, Spanò N (2014) Infaunal community responses to a gradient of trawling disturbance and a long-term fishery exclusion zone in the Southern Tyrrhenian Sea. Cont Shelf Res 76:25–35
McCutchan JH, Lewis WM, Kendall C, McGrath CC (2003) Variation in trophic shift for stable isotope ratios of carbon, nitrogen, and sulfur. Oikos 102:378–390
McHugh PA, McIntosh AR, Jellyman PG (2010) Dual influences of ecosystem size and disturbance on food chain length in streams. Ecol Lett 13:881–890
Morton B (2011) At last, a trawling ban for Hong Kong’s inshore waters. Mar Pollut Bull 62:1153–1154
Morton B, Morton J (1983) the sea shore Ecology of Hong Kong. Hong Kong University Press, Hong Kong
Mullowney DRJ, Morris CJ, Dawe EG, Skanes KR (2012) Impacts of a bottom trawling exclusion zone on Snow Crab abundance and fish harvester behavior in the Labrador Sea, Canada. Mar Policy 36:567–575
Munga C, Ndegwa S, Fulanda B, Manyala J, Kimani E, Ohtomi J, Vanreusel A (2012) Bottom shrimp trawling impacts on species distribution and fishery dynamics; Ungwana Bay fishery Kenya before and after the 2006 trawl ban. Fish Sci 78:209–219
Murawski SA, Brown R, Lai HL, Rago PJ, Hendrickson L (2000) Large-scale closed areas as a fishery-management tool in temperate marine systems: the Georges Bank experience. Bull Mar Sci 66:775–798
Myers RA, Worm B (2005) Extinction, survival or recovery of large predatory fishes. Philos Trans R Soc Lond B Biol Sci 360:13–20
Ng TPT, Cheng MCF, Ho KKY, Lui GCS, Leung KMY, Williams GA (2016) Hong Kong’s rich marine biodiversity: the unseen wealth of South China’s megalopolis. Biodivers Conserv 26:23–36
Nicholson MD, Jennings S (2004) Testing candidate indicators to support ecosystem-based management: the power of monitoring surveys to detect temporal trends in fish community metrics. ICES J Mar Sci 61:35–42
Pauly D, Watson R (2005) Background and interpretation of the ‘Marine Trophic Index’ as a measure of biodiversity. Philos Trans R Soc Lond B Biol Sci 360:415–423
Pauly D, Christensen V, Dalsgaard J, Froese R, Torres F (1998) Fishing down marine food webs. Science 279:860–863
Pipitone C, Badalamenti F, D’Anna G, Patti B (2000) Fish biomass increase after a four-year trawl ban in the Gulf of Castellammare (NW Sicily, Mediterranean Sea). Fish Res 48:23–30
Post DM (2002) Using stable isotopes to estimate trophic position: models, methods, and assumptions. Ecology 83:703–718
Post DM, Takimoto G (2007) Proximate structural mechanisms for variation in food-chain length. Oikos 116:775–782
Rabalais NN, Turner RE, Wiseman WJ Jr (2002) Gulf of Mexico hypoxia, aka “The dead zone.” Annu Rev Ecol Syst 33:235–263
Rice WR (1989) Analyzing tables of statistical tests. Evolution 43:223–225
Ronquillo IA, Llana MEG (1987) Biological effects of fishery management measures in the Philippines. Papers Presented at the Symposium on the Exploitation and Management of Marine Fishery Resources in Southeast Asia, Darwin, Australia, 16–19 February 1987. Regional Office for Asia and the Pacific, Food and Agriculture Organization of the United Nations
Russ GR, Alcala AC (1996) Marine reserves: rates and patterns of recovery and decline of large predatory fish. Ecol Appl 6:947–961
Russ GR, Alcala AC (2004) Marine reserves: long-term protection is required for full recovery of predatory fish populations. Oecologia 138:622–627
Sadovy Y (1998) Patterns of reproduction in marine fishes of Hong Kong and adjacent waters. In: Morton B (ed) The marine biology of the South China Sea: Proceedings of the Third International Conference of the Marine Biology of the South China Sea, Hong Kong. pp 261–274
Sciberras M, Hiddink JG, Jennings S, Szostek CL, Hughes KM, Kneafsey B, Clarke LJ, Ellis N, Rijnsdorp AD, McConnaughey RA, Hilborn R, Collie JS, Pitcher CR, Amoroso RO, Parma AM, Suuronen P, Kaiser MJ (2018) Response of benthic fauna to experimental bottom fishing: a global meta-analysis. Fish Fish 19:698–715
Serrano A, Rodríguez-Cabello C, Sánchez F, Velasco F, Olaso I, Punzón A (2011) Effects of anti-trawling artificial reefs on ecological indicators of inner shelf fish and invertebrate communities in the Cantabrian Sea (southern Bay of Biscay). J Mar Biol Assoc UK 91:623–633
Shannon L, Coll M, Bundy A, Gascuel D, Heymans JJ, Kleisner K, Lynam CP, Piroddi C, Tam J, Travers-Trolet M (2014) Trophic level-based indicators to track fishing impacts across marine ecosystems. Mar Ecol Prog Ser 512:115–140
Shao KT (2019) Taiwan Fish Database WWW Web electronic publication. http://fishdb.sinica.edu.tw
Sheehan EV, Stevens TF, Gall SC, Cousens SL, Attrill MJ (2013) Recovery of a temperate reef assemblage in a marine protected area following the exclusion of towed demersal fishing. PLoS One 8:e83883
Shin YJ, Shannon LJ, Bundy A, Coll M, Aydin K, Bez N, Blanchard JL, de Fatima BM, Diallo I, Diaz E (2010) Using indicators for evaluating, comparing, and communicating the ecological status of exploited marine ecosystems. 2. Setting the scene. ICES J Mar Sci 67:692–716
SMO (2021) Hong Kong geographic data. https://www.landsd.gov.hk/mapping/en/publications/map.htm Accessed 10 Sept 2021
Su L, Chen ZZ, Zhang K, Xu YW, Xu SN, Wang KL (2021) Decadal-scale variation in mean trophic level in Beibu Gulf based on bottom-trawl survey data. Mar Coast Fish 13(3):174–182
Sumaila UR, Cheung WWL, Teh L (2007) Rebuilding Hong Kong's marine fisheries: an evaluation of management options. Fisheries Centre Working Paper #1198–6727, University of British Columbia, Vancouver
Svedäng H (2010) Long-term impact of different fishing methods on the ecosystem in the Kattegat and Öresund. European Parliament, Brussels
Swaleh K, Kaunda-Arara B, Ruwa R, Raburu P (2015) Ecosystem-based assessment of a prawn fishery in coastal Kenya using ecological indicators. Ecol Indicators 50:233–241
Tao LSR, Lui KKY, Lau ETC, Ho KKY, Mak YKY, de Mitcheson YS, Leung KMY (2018) Trawl ban in a heavily exploited marine environment: responses in population dynamics of four stomatopod species. Sci Rep 8:17876
Tao LSR, Lau DCP, Perkins MJ, Hui TTY, Yau JKC, Mak YKY, Lau ETC, Dudgeon D, Leung KMY (2020) Stable-isotope based trophic metrics reveal early recovery of tropical crustacean assemblages following a trawl ban. Ecol Indic 117:106610
Tao LSR, Lui GCS, Wong KJH, Hui TTY, Mak YKY, Sham RC-T, Yau JKC, Cheung WWL, Leung KMY (2020) Does a Trawl Ban Benefit Commercially Important Decapoda and Stomatopoda in Hong Kong? Ecosystems 24:1157–1170
Thrush SF, Dayton PK (2002) Disturbance to marine benthic habitats by trawling and dredging: implications for marine biodiversity. Annu Rev Ecol Syst 33:449–473
Vaquer-Sunyer R, Duarte CM (2008) Thresholds of hypoxia for marine biodiversity. Proc Natl Acad Sci 105:15452–15457
Wang Z, Leung KMY, Sung YH, Dudgeon D, Qiu JW (2021) Recovery of tropical marine benthos after a trawl ban demonstrates linkage between abiotic and biotic changes. Comm Biol 4:212
Worm B, Hilborn R, Baum JK, Branch TA, Collie JS, Costello C, Fogarty MJ, Fulton EA, Hutchings JA, Jennings S, Jensen OP, Lotze HK, Mace PM, McClanahan TR, Minto C, Palumbi SR, Parma AM, Ricard D, Rosenberg AA, Watson R, Zeller D (2009) Rebuilding global fisheries. Science 325:578–585
Xu J, Yin K, Liu H, Lee JH, Anderson DM, Ho AYT, Harrison PJ (2010) A comparison of eutrophication impacts in two harbours in Hong Kong with different hydrodynamics. J Mar Syst 83:276–286
Acknowledgements
This work is largely supported by the Collaborative Research Fund from the Research Grants Council of the Hong Kong Special Administrative Region, China (Project numbers: HKU5/CRF/2013 and C7050-18EF). We are very thankful to colleagues who helped with the field work and laboratory work including Archer Wong, Denis Chan, Kevin Ho, Matthew Perkins, Racliffe Lai, Ronia Sham and Stella Wong. We are grateful to Helen Leung for her technical support, and to Matthew Perkins for advices on stable isotope analysis. We also thank Sun Shuyi who assisted in preparation of the graphic abstract. We thank the Director of the Agriculture, Fisheries and Conservation Department for granting us a scientific research permit (R1710007) to conduct this trawling study.
Author information
Authors and Affiliations
Contributions
K.M.Y. Leung initiated and designed this study. Y.K.Y. Mak, L.S.R. Tao and V.C.M. Ho conducted the experiments. L.S.R. Tao and Y.K.Y. Mak drafted the main manuscript text. K.M.Y. Leung, W.W.L. Cheung and D. Dudgeon supervised the work, and contributed to the data analysis and interpretation, as well as manuscript preparation. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Yanny K. Y. Mak and Lily S. R. Tao are Joint first authors.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Mak, Y.K.Y., Tao, L.S.R., Ho, V.C.M. et al. Initial recovery of demersal fish communities in coastal waters of Hong Kong, South China, following a trawl ban. Rev Fish Biol Fisheries 31, 989–1007 (2021). https://doi.org/10.1007/s11160-021-09685-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11160-021-09685-5