Abstract
Background and aims
Plants have evolved an array of root traits associated with phosphorus (P) acquisition, including morphological and physiological traits. This study aimed to characterize the differences of various root traits in soybean (Glycine max) and explore their roles in P acquisition.
Methods
Root functional traits associated with P acquisition were characterized in 49 cultivated soybean landraces from the North China Plain grown in a glasshouse under low-P condition.
Results
We found a large variation in plant growth and all studied root traits. There was a significant correlation between total plant P content and root morphological traits in all 49 varieties. Hierarchical classification on principal components (HCPC) based on principal component analysis (PCA) indicated that soybean varieties could be grouped into three distinct clusters: total plant P content was positively correlated with some root morphological traits and seed P content in cluster 1 and showed positive correlations with root tissue density and rhizosheath carboxylates in cluster 2, while total P content showed no significant correlation with any root trait in cluster 3.
Conclusion
Root morphology and seed P content generally determined P acquisition of the present soybean varieties, but carboxylates also contributed to P uptake in some P-efficient varieties. We may be able to maximize soybean P acquisition via stacking root morphological and physiological traits, thus allowing plants to access different soil P pools.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Phosphorus (P) is an essential macronutrient for plant growth, and P fertilizers are used in intensive agriculture to achieve high crop yields. However, P is readily sorbed to aluminum (Al) and iron (Fe) (hydr)oxides in acidic soils, or precipitates as calcium (Ca)-P in calcareous soils, which results in 70–90% of P fertilizers becoming unavailable to most crop plants (Hinsinger 2001). This not only causes loss of finite phosphate rock reserves but also threatens environmental quality with the overused P fertilizers running off into lakes and causing eutrophication (Plaxton and Lambers 2015). Therefore, P-efficient crops are urgently needed to reduce the over-reliance of agriculture on unsustainable P fertilizers and to maximize agronomic benefits and long-term global food security in a sustainable manner (Cong et al. 2020).
Plants have evolved strategies to adapt to P-limiting conditions (Cong et al. 2020; Ding et al. 2021; Lynch 2011). They can enhance their internal P-utilisation efficiency by lowering P requirement (functioning at low levels of ribosomal RNA (rRNA)), optimizing P allocation (preferentially allocating P to photosynthetic cells, efficiently translocating P from senescing organs to growing organs), or replacing phospholipids by sulfolipids and galactolipids (Hayes et al. 2018; Lambers et al. 2012; Sulpice et al. 2014; Veneklaas et al. 2012). They can increase P acquisition by adjusting their root architecture or morphology, for example, by forming thinner, longer, shallower roots and more adventitious roots to increase their P-foraging capacity under a low P availability (Cong et al. 2020; Ding et al. 2021; Lynch 2011). They may also enhance P acquisition via root physiological traits, for example, the release of acid phosphatases or exudation of carboxylates (Clarholm et al. 2015; Ding 2022; Neumann et al. 2000). Root exudation of carboxylates is notoriously hard to measure in the field, but leaf manganese concentration ([Mn]) is an easily measurable proxy for carboxylate release in native species in Western Australia and eastern Brazil and in chickpea (Cicer arietinum) genotypes (Lambers et al. 2015; Pang et al. 2018; Zhou et al. 2022). Therefore, it is necessary to explore the correlation between leaf [Mn] and carboxylate release in other crop species including soybean. In addition, plants may interact with microbes, especially symbiotic mycorrhizal fungi, to increase P uptake (Kafle et al. 2019). These strategies allow plants to access different pools of soil P in contrasting ways.
All P-acquisition strategies incur a significant carbon cost, and plants tend to rely mainly on one or very few P-acquisition traits (Lynch et al. 2005). Species on severely P-impoverished sites, like kwongan, fynbos and campos rupestres, generally rely on root exudates (e.g., carboxylates, phosphatases) to acquire P, and these species tend to have thin roots (Lambers et al. 2022). Root morphology of maize (Zea mays), rather than root physiology, tends to respond strongly to low soil P availability (Wen et al. 2017). On the other hand, in faba bean (Vicia faba), physiological traits exhibit a stronger response to P deficiency than morphological traits (Zhang et al. 2016). There might be a trade-off between different P-acquisition strategies at crop species or varieties levels: species with thinner roots mainly rely on root morphological and physiological traits to access P, whereas species with thicker roots exhibit greater colonization by arbuscular mycorrhizal fungi (AMF) or carboxylate and phosphatase release in the rhizosheath under P deficiency (Honvault et al. 2021; Wen et al. 2019). Chickpea varieties with greater carboxylate release on the basis of root dry weight also have thinner roots and higher AMF colonisation, which is different from the findings at the crop species level (Wen et al. 2020). However, we do not know if these trade-offs of P-acquisition strategies at the species or varieties levels also exist in soybean varieties.
Soybean (Glycine max (L.) Merr) is a grain legume that is a major source of protein and vegetable oil for human consumption and a high-quality animal feed (Zhao et al. 2004). The demand for soybean has increased globally, but its growth and production are often limited by a low P availability (Wang et al. 2010). The development of P-efficient soybean varieties is considered the most sustainable and economical approach to soybean production under low-P conditions (Wang et al. 2010). Phosphorus-acquisition-efficient soybean varieties have been identified with specific root morphological or architectural traits (Pan et al. 2008; Zhao et al. 2004; Zhou et al. 2016). Based on a GIS-assisted approach, Zhao et al. (2004) selected an “applied core collection” of soybean germplasm in China and showed that roots with shallow architecture enable soybean to absorb more P. An assessment on P-efficiency related characteristics of 96 soybean genotypes in Northeast China showed that P efficiency is positively correlated with dry weight of shoots and roots, root:shoot ratio, root length and root surface area (Pan et al. 2008). Not only root morphological traits, but also carboxylate exudation and rhizosheath acid phosphatase activities contribute to soybean P efficiency (Tantriani et al. 2023; Zhou et al. 2016). Root-hair development (length and density) is also related to P-uptake efficiency of soybean genotypes from USA, Uganda and Zimbabwe (Vandamme et al. 2013). In addition, root-hair length is highly correlated with rhizosheath (soil adhering to roots) size in wheat, suggesting the possibility of using it as a surrogate of root hairs in exploring P-acquisition efficiency (Delhaize et al. 2012; James et al. 2016).
The North China Plain is one of the major soybean-production areas in China (Wu et al. 2006; Xin and Tao 2020). However, little information is available about P-efficient soybean varieties in this region. The relative contribution of root morphological or physiological traits to P acquisition is also unknown. Therefore, we selected 49 landraces cultivated on the North China Plain from a mini core collection of 256 soybean landraces representing 70% of the entire soybean germplasm resource in China (Song et al. 2010; Wang et al. 2006). Root functional (morphological and physiological) traits associated with P acquisition and [Mn] in mature leaves of these varieties, as a proxy for rhizosphere carboxylates (Lambers et al. 2015; Pang et al. 2018) were assessed under low-P conditions. We aimed to 1) quantify the genotypic variation of a range of root traits; 2) characterize the correlations among root traits involved in P acquisition to explore trade-offs and the main P-acquisition strategies; 3) test if leaf [Mn] can be used as a proxy for rhizosheath carboxylates in soybean.
Materials and methods
Plant material
A mini-core collection of 256 soybean landraces was established based on morphological and simple sequence repeat (SSR) markers (Song et al. 2010; Wang et al. 2006). This number of varieties represents more than 70% of the genetic diversity of the entire germplasm resource, making it effective to use this core collection to explore traits or genes in the entire Chinese soybean germplasm collection, including the search for P-efficient varieties. In this study, 49 landraces from the North China Plain from the core collection were selected (Table S1).
Growth conditions
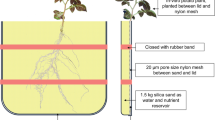
The study was carried out in a glasshouse at China Agricultural University, Beijing, with a day temperature of 25–30℃ and a night temperature of 18–22℃. A field soil was collected from the upper 20 cm of an experimental station at Quzhou, Hebei. River sand was washed, and then both river sand and field soil were air-dried and sieved (2-mm mesh size) to remove coarse fragments and microarthropods. After that, field soil and river sand were sterilized by gamma irradiation (> 25 K Gray). To obtain a low soil P concentration, sterilized river sand and field soil were mixed in a ratio of 3:7 (w/w) and each pot (85 cm × 85 mm × 180 mm) was filled with a 1.2 kg soil mixture. Basal nutrients were not added as we did not expect any nutrient deficiency except P; the field soil used in this study was fertilized in an intensive agriculture system. The soil mixture contained 12, 4.7, 101 and 3.5 µg g−1 inorganic nitrogen (N), ‘plant-available’ P (i.e. Olsen P), available potassium (K) and available manganese (Mn), respectively, and had a pH of 8.3 (1:2.5, soil: water). Soil mineral nitrogen (N) concentration was determined using an automated discrete analyser (SmartChem 450, AMS Alliance, Rome, Italy) after extracting fresh soil with 0.01 M CaCl2 (1:20, soil:water) by shaking for 1 h at 25 °C. Available soil P was determined according to the Olsen-P method, based on the extraction of air dried soil with 0.5 M NaHCO3 at pH 8.5 (180 rpm, 25 °C) (Olsen 1954). Plant-available soil K was determined by a flame photometer (FP6410, INESA, Shanghai, China) after extracting with 1 M NH4OAc by shaking for 30 min at 25 °C (Bao 2000). Available Mn was measured by an atomic absorption spectrophotometer (TAS-990 F, Beijing Purkinje General Instrument Co. Ltd., Beijing, China), after extracting air dried soil with DTPA (0.005 M diethylenetriaminepentaacetic acid, 0.1 M triethanolamine, and 0.01 M CaCl2) at pH 7.3 by shaking for 2 h at 25 °C (Lindsay and Norvell 1978). Exogenous KH2PO4 was mixed with the soil mixture to achieve a ‘plant-available’ P concentration of 10 µg g−1 before filling the pots. The organic P concentration of the field soil was 69.6 µg g−1. The pot experiment followed a complete randomized design with soybean genotype as the main factor. Each genotype was replicated in four pots. Four seeds were directly sown at 20 mm depth and inoculated with soybean rhizobium (Ruichu Biotechnology Co., Ltd, Jiangsu, China). Seedlings were thinned to one plant per pot one week after sowing. Each pot was watered with deionized water to 80% pot capacity by weighing every other day.
Plant harvest and measurement
Plants were harvested 45 days after sowing. Shoots were separated from the roots; mature (fully-expanded) leaves on the main stem were removed from the stem. All plant parts were weighed after drying at 70℃ for 72 h. Root traits associated with P acquisition were analyzed: (a) biomass allocation (root shoot ratio); (b) root morphological traits (total root length, root surface area, mean root diameter, and root tissue density); (3) root physiological traits (the total amount of carboxylates recovered from the rhizosheath soil per plant, the amount of carboxylates in the rhizosheath relative to root dry weight, and acid phosphatase activity).
Root physiological traits
At harvest, each pot was squeezed gently to allow dislodgement of the soil column and loosening of soil around the roots. The entire root system was removed from the soil and shaken gently to remove excess bulk soil; the adhering soil and sand around the roots was defined as rhizosheath soil (Pang et al. 2017). The entire root system was then transferred into a decomposable cup containing 100 mL of 0.2 mM CaCl2 and gently dunked into a solution to remove as much of the rhizosheath soil as possible. Fifteen mL of rhizosheath soil solution was taken and filtered through a 0.22 mm syringe filter. A 1-mL subsample was transferred into a high performance liquid chromatography vial and frozen at 20℃ until analysis of carboxylates according to Shen et al. (2003).
Acid phosphatase in the rhizosheath soil rather than alkaline phosphatase was measured since soybeans can release carboxylates and acidify the rhizosphere. For the determination of acid phosphatase activity, two 0.5 mL subsamples of the filtered rhizosheath extract were transferred into 2-mL centrifuge tubes and measured as described by Zhang et al. (2016). Briefly, 0.4 mL sodium acetate buffer (pH 5.2) and 0.1 mL 0.15 M p-nitrophenyl phosphate (PNP) substrate were added to the 2-mL centrifuge tube and incubated for 30 min at 25–30°C, and 0.5 mL 0.5 M NaOH was added to terminate the reaction. The absorption of supernatants was then measured at 405 nm. After extraction of acid phosphatase and carboxylates from the rhizosheath solution, the rhizosheath soil was oven-dried at 105℃ for 72 h, and the dry weight was recorded as rhizosheath soil dry weight.
Root morphological traits
After collection of rhizosheath exudates, roots were washed free of remaining soil, spread out on a transparent plastic tray, and root images were obtained at a resolution of 300 dpi using an Epson scanner (Epson Expression 1600 pro, Model EU-35, Japan). Root images were analyzed for total root length, root surface area, mean root diameter and root volume with WinRHIZO software (Pro 2009b, Regent Instruments Inc., Quebec City, Canada). After scanning, roots were oven-dried at 70℃ for 72 h until constant weight to measure biomass. Root tissue density was calculated as the ratio of root dry weight to root volume.
Nutrient analyses
All dry plant parts (mature leaves, stems, and roots) were ground into a fine powder using a Teste universal grinder (FW100, Tianjin, China). About 100 mg (mature leaves, stems, and roots) of ground material was digested with HNO3 and H2O2 (3:1) and nutrients were measured by inductively coupled plasma optical emission spectrometry (OPTIMA 3300 DW; Perkin-Elmer, Norwalk, CT, USA).
Calculation of physiological P-use efficiency
Physiological P-use efficiency was calculated as described by Pang et al. (2010). Briefly, physiological P-use efficiency = DW / whole plant P concentration.
Statistics
All statistical analyses were performed using the R software platform (R Core Team 2019). The coefficient of variation (CV) of each plant trait was calculated as the ratio of the standard deviation to the mean. Principal component analysis (PCA) and hierarchical classification on principal components (HCPC) were performed with functions “PCA” and “HCPC” from the FactoMineR package (Honvault et al. 2021). Correlations between traits were calculated with Pearson (data meeting normal distribution) or Spearman (data not meeting normal distribution) tests via the “cor.test” function in R. All figures were plotted also using the R software platform (R Core Team 2019).
Results
Plant growth, root shoot ratio, root morphology, and rhizosheath soil dry weight
We found a large variation among 49 soybean varieties for plant growth, root shoot ratio, and all investigated root morphological traits including total root length and root surface area (Fig. 1). Shoot dry weight was in the range of 0.89–1.74 g (CV = 15.5%, Fig. 1a), root dry weight 0.21–0.44 g (CV = 15.7%, Fig. 1b), total root length 19.8–46.0 m (CV = 19.1%, Fig. 1c), root surface area 221.9–474.1 cm2 (CV = 17. 9%, Fig. 1d), and root shoot ratio 0.18–0.35 (CV = 16.6%, Fig. 1e). The variation for mean root diameter and root tissue density was small, with mean root diameter ranging from 0.31 to 0.37 mm (CV = 4.8%, Fig. 1f), and root tissue density ranging from 0.10 to 0.14 g mm−3 (CV = 8.6%, Fig. 1g). Rhizosheath (rhizosheath mass per unit root length) size varied greatly, with a 2.7-fold difference among the 49 varieties, ranging from 0.14 to 0.38 g m−1 (CV = 21.6%, Fig. 1h).
Boxplots showing (a) shoot dry weight, (b) root dry weight, (c) total root length, (d) root surface area, (e) mean root diameter, (f) root shoot ratio, (g) root tissue density, and (h) rhizosheath size of 49 soybean varieties grown for 45 days in a mixture of sterilized washed river sand and field soil with a low phosphorus (P) availability (10 µg g−1 soil). The central horizontal bar in each box shows the median, the box represents the interquartile range, the whiskers show the location of the most extreme data points that are still within a range of 1.5 of the upper or lower quartiles, and the blue symbols are mean values for each genotype
Carboxylates, acid phosphatases, and mature leaf Mn concentration
We found a 3.4-fold difference in the total amount of carboxylates per plant recovered in the rhizosheath soil among the 49 varieties, ranging from 20.8 to 70.5 µmol per plant (CV = 27.4%, Fig. 2a), and a 4.1-fold difference in the amount of malonate (the major carboxylate) per plant recovered in the rhizosheath soil among the 49 varieties, ranging from 13.9 to 57 µmol per plant (CV = 31.9%, Fig. 2b). Likewise, the total amount of carboxylates and malonate relative to root DW varied greatly, with a 2.9-fold difference for total carboxylates (from 73.7 to 21.4 µmol g−1 root DW, CV = 22.8%, Fig. 2c), and a 3.1-fold difference for malonate (from 53.1 to 162 µmol g−1 root DW, CV = 26.6%, Fig. 2d) among 49 varieties. Acid phosphatase activity in the rhizosheath varied greatly, with a 5.2-fold difference among 49 soybean varieties, ranging from 1.6 to 8.4 nkat g−1 soil (CV = 33.5%, Fig. 2e). The [Mn] in mature leaves also varied greatly, with a 2.5-fold difference, ranging from 44.6 to 113.5 mg kg−1 (CV = 24.1%, Fig. 2f).
Boxplots showing (a) total carboxylates per plant recovered in the rhizosheath soil, (b) malonate per plant recovered in the rhizosheath soil, (c) the total amount of carboxylates relative to root dry weight (DW), (d) the amount of malonate relative to root DW, (e) acid phosphatase activity, (f) manganese (Mn) concentration in mature leaves of 49 soybean varieties grown for 45 days in a mixture of sterilized washed river sand and field soil with a low phosphorus (P) availability (10 µg g−1 soil). The central horizontal bar in each box shows the median, the box represents the interquartile range, the whiskers show the location of the most extreme data points that are still within a range of 1.5 of the upper or lower quartiles, and the blue symbols are mean values for each genotype
Boxplots showing (a) total plant phosphorus (P) content, (b) total plant P concentration, (c) seed P content and (d) physiological P-use efficiency of 49 soybean varieties grown for 45 days in a mixture of sterilized washed river sand and field soil with a low P availability (10 µg g−1). The central horizontal bar in each box shows the median, the box represents the interquartile range, the whiskers show the location of the most extreme data points that are still within a range of 1.5 of the upper or lower quartiles, and the blue symbols are mean values for each genotype
Total plant P content and concentration, seed P content, and physiological P-use efficiency
Total plant P content varied two-fold among 49 varieties, ranging from 0.97 to 1.96 mg per plant for total P content (CV = 16.4%, Fig. 3a), while total plant P concentration varied only slightly, ranging from 0.74 to 0.96 mg g−1 plant DW (CV = 6.5%, Fig. 3b). There was significant variation in seed P content, ranging from 0.74 to 0.96 mg per seed (CV = 29.6%, Fig. 3c), and the variation for physiological P-use efficiency was large, ranging from 1325 to 2594 g2 DW g−1 P (CV = 14.6%, Fig. 3d).
Correlations among traits of all varieties and within clusters
Principal component analysis based on 10 plant traits of 49 soybean varieties explained 62% of the variation in the first two components (Fig. 4a). The first component (PC1) represented 37.9% of the variation and was dominated by root-related traits. The second component (PC2) represented 24.1% variation and accounted primarily for total plant P content, total plant P concentration, and shoot dry weight (Fig. 4a). Among all 49 soybean varieties, total plant P content showed a significant positive correlation with seed P content (r = 0.37, P < 0.01), root surface area (r = 0.36, P < 0.01), root dry weight (r = 0.53, P < 0.001), and shoot dry weight (r = 0.9, P < 0.001). The [Mn] in mature leaves showed a significant positive correlation with total root length (r = 0.66, P < 0.001), root surface area (r = 0.64, P < 0.001), root dry weight (r = 0.54, P < 0.001), the total amount of carboxylates per plant recovered in the rhizosheath soil (r = 0.37, P < 0.01), the amount of malonate per plant recovered in the rhizosheath soil (r = 0.39, P < 0.001), and the amount of malonate in the rhizosheath relative to root dry weight (r = 0.3, P < 0.5) (Table 1).
We divided soybean varieties into three groups via hierarchical classification on principal components (HCPC) (Fig. 4b). The first group mainly comprised varieties with relatively low total plant P content, small root systems (total root length and root surface area), smaller amount of carboxylates released, but a larger amount of acid phosphatase released (Table 2). Their total plant P content showed a significant positive correlation with seed P content (r = 0.48, P < 0.01), root surface area (r = 0.48, P < 0.5), root dry weight (r = 0.6, P < 0.01), mean root diameter (r = 0.46, P < 0.5), and shoot dry weight (r = 0.88, P < 0.001). The [Mn] in mature leaves showed a significant positive correlation with total root length (r = 0.54, P < 0.5) (Table 3). The second group mainly comprised varieties with the highest total plant P content, relatively larger root systems (total root length and root surface area), a larger amount of carboxylates released, but a smaller amount of acid phosphatase released (Table 2). Their total plant P content showed a significant positive correlation with root tissue density (r = 0.48, P < 0.5), the total amount of carboxylates per plant recovered in the rhizosheath soil (r = 0.52, P < 0.5), and shoot dry weight (r = 0.65, P < 0.01). The [Mn] in mature leaves showed a significant positive correlation with total root length (r = 0.46, P < 0.5) and root dry weight (r = 0.5, P < 0.5) (Table 4). The third group mainly included varieties with a relatively lower total plant P content, the largest root system (total root length and root surface area), the largest amount of carboxylates released but a smaller amount of acid phosphatase released (Table 2); its total plant P content showed a significant positive correlation with shoot dry weight (r = 0.9, P < 0.001) (Table 5).
Varieties with high P efficiency
All five varieties within the top 10% of shoot dry weight belonged to cluster 2. One variety (ZDD01612) also belonged to the top 10% of root dry weight, shoot P content, and physiological P-use efficiency. Two varieties (ZDD01612 and ZDD02866) belonged to the top 10% in terms of physiological P-use efficiency. Four varieties (ZDD03570, ZDD01612, ZDD02866, and ZDD10100) belonged to the top 10% of shoot P content (Table 6).
Discussion
Unlike N, which readily moves to the root surface by mass flow, P moves mainly by diffusion, along a gradient as a result by root P uptake (Plaxton and Lambers 2015). Thus, root traits are essential for P acquisition and plant growth. In our study, when pooling all 49 soybean varieties, total plant P content only showed a significant positive correlation with seed P content (r = 0.37, P < 0.01), root surface area (r = 0.36, P < 0.01), root dry weight (r = 0.53, P < 0.001), and shoot dry weight (r = 0.9, P < 0.001). This suggests that seed P content and root morphological traits determined the P acquisition of soybean varieties from the North China Plain. However, if we divide these varieties into three clusters according to hierarchical classification on principal components (HCPC), total plant P content in cluster 2 not only showed a significant positive correlation with root tissue density (r = 0.48, P < 0.5) and shoot dry weight (r = 0.65, P < 0.01), but also with the total amount of carboxylates per plant recovered in the rhizosheath soil (r = 0.52, P < 0.5). In addition, all P-efficient varieties (indicated by shoot dry weight) belonged to this cluster, and this cluster of varieties had the highest mean total plant P content, suggesting that both root morphological and physiological traits (rhizosheath carboxylates) enhanced P acquisition under P-limited conditions. This agrees with the finding that roots of P-efficient soybean varieties release greater quantities of carboxylates and enhance P availability in the rhizosphere (Krishnapriya and Pandey 2016; Tantriani et al. 2023; Zhou et al. 2016). Phosphorus-efficient varieties exuded oxalate at a much faster rate than P-inefficient varieties under P starvation, implying that oxalate is probably involved in P efficiency in soybean (Dong et al. 2004). The measurement of oxalate in our study was not reliable, and hence we cannot explore the contribution of oxalate in P acquisition. There are also studies showing that P deficiency failed to induce exudation of any carboxylate (Nian et al. 2003; Yang et al. 2000), probably because the varieties used in those studies mainly relied on root morphology or seed P content to acquire P, like the varieties belonging to cluster 1 in the present study. Root-hair development (length and density) is related to P-uptake efficiency in wheat grown on acid soils, and rhizosheath size was used as a good surrogate (James et al. 2016; Vandamme et al. 2013; Wang et al. 2004). However, we did not find any correlation between root hairs (represented by rhizosheath size) and total P content, probably because rhizosheath size is not a good surrogate of root hairs in soybean and chickpea (Pang et al. 2018). Future studies are warranted to study the correlation between root-hair length and density and rhizosheath size in soybean. Our study confirms the possibility of breeding P-efficient soybean varieties by genetic improvement of various root morphological and physiological traits. Physiological P-use efficiency was always highly correlated with shoot dry weight which confirms that shoot dry weight can be used as proxy for the selection of varieties with high P efficiency (Table 6).
Seed P is vital for plant germination and initial root growth (Wang et al. 2021; White and Veneklaas 2012). When pooling all 49 soybean varieties, seed P content was positively correlated with mean root diameter and root dry biomass (Table 1). This suggests that seed P content may enhance P uptake by promoting root traits, offering the opportunity to improve soybean P uptake. However, when we investigated the three clusters we recognised, only seed P content was positively correlated with mean root diameter and root dry biomass in cluster 1, and total P uptake in cluster 1 was not as efficient as that in cluster 2 (Table 2). High seed P content may also constrain the release of carboxylates and acid phosphatase (Table 1). A high seed P concentration implies a high phytate content, as most seed P exists in the form of phytate, which lowers the bioavailability of micronutrients (zinc, iron) in food and feed (Raboy 2001). However, we found that seed P concentration was negatively correlated with seed P content. Therefore, we also suggest to use high seed P content as proxy for the selection of varieties with high P efficiency. What should be borne in mind is that this study was only conducted at the seedling stage and the conclusion is based on that. Beyond our expectation, our soybean landraces experienced not only P deficiency, but possibly also some K and Zn deficiency (Adams et al. 2000; Brown and Jones 1977; Kirkby 2023; Makarim and Cox 1983; Ohki 1976; 1977) (Fig. S1). However, P was the most limiting nutrient for soybean growth in this study, so the possible K and Zn deficiencies are unlikely to affect the genotypic ranking of P-efficiency traits according to the Liebig-Sprengel "Law of the Minimum" (Jungk 2009). However, we suggest to add basal nutrients, except P, for future screening of P-efficient species/genotypes to avoid possible deficiencies of nutrients other than P.
Genotypic differences in root traits are important determinants of plant nutrient-use efficiency (Marschner 1998). In this study, we found major genotypic variation in root morphological traits (except mean root diameter) and especially root physiological traits of soybean varieties from the North China Plain. This is consistent with earlier studies; for example, Zhao et al. (2004) found a significant difference in root architecture, growth, P content, and P efficiency (indicated by seed yield) among 308 representative soybean varieties from South China, and showed that root architecture (the structure and organization of a plant's root system) was closely correlated with P efficiency. In a glasshouse study, Pan et al. (2008) found substantial genotypic variation in plant growth, root shoot ratio, root length, root surface area, and P efficiency of 96 soybean varieties from Northwest China, and that P efficiency was positively correlated with these root morphological parameters. Substantial genotypic variation in root-hair development has also been shown for soybean (Vandamme et al. 2013; Wang et al. 2004). A substantial genotypic variation in P efficiency was also detected for 274 soybean varieties from southwest China. A faster rate of carboxylate exudation and acid phosphatase activity was also observed in P-efficient soybean varieties under low-P conditions when compared with P-inefficient varieties (Zhou et al. 2016). Together with our studies, all P-efficient soybean from different regions of China or other countries exhibit substantial genotypic variations in morphological or physiological traits. All these results suggest the potential of breeding varieties that are more P efficient with various P-acquisition strategies.
Leaf [Mn] is a proxy for carboxylate-releasing P-acquisition strategies in P-impoverished conditions, including some Australian native species (Lambers et al. 2015, 2021; Zhou et al. 2022) and crop species, e.g., chickpea (Pang et al. 2018). However, our results do not support the use of leaf [Mn] as a proxy for rhizosheath carboxylates in soybean. This is probably because most soybean varieties in our study predominantly relied on root morphology to acquire P and Mn, with a significant correlation between leaf [Mn] and root morphological traits, such as total root length and root surface area (Tables 3, 5, and 6). The lack of a correlation between leaf [Mn] and rhizosheath carboxylates in soybean in the present study should not prevent further exploration of the use of leaf [Mn] as a proxy for rhizosphere carboxylates in other soybean varieties and other species, particularly crop species.
There is increasing evidence of trade-offs among root functional traits in P acquisition at the species or genotype level (Honvault et al. 2021; Weemstra et al. 2016; Wen et al. 2019, 2020). At crop species levels, thick-rooted species exhibit greater carboxylate release and phosphatase activity in the rhizosheath, while thin-rooted species show a greater response in root morphological traits under low P supply (Honvault et al. 2021; Wen et al. 2019). Therefore, root diameter is considered a good predictor of the relative expression of different root traits in crops (Honvault et al. 2021; Wen et al. 2019). In the present study, the variation in root diameter was very small, and this agrees with another study that intra-species variation for average root diameter is small for soybean (Vandamme et al. 2013). In addition, no coordination or trade-offs among root diameter and other root traits were observed. Furthermore, we found no trade-offs among different P-acquisition strategies in this study, either considering all varieties together or considering different clusters. This suggests that trade-offs among root functional traits in P acquisition may not exist in soybean varieties tested in present study, but this warrants further studies, because such trade-offs do exist among chickpea varieties (Wen et al. 2020). Root architecture, root hairs, and AMF colonization are also involved in P acquisition in soybean (Wang et al. 2004, 2011, 2010; Zhao et al. 2004), but these were not measured in the present study and the use of rhizosheath size as a surrogate for root hairs may not be suitable. Therefore, these parameters should be included in future studies. A combined effect of root tissue density and the total amount of carboxylates recovered in the rhizosheath on P acquisition was found in cluster 2, suggesting the potential for maximising soybean P acquisition via stacking strategies (morphological and physiological traits) thus accessing different P pools in soils.
Conclusions
The present study shows large variation for most root traits of soybean varieties from the North China Plain. In general, seed P content and root morphology was the key determinant of soybean P acquisition. However, the finding that all five varieties within the top 10% of shoot dry weight belonged to cluster 2, demonstrates the possibility of using the soybean reference set in breeding programs to improve both root morphological and physiological traits to enhance P acquisition. Furthermore, we observed no trade-off among root functional traits in the studied soybean varieties. In contrast, there was a complementary correlation between root tissue density and total carboxylate amount recovered in the rhizosheath in soybean P acquisition.
Data availability
This is not applicable to this manuscript.
Code availability
This is not applicable to this manuscript.
References
Adams ML, Norvell WA, Philpot WD, Peverly JH (2000) Spectral detection of micronutrient deficiency in ‘Bragg’soybean. Agron J 92:261–268
Bao SD (2000) Soil Agrochemical Analysis (in Chinese). China Agricultural Press, Beijing
Brown JC, Jones WE (1977) Fitting plants nutritionally to soils. I. soybeans 1. Agron J 69:399–404
Clarholm M, Skyllberg U, Rosling A (2015) Organic acid induced release of nutrients from metal-stabilized soil organic matter – the unbutton model. Soil Biol Biochem 84:168–176
Cong W-F, Suriyagoda LDB, Lambers H (2020) Tightening the phosphorus cycle through phosphorus-efficient crop genotypes. Trends Plant Sci 25:967–975
Delhaize E, James RA, Ryan PR (2012) Aluminium tolerance of root hairs underlies genotypic differences in rhizosheath size of wheat (Triticum aestivum) grown on acid soil. New Phytol 195:609–619
Ding W (2022) The promotion of legume nodulation in plant-soil-microbe systems under phosphorus-limited conditions. Plant Soil 476:251–262
Ding W, Cong W, Lambers H (2021) Plant phosphorus-acquisition and -use strategies affect soil carbon cycling. Trends Ecol Evol 36:899–906
Dong D, Peng X, Yan X (2004) Organic acid exudation induced by phosphorus deficiency and/or aluminium toxicity in two contrasting soybean genotypes. Physiol Plant 122:190–199
Hayes PE, Clode PL, Oliveira RS, Lambers H (2018) Proteaceae from phosphorus-impoverished habitats preferentially allocate phosphorus to photosynthetic cells: an adaptation improving phosphorus-use efficiency. Plant Cell Environ 41:605–619
Hinsinger P (2001) Bioavailability of soil inorganic P in the rhizosphere as affected by root-induced chemical changes: a review. Plant Soil 237:173–195
Honvault N, Houben D, Nobile C, Firmin S, Lambers H, Faucon M-P (2021) Tradeoffs among phosphorus-acquisition root traits of crop species for agroecological intensification. Plant Soil 461:137–150
James RA, Weligama C, Verbyla K, Ryan PR, Rebetzke GJ, Rattey A, Richardson AE, Delhaize E (2016) Rhizosheaths on wheat grown in acid soils: phosphorus acquisition efficiency and genetic control. J Exp Bot 67:3709–3718
Jungk A (2009) Carl Sprengel—The founder of agricultural chemistry: A re-appraisal commemorating the 150th anniversary of his death. J Plant Nutr Soil Sci 172:633–636
Kafle A, Cope KR, Raths R, Krishna Yakha J, Subramanian S, Bücking H, Garcia K (2019) Harnessing soil microbes to improve plant phosphate efficiency in cropping systems. Agronomy 9:127
Kirkby EA (2023) Introduction, definition, and classification of nutrients. Elsevier, Marschner’s Mineral Nutrition of Plants
Krishnapriya V, Pandey R (2016) Root exudation index: screening organic acid exudation and phosphorus acquisition efficiency in soybean genotypes. Crop Pasture Sci 67:1096–1109
Lambers H, Cawthray GR, Giavalisco P, Kuo J, Laliberté E, Pearse SJ, Scheible W-R, Stitt M, Teste F, Turner BL (2012) Proteaceae from severely phosphorus-impoverished soils extensively replace phospholipids with galactolipids and sulfolipids during leaf development to achieve a high photosynthetic phosphorus use efficiency. New Phytol 196:1098–1108
Lambers H, de Britto CP, Cawthray GR, Denton MD, Finnegan PM, Hayes PE, Oliveira RS, Power SC, Ranathunge K, Shen Q, Wang X, Zhong H (2022) Strategies to acquire and use phosphorus in phosphorus-impoverished and fire-prone environments. Plant Soil 476:133–160
Lambers H, Hayes PE, Laliberté E, Oliveira RS, Turner BL (2015) Leaf manganese accumulation and phosphorus-acquisition efficiency. Trends Plant Sci 20:83–90
Lambers H, Wright IJ, Guilherme Pereira C, Bellingham PJ, Bentley LP, Boonman A, Cernusak LA, Foulds W, Gleason SM, Gray EF, Hayes PE, Kooyman RM, Malhi Y, Richardson SJ, Shane MW, Staudinger C, Stock WD, Swarts ND, Turner BL, Turner J, Veneklaas EJ, Wasaki J, Westoby M, Xu Y (2021) Leaf manganese concentrations as a tool to assess belowground plant functioning in phosphorus-impoverished environments. Plant Soil 461:43–61
Lindsay WL, Norvell WA (1978) Development of a DTPA soil test for zinc, iron, manganese, and copper. Soil Sci Soc Am J 42:421–428
Lynch JP (2011) Root phenes for enhanced soil exploration and phosphorus acquisition: tools for future crops. Plant Physiol 156:1041–1049
Lynch JP, Ho MD, Phosphorus L (2005) Rhizoeconomics: carbon costs of phosphorus acquisition. Plant Soil 269:45–56
Makarim AK, Cox FR (1983) Evaluation of the need for copper with several soil extractants. Agron J 75:493–496
Marschner H (1998) Role of root growth, arbuscular mycorrhiza, and root exudates for the efficiency in nutrient acquisition. Field Crops Res 56:203–207
Neumann G, Massonneau A, Langlade N, Dinkelaker B, Hengeler C, Römheld V, Martinoia E (2000) Physiological aspects of cluster root function and development in phosphorus-deficient white lupin (Lupinus albus L.). Ann Bot 85:909–919
Nian H, Ahn SJ, Yang ZM, Matsumoto H (2003) Effect of phosphorus deficiency on aluminium-induced citrate exudation in soybean (Glycine max). Physiol Plant 117:229–236
Ohki K (1976) Manganese deficiency and toxicity levels for ‘Bragg’ soybeans 1. Agron J 68:861–864
Ohki K (1977) Critical zinc levels related to early growth and development of determinate soybeans 1. Agron J 69:969–974
Olsen SR (1954) Estimation of available phosphorus in soils by extraction with sodium bicarbonate. US Department of Agriculture, Washington DC
Pan X, Li W, Zhang Q, Li Y, Liu M (2008) Assessment on phosphorus efficiency characteristics of soybean genotypes in phosphorus-deficient soils. Agric Sci Chin 7:958–969
Pang J, Bansal R, Zhao H, Bohuon E, Lambers H, Ryan MH, Ranathunge K, Siddique KHM (2018) The carboxylate-releasing phosphorus-mobilizing strategy can be proxied by foliar manganese concentration in a large set of chickpea germplasm under low phosphorus supply. New Phytol 219:518–529
Pang J, Ryan MH, Siddique KHM, Simpson RJ (2017) Unwrapping the rhizosheath. Plant Soil 418:129–139
Pang J, Ryan MH, Tibbett M, Cawthray GR, Siddique KHM, Bolland MDA, Denton MD, Lambers H (2010) Variation in morphological and physiological parameters in herbaceous perennial legumes in response to phosphorus supply. Plant Soil 331:241–255
Plaxton W, Lambers H (2015) Annual Plant Reviews, Volume 48, Phosphorus Metabolism in Plants. John Wiley & Sons, Perth
R Core Team (2019) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria
Raboy V (2001) Seeds for a better future: ‘low phytate’ grains help to overcome malnutrition and reduce pollution. Trends Plant Sci 6:458–462
Shen J, Rengel Z, Tang C, Zhang F (2003) Role of phosphorus nutrition in development of cluster roots and release of carboxylates in soil-grown Lupinus albus. Plant Soil 248:199–206
Song X, Li Y, Chang R, Guo P, Qiu L-J (2010) Population structure and genetic diversity of mini core collection of cultivated soybean (Glycine max) in China. Sci Agric Sin 43:2209–2219
Sulpice R, Ishihara H, Schlereth A, Cawthray GR, Encke B, GiavaliscoI P, Ivakov A, Arrivault S, Jost R, Krohn N, Kuo J, Laliberte E, Pearse SJ, Raven JA, Scheible W-R, Teste F, J. VE, Stitt M, Lambers H, (2014) Low levels of ribosomal RNA partly account for the very high photosynthetic phosphorus-use efficiency of Proteaceae species. Plant Cell Environ 37:1276–1298
Tantriani CW, Oikawa A, Tawaraya K (2023) Low phosphorus tolerance mechanisms in soybean cultivars grown in soil. J Soil Sci Plant Nutr 23:6331–6344
Vandamme E, Renkens M, Pypers P, Smolders E, Vanlauwe B, Merckx R (2013) Root hairs explain P uptake efficiency of soybean genotypes grown in a P-deficient Ferralsol. Plant Soil 369:269–282
Veneklaas EJ, Lambers H, Bragg J, Finnegan PM, Lovelock CE, Plaxton WC, Price CA, Scheible W-R, Shane MW, White PJ, Raven JA (2012) Opportunities for improving phosphorus-use efficiency in crop plants. New Phytol 195:306–320
Wang L, Guan Y, Guan R, Li Y, Ma Y, Dong Z, Liu X, Zhang H, Zhang Y, Liu Z, Chang R, Xu H, Li L, Lin F, Luan W, Yan Z, Ning X, Zhu L, Cui Y, Piao R, Liu Y, Chen P, Qiu L (2006) Establishment of Chinese soybean Glycine max core collections with agronomic traits and SSR markers. Euphytica 151:215–223
Wang L, Liao H, Yan X, Zhuang B, Dong Y (2004) Genetic variability for root hair traits as related to phosphorus status in soybean. Plant Soil 261:77–84
Wang X, Pan Q, Chen F, Yan X, Liao H (2011) Effects of co-inoculation with arbuscular mycorrhizal fungi and rhizobia on soybean growth as related to root architecture and availability of N and P. Mycorrhiza 21:173–181
Wang X, Pang J, Wen Z, Gadot G, de Borda A, Siddique KHM, Lambers H (2021) Lower seed P content does not affect early growth in chickpea, provided starter P fertiliser is supplied. Plant Soil 463:113–24
Wang X, Yan X, Liao H (2010) Genetic improvement for phosphorus efficiency in soybean: a radical approach. Ann Bot 106:215–222
Weemstra M, Mommer L, Visser EJW, van Ruijven J, Kuyper TW, Mohren GMJ, Sterck FJC (2016) Towards a multidimensional root trait framework: a tree root review. New Phytol 211:1159–1169
Wen Z, Li H, Shen J, Rengel Z (2017) Maize responds to low shoot P concentration by altering root morphology rather than increasing root exudation. Plant Soil 416:377–389
Wen Z, Li H, Shen Q, Tang X, Xiong C, Li H, Pang J, Ryan MH, Lambers H, Shen J (2019) Tradeoffs among root morphology, exudation and mycorrhizal symbioses for phosphorus-acquisition strategies of 16 crop species. New Phytol 223:882–895
Wen Z, Pang J, Tueux G, Liu Y, Shen J, Ryan MH, Lambers H, Siddique KHM (2020) Contrasting patterns in biomass allocation, root morphology and mycorrhizal symbiosis for phosphorus acquisition among 20 chickpea genotypes with different amounts of rhizosheath carboxylates. Funct Ecol 34:1311–1324
White PJ, Veneklaas EJ (2012) Nature and nurture: the importance of seed phosphorus content. Plant Soil 357:1–8
Wu D, Yu Q, Lu C, Hengsdijk H (2006) Quantifying production potentials of winter wheat in the North China Plain. Eur J Agron 24:226–235
Xin Y, Tao F (2020) Developing climate-smart agricultural systems in the North China Plain. Agric, Ecosyst Environ 291:106791
Yang ZM, Sivaguru M, Horst WJ, Matsumoto H (2000) Aluminium tolerance is achieved by exudation of citric acid from roots of soybean (Glycine max). Physiol Plant 110:72–77
Zhang D, Zhang C, Tang X, Li H, Zhang F, Rengel Z, Whalley WR, Davies WJ, Shen J (2016) Increased soil phosphorus availability induced by faba bean root exudation stimulates root growth and phosphorus uptake in neighbouring maize. New Phytol 209:823–831
Zhao J, Fu J, Liao H, He Y, Nian H, Hu Y, Qiu L, Dong Y, Yan X (2004) Characterization of root architecture in an applied core collection for phosphorus efficiency of soybean germplasm. Chin Sci Bull 49:1611–1620
Zhou T, Du Y, Ahmed S, Liu T, Ren M, Liu W, Yang W (2016) Genotypic differences in phosphorus efficiency and the performance of physiological characteristics in response to low phosphorus stress of soybean in southwest of China. Front Plant Sci 7:1776
Zhou XM, Ranathunge K, Cambridge ML, Dixon KW, Hayes PE, Nikolic M, Shen Q, Zhong H, Lambers H (2022) A cool spot in a biodiversity hotspot: why do tall Eucalyptus forests in Southwest Australia exhibit low diversity? Plant Soil 476:669–688
Acknowledgements
This research was supported by National Natural Science Foundation of China (32072676, 32271610, 32102468), and the Australian Research Council (LP200100341). Wenli Ding was supported by the International Postdoctoral Exchange Fellowship Program (Talent-Introduction Program YJ20200203), which is co-funded by the Office of China Postdoc Council and China Agricultural University, and by China Postdoctoral Science Foundation (2021M700165). We thank Advanced Discipline Construction in Beijing (Agriculture Green Development) and Introducing Talents of Discipline to Universities (Plant-soil interactions innovative research platform 1031-00100701) to support this research. In addition, we thank Lijuan Qiu and Yongzhe Gu for providing soybean seeds as well as related information which was funded by the Ministry of Science and Technology, Platform of National Crop Germplasm Resources of China (NICGR-2019-04), Ministry of Agriculture and Rural Affairs: Crop Germplasm Resources Protection Platform (2019NWB036-05). We thank Zhihui Wen for the internal review. We also thank two anonymous reviewers for their highly valuable comments.
Funding
The study was financially supported by National Natural Science Foundation of China (32271610, 32072676, 32102468), International Postdoctoral Exchange Fellowship Program (Talent-Introduction Program YJ20200203), China Postdoctoral Science Foundation (2021M700165), the Australian Research Council (LP200100341), Advanced Discipline Construction in Beijing (Agriculture Green Development) and Introducing Talents of Discipline to Universities (Plant-soil interactions innovative research platform 1031–00100701).
Author information
Authors and Affiliations
Contributions
Wenfeng Jiao, Wen-Feng Cong, Jiayin Pang and Hans Lambers designed the study. Wenli Ding, Wenfeng Jiao and Boyu Zheng performed the experiment and collected the data. Wenli Ding analysed the data. Wenli Ding, Jiayin Pang, Wen-Feng Cong and Hans Lambers wrote this manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflicts of interest/Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Responsible Editor: Peter J. Gregory.
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ding, W., Jiao, W., Pang, J. et al. Clusters of cultivated soybean landraces from the North China Plain coordinate root morphology and rhizosheath carboxylates enhancing phosphorus acquisition. Plant Soil (2024). https://doi.org/10.1007/s11104-024-06717-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11104-024-06717-4