Abstract
The aim of this work was to evaluate the influence of enzymatic hydrolysis on dietary fiber, phenolic compounds and technological properties of apple pomace as wheat bread supplement. Apple pomace was hydrolyzed with Viscozyme® L, Pectinex® Ultra Tropical, Celluclast® 1.5 L for 1 and 5 h. Soluble (SDF) and insoluble (IDF) dietary fiber, reducing sugars and the total phenolic contents (TPC), along with the technological properties (water and oil retention capacities, solubility index, emulsion stability) of treated apple pomace were evaluated. The prebiotic activity of apple pomace water-soluble fraction on two probiotic strains Lactobacillus acidophilus DSM 20079 and Bifidobacterium animalis DSM 20105 was investigated. Treatment with Celluclast® 1.5 L increased SDF, reducing sugars, SDF/IDF ratio and decreased IDF of apple pomace. While treatment with Viscozyme® L, Pectinex® Ultra Tropical increased reducing sugars, solubility index and TPC, but in most cases reduced oil and water retention capacities, decreased SDF and IDF content. All apple pomace extracts promoted growth of probiotic strains. Addition of 5% of apple pomace hydrolyzed with Celluclast® 1.5 L did not have negative impact on wheat bread, while addition of other enzymatically hydrolyzed apple pomaces decreased pH, specific volume and porosity of wheat bread. Obtained results suggest that apple pomace enzymatically hydrolyzed with Celluclast® 1.5 L can be potentially used for wheat bread supplementation with dietary fiber.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Nowadays consumers are looking for healthy food products with high dietary fiber (DF) content that increases the interest in application of fruit pomace [1]. Bakery products are mostly used for supplementation with DF and apple pomace can be used as source of DF for production of various extruded or baked goods [2]. Apple pomace, mainly obtained after production of juice, represents approximately 30% of the original fruit and contains peel, core, seed, and other part of fruit mostly composed of insoluble carbohydrates [3]. DF is an important nutritional component and it is recommended by the Food and Drug Administration to use DF in the diet approximately 25–35 g per day [4]. However, the composition of DF and soluble dietary fiber (SDF)/insoluble dietary fiber (IDF) ratio are important for nutrition and SDF/IDF ratio is recommended at 1:2 [5]. SDF intake of about 6 g/day is associated with several health benefits such as reduction of LDL-cholesterol in serum and estimated risk for coronary heart disease [4].
The increasing level of pomace in bakery products negatively effects its properties by decreasing loaf volume and increasing hardness [6, 7]. Nevertheless, DF due to the ability to retain water may prolong bread freshness and increase dough viscosity [7, 8]. Enzymatic treatment may change the DF composition and SDF/IDF ratio as well as technological properties and improve quality of bread [8, 9], also may increase phenolic compound content [10]. Increased level of SDF in baked products impacts digestion of starch and other carbohydrates (with high glycemic index) which are present in bread and other cereal-based goods [7]. Incorporation of SDF in food products is easier than IDF, moreover SDF also does not have negative impact for texture or taste, and reduces negative effects of DF for bakery goods [7, 8].
Previously studies indicated apple pomace as a good source for the isolation of compounds such as phenols, pectin, or as a substrate for ethanol production [11,12,13]. Cellulases (for example, commercially available Celluclast 1.5 L) are mostly used for pectin extraction and the yield increases with the increase of both the extraction time and the enzyme dose [13, 14], however long hydrolysis duration and high amount of enzyme may have negative impact for content of SDF [15]. Pectinases increase the amount of phenolic compounds and oligosaccharides [16]. Viscozyme shows good ability to release and solubilize phenolic compounds [9] and produce pectic oligosaccharides [17, 18]. In several works, Viscozyme was also used for pectin extraction [19, 20]. The SDF, pectic oligosaccharides and polyphenols show prebiotic properties and can modulate the profile of the gut microbiota [17, 18, 21].
In this study enzymatic hydrolysis with several commercially available enzymes, such as Viscozyme® L, Pectinex® Ultra Tropical, Celluclast® 1.5 L, was used for modification of DF composition and technological properties of apple pomace, and enzymatically treated apple pomace was used for wheat bread supplementation with DF. Enzymes used for this study are usually used for pectin and phenolic compound extraction, however, there is lack of information about the effect of enzymatic hydrolysis on whole apple pomace technological properties. There are several studies describing apple pomace effect on wheat bread quality, while information about the effect of enzymatically treated apple pomace on wheat bread quality is missing.
Materials and Methods
Apple pomace obtained after juice processing was purchased from “UOGUSULTYS” (Giedriai, Lithuania), the moisture content was 8.46%, particle size < 1 mm. The enzymatic hydrolysis of apple pomace was carried out using commercial enzyme preparations Viscozyme® L, Pectinex® Ultra Tropical, Celluclast® 1.5 L (Novozyme, Denmark) and 5% of enzymatically treated pomace was used for wheat bread supplementation. The influence of enzymatic treatment on chemical composition and technological properties (water retention capacity (WRC), oil retention capacity (ORC), solubility index (SI), emulsion stability), total phenolic content (TPC), in vitro prebiotic activity, FT-IR spectra as well as bread quality was evaluated. Detailed descriptions of pomace hydrolysis, bread preparation and methods used for analysis are shown in the Supplementary material.
Results and Discussion
Apple Pomace Chemical Composition
The proximate chemical composition of apple pomace was determined and presented in supplementary Table 1. The total carbohydrate content was predominant in apple pomace, and RS (46.59 ± 0.93 g/100 g) composed a major part of carbohydrates. TDF content was 35.66 ± 1.25 g/100 g and SDF/IDF ratio was determined at 0.40 (1:2.66), what is close to 1:2. Borujeni et al. [11] also reported high levels of free sugars in apple pomace, where glucose (21.7%) and fructose (24.3%) were the major free sugars. Antonic et al. [22] reported TDF variation in apple pomace from 26.8 to 82.0%. Similar TDF, IDF and SDF of apple pomace were reported by Alongi et al. [23]. TDF content and SDF/IDF ratio depend on variety of apple [24], type of manufacturing process [25].
The TPC of apple pomace was 3.85 mg GAE/g and was within the range of the mixed cultivar of apple pomace reported by Diñeiro García et al. [26]. However, Toledo et al. [27] reported higher amount of TPC in apple by-products (5.92 ± 1.78 mg GAE/g).
DF, RS and TPC of Enzymatically Treated Apple Pomace
DF, RS and TPC content in enzymatically treated apple pomace are presented in Table 1. The enzymatic hydrolysis changed TDF, IDF, SDF content and SDF/IDF ratio. The TDF content significantly (p ≤ 0.05) decreased after enzymatic treatment in all of the samples. In most cases, the duration of hydrolysis did not have significant impact (p ≤ 0.05) on the IDF and SDF content obtained using the same enzyme. It is known, that the changes of pomace cell wall composition and increase of soluble oligomeric and polymeric components depend on the composition and activity of enzyme preparations [28]. The enzymatic treatment with the Celluclast® 1.5 L increased SDF and the SDF/IDF ratio, which was close to the recommended (1:2) [5]. Li et al. [15] reported the increased SDF content in apple pomace after treatment with cellulase. Commercial Celluclast also can be used for enzymatic extraction of pectin [13, 29]. Viscozyme® L and Pectinex® Ultra Tropical decreased the content of the SDF compared to the control and the lowest amount was obtained in sample treated with Pectinex® Ultra Tropical for 5 h. Other studies showed that enzymatic hydrolysis with pectinases increase oligosaccharides [16,17,18]. However, dosage of enzyme and treatment duration can negatively affect SDF by increasing content of oligomers and monomers [15] which do not precipitate with ethanol [30]. Enzymatic treatment with Pectinex® Ultra Tropical and Viscozyme® L significantly (p ≤ 0.05) increased RS content in apple pomace and the highest content was determined in sample hydrolyzed with Pectinex® Ultra Tropical for 5 h (50% higher compared to the cntrol). Enzymatic hydrolysis released not only RS but also organic acids and pH significantly decreased after treatment with enzymes containing pectinases. Gama et al. [31] also reported decreased pH and increased RS and galacturonic acid content after enzymatic treatment of apple pomace with combination of Viscozyme and Celluclast.
Enzymatic hydrolysis with Pectinex® Ultra Tropical and Viscozyme® L significantly (p ≤ 0.05) increased TPC up to 5.15 mg GAE/g, while treatment with Celluclast® 1.5 L did not have significant (p ≤ 0.05) impact on TPC in apple pomace. Duration of hydrolysis also did not have significant (p ≤ 0.05) impact on TPC content. Zheng et al. [16] reported Viscozyme® L as suitable enzyme for polyphenols extraction. The carbohydrate hydrolyzing enzymes (cellulase, hemicellulase, and pectinase) disrupt the cell wall structure, by releasing polyphenolic compounds which are entrapped within polysaccharide complexes [32]. Results indicated significant negative correlation between TPC and TDF content (Pearson correlation coefficient: -0.9471, p = 0.0000).
Obtained results suggest that apple pomace could be treated at the selected conditions with Celluclast® 1.5 L to increase SDF and with other enzymes to increase TPC.
In vitro Prebiotic Activity
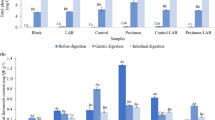
The effect of apple pomace water-soluble fraction (WSF) obtained after enzymatic hydrolysis for 1 h on probiotic bacteria growth are shown in Supplementary Fig. 1. L. acidophilus DSM 20079 and B. animalis DSM 20105 showed an ability to use different carbohydrate as their carbon sources, and the growth of probiotic bacteria was significantly (p ≤ 0.05) higher than in carbohydrate-free media. The growth of L. acidophilus DSM 20079 and B. animalis DSM 20105 after 24 h was significantly higher in media supplemented with all of tested apple pomace WSFs than in media supplemented with glucose or inulin. However, after 48 h the highest cell viability was in medium supplemented with glucose, but L. acidophilus DSM 20079 viability in media supplemented with all of the apple pomace WSFs were significant (p ≤ 0.05) higher than in medium supplemented with inulin. Comparing different apple pomace WSFs obtained after enzymatic hydrolysis no statistically significant difference (p ≤ 0.05) was observed for both probiotic bacteria growths. Enzymatically hydrolyzed apple pomace showed high content of RS (Table 1), good prebiotic effect in samples with higher sugar content were also reported in other studies [33]. Higher probiotic cell viability after 24 h in media supplemented with apple pomace WSF than in medium with glucose may be due to other soluble carbohydrates such as SDF and oligosaccharides. Pectin and pectic oligosaccharides obtained from apple pomace proliferates probiotic bacteria growth [34]. Various carbohydrates could be used as carbon source for L. acidophilus [35]. Bifidobacterium shows more specific preference for carbon source within the genus, but most of them may use a wide range of different carbohydrates [S1]. Sembries et al. [S2] reported that administration of juices extracted from apple pomace increases Lactobacillus and Bifidobacterium counts.
Technological Properties
WRC, SI and ORC are presented in Table 1. The hydration properties are affected by many different factors such as chemical structure, porosity, particle size, pH, ionic form, ionic strength, temperature [8]. The WRC of apple pomace was 16.20–16.83 g/g, Wang et al. [S3] reported similar WRC of apple pomace (16.3 ± 0.6 g/g). Enzymatic hydrolysis in most cases decreased WRC and increased SI, however treatment with Viscozyme® L for 1 h and with Celluclast® 1.5 L for 1 and 5 h did not have significant influence on WRC compared with control samples (p ≤ 0.05). The lowest WRC (9.88 ± 0.03 g/g) and the highest SI (43.80 ± 2.10%) were obtained using Pectinex® Ultra Tropical where pectinases are the main enzymes. Pectin can increase hydration properties in samples due to its hydroxyl groups which allow water associations through hydrogen bonding [S4]. Enzymatic hydrolysis changes technological properties of pomace due to the changes and destruction of cell wall material [28]. In this study the TDF content and WRC correlated significantly (Pearson correlation coefficient: +0.8318, p = 0.0001).
ORC show ability of fibers to retain oil after mixing, incubation with oil and centrifugation [8]. In most cases enzymatic treatment decreased ORC and varied from 2.15 to 5.78 g/g. Cerda-Tapia et al. [S5] reported lower ORC (1.69 ± 0.13 g/g) of apple pomace. Results demonstrated that longer hydrolysis duration had negative impact on ORC. The highest ORC was in control samples and pomace treated with Celluclast 1.5 L for 1 h. The lowest ORC was obtained after 5 h treatment with Pectinex® Ultra Tropical. This sample also have the lowest TDF content, TDF and ORC correlated significantly (Pearson correlation coefficient: +0.8561, p = 0.0000). ORC depends on the surface properties, overall charge density and to the hydrophilic nature of the components [8]. Enzymatic hydrolysis may change structure and decrease capillarity of DF which leads to decreased ORC [S6].
The effect of enzymatically hydrolyzed apple pomace on emulsion stability was determined (Supplementary Fig. 2). The static stability of the emulsion decreased during storage in all samples, however, in most cases the stability changes were insignificant (p ≤ 0.05) during 24 h (Supplementary Fig. 2a). Huc-Mathis et al. [S7] reported slight coalescence during first the 3 days storage of emulsion prepared with apple pomace, after which emulsion remained constant until 15 days. Emulsion with pomace hydrolyzed with Viscozyme® L for 1 h showed the highest (81.5%) static stability (Supplementary Fig. 2b). The highest thermal stability was obtained in emulsions with apple pomace treated with Pectinex® Ultra Tropical for 1 and 5 h (72.8 and 71.5%, respectively), which also had the highest SI. The lowest thermal stability was observed in emulsion with pomace hydrolyzed with Celluclast® 1.5 L for 5 h. Enzymatic hydrolysis of apple pomace could change particle size, by decreasing TDF and IDF to lower molecular weight components. Particle size has influence for emulsion stabilization, the smaller particles enhance emulsion stability [S8].
In most cases apple pomace treated enzymatically for 1 h showed better technological properties comparing with pomace treated for 5 h, therefore pomace treated for 1 h were chosen for further analysis.
FT-IR Analysis of Apple Pomace
FT-IR are used for understanding the structure and chemical composition of fibers [S9]. FT-IR spectra of enzymatically treated apple pomace for 1 h are shown in Supplementary Fig. 3. The strong and broad peak near 3400 cm− 1 characterizes the O-H or N-H groups in pomace. O–H stretching of hydrogen bound to the hydroxyl groups is originating mainly from cellulose and hemicellulose [S10]. This peak also is associated with inter- and intramolecular hydrogen bonding of the GalA backbone [S11]. The peak near 2900 cm− 1 is representative of C–H vibrations which typically includes C–H, C–H2 and C–H3 stretching vibrations [S11]. The peak at around 1744 cm− 1 corresponds to the carbonyl group (C = O). This peak is originating mainly from aldehyde [S12], but it also can indicate the presence of uronic acid [S13]. C–O stretching around 1400 cm− 1 is assigned to alcohols, ethers, and esters 1400 cm− 1 [S12]. The peak at near 1,630 cm− 1 corresponds to characteristic bending or stretching of aromatic hydrocarbons of lignin [S14]. In most cases, all non-hydrolyzed and hydrolyzed pomaces had identical spectra and new chemical groups were not produced, nevertheless, some decreases or shifts were observed at peaks around 1743, 1438, 1147, 617 cm− 1.
Wheat Bread Quality Characteristics
Wheat breads made with 5% of 1 h enzymatically treated (VAB, PAB, CeAB) and non-treated (CAB) apple pomace were compared to wheat bread made without the addition of apple pomace (CWB) (Supplementary Fig. 4) and the characteristics of bread quality were investigated (Table 2). The addition of apple pomace had impact on bread pH and TTA values. Bread supplementation with apple pomace significantly decreased (p ≤ 0.05) pH and increased TTA compared to the CWB. The lowest pH and highest TTA was determined in PAB and VAB. The TTA of PAB was 2.33 times higher than the TTA of CWB and 1.45 times higher than of CeAB. Other authors also reported increased acidification of wheat bread after addition of berry pomace [S15, S16]. The decrease of pH and increase of TTA may be due to acids present in apple pomace.
The addition of apple pomace caused a decrease in bread specific volume (from 3.75 to 2.32 cm3/g) and porosity (from 80.06 to 70.71%) compared to CWB (Table 2), except apple pomace hydrolyzed with Celluclast® 1.5 L which had statistically insignificant impact on bread porosity. The decrease of porosity and specific volume may be due to decreased content of gluten and increased content of DF. DF has a negative effect on bread volume, which is caused by lowered gas retention [S17]. Polysaccharides affect changes in the secondary structure of gluten proteins by changing conformation of disulphide bridges. Polysaccharides may partially dehydrate gluten network due to competitive water binding [S18], which can be counteracted by adjusting the water level in formulations [S15]. Low pH also may have influence for structural, thermal and rheological properties of gluten dough, gluten structure may become weaker [S19]. The lowest specific volume and porosity were observed in VAB and PAB, which had also the lowest pH. Comparing bread samples supplemented with apple pomace the highest specific volume was observed in CAB, CeAB, and the highest porosity in CeAB, while pomace enzymatically hydrolyzed with Viscozyme® L and Pectinex® Ultra Tropical significantly decreased specific volume and porosity.
The effect of enzymatically treated apple pomace on the wheat bread texture characteristics (hardness, springiness, gumminess, chewiness, and resilience) and their changes during storage are shown in Supplementary Fig. 5. Apple pomace non-treated and enzymatically treated with Celluclast® 1.5 L did not have significant (p ≤ 0.05) impact on wheat bread hardness (CWB – 36.7 ± 1.5 N; CAB – 26.9 ± 3.0 N; CeAB – 22.7 ± 1.1 N), however apple pomace enzymatically treated with Viscozyme® L and Pectinex® Ultra Tropical significantly increased (p ≤ 0.05) bread hardness. SDF can promote bread quality [S20], apple pomace treated with Celluclast® 1.5 L and non-treated apple pomace had higher amount of SDF compared with apple pomace treated with enzymes containing pectinases (Viscozyme® L and Pectinex® Ultra Tropical). The rheological properties of dough can be improved by adding pectin which enhance a dense network of gluten [S21]. Addition of apple pomace treated with Viscozyme® L and Pectinex® Ultra Tropical significantly (p ≤ 0.05) decreased cohesiveness and resilience, but increased gumminess and chewiness of fresh bread. While apple pomace non-treated and treated with Celluclast® 1.5 L did not have significant (p ≤ 0.05) influence for wheat bread gumminess and chewiness. During 4-day storage the hardness, gumminess and chewiness significantly (p ≤ 0.05) increased, and cohesiveness and resilience significantly (p ≤ 0.05) decreased in all of the same bread samples. Apple pomace did not have significant (p ≤ 0.05) influence on wheat bread springiness and its changes during 4-day storage.
The addition of enzymatically treated apple pomace slightly changed bread sensory attributes: increased acidity and decreased sweetness and porosity, however, in most cases, these differences were no statistically significant (p ≤ 0.05) (Supplementary Fig. 6).
The results suggested that wheat bread supplemented with apple pomace without enzymatically treatment and treated with Celluclast® 1.5 L in most cases did not have negative impact on bread texture characteristic comparing to wheat bread without apple pomace. While Viscozyme® L and Pectinex® Ultra Tropical had negative impact for wheat bread texture characteristic.
Conclusion
Apple pomace obtained after juice production contains 35.66 g/100 g of dry weight TDF (SDF/IDF ratio 0.4). Enzymatic hydrolysis with enzymes mostly used for pectin and TPC extraction changed SDF/IDF ratio and technological properties of apple pomace. Hydrolysis with Celluclast® 1.5 L increased content of SDF and SDF/IDF ratio in apple pomace. While apple pomace treatment with Viscozyme® L and Pectinex® Ultra Tropical resulted in decreased SDF/IDF ratio, but increased TPC. Enzymatically hydrolyzed apple pomace showed increased SI, RS content and, in most cases, decreased WRC and ORC. Hydrolyzed as well as non-hydrolyzed apple pomace water-soluble fractions promoted growth of Lactobacillus acidophilus DSM 20079 and Bifidobacterium animalis DSM 20105. FT-IR analysis showed that in most cases there were no changes in the composition of chemical groups comparing non-treated and enzymatically treated apple pomaces. Wheat bread supplemented with 5% of non-treated and enzymatically treated apple pomace had lower specific volume, porosity, pH and higher TTA compared to the wheat bread. Apple pomace enzymatically hydrolyzed with Celluclast® 1.5 L did not show negative influence for bread texture properties. The addition of apple pomace also did not have significant impact for wheat bread sensory properties. Obtained results suggested that apple pomace enzymatically treated with Celluclast® 1.5 L which increased SDF/IDF ratio could be used for wheat bread supplementation with dietary fiber.
Data Availability
All data generated or analyzed during this study are included in this published article and its supplementary information files.
References
Kruczek M, Drygas B, Habryka C (2016) Pomace in fruit industry and their contemporary potential application. World Sci News 48:259–265
Reis SF, Rai DK, Abu-Ghannam N (2014) Apple pomace as a potential ingredient for the development of new functional foods. Int J Food Sci Technol 49:1743–1750. https://doi.org/10.1111/IJFS.12477
Vendruscolo F, Albuquerque PM, Streit F et al (2008) Apple pomace: a versatile substrate for biotechnological applications. Crit Rev Biotechnol 28:1–12. https://doi.org/10.1080/07388550801913840
FDA authors (2008) Health claims: fruits, vegetables, and grain products that contain fiber, particularly soluble fiber, and risk of coronary heart disease. In: Code of Federal Regulations. https://www.ecfr.gov/current/title-21/chapter-I/subchapter-B/part-101/subpart-E/section-101.77. Accessed 28 May 2022
Schneeman BO (1987) Soluble vs insoluble fiber: different physiological responses. Food Technol 47:81–82
Masoodi FA, Chauhan GS (1998) Use of apple pomace as a source of dietary fiber in wheat bread. J Food Process Preserv 22:255–263. https://doi.org/10.1111/J.1745-4549.1998.TB00349.X
Gómez M, Martinez MM (2018) Fruit and vegetable by-products as novel ingredients to improve the nutritional quality of baked goods. Crit Rev Food Sci Nutr 58:2119–2135. https://doi.org/10.1080/10408398.2017.1305946
Elleuch M, Bedigian D, Roiseux O et al (2011) Dietary fibre and fibre-rich by-products of food processing: characterisation, technological functionality and commercial applications: a review. Food Chem 124:411–421. https://doi.org/10.1016/J.FOODCHEM.2010.06.077
Garcia-Amezquita LE, Tejada-Ortigoza V, Serna-Saldivar SO, Welti-Chanes J (2018) Dietary fiber concentrates from fruit and vegetable by-products: processing, modification, and application as functional ingredients. Food Bioprocess Technol 11:1439–1463. https://doi.org/10.1007/S11947-018-2117-2
de Camargo AC, Regitano-D’Arce MAB, Biasoto ACT, Shahidi F (2016) Enzyme-assisted extraction of phenolics from winemaking by-products: antioxidant potential and inhibition of alpha-glucosidase and lipase activities. Food Chem 212:395–402. https://doi.org/10.1016/J.FOODCHEM.2016.05.047
Borujeni NE, Karimi K, Denayer JFM, Kumar R (2022) Apple pomace biorefinery for ethanol, mycoprotein, and value-added biochemicals production by Mucor indicus. Energy 240:122469. https://doi.org/10.1016/J.ENERGY.2021.122469
Barreira JCM, Arraibi AA, Ferreira ICFR (2019) Bioactive and functional compounds in apple pomace from juice and cider manufacturing: potential use in dermal formulations. Trends Food Sci Technol 90:76–87. https://doi.org/10.1016/J.TIFS.2019.05.014
Dranca F, Oroian M (2019) Optimization of pectin enzymatic extraction from malus domestica ‘fălticeni’ apple pomace with celluclast 1.5L. Molecules 24:2158. https://doi.org/10.3390/MOLECULES24112158
Wikiera A, Mika M, Starzyńska-Janiszewska A, Stodolak B (2015) Application of Celluclast 1.5L in apple pectin extraction. Carbohydr Polym 134:251–257. https://doi.org/10.1016/J.CARBPOL.2015.07.051
Li X, He X, Lv Y, He Q (2014) Extraction and functional properties of water-soluble dietary fiber from apple pomace. J Food Process Eng 37:293–298. https://doi.org/10.1111/JFPE.12085
Zheng HZ, Hwang IW, Chung SK (2009) Enhancing polyphenol extraction from unripe apples by carbohydrate-hydrolyzing enzymes. J Zhejiang Univ Sci B 10:912–919. https://doi.org/10.1631/JZUS.B0920186
Martínez Sabajanes M, Yáñez R, Alonso JL, Parajó JC (2012) Pectic oligosaccharides production from orange peel waste by enzymatic hydrolysis. Int J Food Sci Technol 47:747–754. https://doi.org/10.1111/J.1365-2621.2011.02903.X
Gómez B, Yáñez R, Parajó JC, Alonso JL (2016) Production of pectin-derived oligosaccharides from lemon peels by extraction, enzymatic hydrolysis and membrane filtration. J Chem Technol Biotechol 91:234–247. https://doi.org/10.1002/JCTB.4569
Min B, Lim J, Ko S et al (2011) Environmentally friendly preparation of pectins from agricultural byproducts and their structural/rheological characterization. Bioresour Technol 102:3855–3860. https://doi.org/10.1016/J.BIORTECH.2010.12.019
Lim J, Yoo J, Ko S, Lee S (2012) Extraction and characterization of pectin from Yuza (Citrus junos) pomace: a comparison of conventional-chemical and combined physical–enzymatic extractions. Food Hydrocoll 29:160–165. https://doi.org/10.1016/J.FOODHYD.2012.02.018
Liu H, Wei X, Zu S et al (2021) Separation and identification of neutral oligosaccharides with prebiotic activities from apple pectin. Food Hydrocoll 121:107062. https://doi.org/10.1016/J.FOODHYD.2021.107062
Antonic B, Jancikova S, Dordevic D, Tremlova B (2020) Apple pomace as food fortification ingredient: a systematic review and meta-analysis. J Food Sci 85:2977–2985. https://doi.org/10.1111/1750-3841.15449
Alongi M, Melchior S, Anese M (2019) Reducing the glycemic index of short dough biscuits by using apple pomace as a functional ingredient. LWT-Food Sci Technol 100:300–305. https://doi.org/10.1016/J.LWT.2018.10.068
Figuerola F, Hurtado ML, Estévez AM et al (2005) Fibre concentrates from apple pomace and citrus peel as potential fibre sources for food enrichment. Food Chem 91:395–401. https://doi.org/10.1016/J.FOODCHEM.2004.04.036
Paganini C, Nogueira A, Silva NC, Wosiacki G (2005) Utilization of apple pomace for ethanol production and food fiber obtainment. Ciência e Agrotecnologia 29:1231–1238. https://doi.org/10.1590/S1413-70542005000600018
Diñeiro García Y, Valles BS, Picinelli Lobo A (2009) Phenolic and antioxidant composition of by-products from the cider industry: Apple pomace. Food Chem 117:731–738. https://doi.org/10.1016/J.FOODCHEM.2009.04.049
Toledo NMV, Mondoni J, Harada-Padermo SS et al (2019) Characterization of apple, pineapple, and melon by-products and their application in cookie formulations as an alternative to enhance the antioxidant capacity. J Food Process Preserv 43:e14100. https://doi.org/10.1111/JFPP.14100
Dongowski G, Sembries S (2001) Effects of commercial pectolytic and cellulolytic enzyme preparations on the apple cell wall. https://doi.org/10.1021/JF001410
Sabater C, Corzo N, Olano A, Montilla A (2018) Enzymatic extraction of pectin from artichoke (Cynara scolymus L.) by-products using Celluclast®1.5L. Carbohydr Polym 190:43–49. https://doi.org/10.1016/J.CARBPOL.2018.02.055
Yoon KY, Cha M, Shin SR, Kim KS (2005) Enzymatic production of a soluble-fibre hydrolyzate from carrot pomace and its sugar composition. Food Chem 92:151–157. https://doi.org/10.1016/J.FOODCHEM.2004.07.014
Gama R, van Dyk JS, Pletschke BI (2015) Optimisation of enzymatic hydrolysis of apple pomace for production of biofuel and biorefinery chemicals using commercial enzymes. 3 Biotech 5:1075–1087. https://doi.org/10.1007/s13205-015-0312-7
Nadar SS, Rao P, Rathod VK (2018) Enzyme assisted extraction of biomolecules as an approach to novel extraction technology: a review. Food Res Int 108:309–330. https://doi.org/10.1016/J.FOODRES.2018.03.006
Huang F, Liu H, Zhang R et al (2019) Physicochemical properties and prebiotic activities of polysaccharides from longan pulp based on different extraction techniques. Carbohydr Polym 206:344–351. https://doi.org/10.1016/J.CARBPOL.2018.11.012
Calvete-Torre I, Sabater C, Antón MJ et al (2022) Prebiotic potential of apple pomace and pectins from different apple varieties: modulatory effects on key target commensal microbial populations. Food Hydrocoll 133:107958. https://doi.org/10.1016/J.FOODHYD.2022.107958
Altermann E, Russell WM, Azcarate-Peril MA et al (2005) Complete genome sequence of the probiotic lactic acid bacterium Lactobacillus acidophilus NCFM. Proc Natl Acad Sci USA 102:3906–3912. https://doi.org/10.1073/pnas.0409188102
Acknowledgements
The authors acknowledge Novozymes A/S, Denmark, for kindly providing the enzymes required for this research.
Funding
No funds, grants, or other support was received.
Author information
Authors and Affiliations
Contributions
Jolita Jagelaviciute: Conceptualization, methodology, investigation, original draft preparation, review and editing; Guste Staniulyte: Investigation; Dalia Cizeikiene: methodology, review and editing; Loreta Basinskiene: Supervision, review and editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
Not applicable.
Conflict of Interest
The authors declare no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jagelaviciute, J., Staniulyte, G., Cizeikiene, D. et al. Influence of Enzymatic Hydrolysis on Composition and Technological Properties of Apple Pomace and Its Application for Wheat Bread Making. Plant Foods Hum Nutr 78, 307–313 (2023). https://doi.org/10.1007/s11130-023-01054-w
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11130-023-01054-w