Abstract
Purpose
The proportion of very old people is rising, and so, describing their health-related quality of life (HRQoL) is an important point of interest. The aim of this study was to analyse the predictive factors on HRQoL throughout a 3-year follow-up period, in a community-based cohort of octogenarian people.
Methods
From 290 subjects aged 85 and over, sociodemographic and geriatric data, including levels of frailty phenotype assessment, and HRQoL using the EuroQol 5D3L (EQ-5D) instrument were collected. A longitudinal analysis was performed by generalized estimating equations (jointly testing the bivariate effect of variables and its time dependence) and regression mixed models to evaluate the adjusted effect of variables on HRQoL after a 3-year follow-up.
Results
In the EQ-5D baseline assessment, the average visual analogue self-rating scale value was 63.82 (SD ± 19.45), the EQ-5D index was 0.67 (0.34) and the most significant issues were pain/discomfort (61.2 %), depression (45.3 %) and mobility (44.6 %). The third year index was 0.55 (0.38). Independent predictive factors of a lower HRQoL identified by the regression mixed models were female gender (marginal effect ME = −0.101; p = 0.003), being pre-frail (ME = −0.142; p = 0.011) or frail (ME = −0.071; p = 0.030), having heart failure (ME = −0.081; p = 0.037) and having a high social risk score (ME = −0.020; p = 0.015). In contrast, higher functional status (ME = 0.050; p < 0.001) and nutritional score (ME = 0.013; p = 0.011) appeared to be predictive factors of an enhanced HRQoL. The adjusted effect of “time of follow-up” had no statistical significance.
Conclusion
Frail individuals at baseline have a significant lower HRQoL, whereas a higher functional status and nutritional status are independent predicting factors of an enhanced HRQoL after 3 years of follow-up. These findings may encourage clinicians in order to asses HRQoL.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In recent decades, the oldest old group (80 years or older) has been the most rapidly expanding population segment in developed countries [1]. The proportion of very old people is projected to increase even more, tripling from 22 million in 2008 to 61 million in 2060 in Europe [2]. Although the proportion of elderly with severe disabilities has decreased, more mild levels of disability have increased in people older than 65 in the UK [2, 3]. Therefore, with elderly populations continuing to rise, maintaining and improving health-related quality of life (HRQoL) has become a major focus of interest [4–7].
The HRQoL is not a single entity. It is a complex state to measure because several domains, including physical, emotional, spiritual, cognitive and social well-being, play important roles [4, 5, 8]. A multidisciplinary approach is required to manage the heterogeneity of several health outcomes associated with ageing such as visual and hearing impairment, loneliness, disability and cognitive changes that impact the HRQoL measurements [8–10]; especially, in elderly the frailty syndrome, a diminished capacity to compensate effectively for external stresses [11], may also impact on HRQoL as was described [12].
Furthermore, the oldest old have been considered to have a “poorer” HRQoL than younger elderly people because of the co-morbid medical conditions and multiple medications [13, 14]. However, among nonagenarians in our area, we reported a relatively good HRQoL despite the presence of functional decline and nutritional risk co-morbidities [15], and similarly, other groups reported that the increase in longevity can be associated with better HRQoL, especially if accompanied by reasonable levels of mental and physical health [14]. Therefore, the relationship of these factors to QoL has not been well delineated, and the information available about the oldest old people is limited. We hypothesize that oldest old people of the community will demonstrate characteristic predictive factors which could account for the loss of global HRQOL. Thus, the aim of this study was to analyse the predictive factors on HRQoL based on individual evaluations throughout a 3-year follow-up period, in a community cohort of octogenarian people.
Methods
The current data were gathered within the framework of the Octabaix study, a prospective population-based study of 328 community-dwelling older adults. All subjects were Spanish Caucasians born in 1924 (85-years-old at the time of inclusion, in 2009) who were registered in one of the seven primary healthcare centres in the geographical area of Baix Llobregat (Barcelona, Spain). The sampling procedures and data collection methods have been described elsewhere [16]. The combined population served includes approximately 210,000 inhabitants, and the referral hospital is the Hospital Universitari de Bellvitge. The only exclusion criterion was to be institutionalized (24 h of professional care available). From the total 696 potential subjects, 487 were eligible individuals after cleaning the data, according to telephone citation and verification of the listings of the computer services of primary health. Different reasons for non-inclusion were as follows: 76 (10.9 %) living in unregistered nursing homes as listed, 67 (9.6 %) died before the study started and 66 (9.4 %) displaced to another city. The participation rate was 67.5 %, and there were no differences among respondents and non-respondents in terms of gender, healthcare centre or physician in charge. Subjects were examined at home or at the primary healthcare centre if they were ambulatory, by trained teams (medical doctors and nurses) with extensive experience in working with older adults. The institutional ethics committee of the Jordi Gol Institute for Primary Care Research approved the study. All patients gave written informed consent.
At baseline, we assessed sensory status (near vision measured using the Jaeger test and hearing ability measured using the whisper test), geriatric assessment tests and chronic health problems. The list of chronic health problems performed according to medical records was as follows: hypertension (≥140/90 mmHg), diabetes mellitus (DM), ischaemic cardiopathy, stroke, heart failure, atrial fibrillation (determined by electrocardiogram), dementia, Parkinson disease, chronic obstructive lung disease, thyroid disease, cancer and anaemia. The geriatric assessment included the functional status measured using the Barthel Index score (BI) [17] for basic activities of daily living (ADL). The BI measures performance in seven areas: feeding, bathing, personal cleanliness, dressing, bowel/bladder control, getting on/off the toilet and locomotion. A maximum score of 100 indicates that the subject is independent, and a score of 0 is completely dependent. The Lawton Index (LI) score [18] for instrumental ADL ranges from 0 (low function, dependent) to 8 (high function, independent). Cognitive status was evaluated with the mini-mental state examination a widely used cognitive impairment screening test. The version of the mini-mental state examination that has been adapted and validated for use in Spain, known as the MEC [19], takes particular account of the very low educational level of the elderly Spanish population. Cognitive status was evaluated using this MEC, which provides a score of up to 35 (a score of 23 or below indicates cognitive impairment; a score of 19 or below indicates sever cognitive impairment). The MEC assesses orientation, memory, concentration, language and praxis. The Charlson comorbidity index [20] was used to measure overall comorbidity. Scores here range from 0 to a theoretical maximum of 33, depending on the presence of certain diseases (dementia, cancer lymphoma, stroke, sever renal impairment, etc.) with assigned values. This study considered as a significant comorbidity a Charlson Index score of two or more and disability as a BI < 90 or a LI < 7. Nutritional status was assessed using the mini nutritional assessment (MNA) [21], which has a maximum score of 30 and scores <23.5 classified as a risk of malnutrition. Social risk was measured using the Gijon Test [22] whose maximum score is 24 points (social risk between 10 and 14; social problems with >15) exploring five dimensions (family, housing, income, loneliness, and dependence on a caregiver and social relations). A five-component examination modification of Fried’s definition was applied to identify frail individuals [11]. Frailty was coded on three levels, based on the sum of the number of individual criteria present (frail 3–5, pre-frail 1–2, non-frail 0). The screening criteria of frailty used were as follows: 1/Weakness, measured using a handgrip dynamometer; 2/Slowness, measured using walking speed with the Get up and Go Test; 3/Weight Loss, individuals reporting more than 5 % (≥3 kg); 4/Exhaustion, based on the question, “In the last month, have you had too little energy to do the things you wanted to do?”; 5/Low Physical Activity, based on the question “Do you engage in activities such as gardening, dancing, going for a walk?” The number of falls in the year previous to the study, the total number of chronic medications (polypharmacy ≥5) and the number of hospitalizations were also recorded. The patients’ HRQoL was measured with a generic instrument, the EuroQol 5D3L (EQ-5D) [23], which is a self-administered, instrument with a 5-question multi-attribute questionnaire (EQ-5D descriptive system) and a visual analogue self-rating scale (VAS). Respondents were asked to rate the severity of their current problems (level 1 = no problems, level 2 = moderate problems and level 3 = severe problems) for five dimensions of health: mobility, self-care, usual activities, pain/discomfort and anxiety/depression. Using this information, patients were classified into 243 (35) possible health statuses. EQ-5D health statuses can be converted into a EQ-5D index score ranging from −0.594 to 1.0 (in which 1 is full health, 0 is dead and negative values worse than death), by using a set of weighted preferences. The VAS consists of a 20-cm, vertical 0–100 scale, in which 0 represents the worst imaginable health status and 100 represents the best imaginable health status. The respondents marked a point on the scale to reflect their overall health on the day of the interview.
Procedure
All patients were included for analysis, except those with cognitive impairment at baseline (MEC score <19) because of the lack of reliability and validity of EQ-5D index among these subjects. After 3 years of follow-up, those who did not die, were not institutionalized, did not move to other areas or did not decline to participate were reassessed. All individuals included at baseline were considered for the multivariate analysis. Those who died at follow-up were coded as zero values EQ-5D index (given EQ-5D index score interpretation). Remaining individuals not reassessed, and those developing severe cognitive impairment (MEC < 19) during the follow-up were considered as missing values.
Statistical analysis
Standard descriptives were obtained for the sample. For the bivariate analysis, the McNemar test was used to compare categorical variables, and Student’s paired t test was used to compare continuous variables. A comparative analysis was performed between baseline and the final assessment after 3 years of follow-up. In addition to calculating EQ-5D averages by subgroups according to potential associated factors, multiple generalized estimating equation (GEE) models were used to explore the bivariate unadjusted effect of variables on HRQoL taking into account the time of follow-up (“interaction with time”). The issue of missing data was handled by the multiple imputations (also for the outcome) a highly recommended statistical procedure in front of the complete case analysis [24]. For multivariate regression analysis, mixed models were fit to evaluate the adjusted effect of variables on HRQoL taking into account the longitudinal nature of data. Two mixed models were analysed: the mixed model by multiple imputation (including missing data) and the complete cases mixed model (complete cases mixed models provided in “Appendix”). A complete model was adjusted in which all variables were considered relevant a priori. A significant final model was obtained by eliminating the variables that did not add information (according to the Akaike information criterion) from the complete model. This procedure was used to estimate the adjusted coefficient of the marginal effect (SD). The results were interpreted as statistically significant if p < 0.05. All analyses were performed using R.2.14.2 Statistics.
Results
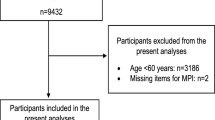
The final study sample comprised 290 subjects from a total of 328 in the original cohort, after excluding 38 due to a cognitive impairment (MEC score <19) present at baseline. After 3 years of follow-up, 30 (10.3 %) subjects died, 19 were institutionalized, nine moved to another area and 28 declined to participate. Therefore, 204 (70.3 %) people could be reassessed at the end of the third year of follow-up (Fig. 1). Of these 290 included at baseline, 174 were women (60.0 %), and 233 (80.3 %) had <6 years of education. Impaired visual acuity was present in 230 (79 %) subjects, and 107 (37 %) had impaired auditory acuity. Hypertension was recorded in 222 cases (79 %), dyslipidaemia in 147 (501 %) and DM in 47 (16 %). The most prevalent chronic diseases were anaemia 49 (17 %), presence of previous stroke 42 (15 %), heart failure 41 (14 %), atrial fibrillation 38 (13 %) and cancer 37 (13 %). The mean (±SD) values at baseline were the following: BI 89.79 (15.84), LI 5.68 (2.38), MEC 28.52 (4.49), Charlson comorbidity index 1.38 (1.57), MNA 24.88 (3.56) and social risk test 9.61 (2.63). One hundred and sixty (55.0 %) people were pre-frail, and 49 (16.7 %) were frail. Concerning HRQoL, baseline prevalence of various issues was the following: mobility 129 (44.6 %), self-care 63 (21.8 %), usual activities 102 (35.3 %), pain/discomfort 177 (61.2 %) and anxiety/depression 131 (45.3 %). The mean VAS value was 63.82 (19.45), and the EQ-5D index was 0.67 (0.34).
Table 1 shows the comparison of the sociodemographic and co-morbidity characteristics at baseline and after the 3 years of follow-up. There were statistically significant differences with p < 0.001 in the BI, LI, MEC, Charlson Index, MNA, frailty, hospital emergencies and heart failure. Additional variables that reached statistical significance were previous stroke (p = 0.003), dementia (p = 0.008), ischaemic cardiomyopathy (p = 0.009) and atrial fibrillation (p = 0.015).
Table 2 shows the effect of variables along three years of follow-up on HRQoL status and tested their interaction with time. The variables in which time did not modify their statistically significant effect on HRQoL were gender, education level, visual acuity, anaemia diagnosis, ischaemic cardiomyopathy, dementia, frailty, Charlson Index, LI, BI, MEC, MNA, social risk, medications and number of hospital emergencies.
Independent predictors of lower HRQoL identified by the mixed models are shown in Table 3. These included female gender, pre-frail or frail, heart failure and a high social risk score. However, higher LI scores and higher MNA at baseline were independent predictors of a higher HRQoL at the third year of follow-up. The adjusted effect of “time itself” was not found to be significant to explain changes of global HRQoL when taking into account the presented explanatory variables.
Discussion
This longitudinal study revealed that a high level of HRQoL was maintained in most of our community-dwelling octogenarians despite a decline after 3 years of follow-up. Frailty was one of the main predicting factors for a reduced HRQoL while, interestingly enough, having a higher functional and nutritional status appeared as independent factors which predicted a higher HRQoL at the 3-year follow-up assessment.
The baseline cohort was a healthy community group with high scores on functional activities (BI 89.8), no cognitive impairment (MEC 28.5) and a low global co-morbidity index (1.4). The assessment of EQ-5D revealed a good VAS (63.8) and a high EQ-5D index (0.67). Accordingly, in a 61-year-old diabetic group, Fritz et al. [25] also reported a high proportion (88 %) of individuals that assessed their HRQoL as good, in addition to other studies carried out in elderly populations but with less favourable percentages such as of the 85-year-olds in Newcastle community (77 %) and of the 78-year-old community group (76 %) described by Szaton et al. [10, 26]. One explanation for these different values may be the heterogeneity of the different instruments used for measuring HRQoL. Therefore, the Newcastle study from the UK [26] utilized one single self-rated general health question. In our study group, HRQoL was estimated by using the EQ-5D index, and Szanton et al. [10] from US reported the EQ-5D descriptive system scores but not the EQ-5D index, whereas the Swedish study [25] described a profile of individuals’ health by a generic HRQoL questionnaire with thirteen sub-scales (physical functioning, pain, sleep, marital status, sexual activity, etc.) of psychometric measures, different from the overall health score provided by EQ-5D index of econometric measures. Thus, these heterogeneity HRQoL measures offer different discriminant abilities which could be explained by the methodological development of instruments and different aspects related to HRQoL.
In our study, the characteristics of the sample showed that being female appeared as a predictor of a lower HRQoL as reported by studies performed in similar-aged community groups [7, 27] and contrasts with other younger groups [8, 10]. Exploring other sociodemographic characteristics, we found that in this octogenarian group, the EQ-5D index at baseline (0.67) was higher than in nonagenarians (0.58) of the same community [15], as expected given the influence of ageing on HRQoL [28]. However, this community sample presents similar HRQoL to that reported in patients aged 75 (0.63) with chronic heart failure [29]. Therefore, despite a global decrease from 0.67 to 0.55 in a three-year period in our group, social, functional and cognitive outcomes were able to explain such decrease as shown the non-significance of “time” in the multivariate regression analyses. These results emphasize that the decline in health status may be attributed to geriatric conditions rather than to the temporary or chronological effect of ageing itself [30, 31].
Interestingly, frailty phenotype—a state that reflects the vulnerability to stresses and increased risk of adverse outcomes—has emerged as determinant HRQoL [13, 32]. In our study, frailty also appeared as a strong predicting factor. In terms of quality and quantity of life, by using the quality-adjusted life years (QALYs) [33], the estimated impact of pre-frailty in our octogenarians was 0.92 of QALYs per individual (calculated as 0.142—utility score for pre-frailty—multiplied by 6.5—life expectancy years at 85-years-old) and 0.46 of QALYs for frailty. The relationship was confirmed after adjusting for gender, education and relevant health behaviours (social risk and physical activity), and persisted when co-morbidities were included in the analysis. Thus, this is an important point, because it is preciously in this pre-frail group (unlike the frail group) which has a higher capacity to compensate external stresses (falls) where the capacity to compensate internal stress (anxiety, pain) probably has a higher impact on the loss of HRQoL. Therefore, in order to keep the incidence of frailty phenotype to a minimum and to avoid a decline in HRQoL over time, interventions should be strongly targeted towards pre-frail elderly people. This, together with our results concerning instrumental activities as another important determining factor of HRQoL as described [7], emphasizes the relevant importance of conducting comprehensive multidimensional examinations not only for detecting early stages of deterioration or latent diseases but also for implementing strategies to promote a more favourable HRQoL.
In contrast to other groups [9], our study did not show global co-morbidity as a reliable predicting factor for a lower HRQoL. Despite a close association between HRQoL and individual co-morbidities such as nutritional risk and heart failure, no clear relationship was found in this group with other diseases [34], such as myocardial infarction, angina, hypertension and diabetes [25, 31]. Therefore, in this elderly group, the association between HRQoL and some individual co-morbidities may become weaker since there are other different levels of severity and related medical conditions that can impact. Consequently, these findings should be treated with caution and further studies are needed to complete these results. Additionally, a remarkable inverse association was found in the bi-variant analysis between multiple medications and HRQoL although it did not persist. This in our opinion reinforces the well-described patient preferences and should be taken into account upon considering different therapy management options [8, 25].
Finally, a peculiar result of this study was the mediating effect of social risk over HRQoL. This finding seems to be especially important in this particular community group which had homogeneous socio-economic characteristics that may be indirectly related to other potential social factors, such as caregiver influence [35] and the support provided for mobility, nutrition, hygienic conditions, transport facilities or pharmacy surveillance [13]. Accordingly, future strategies to encourage receiving examinations, such as appealing announcements, transportation service facilities and home visit examinations, should be considered to determine frailty and social risk early in elderly people.
A main strength of the present study is the baseline sample size of the community-dwelling population. They had similar characteristics in terms of age, education and socio-economic environment; however, this strength might also constitute a weakness by limiting the generalization of the results to other areas or countries. Nevertheless, the use of EQ-5D, which is a validated instrument, may allow other authors to compare our results to other patient groups to help develop broad health service resource allocation decisions in other settings. There are several limitations in this study. The cohort was drawn from a relatively small number of centres. This study was drawn as an observational cohort not specifically designed to test the effect of a specific variable on the outcome; thus, these results cannot be considered more than exploratory, requiring further studies to specifically determine the effect of any of these explanatory variables on HRQoL (i.e. a well-planified controlled trial). Finally, the percentage of missing data is high in our study, which is probably due to the high percentage of frail elderly people with higher level of morbidities at the follow-up than that observed at baseline, who were subsequently institutionalized, declined to participate or moved from the area. Therefore, we utilized multiple imputations for the statistical analyses to handle this issue of missing data.
In conclusion, a high level of HRQoL was maintained in this group of octogenarians compared with other population groups despite a decline at the follow-up assessment. Frailty was a significant predictor of lower HRQoL, while functional and nutritional statuses were independent predictors of a better HRQoL after 3 years of follow-up. These findings may encourage clinicians to recognize frail individuals to initiate measures for preventing and treating frailty at an earlier date in order to assess the HRQoL.
References
Christensen, K., Doblhammer, G., Rau, R., & Vaupel, J. W. (2009). Ageing populations: The challenges ahead. Lancet, 374, 1196–1208.
Rechel, B., Grundy, E., Robine, J. M., Cylus, J., Mackenbach, J. P., Knai, C., et al. (2013). Ageing in the European Union. Lancet, 381, 1312–1322.
Jagger, C., Matthews, R., Matthews, F., Robinson, T., Robine, J. M., & Brayne, C. (2007). Medical Research Council Cognitive Function and Ageing Study Investigators. The burden of diseases on disability-free life expectancy in later life. Journals of Gerontology. Series A, Biological Sciences and Medical Sciences, 62, 408–414.
Dening, T. R., Chi, L. Y., Brayne, C., Huppert, F. A., Paykel, E. S., & O’Connor, D. W. (1998). Changes in self-rated health, disability and contact with services in a very elderly cohort: A 6-year follow-up study. Age and Ageing, 27, 23–33.
Ploeg, J., Brazil, K., Hutchison, B., Kaczorowski, J., Dalby, D. M., Goldsmith, C. H., et al. (2010). Effect of preventive primary care outreach on health related quality of life among older adults at risk of functional decline: Randomised controlled trial. BMJ, 16(340), c1480.
Lapid, M. I., Rummans, T. A., Boeve, B. F., McCormick, J. K., Pankratz, V. S., Cha, R. H., et al. (2011). What is the quality of life in the oldest old? International Psychogeriatrics, 23, 1003–1010.
Liao, Y., McGee, D. L., Cao, G., & Cooper, R. S. (2000). Quality of the last year of life of older adults: 1986 versus 1993. JAMA, 283, 512–518.
O’Boyle, C. A. (1997). Measuring the quality of later life. Philosophical Transactions of the Royal Society of London. Series B, Biological Sciences, 352, 1871–1879.
Orfila, F., Ferrer, M., Lamarca, R., & Alonso, J. (2000). Evolution of self-rated health status in the elderly: Cross-sectional versus longitudinal estimates. Journal of Clinical Epidemiology, 53, 563–570.
Szanton, S. L., Thorpe, R. J., Boyd, C., Tanner, E. K., Leff, B., Agree, E., et al. (2011). Community aging in place, advancing better living for elders: A bio-behavioral-environmental intervention to improve function and health-related quality of life in disabled older adults. Journal of the American Geriatrics Society, 59, 2314–2320.
Boyd, C. M., Xue, Q. L., Simpson, C. F., Guralnik, J. M., & Fried, L. P. (2005). Frailty, hospitalization, and progression of disability in a cohort of disabled older women. American Journal of Medicine, 118, 1225–1231.
Chou, C. H., Hwang, C. L., & Wu, Y. T. (2012). Effect of exercise on physical function, daily living activities, and quality of life in the frail older adults: A meta-analysis. Archives of Physical Medicine and Rehabilitation, 93, 237–244.
Ishimoto, Y., Wada, T., Hirosaki, M., Kasahara, Y., Kimura, Y., Konno, A., et al. (2009). Health-related differences between participants and nonparticipants in community-based geriatric examinations. Journal of the American Geriatrics Society, 57, 360–362.
Layte, R., Sexton, E., & Savva, G. (2013). Quality of life in older age: Evidence from an Irish cohort study. Journal of the American Geriatrics Society, 61(s2), S299–S305.
Ferrer, A., Formiga, F., Almeda, J., Alonso, J., Brotons, C., & Pujol, R. (2010). Health-related quality of life in nonagenarians: Gender, functional status and nutritional risk as associated factors. Medicina Clinica (Barc), 134, 303–306.
Ferrer, A., Padrós, G., Formiga, F., Rojas-Farreras, S., Perez, J. M., & Pujol, R. (2012). Diabetes mellitus: Prevalence and impact of morbidities in the oldest old. The octabaix study. Journal of the American Geriatrics Society, 60, 462–467.
Mahoney, F. I., & Barthel, D. W. (1965). Functional evaluation: The Barthel Index. A simple index of independence useful in scoring improvement in the rehabilitation of the chronically ill. Md State Medical Journal, 14, 61–65.
Lawton, M. P., & Brody, E. M. (1969). Assessment of older people: Self-maintaining and instrumental activities of daily living. Gerontologist, 9, 179–186.
Lobo, A., Saz, P., Marcos, G., Día, J. L., de la Cámara, C., Ventura, T., et al. (1999). Revalidación y normalización del Mini-Examen Cognoscitivo (primera versión en castellano del Mini-mental Status Examination) en la población general geriátrica. Medicina Clinica (Barc), 112, 767–774.
Charlson, M. E., Pompei, P., Ales, K. L., & MacKenzie, C. R. (1987). A new method of classifying prognostic comorbidity in longitudinal studies: Development and validation. Journal of Chronic Diseases, 40, 373–375.
Guigoz, Y. (2006). The mini nutritional assessment (MNA) review of the literature–What does it tell us? Journal of Nutrition Health and Aging, 10, 466–485.
Alarcón, M. T., & González, J. I. (1998). La escala sociofamiliar de Gijón, instrumento útil en el hospital general. Review Esp Geriatr Gerontol, 33, 178–180.
Badia, X., Roset, M., Herdman, M., & Kind, P. (2001). A comparison of United Kingdom and Spanish general population time trade-off values for EQ-5D health states. Medical Decision Making, 21, 7–16.
Schafer, J. L. (1999). Multiple imputation: A primer. Statistical Methods in Medical Research, 8, 3–15.
Fritz, T., Caidahl, K., Krook, A., Lundström, P., Mashili, F., Osler, M., et al. (2013). Effects of Nordic walking on cardiovascular risk factors in overweight individuals with type 2 diabetes, impaired or normal glucose tolerance. Diabetes Metabolism Research Reviews, 29, 25–32.
Collerton J, Davies K, Jagger C, Kingston A, Bond J, Eccles MP et al. (2009) Health and disease in 85 year olds: Baseline findings from the Newcastle 85+ cohort study. BMJ 339 b4904.
Pepine, C. J. (2013). Effects of pharmacologic therapy on health-related quality of life in elderly patients with atrial fibrillation: A systematic review of randomized and nonrandomized trials. Clinical Medicine Insights Cardiology, 7, 1–20.
Azpiazu, M., Cruz, A., Villagrasa, J. R., Abanades, J. C., García, N., & Alvarez, C. (2003). Quality of life in noninstitutionalized persons older than 65 years in two health care districts in Madrid. Atencion Primaria, 31, 285–292.
De Rivas, B., Permanyer-Miralda, G., Brotons, C., Brotons, C., Aznar, J., & Sobreviela, E. (2008). Health-related quality of life in unselected outpatients with heart failure across Spain in two different health care levels. Magnitude and determinants of impairment: The INCA study. Quality of Life Research, 17, 1229–1238.
Westlake, C., Dracup, K., Creaser, J., et al. (2002). Correlates of health-related quality of life in patients with heart failure. Heart and Lung, 3, 85–93.
Van der Weele, G. M., Gussekloo, J., De Waal, M. W., De Craen, A. J., & Van der Mast, R. C. (2008). Co-occurrence of depression and anxiety in elderly subjects aged 90 years and its relationship with functional status, quality of life and mortality. International Journal of Geriatric Psychiatry, 21, 2162.
Kanauchi, M., Kubo, A., Kanauchi, K., & Saito, Y. (2008). Frailty, health-related quality of life and mental well-being in older adults with cardiometabolic risk factors. International Journal of Clinical Practice, 62, 1447–1451.
Whitehead, S. J., & Ali, S. (2010). Health outcomes in economic evaluation: The QALY and utilities. British Medical Bulletin, 96, 5–21.
Erceg, P., Despotovic, N., Milosevic, D. P., Soldatovic, I., Zdravkovic, S., Tomic, S., et al. (2013). Health-related quality of life in elderly patients hospitalized with chronic heart failure. Clinical Interventions in Aging, 8, 1539–1546.
Shyu, Y. I., Liang, J., Tseng, M. Y., Li, H. J., Wu, C. C., Cheng, H. S., et al. (2013). Comprehensive and subacute care interventions improve health-related quality of life for older patients after surgery for hip fracture: A randomised controlled trial”. CT. International Journal of Nursing Studies, 50, 1013–1024.
Conflict of interest
None.
Members of the Octabaix Study
J Almeda (Unitat de Suport a la Recerca de Costa de Ponent, IDIAP J Gol), Oriol Cunillera (Unitat de Suport a la Recerca de Costa de Ponent, IDIAP J Gol), C Fernández (CAP Rambla), A Gil (ABS Sant Andreu de la Barca), C Llopart (ABS Sant Andreu de la Barca), MJ Megido (ABS Just Oliveras), G Padrós (Laboratori Clínic L’Hospitalet), M Sarró (CAP Florida Nord) and A Tobella (ABS Martorell Rural) data collection.
Author information
Authors and Affiliations
Consortia
Corresponding author
Appendix
Appendix
See Table 4.
Rights and permissions
About this article
Cite this article
Ferrer, A., Formiga, F., Cunillera, O. et al. Predicting factors of health-related quality of life in octogenarians: a 3-year follow-up longitudinal study. Qual Life Res 24, 2701–2711 (2015). https://doi.org/10.1007/s11136-015-1004-9
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11136-015-1004-9