Abstract
Objectives
Health state utilities (HSUs) are an input metric for estimating quality-adjusted life-years (QALY) in cost–utility analyses. Currently, there is a paucity of data on association of knee symptoms with HSUs for middle-aged populations. We aimed to describe the association of knee symptoms and change in knee symptoms with SF-6D HSUs and described the distribution of HSUs against knee symptoms’ severity.
Methods
Participants (36–49-years) were selected from the third follow-up (completed 2019) of Australian Childhood Determinants of Adult Health study. SF-6D HSUs were generated from the participant-reported SF-12. Association between participant-reported WOMAC knee symptoms’ severity, change in knee symptoms over 6–9 years, and HSUs were evaluated using linear regression models.
Results
For the cross-sectional analysis, 1,567 participants were included; mean age 43.5 years, female 54%, BMI ± SD 27.18 ± 5.31 kg/m2. Mean ± SD HSUs for normal, moderate, and severe WOMAC scores were 0.820 ± 0.120, 0.800 ± 0.120, and 0.740 ± 0.130, respectively. A significant association was observed between worsening knee symptoms and HSUs in univariable and multivariable analyses after adjustment (age and sex). HSU decrement for normal-to-severe total-WOMAC and WOMAC-pain was − 0.080 (95% CI − 0.100 to − 0.060, p < 0.01) and − 0.067 (− 0.085 to − 0.048, p < 0.01), exceeding the mean minimal clinically important difference (0.04). Increase in knee pain over 6–9 years was associated with a significant reduction in HSU.
Conclusion
In a middle-aged population-based sample, there was an independent negative association between worse knee symptoms and SF-6D HSUs. Our findings may be used by decision-makers to define more realistic and conservative baseline and ongoing HSU values when assessing QALY changes associated with osteoarthritis interventions.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Joint pain is the most prevalent musculoskeletal condition causing physical and psychosocial disability, and affecting millions of people and societies globally from both a humanistic and economic perspective [1, 2]. Knee pain is the most pervasive type of joint pain among adults older than 18 years and is reported by one in every two adults aged 50 years or older [3, 4]. A common cause of knee pain in the elderly population is knee osteoarthritis (OA), a chronic, progressive musculoskeletal disorder [5]. Importantly, the health-related quality-of-life (HRQoL) burden for knee pain in a younger (namely middle-aged) population worldwide is not well investigated and a younger cohort’s knee pain may translate to knee OA for many people who suffer knee pain, particularly as they reach older age and older age cohorts.
The global prevalence of knee OA was estimated to be more than 350 million people in 2019, and more than 2 million people had knee OA in Australia in the year 2019 [5, 6]. The economic burden of joint pain is also substantial [7]. Among patients with knee OA, knee pain has been recognized as a significant reason for knee replacement [8]. OA imparts a substantial economic burden to health systems globally; in Australia alone, it accounted for $3.5 billion of direct healthcare costs and $23 billion for total costs, including the indirect and intangible costs (for example, lost work productivity and loss of well-being) [5, 9, 10].
Knee OA is characterized by knee pain, stiffness, and physical dysfunction leading to limitations for activities of daily living and consequent diminution of HRQoL [11]. Various studies using both disease-specific patient-reported outcome (PRO) instruments such as the Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) [12, 13], Knee injury and Osteoarthritis Outcome Score (KOOS) [8, 14], and generic PRO instruments such as the EuroQol-5 suite of instruments (EQ-5D-3L and 5L) [14], Short-Form-6 Dimension (SF-6D) [15], 12-item Short-Form Survey (SF-12) [8, 16], and 36-item Short-Form Health Survey (SF-36) [13] have reported a severe impact of the presence of knee pain on HRQoL in patients with knee OA. Disease-specific HRQoL instruments (such as the WOMAC and KOOS) provide valuable disease-specific information from the patient's perspective and are highly sensitive to detect the minimal clinically important difference (MCID) in knee pain in OA [17, 18]. However, these instruments are profile-based measures and, therefore, not recommended to generate preference-based scores called health state utility (HSU) values needed for health economic analyses [19]. Generic PRO instruments, also called preference-based HRQoL instruments or multi-attribute utility instruments (such as the SF-6D [20], EQ-5D-5L and 3L [21], AQoL-8D [22], and HUI [23]), generate these patient-reported quantitative preference-based measures of HRQoL called HSU [24,25,26]. More specifically, HSUs are a health economic measure on a scale of 0.0–1.0 that provide the strength of preference an individual places on a particular health state relative to the states of perfect health (namely HSU = 1.0) and death (HSU anchored at a value of 0 and some instruments assign HSUs at less than 0 for health states considered worse than death) [25, 27, 28]. Importantly, multi-attribute utility instruments that assess HSUs are routinely used to evaluate the quality of life for patients with knee OA and are now gaining importance as the preferred outcome measure in patients reporting knee pain [8].
Quality-adjusted life-years (QALYs) are an outcome metric routinely used in health economic evaluations, particularly cost–utility analysis (CUA) [29,30,31]. QALYs incorporate both the quantity and quality of life, and therefore allow a broader comparison across varying patient populations, treatment strategies, and clinical settings [29, 30]. The duration spent in a particular health state (survival) provides the quantity component, and the HSU assigned to the specific health state provides the quality (HRQoL) component for QALY calculation, which is simply a product of survival and HSU (i.e., quantity and quality) [29, 30].
Markov modelling is often used as a tool to estimate the economic and humanistic burden for chronic conditions such as knee pain and knee OA [27, 28]. Importantly, for accurate estimation of the humanistic burden, it is essential to assign evidence-based HSUs to patients entering the model, rather than simply assuming these patients to be at an HSU of 1.0 (that is perfect health) [32]. A typical Markov model for CUA starts with a cohort of people at risk of a disease (knee pain, knee OA) and simulates the transition of the study population between discrete health states [33, 34]. The transition is governed by transition probabilities and has an associated HSU for each health state occurring within a defined period called the Markov cycle [35]. Hence, evidence on HSUs for this cohort and how knee-symptom severity affects HSUs may enable appropriate assumptions for CEA models and health economic decision making. Utilities have also been shown to be independent predictors of patient outcomes, including all-cause mortality and development of complications [36]. Additionally, clinicians have found that measuring health utilities is of benefit to patients regarding clinical assessment, relationships, communication, and management [37]. Thus, understanding the characteristics of the disease, such as knee pain and its impact on HSUs, is of utmost importance to promote value-based care and informed resource allocation.
Although previous studies have reported HSUs for knee OA patients and highlighted the detrimental effect of knee pain on quality of life in older adults and patients with knee OA [38], there is a paucity of literature regarding the HSUs for a general population entering an OA health economic model and little is known about the association of knee pain, stiffness, and dysfunction with HSU in younger adults [39,40,41,42,43].
Against the backdrop of a paucity of literature that assesses HSUs for a relatively younger population with knee pain, to our knowledge, this will be the first study to explore the association of knee symptoms and HSUs in a sample largely representative of the Australian middle-aged population. Therefore, we aimed to describe the distribution of HSUs against knee-symptom severity classifications and the impact of change in knee symptoms over 6–9 years on HRQoL using the SF-6D’s HSUs.
Methods
Study participants and data collection
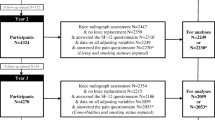
The study sample was selected from the Childhood Determinants of Adult Health (CDAH) study, a three-phase follow-up of participants in the Australian Schools Health and Fitness Survey (ASHFS). The study designs of CDAH and ASHFS have been described elsewhere; here, we provide a summary [44, 45]. ASHFS was a nationwide survey comprising a sample of 8498 school children aged 7–15 years (mean age 11) for whom a wide range of health-related measures was collected in the year 1985. Figure 1 shows the flow of participants from the ASHFS into the CDAH cohorts. The first follow-up of the ASHFS sample for the CDAH study comprised 3521 participants and was conducted in 2004–2006 (referred to as the CDAH-1) [46,47,48]. The second follow-up (referred to as CDAH-2) consisted of 2815 participants and was conducted during 2009–2011 [47, 48]. During the same time, an ancillary study (referred to as CDAH-knee study) consisting of 449 CDAH participants was introduced to assess the impact of early life risk factors on knee structure and symptoms. The third follow-up (referred to as CDAH-3) comprised a sample of 1568 participants and was conducted in 2014–2019.
Our cross-sectional study sample (Fig. 1, bottom curly bracket) was derived from CDAH-3 (n = 1567: aged 36–49 years, female 54%). We included data for participants who completed the SF-12 and knee WOMAC questionnaires and provided other clinical characteristics at the CDAH-3 follow-up study. For our longitudinal analysis (Fig. 1, right curly bracket), participants were selected who completed the CDAH-knee study [49] (n = 313; age mean ± standard deviation (SD) = 34.94 ± 2.72 years; female = 48%) and were matched to CDAH-3 data after 6–9 years (n = 313; age mean ± SD = 42.84 ± 3.38 years).
All the participants of the CDAH-knee and CDAH-3 studies provided written informed consent, and the studies were approved by the Southern Tasmania Health and Medical Human Research Ethics Committee, Monash University Human Research Ethics Committee, and the Northern Sydney and Central Coast Area Human Research Ethics Committee.
Anthropometric measurements
At the CDAH study, weight was measured to the nearest 0.1 kg and with shoes, socks, and bulky clothing removed. Height was measured to the nearest 0.1 cm with shoes and socks removed using a Leicester stadiometer (Invicta, Leicester, UK) [49]. Body mass index (BMI) was calculated from height and weight as kilograms of weight per square meter of height (kg/m2) [50]. BMI was categorized as underweight (< 18.5 kg/m2), healthy weight (≥ 18.5 and ≤ 24.9 kg/m2), overweight (≥ 25.0 and ≤ 29.9 kg/m2), and obese (≥ 30 kg/m2) [51]. Sociodemographic and clinical data on other participant characteristics such as education and co-morbidity were self-reported in childhood and follow-up.
Knee symptom measurements
Data on knee pain and symptoms were collected through questionnaires in CDAH 3 study and computer-assisted telephone interviews (CATI) in the CDAH-Knee study. The WOMAC instrument was used to assess the knee pain, stiffness, and physical dysfunction captured on a scale of “0–9”, where “0” indicated no pain, stiffness, or dysfunction, and “9” indicated the maximum observed pain, stiffness, and dysfunction [18]. Although the WOMAC is a preferred disease-specific PRO instrument for patients with OA, it is also validated for responsiveness to knee pain and symptoms in young study populations without OA [17, 52]. The WOMAC instrument captured and assessed knee pain under five subscales with the highest possible score of 45, and stiffness and dysfunction were captured under 2 and 17 subscales with the highest possible scores of 18 and 153, respectively. The overall WOMAC score was calculated as a sum of scores for each subscale with the highest possible score of 216. Score ≥ 1 for respective domains of WOMAC was indicative of pain, stiffness, and dysfunction. The knee pain was categorized as: no pain (WOMAC-pain score = 0), moderate pain (score ≤ 5), and severe pain (score > 5) [53, 54]. Stiffness was categorized as, no stiffness (WOMAC-stiffness score = 0), moderate stiffness (score ≤ 2), and severe stiffness (score > 2). Physical dysfunction was categorized as: no function limitation (WOMAC-function score = 0), moderate function limitation (score ≤ 17), and severe function limitation (score > 17) [55].
SF-6D HSUs for people with knee pain
We used the SF-12 (version 2) questionnaire to capture the patient-based assessments of HRQoL at the CDAH-knee and CDAH-3 follow-ups. The SF-6D is a globally prevalent multi-attribute utility instrument derived from the patient-reported responses to the SF-36 or SF-12 questionnaire [24]. More specifically, the SF-6D assesses HSUs for 18,000 health states, and has been well validated for complex and chronic disease states and has an equal preponderance to both physical and psychosocial health needs [56]. HSUs for our study population were derived from the patient-reported response to the SF-12 and then calculated through the SF-6D’s algorithm based on the UK value set in the absence of an Australian value set for the SF-12 [24, 57]. The HSU value generated by the SF-6D’s algorithm ranges from 1.00 (best HRQoL state or perfect health) to 0.30 (worst HRQoL state measured by the SF-6D) [24].
We adopted the previously reported minimal important difference (MID) (or minimal clinically important difference (MCID)) for the SF-6D’s HSUs at a mean of 0.04 to assess the significance of the HSUs variation across the knee-symptom severity groups [58, 59]. In the absence of knee pain-specific MID or MCIDs for the SF-12 variant of the SF-6D, we have adopted a slightly more conservative estimate of composite measures that have used the SF-36 variant of the SF-6D. To illustrate, the reported measure for knee OA by Brazier et al. (2005) is 0.035 utility points, and for rheumatoid arthritis is 0.037 utility points [59, 60]. Additionally, a paper that investigated both the SF-12 and SF-36 variants of the SF-6D for a study population with spinal cord injuries suggested that the SF-6D of either derivation was suitable for detecting the clinical change for that study [61]. We also adopted the previously reported UK and Australian population norms, including population norms for the UK middle-aged cohort of 45–49 (0.79 utility points), and Australian middle-aged cohort deciles of 31–40 years (0.79 utility points) and 41–50 years (0.77 utility points), and these mean HSUs are similar for both countries [62, 63].
Statistical methods
Summary data describing the sociodemographic characteristics of the participants at CDAH-knee and CDAH-3 follow-ups are presented as means with standard deviations (SDs) for continuous variables, and as percentages with frequency counts for categorical variables. Comparison of the mean values of age, weight, and BMI of two groups of participants (those for whom an SF-6D HSU could be generated or not—i.e., with and without SF-6D HSU) were performed using two-sample t-tests. Univariable and multivariable linear regression was performed to estimate the association between WOMAC knee symptom (knee pain, stiffness, and dysfunction) scores and SF-6D HSU before and after adjustment for confounders. The outcome variable (SF-6D HSU) was transformed (for example by taking logarithms) prior to analysis to reduce positive skewness. Importantly, the estimates reported have been back-transformed to the original scale of the HSU. In the development of the multivariable regression models, confounding and statistical interaction were assessed with covariates for age, sex, BMI, education, and co-morbidities including diabetes and hypertension. Results are reported without and with adjustment for age and sex. The cross-sectional analyses are reported with additional adjustments for BMI and education. Tests of interaction did not reach statistical significance, but results for subgroup analyses for sex (male and female), age (36–40 years, 41–45 years, and > 45 years), and BMI (underweight, healthy weight, overweight, and obese) are reported. In longitudinal analyses, covariates for education, diabetes, and hypertension were significant predictors of the outcome, but adjustment with these factors did not markedly alter the estimated coefficients of the covariates for the WOMAC measures. The longitudinal analyses included regression of HSU on change in WOMAC scores, and of change in HSU on WOMAC scores and on change in WOMAC scores. The changes were calculated by subtracting the values of HSU and WOMAC at CDAH-knee from the respective values at CDAH-3. To check the robustness of the findings to the statistical methods used, the linear regression estimates were compared with those from truncated regression models that acknowledged the upper and lower limits of the measured range of the SF-6D HSUs. Providing confidence in the robustness of the results, the estimates were barely changed. Only participants who had complete data on all covariates and the outcome of interest were included in the models; no data were imputed. A p-value of < 0.05 was considered statistically significant. All analyses were performed using STATA software, version 16.0 (Stata Corp.).
Results
Participant characteristics
Our cross-sectional analysis sample included 1567 participants from the CDAH-3 study. The anthropometric characteristics of participants in the cross-sectional sample are shown in Table 1. Briefly, the mean ± SD age of the population was 43.5 ± 2.9 years, with females constituting 54% of the sample, and overall mean ± SD BMI was 27.18 ± 5.31 kg/m2. The mean (SD) total-WOMAC score was 10.20 ± 21.47, and the mean (SD) HSU value was 0.79 ± 0.12 ranging from 0.39 to 1.
Our longitudinal analysis sample included 313 participants followed from CDAH-knee to CDAH-3 (Online Appendix 1, Table 1). The mean ± SD age was 34.94 ± 2.72 and 42.84 ± 3.38, at CDAH-knee and CDAH-3, respectively. The mean ± SD total-WOMAC scores were 6.26 ± 12.64 and 6.62 ± 13.99, and the mean ± SD HSU values were 0.790 ± 0.120 and 0.800 ± 120, at CDAH-knee and CDAH-3, respectively. The participant characteristics were comparable in terms of age, weight, and BMI for males and females for whom an SF-6D HSU could be generated or not generated from the SF-12 patient-reported responses (p < 0.05) (Online Appendix 1, Table 6).
HSUs and WOMAC categories
Table 2 presents the WOMAC score for various categories and the corresponding HSUs for the cross-sectional analysis. The mean ± SD HSU for normal, moderate, and severe total-WOMAC scores were 0.820 ± 0.120, 0.800 ± 0.120, and 0.740 ± 0.130, respectively. Demonstrating a consistent trend, participants with normal WOMAC score (WOMAC = 0) had a consistently higher HSU value than participants in moderate and severe WOMAC score groups across the categories of total-WOMAC, WOMAC-pain, WOMAC-stiffness, and WOMAC-function. Additionally, the difference between participants classified in the normal or mild disease severity classification compared to the severe disease severity classification exceeded the MCID of 0.04 utility points for the SF-6D [58, 59]. Importantly, this demonstrates the discriminatory power of the SF-6D to appropriately differentiate according to the disease severity classifications with reduced HSUs for increasing disease severity confirming the discriminatory validity of the SF-6D HSU values for this population. Although a smaller sample size, similar trends were observed for the longitudinal sample (Table 2).
Association of knee symptoms and HSUs
Table 3 shows the associations between various WOMAC groups and SF-6D HSU values for the CDAH-3 cross-sectional analysis. Importantly, a significant association was observed between worse knee symptoms and HSUs in univariable and multivariable analyses after adjustment for age and sex. The HSU decrement for normal-to-severe total-WOMAC and WOMAC-pain groups was − 0.080 (95% CI − 0.100 to − 0.060, p < 0.01) and − 0.067 (− 0.085 to − 0.048, p < 0.01), exceeding the MCID (0.04 utility points). Based on the linear regression coefficient, the SF-6D HSU value exceeded the MCID of 0.04 at the WOMAC score of 33, 9, 5, and 22 for total-WOMAC, WOMAC-pain, WOMAC-stiffness, and WOMAC-function. Consistently across groups (i.e., total-WOMAC, WOMAC-pain, WOMAC-stiffness, and WOMAC-function), a worse WOMAC score was associated with a statistically significantly stronger detrimental impact on HSUs demonstrated by lower beta coefficient value. Table 4 shows the longitudinal association between WOMAC score at the CDAH-knee and CDAH-3 follow-ups and HSU value at CDAH -3. Knee pain at CDAH-knee was significantly associated with a reduction in HSU 6–9 years later at the CDAH-3 follow-up [adjusted regression coefficient = − 0.004 (− 0.008 to − 0.001; p = 0.038)]. Similarly, a consistent trend was observed across the knee symptoms (WOMAC total, pain, stiffness, and function) at CDAH-knee and HSU at CDAH-3 follow-up. Participants with worsening WOMAC scores over 6–9 years had a lower HSU, compared to those with stable/decreased WOMAC over the same period (Fig. 2). Participants with increased knee pain compared to those with stable knee pain had a statistically and clinically significant negative impact on HSU at the end of follow-up at CDAH-3 [adjusted regression coefficient = − 0.040 (− 0.073 to − 0.008; p = 0.015)] (Table 4).
The change in WOMAC scores over 6–9 from CDAH-knee to CDAH-3 was also negatively associated with a change in HSU value during the same period, although not statistically significant (Online Appendix 1, Table 2). Similarly, WOMAC at CDAH-knee was negatively associated with a change in HSU from CDAH-knee to CDAH-3 follow-up.
Sub-group analysis
Sub-group analysis of cross-sectional data based on sex showed a consistently stronger effect of reduced WOMAC scores on HSU decrement in women compared to men across the WOMAC symptom severity groups for all WOMAC domains, except WOMAC-function (Online Appendix 1, Table 3). Similarly, younger participants (36–40 years) had consistently more substantial effects of worse WOMAC scores on HSU decrement compared to older participants in the age groups of 41–45 years, and ≥ 45 years, across the WOMAC symptom severity groups for various WOMAC domains (Online Appendix 1, Table 4). No specific pattern was observed for the sub-group analysis based on BMI (Online Appendix 1, Table 5).
Discussion
To our knowledge, this is the first study examining both the cross-sectional and longitudinal relationship between knee symptoms (pain, stiffness, and dysfunction) and HRQoL assessed with SF-6D HSUs in middle-aged adults. In this population-based cohort of Australian middle-aged adults that is broadly representative of a middle-aged Australian population, we found that WOMAC knee symptoms scores were negatively associated with HSU, and an increase in knee pain over 6–9 years was associated with a reduction of HSU as compared to those with no or stable knee pain [64]. We also found that the SF-6D had good discriminatory power for the middle-aged adult cohort, particularly when we examined the decrements for disease-severity classifications.
It is crucial to consider the public health implications of knee pain in a younger (middle-aged) population. Previous studies have shown that the reduction in HRQoL due to knee pain leads to reduced productivity [65,66,67]. However, there has been less focus on knee pain in the relatively younger population [2]. Therefore, our novel findings and the SF-6D decrements associated with diminished knee health across disease-severity classifications can be used to populate health economic models. Additionally, our HSU findings could also be adopted as baseline measures for clinical assessment and comparisons.
Health economics modelling of a middle-aged cohort’s knee health
Our study addresses an important evidence gap in the literature by providing HSUs associated with knee pain and symptoms for a younger population cohort where treatments or interventions (particularly for severe disease severity, or to avoid increased disease severity) are of humanistic and economic interest to the individual, health payers, and broader society. We suggest that our large Australian sample of middle-aged people with knee pain is a largely representative of middle-aged knee cohorts who suffer knee pain in other jurisdictions [64]. Therefore, an important application of our results would be as a baseline value in an economic evaluation assessing health economic and humanistic burden of knee pain and knee OA for health technology assessment [10, 68]. For instance, in patients with knee OA, to assess the benefits of a treatment alleviating pain, researchers need the average HSU for a cohort that has knee OA (and its associated knee pain) and the average HSU for a cohort that does not have knee OA but may have underlying knee pain (i.e., the baseline) [69,70,71]. Consequently, in a health economic evaluation of treatment for knee OA assigning an HSU value of ‘1’ for the starting health state of a patient with no knee OA associated pain may not correctly capture the state and is likely to overestimate the effect of the intervention [70,71,72]. The mean HSUs corresponding to the various knee symptoms (knee pain, stiffness, function, and total-WOMAC) categories (no symptom, moderate, and severe symptoms and the associated utility decrement) and the regression coefficient can inform cost-effectiveness analyses of knee pain and knee OA [68, 73]. For example, our result provides HSUs that can be appropriately used in economic evaluations, such as the investigation conducted by Karmarkar et al., as a baseline value [32]. Similarly, for studies assessing the humanistic burden of knee OA, assigning a baseline HSU value and the longitudinal change in the HSU over the years, for those who do not develop knee OA, based on our data will help in the realistic estimation of the humanistic burden over the life course [32, 74, 75]. Hence, the use of baseline HSU and change in HSU from our study is likely to represent a more accurate prediction of health state trajectory than assuming an HSU of ‘1’ for the individuals without knee OA [32, 74].
SF-6D discriminatory power for a middle-aged cohort’s knee health
Another key finding of our study was the validation of the discriminatory power of the SF-6D multi-attribute utility instrument’ for a younger knee health cohort. The choice of the correct multi-attribute utility instrument that has discriminatory power to assess health states and the changes across health states is crucial [56]. Our study has shown that as knee health diminishes from normal to severe, the SF-6D’s HSU decrease was clinically meaningful.
Consistent with our findings for the SF-6D, earlier studies using older age cohorts have compared the association of knee pain with HRQoL in patients with established knee OA [8]. In two population-based cohort studies, Muraki et al., using the SF-8D and EQ-5D-3L, reported that knee pain was associated with reduced quality of life in both men and women [2, 76]. Using the SF-36 score only (not SF-6D HSUs), Antonopoulou et al. reported the detrimental effect of knee pain on quality of life for patients with musculoskeletal disorders attending the primary care center [77]. Similarly, longitudinal studies have reported that worsening of knee pain is one of the significant factors associated with reduced quality of life (assessed using KOOS, Japanese Knee Osteoarthritis Measure, and SF-12) in patients with knee OA [42, 78, 79]. In patients with knee OA, knee pain is shown to cause avoidance of physical activity and leading to muscle weakness and disability [80]. Similarly, knee pain in young adults can affect various aspects of daily life activity and hence impacting overall HRQoL [81, 82]. Several factors may be impacting HRQoL in this population; however, in our study, the multivariate analysis adjustment with BMI, education, and co-morbidity did not markedly alter the estimated coefficients of the covariates. The effect of BMI on HSU was not significant, with the standardized linear regression coefficients of − 0.07 as compared to − 0.24 for the total-WOMAC, which may be the reason that BMI did not impact our study’s estimates. Hence, our interpretation is based on estimates adjusted for age and sex only, although we also presented data on other adjustments.
Regarding comparisons with both the UK and Australian population norms for a middle-aged cohort, our HSU values for participants who reported severe knee health were diminished from both the UK and Australian population norms by the MCID (reduced by 0.05 utility points) for the 45–49 year (UK) and 31–40 year (Australia) age category of population norms (0.79 utility points) [62]. Nevertheless, we also note that the overall mean HSU values in our study correspond to the previously reported population norm for both countries [62]. The pattern of differences in HSU between sex, with female reporting lower HSU, was also consistent with previously reported values from both the UK and the Australian population [62]. Previous studies have cited sociodemographic and socioeconomic differentials as a possible explanation for the sex differences in HRQoL [83, 84].
Strengths and limitations
The strengths of our study included assessment of cross-sectional and longitudinal association using 6–9 year follow-up data from a population-based cohort of Australian middle-aged adults. The large cross-sectional sample enabled the statistical power to examine the association of knee-symptom severity and the HSU. This is important as the younger population is less likely studied in relation to joint pain and HRQoL. We also suggest that this large Australian sample of middle-aged people with knee pain is broadly representative of middle-aged knee cohorts who suffer knee pain in other jurisdictions. Second, we used the disease-specific HRQoL (WOMAC scale) instrument to classify knee pain to enable us to assess the discriminatory sensitivity of the SF-6D. Third, using the SF-6D, we generated baseline HSUs from a large representative cohort of a middle-aged population.
Our study also has certain limitations to be considered while interpreting the findings. We categorized the WOMAC score into subgroups based on the severity of symptoms to allow easy interpretation of the results. Similar approaches have been used by other researchers earlier [53,54,55]. While we argue that these subgroups may be useful for health economic analyses (such as studies may subgroup patients based on knee-symptom severity and corresponding HSU), some information is lost when continuous measures are converted to categorical subgroups. Although this approach may have limited some information and statistical power, it improved the clinical interpretation and applicability of findings. We also acknowledge that we used the UK value set for this study; however, this is the most comparable value set that can be derived using the SF-12 variant of the SF-6D algorithm, and we have assumed that the derived value set is broadly comparable to the Australian middle-aged cohort for knee health. Another limitation of our study is that there is no MID or MCID for knee pain for the SF-6D tariff generated from the patient-reported responses to SF-12 questionnaire. Nevertheless, there are some studies that have estimated a composite or knee OA MID or MCID for the SF-36 variant of the SF-6D algorithm and we subsequently adopted a conservative estimate of 0.04 utility points for our study. We note that there is one study that investigated rheumatoid arthritis of the hand that used the SF-12 variant for SF-6D and this study estimated a benefit at 4 and 12 months of 0.06 utility points. A final limitation is that there are no Australian population norms for the SF-12 SF-6D algorithm. Therefore, to examine general trends and to provide further contextualisation to our study we also considered the population norms for both the UK and Australia which are similar for the general population and for the middle-aged cohorts.
Conclusion
In a middle-aged population-based sample, WOMAC scores and increasing WOMAC scores over 6–9 years for knee pain were negatively associated with HSUs, including clinically meaningful differences in SF-6D HSUs from normal to moderate/severe knee symptoms. Our findings may be used by decision-makers to define a more realistic and conservative baseline and ongoing HSU values when assessing QALY changes associated with OA interventions. Our findings also validate the discriminatory power of the SF-6D for assessing knee health for a middle-aged cohort.
References
Woolf, A. D., & Pfleger, B. (2003). Burden of major musculoskeletal conditions. Bulletin of the World Health Organization, 81(9), 646–656
Muraki, S., Akune, T., Oka, H., En-yo, Y., Yoshida, M., Saika, A., et al. (2010). Impact of knee and low back pain on health-related quality of life in Japanese women: The Research on Osteoarthritis Against Disability (ROAD). Modern Rheumatology., 20(5), 444–451
Nguyen, U. S. D. T., Zhang, Y., Zhu, Y., Niu, J., Zhang, B., & Felson, D. T. (2011). Increasing prevalence of knee pain and symptomatic knee osteoarthritis: Survey and cohort data. Annals of Internal Medicine, 155(11), 725–732
Jinks, C., Jordan, K., Ong, B. N., & Croft, P. (2004). A brief screening tool for knee pain in primary care (KNEST). 2. Results from a survey in the general population aged 50 and over. Rheumatology (Oxford), 43(1), 55–61
Health AIo, Welfare. (2019). Osteoarthritis. AIHW.
Vos, T., Lim, S. S., Abbafati, C., Abbas, K. M., Abbasi, M., Abbasifard, M., et al. (2020). Global burden of 369 diseases and injuries in 204 countries and territories, 1990–2019: A systematic analysis for the Global Burden of Disease Study 2019. The Lancet, 396(10258), 1204–1222
Gaskin, D. J., & Richard, P. (2012). The economic costs of pain in the United States. The Journal of Pain, 13(8), 715–724
Bindawas, S. M., Vennu, V., & Al, S. S. (2015). Differences in health-related quality of life among subjects with frequent bilateral or unilateral knee pain: Data from the Osteoarthritis Initiative study. Journal of Orthopaedic and Sports Physical Therapy, 45(2), 128–136
Hunter, D. J., Nicolson, P. J. A., Little, C. B., Robbins, S. R., Wang, X., & Bennell, K. L. (2019). Developing strategic priorities in osteoarthritis research: Proceedings and recommendations arising from the 2017 Australian Osteoarthritis Summit. BMC Musculoskeletal Disorders, 20(1), 74
Xie, F., Kovic, B., Jin, X., He, X., Wang, M., & Silvestre, C. (2016). Economic and humanistic burden of osteoarthritis: A systematic review of large sample studies. PharmacoEconomics, 34(11), 1087–1100
Arden, N., Blanco, F., Cooper, C., Guermazi, A., Hayashi, D., Hunter, D., et al. (2014). Atlas of osteoarthritis. (1st ed.). Springer Healthcare Communications.
Salaffi, F., Carotti, M., & Grassi, W. (2005). Health-related quality of life in patients with hip or knee osteoarthritis: Comparison of generic and disease-specific instruments. Clinical Rheumatology, 24(1), 29–37
Pang, J., Cao, Y. L., Zheng, Y. X., Gao, N. Y., Wang, X. Z., Chen, B., et al. (2015). Influence of pain severity on health-related quality of life in Chinese knee osteoarthritis patients. International Journal of Clinical and Experimental Medicine, 8(3), 4472–4479
Kiadaliri, A. A., Lamm, C. J., de Verdier, M. G., Engström, G., Turkiewicz, A., Lohmander, L. S., et al. (2016). Association of knee pain and different definitions of knee osteoarthritis with health-related quality of life: A population-based cohort study in southern Sweden. Health and Quality of Life Outcomes, 14(1), 121
Törmälehto, S., Mononen, M. E., Aarnio, E., Arokoski, J. P., Korhonen, R. K., & Martikainen, J. (2018). Health-related quality of life in relation to symptomatic and radiographic definitions of knee osteoarthritis: Data from Osteoarthritis Initiative (OAI) 4-year follow-up study. Health and Quality of Life Outcomes, 16(1), 154
Bindawas, S. M., Vennu, V., & Auais, M. (2015). Health-related quality of life in older adults with bilateral knee pain and back pain: Data from the Osteoarthritis Initiative. Rheumatology International, 35(12), 2095–2101
Bellamy, N. (2002). WOMAC: A 20-year experiential review of a patient-centered self-reported health status questionnaire. Journal of Rheumatology, 29(12), 2473–2476
McConnell, S., Kolopack, P., & Davis, A. M. (2001). The Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC): A review of its utility and measurement properties. Arthritis and Rheumatism, 45(5), 453–461
Xie, F., Pullenayegum, E. M., Li, S. C., Hopkins, R., Thumboo, J., & Lo, N. N. (2010). Use of a disease-specific instrument in economic evaluations: Mapping WOMAC onto the EQ-5D utility index. Value in Health, 13(8), 873–878
Kharroubi, S. A., Brazier, J. E., Roberts, J., & O’Hagan, A. (2007). Modelling SF-6D health state preference data using a nonparametric Bayesian method. Journal of Health Economics, 26(3), 597–612
Mulhern, B., Feng, Y., Shah, K., Janssen, M. F., Herdman, M., van Hout, B., et al. (2018). Comparing the UK EQ-5D-3L and English EQ-5D-5L value sets. PharmacoEconomics, 36(6), 699–713
Richardson, J., Sinha, K., Iezzi, A., & Khan, M. A. (2014). Modelling utility weights for the Assessment of Quality of Life (AQoL)-8D. Quality of Life Research, 23(8), 2395–2404
Feeny, D., Furlong, W., Torrance, G. W., Goldsmith, C. H., Zhu, Z., DePauw, S., et al. (2002). Multiattribute and single-attribute utility functions for the health utilities index mark 3 system. Medical Care, 40(2), 113–128
Brazier, J. E., & Roberts, J. (2004). The estimation of a preference-based measure of health from the SF-12. Medical Care, 42, 851–859
Horsman, J., Furlong, W., Feeny, D., & Torrance, G. (2003). The Health Utilities Index (HUI®): Concepts, measurement properties and applications. Health and Quality of Life Outcomes, 1(1), 54
Brooks R, Group E. (1996). EuroQol: The current state of play. Health Policy, 37(1), 53–72
Edlin, R., McCabe, C., Hulme, C., Hall, P., & Wright, J. (2015). Cost effectiveness modelling for health technology assessment. Springer.
Muennig, P., & Bounthavong, M. (2016). Cost-effectiveness analysis in health: A practical approach. Wiley.
National Institute for Health and Care Excellence. (2015). Single technology appraisal: User guide for company evidence submission template-Process and methods-3 Cost effectiveness. National Institute for Health and Care Excellence. [updated 01 April 2017]. Process and methods [PMG24]. Retrieved from https://www.nice.org.uk/process/pmg24/chapter/cost-effectiveness#economic-analysis
Government A. (2020). Quality-Adjusted-Life-Years (QALYs) Australia: The Department of Health. Retrieved from https://www1.health.gov.au/internet/publications/publishing.nsf/Content/illicit-pubs-needle-return-1-rep-toc~illicit-pubs-needle-return-1-rep-5~illicit-pubs-needle-return-1-rep-5-2
Torrance, G. W., & Feeny, D. (1989). Utilities and quality-adjusted life years. International Journal of Technology Assessment in Health Care, 5(4), 559–575
Karmarkar, T. D., Maurer, A., Parks, M. L., Mason, T., Bejinez-Eastman, A., Harrington, M., et al. (2017). A fresh perspective on a familiar problem: Examining disparities in knee osteoarthritis using a Markov model. Medical Care, 55(12), 993
Beck, J. R., & Pauker, S. G. (1983). The Markov process in medical prognosis. Medical Decision Making, 3(4), 419–458
Sonnenberg, F. A., & Beck, J. R. (1993). Markov models in medical decision making: A practical guide. Medical Decision Making, 13(4), 322–338
Graves, N., Wloch, C., Wilson, J., Barnett, A., Sutton, A., Cooper, N., et al. (2016). A cost-effectiveness modelling study of strategies to reduce risk of infection following primary hip replacement based on a systematic review. Health Technology Assessment. https://doi.org/10.3310/hta20540
Clarke, P. M., Hayes, A. J., Glasziou, P. G., Scott, R., Simes, J., & Keech, A. C. (2009). Using the EQ-5D index score as a predictor of outcomes in patients with type 2 diabetes. Medical Care, 47(1), 61–68
Skinner, E. H., Denehy, L., Warrillow, S., & Hawthorne, G. (2013). Comparison of the measurement properties of the AQoL and SF-6D in critical illness. Critical Care and Resuscitation, 15(3), 205–212
Zhao, T., Winzenberg, T., de Graaff, B., Aitken, D., Ahmad, H., & Palmer, A. J. (2020). A systematic review and meta-analysis of health state utility values for osteoarthritis-related conditions. Arthritis Care & Research. https://doi.org/10.1002/acr.24478
Jinks, C., Jordan, K., & Croft, P. (2002). Measuring the population impact of knee pain and disability with the Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC). Pain, 100(1–2), 55–64
Wilkie, R., Peat, G., Thomas, E., & Croft, P. (2007). Factors associated with restricted mobility outside the home in community-dwelling adults ages fifty years and older with knee pain: An example of use of the International Classification of Functioning to investigate participation restriction. Arthritis and Rheumatism, 57(8), 1381–1389
Jinks, C., Jordan, K., & Croft, P. (2007). Osteoarthritis as a public health problem: The impact of developing knee pain on physical function in adults living in the community: (KNEST 3). Rheumatology (Oxford), 46(5), 877–881
Fransen, M., Su, S., Harmer, A., Blyth, F. M., Naganathan, V., Sambrook, P., et al. (2013). A longitudinal study of knee pain in older men: Concord Health and Ageing in Men Project. Age and Ageing, 43(2), 206–212
Woo, J., Leung, J., & Lau, E. (2009). Prevalence and correlates of musculoskeletal pain in Chinese elderly and the impact on 4-year physical function and quality of life. Public Health, 123(8), 549–556
Venn, A. J., Thomson, R. J., Schmidt, M. D., Cleland, V. J., Curry, B. A., Gennat, H. C., et al. (2007). Overweight and obesity from childhood to adulthood: A follow-up of participants in the 1985 Australian Schools Health and Fitness Survey. Medical Journal of Australia, 186(9), 458–460
Magnussen, C. G., Raitakari, O. T., Thomson, R., Juonala, M., Patel, D. A., Viikari, J. S., et al. (2008). Utility of currently recommended pediatric dyslipidemia classifications in predicting dyslipidemia in adulthood: evidence from the Childhood Determinants of Adult Health (CDAH) study, Cardiovascular Risk in Young Finns Study, and Bogalusa Heart Study. Circulation, 117(1), 32–42
Smith, K. J., Sanderson, K., McNaughton, S. A., Gall, S. L., Dwyer, T., & Venn, A. J. (2014). Longitudinal associations between fish consumption and depression in young adults. American Journal of Epidemiology, 179(10), 1228–1235
Tian, J., Gall, S., Patterson, K., Otahal, P., Blizzard, L., Patton, G., et al. (2019). Socioeconomic position over the life course from childhood and smoking status in mid-adulthood: Results from a 25-year follow-up study. BMC Public Health, 19(1), 169
Tian, J., Venn, A. J., Blizzard, L., Patton, G. C., Dwyer, T., & Gall, S. L. (2016). Smoking status and health-related quality of life: A longitudinal study in young adults. Quality of Life Research, 25(3), 669–685
Antony, B., Jones, G., Venn, A., Cicuttini, F., March, L., Blizzard, L., et al. (2015). Association between childhood overweight measures and adulthood knee pain, stiffness and dysfunction: A 25-year cohort study. Annals of the Rheumatic Diseases, 74(4), 711–717
Obese, H. (1998). Body Mass Index (BMI). Obesity Research, 6(2), 51S-209S
World Health Organization. (2000). Obesity: Preventing and managing the global epidemic. World Health Organization.
Heintjes, E., Bierma-Zeinstra, S., Berger, M., & Koes, B. (2008). Lysholm scale and WOMAC index were responsive in prospective cohort of young general practice patients. Journal of Clinical Epidemiology, 61(5), 481–488
Antony, B., Venn, A., Cicuttini, F., March, L., Blizzard, L., Dwyer, T., et al. (2016). Correlates of knee bone marrow lesions in younger adults. Arthritis Research & Therapy, 18, 31
Zhang, B., Lin, H., Hunter, D. J., Neogi, T., Wise, B., Choy, E., et al. (2009). A multistate transition model for osteoarthritis pain change. Communications in Statistics – Theory and Methods, 38(18), 3297–3306
Obradovic, M., Lal, A., & Liedgens, H. (2013). Validity and responsiveness of EuroQol-5 dimension (EQ-5D) versus Short Form-6 dimension (SF-6D) questionnaire in chronic pain. Health and Quality of Life Outcomes, 11(1), 110
Campbell, J. A., Jelinek, G. A., Weiland, T. J., Nag, N., Neate, S. L., Palmer, A. J., et al. (2020). SF-6D health state utilities for lifestyle, sociodemographic and clinical characteristics of a large international cohort of people with multiple sclerosis. Quality of Life Research, 29(9), 2509–2527
Dritsaki, M., Petrou, S., Williams, M., & Lamb, S. E. (2017). An empirical evaluation of the SF-12, SF-6D, EQ-5D and Michigan Hand Outcome Questionnaire in patients with rheumatoid arthritis of the hand. Health and Quality of Life Outcomes, 15(1), 20
Jayadevappa, R., Cook, R., & Chhatre, S. (2017). Minimal important difference to infer changes in health-related quality of life—A systematic review. Journal of Clinical Epidemiology, 89, 188–198
Walters, S. J., & Brazier, J. E. (2005). Comparison of the minimally important difference for two health state utility measures: EQ-5D and SF-6D. Quality of Life Research, 14(6), 1523–1532
Luo, N., Johnson, J. A., & Coons, S. J. (2010). Using instrument-defined health state transitions to estimate minimally important differences for four preference-based health-related quality of life instruments. Medical Care, 48, 365–371
Lee, B. B., King, M. T., Simpson, J. M., Haran, M. J., Stockler, M. R., Marial, O., et al. (2008). Validity, responsiveness, and minimal important difference for the SF-6D health utility scale in a spinal cord injured population. Value in Health, 11(4), 680–688
Norman, R., Church, J., van den Berg, B., & Goodall, S. (2013). Australian health-related quality of life population norms derived from the SF-6D. Australian and New Zealand Journal of Public Health, 37(1), 17–23
Van den Berg, B. (2012). SF-6D population norms. Health Economics, 21(12), 1508–1512
Antony, B., Venn, A., Cicuttini, F., March, L., Blizzard, L., Dwyer, T., et al. (2015). Association of physical activity and physical performance with tibial cartilage volume and bone area in young adults. Arthritis Research & Therapy. https://doi.org/10.1186/s13075-015-0813-0
Agaliotis, M., Fransen, M., Bridgett, L., Nairn, L., Votrubec, M., Jan, S., et al. (2013). Risk factors associated with reduced work productivity among people with chronic knee pain. Osteoarthritis and Cartilage, 21(9), 1160–1169
Sayre, E. C., Li, L. C., Kopec, J. A., Esdaile, J. M., Bar, S., & Cibere, J. (2010). The effect of disease site (knee, hip, hand, foot, lower back or neck) on employment reduction due to osteoarthritis. PLoS ONE, 5(5), e10470
Hubertsson, J., Petersson, I. F., Thorstensson, C. A., & Englund, M. (2013). Risk of sick leave and disability pension in working-age women and men with knee osteoarthritis. Annals of the Rheumatic Diseases, 72(3), 401–405
Afzali, T., Fangel, M. V., Vestergaard, A. S., Rathleff, M. S., Ehlers, L. H., & Jensen, M. B. (2018). Cost-effectiveness of treatments for non-osteoarthritic knee pain conditions: A systematic review. PLoS ONE, 13(12), e0209240
Ara, R., Brazier, J., & Zouraq, I. A. (2017). The use of health state utility values in decision models. PharmacoEconomics, 35(1), 77–88
Ara, R., & Brazier, J. E. (2010). Populating an economic model with health state utility values: Moving toward better practice. Value in Health, 13(5), 509–518
Ara, R., & Wailoo, A. (2012). Using health state utility values in models exploring the cost-effectiveness of health technologies. Value in Health, 15(6), 971–974
Paxton, E. S., Kymes, S. M., & Brophy, R. H. (2010). Cost-effectiveness of anterior cruciate ligament reconstruction: A preliminary comparison of single-bundle and double-bundle techniques. The American Journal of Sports Medicine, 38(12), 2417–2425
Brazier, J. (2008). Valuing health states for use in cost-effectiveness analysis. PharmacoEconomics, 26(9), 769–779
Wilson, R., & Abbott, J. H. (2018). Development and validation of a new population-based simulation model of osteoarthritis in New Zealand. Osteoarthritis Cartilage, 26(4), 531–539
Abbott, J. H., Usiskin, I. M., Wilson, R., Hansen, P., & Losina, E. (2017). The quality-of-life burden of knee osteoarthritis in New Zealand adults: A model-based evaluation. PLoS ONE, 12(10), e0185676
Muraki, S., Akune, T., Oka, H., En-Yo, Y., Yoshida, M., Saika, A., et al. (2011). Health-related quality of life in subjects with low back pain and knee pain in a population-based cohort study of Japanese men: The Research on Osteoarthritis Against Disability study. Spine (Phila Pa 1976), 36(16), 1312–1319
Antonopoulou, M. D., Alegakis, A. K., Hadjipavlou, A. G., & Lionis, C. D. (2009). Studying the association between musculoskeletal disorders, quality of life and mental health. A primary care pilot study in rural Crete, Greece. BMC Musculoskeletal Disorders, 10, 143
Norimatsu, T., Osaki, M., Tomita, M., Ye, Z., Abe, Y., Honda, S., et al. (2011). Factors predicting health-related quality of life in knee osteoarthritis among community-dwelling women in Japan: The Hizen-Oshima study. Orthopedics, 34(9), e535–e540
Hoogeboom, T. J., den Broeder, A. A., de Bie, R. A., & van den Ende, C. H. M. (2013). Longitudinal impact of joint pain comorbidity on quality of life and activity levels in knee osteoarthritis: Data from the Osteoarthritis Initiative. Rheumatology (Oxford, England), 52(3), 543–546
Kauppila, A. M., Kyllonen, E., Mikkonen, P., Ohtonen, P., Laine, V., Siira, P., et al. (2009). Disability in end-stage knee osteoarthritis. Disability and Rehabilitation, 31(5), 370–380
Adegoke, B. O., Babatunde, F. O., & Oyeyemi, A. L. (2012). Pain, balance, self-reported function and physical function in individuals with knee osteoarthritis. Physiotherapy Theory and Practice, 28(1), 32–40
Jordan, J., Luta, G., Renner, J., Dragomir, A., Hochberg, M., & Fryer, J. (1997). Knee pain and knee osteoarthritis severity in self-reported task specific disability: The Johnston County Osteoarthritis Project. Journal of Rheumatology, 24(7), 1344–1349
Cherepanov, D., Palta, M., Fryback, D. G., & Robert, S. A. (2010). Gender differences in health-related quality-of-life are partly explained by sociodemographic and socioeconomic variation between adult men and women in the US: Evidence from four US nationally representative data sets. Quality of Life Research, 19(8), 1115–1124
Lundberg, L., Johannesson, M., Isacson, D. G. L., & Borgquist, L. (1999). Health-state utilities in a general population in relation to age, gender and socioeconomic factors. European Journal of Public Health, 9(3), 211–217
Acknowledgements
The authors would like to acknowledge the contributions of the CDAH project manager, Marita Dalton, CDAH participants, funders, and all other project staff.
Funding
AS is supported by the International Graduate Research Scholarship, University of Tasmania. BA is supported by the National Health and Medical Research Council of Australia Fellowship. The CDAH study was supported by the National Health and Medical Research Council Project Grant 211316. CDAH-knee sub-study was supported by the Royal Hobart Hospital Research Foundation (RHHRF) Grant 18-202 RHHRF.
Author information
Authors and Affiliations
Contributions
AS, BA, and AP conceived the Study. AS, JC, and LB conceived the present analysis. AS and LB cleaned and prepared the data and performed the analysis. AS and JC undertook HSU estimation using the algorithm. AS drafted the first draft of the manuscript, and JC, AV, GJ, LB, AP TD, FC, CD, and BA edited the manuscript. All authors commented on and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that there is no conflict of interest.
Ethical approval
The Southern Tasmania Health and Medical Human Research Ethics Committee, Monash University Human Research Ethics Committee, and the Northern Sydney and Central Coast Area Human Research Ethics Committee provided ethical approval for the study (Ethics ID: H0018491).
Informed consent
Participants were asked to read the participant information and to consent before entering the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Singh, A., Campbell, J.A., Venn, A. et al. Association between knee symptoms, change in knee symptoms over 6–9 years, and SF-6D health state utility among middle-aged Australians. Qual Life Res 30, 2601–2613 (2021). https://doi.org/10.1007/s11136-021-02859-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11136-021-02859-5