Abstract
Background and aims
Leaf nutrient resorption process is an indispensable aspect of plant nutrient utilization strategies in boreal peatlands. However, how nitrogen (N) enrichment alters leaf nutrient resorption is unclear at both species and community levels, and the underlying mechanisms remain unknown in these ecosystems.
Methods
Using a fertilization experiment with four N addition levels, we investigated the influence of increased plant-available N on leaf nutrient resorption efficiency and proficiency at both species and community levels, and disentangled the relative contributions of intraspecific variability and altered plant composition to the shifts in community-level nutrient resorption in a poor fen, Northeast China.
Results
Leaf nutrient resorption exhibited divergent responses to N addition at species and community levels. For most species, leaf N resorption efficiency remained unchanged, but leaf N resorption proficiency declined after N addition. In contrast, N addition increased leaf phosphorus (P) resorption of most species. At the species level, leaf N resorption declined with increasing green leaf N:P ratio, while leaf P resorption showed an opposite changing trend. Nitrogen addition reduced community-level leaf N resorption efficiency and proficiency, but increased community-level leaf P resorption efficiency and proficiency. Compared with intraspecific variability, altered plant composition contributed much more to N addition-induced shifts in community-level leaf nutrient resorption.
Conclusion
The plasticity in leaf N:P stoichiometry determines the interspecific responses of leaf nutrient resorption to N addition and shifted plant composition exerts a critical control over N enrichment effect on plant nutrient resorption at the community level in boreal peatlands.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Nutrient resorption from senescing leaves is a key determinant of plant internal nutrient cycling and nutrient conservation strategy in most terrestrial ecosystems (Aerts and Chapin 2000; Vergutz et al. 2012; Estiarte et al. 2022). In general, leaf nutrient resorption is expressed either as the fraction of nutrients that can be resorbed from the mature leaves before senescence (i.e., nutrient resorption efficiency, Aerts 1996), or as the degree to which leaf nutrient concentrations can be reduced during senescence (i.e., nutrient resorption proficiency, Killingbeck 1996). Leaf nutrient resorption process can increase plant competition and fitness by reducing plant’s reliance on soil nutrient status (Eckstein et al. 1999; Yuan and Chen 2015) and determine litter decomposition and nutrient release through its effects on litter chemical composition (Deng et al. 2018; Xu et al. 2020). Therefore, leaf nutrient resorption is of great significance to vegetation community composition, soil organic matter sequestration, and nutrient cycling in terrestrial ecosystems.
Boreal peatlands are often nutrient-poor, carbon-rich ecosystems with about 30% of the world’s soil organic carbon pool as a result of slow organic matter decomposition rates (Limpens et al. 2008; Yu 2011). In these ecosystems, plants usually adapt to soil nutrient deficiency by increasing leaf nutrient resorption capacity, which can reduce nutrient loss during leaf senescence (Eckstein et al. 1999; Xu et al. 2020). Boreal peatlands have recently experienced increasing plant-available nitrogen (N) primarily because of atmospheric N deposition, warming-accelerated soil organic matter mineralization, and permafrost thaw (Rustad et al. 2001; Gruber and Galloway 2008; Finger et al. 2016). The elevated reactive N inputs are believed to reduce leaf N resorption by increasing bioavailable N but enhanced leaf phosphorus (P) resorption due to the intensified P limitation in boreal peatlands. In the limited empirical studies, however, the effect of increased plant-available N on leaf nutrient resorption varies with plant growth forms (van Heerwaarden et al. 2003; Wang et al. 2014; Gao et al. 2022) probably due to the differences in leaf lifespan (Eckstein et al. 1999; Zhang et al. 2013), leaf economic spectrum traits (Yu et al. 2022a, b), plant growth performance (Zhang et al. 2015), the size of non-leaf nutrient pool (Vergutz et al. 2012), and nutrient allocation patterns (Tanabe et al. 2022). Therefore, additional studies are conducted to uncover the general pattern of leaf nutrient resorption responses to altered N availability and its underlying mechanisms in boreal peatlands.
Considering the species-specific responses of leaf nutrient resorption to increased plant-available N, the consequence of N enrichment on leaf nutrient resorption should be assessed at the community level in boreal peatlands. Community-level leaf nutrient resorption is generally controlled by species resorption and plant community composition (See et al. 2015; Lü et al. 2020; Yu et al. 2022a, b). In boreal peatlands, increased plant-available N can substantially change species composition and community structure (Kirkham et al. 1996; Levy et al. 2019; Lin et al. 2020). For instance, N addition is observed to increase the abundances of deciduous shrubs and graminoids at the expense of evergreen shrubs and mosses in a poor fen (Shi et al. 2022). Moreover, the interspecific differences in plant nutrient resorption are even greater than N addition-induced intraspecific variation of plant nutrient resorption in these nutrient-poor ecosystems (e.g., van Heerwaarden et al. 2003; Gao et al. 2022). Therefore, shifted plant community composition after N addition may play a critical part in modulating the direction and magnitude of the changes in community-level plant nutrient resorption. However, the contribution of N enrichment-induced altered plant composition and intraspecific variability to community-level plant nutrient resorption has rarely been disentangled in boreal peatlands.
Here, we established a multi-level N fertilization experiment in a poor fen in the Daxing’anling Mountain, Northeast China, and investigated the shifts in leaf N and P resorption at both species and community levels after six years of N fertilization. The main objectives were to assess the effects of N addition on leaf nutrient resorption efficiency and proficiency and their potential underlying mechanisms in boreal peatlands. Accordingly, we hypothesized that: (1) the impacts of N addition on leaf nutrient resorption would be species-specific due to the interspecific variations in genetic regulation of nutrient use strategies (Yuan and Chen 2015; Estiarte et al. 2022); (2) at the community level, N addition would reduce N resorption but increased P resorption because of the shifts in limiting nutrients from N to P (You et al. 2018; Chen et al. 2021); and (3) altered species composition would make a greater contribution to N addition-induced shifts in community-level nutrient resorption than the intraspecific changes in nutrient resorption due to the substantial interspecific differences in nutrient resorption (e.g., van Heerwaarden et al. 2003; Wang et al. 2014; Gao et al. 2022).
Materials and methods
Study area and experiment design
This experiment was conducted in a poor fen (52°56′36'N, 122°51′19'E) which is located at the north slope of Daxing’anling Mountain in Northeast China. This peatland lies in the south margin of the Eurasian continuous permafrost area with a cold temperate climate. The mean annual temperature is –3.9 °C and mean annual precipitation is about 450 mm. The average depth of the peat layer is about 60 cm, and the maximum thickness of permafrost active layer is about 55 cm. Soil NH4+-N, NO3−-N, and total N concentrations in the 0–10 cm depth are 37 μg g−1, 3.2 μg g−1, and 15.6 mg g−1, respectively. This peatland lies in an intermontane basin and is primarily fed by precipitation and groundwater from the surrounding uplands. The plant community in this peatland consists of deciduous shrubs, evergreen shrubs, graminoids, and mosses. The dominant deciduous shrubs are Betula fruticosa, Salix myrtilloides, Salix rosmarinifolia, and Vaccinium uliginosum, the dominant evergreen shrubs are Ledum palustre and Rhododendron lapponicum, the dominant graminoid species are Deyeuxia angustifolia, Carex globularis, and Eriophorum vaginatum, and the dominant moss species are Sphagnum palustre, Sphagnum beccarii, and Sphagnum magellanicum (Shi et al. 2022). The ambient atmospheric N deposition rate in the study site is approximately 4.5 g N m−2 yr−1 (Zhan et al. 2014).
This N fertilization experiment followed a completely randomized block design. In August 2010, four blocks with similar vegetation and microtopography were randomly chosen and four plots (4 m × 4 m) per block were established in this peatland. Because atmospheric deposited N input is extremely low in the study area, 3 (N3), 6 (N6), and 12 (N12) g N m−2 yr−1 were set up to simulate warming-induced soil organic N mineralization and associated increases in plant-available N (Lin et al. 2020). Meanwhile, the plots without N addition were established as the control treatment. The plots were fenced with stainless steel (40 cm in depth) to avoid the lateral exchange of water and nutrients, and the distance between the adjacent plots was about 1 m. From 2011 to 2016, urea solution was applied monthly from May to July, and the same amount of purified water was added to the control treatments. A detailed description of this experiment was introduced by Lin et al. (2020).
Sample collection and measurements
Because N addition caused the disappearance of moss species (Shi et al. 2022), eight vascular plants that occurred in each plot were selected: three deciduous shrubs (B. fruticosa, S. myrtilloides, and V. uliginosum), two evergreen shrubs (R. lapponicum and L. palustre), and three graminoid species (C. globularis, D. angustifolia, and E. vaginatum). These selected eight species accounted for 87.1% of total aboveground plant biomass in the control treatment.
In early August 2016, we measured the cover of each species with the visual-estimated method (Lin et al. 2020; Shi et al. 2022) and then calculated the relative cover of the selected species in the plot. In each plot, we marked 20 shoots for each shrub species and 60 shoots for each graminoid species. In early August, 30 healthy mature green leaves per species were collected from the marked shoots. Subsequently, 30 newly-senesced leaves per species were collected from late August to late September. Both mature and senesced leaves were oven-dried (45 °C), weighed, ground to pass 0.15-mm sieves, and then used to determine N and P concentrations. Leaf N and P concentrations were respectively measured with sodium hypochlorite-salicylic acid spectrophotometry and molybdate blue spectrophotometry (Temminghoff and Houba 2004) on an AA3 autoanalyzer (Seal Analytical, Germany) after digestion with concentrated sulphuric acid. In addition, we collected soil pore water at 10 cm depth using the hand pumps in each plot in early August 2016. Water samples were filtered, and NH4+-N and NO3−-N concentrations in the filters were measured spectrometrically with sodium hypochlorite-salicylic acid (Temminghoff and Houba 2004) and hydrazine reduction (Kamphake et al. 1967) methods with an AA3 autoanalyzer, respectively.
For each species, nutrient resorption efficiency (%) was obtained with the following formula (Vergutz et al. 2012; Xu et al. 2020):
where Ngreen and Nsenesced are nutrient concentrations of green and senesced leaves, respectively, and Massgreen and Masssenesced are the mass of green and senesced leaves, respectively. Senesced leaf nutrient concentration can reflect the ultimate nutrient resorption potential and thus is used to characterize nutrient resorption proficiency; a high nutrient concentration in senesced leaves is associated with a low nutrient resorption proficiency (Killingbeck 1996). Community-level nutrient resorption was obtained based on the relative cover and nutrient resorption of the selected species in each plot (Yu et al. 2022a, b).
Statistical analyses
Statistical analyses were performed with the R package (version 4.1.1, R Development Core Team 2021), and the significance level was set to 0.05. The effects of species and N addition level on nutrient resorption were tested with linear mixed models in the “nlme” package. The significant differences in nutrient resorption among N addition treatments were assessed with Tukey’s multiple tests in the “multcomp” package. At the species level, the relationship between nutrient resorption and green leaf N:P ratio was assessed with simple linear regression analysis.
According to the procedure of Lepš et al. (2011), we disentangled the relative contributions of intraspecific variability, altered plant composition, and their covariation to community-level nutrient resorption. First, we calculated the specific (including both intraspecific variability and altered plant composition) and fixed (including altered plant composition) community-level nutrient resorption, and then obtained intraspecific community-level nutrient resorption based on the difference between specific and fixed community-level nutrient resorption. Second, the general linear model was performed respectively for specific, fixed, and intraspecific community-level nutrient resorption with N addition level as an explanatory variable. Subsequently, the sums of squares (SS) were extracted for specific (SSspecific), fixed (SSfixed), and intraspecific (SSintra) community-level nutrient resorption explained by the N addition level. Third, the covariation between intraspecific variability and altered plant composition (SScov) was calculated as SScov = SSspecific – SSfixed – SSintra. Thus, the total variation (SSspecific) can be decomposed into intraspecific variability (SSintra), altered plant composition (SSfixed), and their covariation (SScov).
Results
Compared with the control treatments, NH4+-N and NO3−-N concentrations in the soil pore water were higher in the N addition treatments (Fig. S1). Regardless of N addition levels, N addition enhanced the relative cover of B. fruticosa and D. angustifolia, but reduced the relative cover of V. uliginosum, L. palustre, C. globularis, and E. vaginatum (Table S2). However, the relative cover of S. myrtilloides and R. lapponicum remained unchanged after N addition (Table S2).
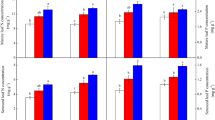
For each species, N addition caused an increase in green leaf N concentration, but did not affect green leaf P concentration (Table S3 and Fig. 1). Irrespective of species, N addition often increased senesced leaf N concentration (Fig. 2), indicating declined N resorption proficiency. Except for C. globularis, N addition decreased P concentration of senesced leaves (Fig. 2), exhibiting increased P resorption proficiency. The response of leaf N resorption efficiency to N addition differed among species (Table S3 and Fig. 3). Nitrogen addition significantly decreased N resorption efficiency of three species (i.e., B. fruticosa, S. myrtilloides, and D. angustifolia), and generally did not change leaf N resorption efficiency of the other five species (Fig. 3). In contrast, N addition increased leaf P resorption efficiency of the selected eight species (Fig. 3).
Effect of N addition level on green leaf nutrient concentrations of selected eight species in a boreal peatland. Error bars are standard errors of the means (n = 4). For each species, different lowercase letters indicate the significant differences (P < 0.05) among treatments. N3, N addition at 3 g N m−2 yr−1; N6, N addition at 6 g N m−2 yr−1; N12, N addition at 12 g N m−2 yr−1
Effect of N addition level on senesced leaf nutrient concentrations of selected eight species in a boreal peatland. Error bars are standard errors of the means (n = 4). For each species, different lowercase letters indicate the significant differences (P < 0.05) among treatments. N3, N addition at 3 g N m−2 yr−1; N6, N addition at 6 g N m−2 yr−1; N12, N addition at 12 g N m−2 yr−1
Effect of N addition level on leaf nutrient resorption efficiency of selected eight species in a boreal peatland. Error bars are standard errors of the means (n = 4). For each species, different lowercase letters indicate the significant differences (P < 0.05) among treatments. N3, N addition at 3 g N m−2 yr−1; N6, N addition at 6 g N m−2 yr−1; N12, N addition at 12 g N m−2 yr−1
At the species level, N:P ratio in green leaves showed a negative relationship with leaf N resorption efficiency (r = –0.447, P < 0.01; Fig. 4), but exhibited a positive relationship with leaf P resorption efficiency (r = 0.462, P < 0.01; Fig. 4). In contrast, green leaf N:P ratio correlated positively with senesced leaf N concentration (r = 0.737, P < 0.01; Fig. 4), but correlated negatively with senesced leaf P concentration (r = –0.576, P < 0.01; Fig. 4). In addition, species-level leaf N:P resorption ratio correlated negatively with N:P ratio in green leaves (r = –0.530, P < 0.01; Fig. S2).
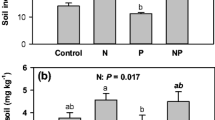
Nitrogen addition caused an increase in community-level green leaf N concentration, but caused a reduction in community-level leaf N resorption efficiency (Table 1). Accordingly, community-level senesced leaf N concentration was increased by N addition, exhibiting declined N resorption proficiency (Table 1). However, N addition did not affect green leaf P concentration, but increased leaf P resorption efficiency at the community level (Table 1). Therefore, community-level senesced leaf P concentration decreased after N addition (Table 1), exhibiting an increase in leaf P resorption proficiency. At the community level, senesced leaf N:P ratio significantly increased with increasing N addition levels, whereas leaf N:P resorption ratio significantly declined with increasing N addition levels (Table 1).
Compared with intraspecific variability, altered plant composition contributed higher to N addition-induced shifts in community-level leaf nutrient resorption (Fig. 5). The relative contributions of intraspecific variability, altered plant composition, and their covariation were respectively 3.1%, 64.4%, and 22.0% for green leaf N concentration, 19.7%, 32.3%, and 45.1% for leaf N resorption efficiency, and 9.8%, 44.8%, and 41.5% for leaf N resorption proficiency (Fig. 5). Similarly, the relative contributions of intraspecific variability, altered plant composition, and their covariation were respectively 9.7%, 10.5%, and –11.4% for green leaf P concentration, 1.0%, 92.2%, and 0.7% for leaf P resorption efficiency, and 0.6%, 105.5%, and –15.3% for leaf P resorption proficiency (Fig. 5).
Decomposition of total variability in community-level leaf N resorption parameters explained by N addition into intraspecific variability, altered plant composition, and covariance effects. Covariance effects are the intervals between the total variability and the sum of intraspecific variability and altered plant composition. *** indicates the significant effects of intraspecific variability or altered plant composition or total variability at P < 0.001
Discussion
Consistent with the first hypothesis, leaf nutrient resorption responses to N addition were species-specific in this peatland. This result was in line with previous studies which found that leaf nutrient resorption exhibited divergent responses to N addition at the species level in temperate and boreal peatlands (van Heerwaarden et al. 2003; Wang et al. 2014; Gao et al. 2022). These different responses would be primarily caused by the differences in genetic adaptation strategies and associated nutrient uptake and storage potentials among species (Yuan and Chen 2015; You et al. 2018; Estiarte et al. 2022). In general, plants often preferentially resorb more limiting nutrient elements from senescing leaves (See et al. 2015; Chen et al. 2021). Leaf N:P ratio can reflect the limiting nutrients for plant growth (Güsewell et al. 2003; You et al. 2018), and thus may be a potential modulator of plant N and P resorption processes. However, no empirical studies have been performed to test the role of leaf N:P ratio in controlling the shifts in leaf nutrient resorption process in boreal peatlands. In this study, the tight correlation between green leaf N:P ratio and leaf nutrient (i.e., N and P) resorption (Fig. 4) confirms that the plasticity in N:P stoichiometry after N enrichment determines the intraspecific variability of plant N and P resorption in boreal peatlands.
Notably, leaf N resorption efficiency of B. fruticosa, S. myrtilloides, and D. angustifolia was more responsive to N addition than that of the other five species. These species-specific responses of N resorption efficiency to N addition would be partly explained by mycorrhizal type and associated nutrient economy (Phillips et al. 2013; Xu et al. 2020). Compared to ericoid mycorrhizal shrubs (V. uliginosum, R. lapponicum, and L. palustre) and non-mycorrhizal sedges (C. globularis and E. vaginatum), ectomycorrhizal shrubs (B. fruticosa and S. myrtilloides) and non-mycorrhizal grass (D. angustifolia) are well-adapted to N-rich habitats (Lin et al. 2020; Shi et al. 2022), and thus can effectively utilize the added available N in this peatland. We also observed that the increase magnitudes of green leaf N concentration after N addition were much greater for B. fruticosa, S. myrtilloides, and D. angustifolia than for the other five species (Fig. 1). Considering the tight relationship between leaf N resorption and leaf N status (Yuan and Chen 2015), N addition only reduced leaf N resorption efficiency of B. fruticosa, S. myrtilloides, and D. angustifolia. Because the plasticity in nutrient resorption can reinforce plant-soil feedbacks and increase plant fitness (Lü et al. 2012), B. fruticosa, S. myrtilloides, and D. angustifolia gained a competitive advantage over the other five species after N addition in this peatland (Table S1). Tsujii et al. (2017) have observed that leaf N resorption is tightly linked to N allocation among low molecular weight compounds, nucleic acids, lipids, and residuals. Due to the difference in nutrient economy, plants with different mycorrhizal associations may have distinct nutrient allocation among different chemical fractions. Therefore, to uncover the mechanisms driving the divergent responses of leaf N resorption, further studies should be performed to assess the effect of N enrichment on leaf N fractions among mycorrhizal associations in boreal peatlands.
Despite the inconsistent responses of leaf nutrient resorption efficiency, N addition often increased N concentration and reduced P concentration in senesced leaves, indicating decreased plant N resorption proficiency and increased plant P resorption proficiency in this peatland. These observations supported the assumption that, compared with nutrient resorption efficiency, nutrient resorption proficiency was more responsive to increased N availability (e.g., van Heerwaarden et al. 2003; Mao et al. 2013; Lü et al. 2020). These results also imply that leaf nutrient resorption proficiency (i.e., the terminal senesced leaf nutrient concentration) would be a powerful indicator reflecting the adaptation of plant nutrient strategies to altered soil N status. Leaf nutrient resorption proficiency determines the initial chemical quality of leaf litter that controls litter decomposability (Deng et al. 2018; Xu et al. 2020). Therefore, N enrichment could further intensify the P limitation of leaf litter decomposition through its contrasting effects on N and P resorption proficiency in boreal peatlands.
At the community level, leaf N resorption declined, but leaf P resorption increased after N addition in this peatland. Thus, these findings supported our second hypothesis. Increased N availability has also been observed to decrease community-level plant N resorption in temperate forests, grasslands, and wetlands (See et al. 2015; Lü et al. 2020; Zhang et al. 2021). However, N addition effect on community-level plant P resorption was contrasting; negative effects were observed in a temperate grassland (Lü et al. 2020) but neutral effects were found in a temperate wetland (Zhang et al. 2021). These observations imply that the responses to N addition were more complex for P resorption than for N resorption. Considering the idiosyncratic responses of species-level nutrient resorption to N addition among species, these observations imply that assessing community-level leaf nutrient resorption responses can fully understand and predict the consequence of altered N availability on plant internal nutrient cycling in boreal peatlands.
In accordance with the third hypothesis, the relative contribution to N addition-induced shifts in community-level nutrient resorption, especially for P resorption, was much greater for altered plant composition than for intraspecific variability. Zhang et al. (2021) also observed that, despite unchanged species-level N resorption efficiency, shifted plant composition after N addition decreased community-level N resorption efficiency in a temperate marsh. For both leaf N and P resorption, N addition-induced intraspecific variability was much lower than the intrinsic difference among species in this peatland (Figs. 3 and 4). Moreover, N addition increased the dominance of B. fruticosa, S. myrtilloides, and D. angustifolia (Shi et al. 2022), leaf nutrient resorption of which was more responsive to N addition than the other five species (Figs. 3 and 4). Accordingly, altered plant composition made a greater contribution to N addition-induced shifts in community-level leaf nutrient resorption than intraspecific variability. These observations indicate the crucial role of altered species composition in regulating community-level nutrient resorption process in boreal peatlands.
Leaf N:P resorption stoichiometry is proposed as an effective indicator of nutrient limitation of plant growth in terrestrial ecosystems (Reed et al. 2012). At the community level, the declined leaf N:P resorption ratio after N addition showed the shift in N limitation to P limitation in this boreal peatland. Boreal peatlands will undergo increased bioavailable N in soils due to climate warming, permafrost thaw, and atmospheric N deposition (Rustad et al. 2001; Gruber and Galloway 2008; Finger et al. 2016). This continuous increase in bioavailable N will further cause the decouple of N and P cycles in soils through its contrasting influences on plant N and P resorption, and thus aggravate the P limitation of biological processes in boreal peatlands.
Boreal peatlands store a vast amount of soil organic carbon mainly because of the extremely slow litter decomposition rates (Limpens et al. 2008; Yu 2011). Considering the critical role of plant nutrient resorption in controlling litter decomposability (Deng et al. 2018; Xu et al. 2020), our observations are key to predicting the consequence of increased N availability on soil organic carbon accumulation in these carbon-rich ecosystems. Nitrogen addition-induced increases in plant P resorption can reduce the amount of litter P return to soils, further causing N:P imbalance in plant litter (You et al. 2018). Meanwhile, increased dominance of B. fruticosa and D. angustifolia after N addition will amplify the N addition effect on N:P imbalance due to their relatively high responsiveness of leaf nutrient resorption. Intensified N:P imbalance after N addition will prime the decomposition of soil organic carbon by promoting the microbial investment in P-degrading enzymes (Bragazza et al. 2013). However, N addition is observed to enhance plant primary productivity (Shi et al. 2022), resulting in increased soil organic carbon inputs via plant litter. Accordingly, further studies are needed to clarify the effect of N enrichment on soil organic carbon budget in boreal peatlands.
In summary, we examined the response of leaf nutrient resorption to N addition at species and community levels and disentangled the contribution of intraspecific variability and shifted plant composition to community-level nutrient resorption in a poor fen, Northeast China. Despite the species-specific responses of leaf nutrient resorption, N addition decreased leaf N resorption but increased leaf P resorption at the community level. The intraspecific variations of leaf nutrient resorption efficiency and proficiency after N addition were largely determined by plasticity in green leaf N:P stoichiometry, and N addition effects on community-level leaf nutrient resorption, particularly P resorption, were primarily caused by the altered plant composition. These findings highlight the key determinants of leaf nutrient resorption at species and community levels and suggest the predominance of changed species composition after N addition in regulating the variations of community-level plant nutrient resorption in boreal peatlands.
Data Availability
Data is available within the electronic supplementary materials of the article.
References
Aerts R (1996) Nutrient resorption from senescing leaves of perennials: are there general patterns? J Ecol 84:597–610. https://doi.org/10.2307/2261481
Aerts R, Chapin FS (2000) The mineral nutrition of wild plants revisited: a re-evaluation of processes and patterns. Adv Ecol Res 30:1–67. https://doi.org/10.1016/S0065-2504(08)60016-1
Bragazza L, Parisod J, Buttler A, Bardgett RD (2013) Biogeochemical plant-soil microbe feedback in response to climate warming in peatlands. Nat Clim Chang 3:273–277. https://doi.org/10.1038/nclimate1781
Chen H, Reed SC, Lü X, Xiao K, Wang K, Li D (2021) Coexistence of multiple leaf nutrient resorption strategies in a single ecosystem. Sci Total Environ 772:144951. https://doi.org/10.1016/j.scitotenv.2021.144951
Deng MF, Liu LL, Jiang L, Liu WX, Wang X, Li SP, Yang S, Wang B (2018) Ecosystem scale trade-off in nitrogen acquisition pathways. Nat Ecol Evol 11:1724–1734. https://doi.org/10.1038/s41559-018-0677-1
Eckstein RL, Karlsson PS, Weih M (1999) Leaf life span and nutrient resorption as determinants of plant nutrient conservation in temperate-arctic regions. New Phytol 143:177–189. https://doi.org/10.1046/j.1469-8137.1999.00429.x
Estiarte M, Campioli M, Mayol M, Penuelas J (2022) Variability and limits of nitrogen and phosphorus resorption during foliar senescence. Plant Commun 4:100503. https://doi.org/10.1016/j.xplc.2022.100503
Finger RA, Turetsky MR, Kielland K, Ruess RW, Mack MC, Euskirchen ES (2016) Effects of permafrost thaw on nitrogen availability and plant-soil interactions in a boreal Alaskan lowland. J Ecol 104:1542–1554. https://doi.org/10.1111/1365-2745.12639
Gao S, Song Y, Song C, Wang X, Gong C, Ma X, Gao J, Cheng X, Du Y (2022) Long-term nitrogen addition alters peatland plant community structure and nutrient resorption efficiency. Science of The Total Environment 844:157176. https://doi.org/10.1016/j.scitotenv.2022.157176
Gruber N, Galloway JN (2008) An Earth-system perspective of the global nitrogen cycle. Nature 451:293–296. https://doi.org/10.1038/nature06592
Güsewell S, Koerselman W, Verhoeven JTA (2003) Biomass N: P ratios as indicators of nutrient limitation for plant populations in wetlands. Ecol Appl 13:372–384. https://doi.org/10.1890/1051-0761(2003)013[0372:BNRAIO]2.0.CO;2
Kamphake LJ, Hannah SA, Cohen JM (1967) Automated analysis for nitrate by hydrazine reduction. Water Res 1:205–216. https://doi.org/10.1016/0043-1354(67)90011-5
Killingbeck K (1996) Nutrients in senesced leaves: keys to the search for potential resorption and resorption proficiency. Ecology 77:1716–1727. https://doi.org/10.2307/2265777
Kirkham FW, Mountford JO, Wilkins RJ (1996) The effects of nitrogen, potassium and phosphorus addition on the vegetation of a Somerset peat moor under cutting management. J Appl Ecol 33:1013–1029. https://doi.org/10.2307/2404682
Lepš J, de Bello F, Šmilauer P, Dolezal J (2011) Community trait response to environment: disentangling species turnover vs intraspecific trait variability effects. Ecography 34:856–863. https://doi.org/10.1111/j.1600-0587.2010.06904.x
Levy P, van Dijk N, Gray A, Sutton M, Jones M, Leeson S, Dise N, Leith I, Sheppard L (2019) Response of a peat bog vegetation community to long-term experimental addition of nitrogen. J Ecol 107:1167–1186. https://doi.org/10.1111/1365-2745.13107
Limpens J, Berendse F, Blodau C, Canadell JG, Freeman C, Holden J, Roulet N, Rydin H, Schaepman-Strub G (2008) Peatlands and the carbon cycle: from local processes to global implications – a synthesis. Biogeosciences 5:1475–1491. https://doi.org/10.5194/bg-5-1475-2008
Lin G, Zeng DH, Mao R (2020) Traits and their plasticity determine responses of plant performance and community functional property to nitrogen enrichment in a boreal peatland. Plant Soil 449:151–167. https://doi.org/10.1007/s11104-020-04478-4
Lü XT, Freschet GT, Flynn DFB, Han XG (2012) Plasticity in leaf and stem nutrient resorption proficiency potentially reinforces plant-soil feedbacks and microscale heterogeneity in a semi-arid grassland. J Ecol 100:144–150. https://doi.org/10.1111/j.1365-2745.2011.01881.x
Lü XT, Hou SL, Reed S, Yin JX, Hu YY, Wei HW, Wang ZW, Yang GJ, Liu ZY, Han XG (2020) Nitrogen enrichment reduces nitrogen and phosphorus resorption through changes to species resorption and plant community composition. Ecosystems 24:602–612. https://doi.org/10.1007/s10021-020-00537-0
Mao R, Song CC, Zhang XH, Wang XW, Zhang ZH (2013) Response of leaf, sheath and stem nutrient resorption to 7 years of N addition in freshwater wetland of Northeast China. Plant Soil 364:385–394. https://doi.org/10.1007/s11104-012-1370-9
Phillips RP, Brzostek E, Midgley MG (2013) The mycorrhizal-associated nutrient economy: a new framework for predicting carbon-nutrient couplings in temperate forests. New Phytol 199:41–51. https://doi.org/10.1111/nph.12221
R Development Core Team (2021) R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna, Austria
Reed SC, Townsend AR, Davidson EA, Cleveland CC (2012) Stoichiometric patterns in foliar nutrient resorption across multiple scales. New Phytol 196:173–180. https://doi.org/10.1111/j.1469-8137.2012.04249.x
Rustad LE, Campbell JL, Marion GM, Norby RJ, Mitchell MJ, Hartley AE, Cornelissen JHC, Gurevitch J, GCTE-NEWS (2001) A meta-analysis of the response of soil respiration, net nitrogen mineralization, and aboveground plant growth to experimental ecosystem warming. Oecologia 126:543–562. https://doi.org/10.1007/s004420000544
See CR, Yanai RD, Fisk MC, Vadeboncoeur MA, Quitero BA, Fahey TJ (2015) Soil nitrogen affects phosphorus recycling: foliar resorption and plant-soil feedbacks in a northern hardwood forest. Ecology 96:2488–2498. https://doi.org/10.1890/15-0188.1
Shi FX, Wang XW, Lin GG, Zhang XH, Chen HM, Mao R (2022) Cryptogams and dwarf evergreen shrubs are vulnerable to nitrogen addition in a boreal permafrost peatland of Northeast China. Appl Veg Sci 25:e12691. https://doi.org/10.1111/avsc.12691
Tanabe R, Miyazawa S, Kitade O, Oikawa S (2022) Effect of symbiotic N2 fixation on leaf protein contents, protein degradation and nitrogen resorption during leaf senescence in temperate deciduous woody species. Oecologia 200:79–87. https://doi.org/10.1007/s00442-022-05264-y
Temminghoff EEJM, Houba VJG (2004) Plant analysis procedures, 2nd edn. Kluwer Academic Publishers, Dordrecht
Tsujii Y, Onoda Y, Kitayama K (2017) Phosphorus and nitrogen resorption from different chemical fractions in senescing leaves of tropical tree species on Mount Kinabalu, Borneo. Oecologia 185:171–180. https://doi.org/10.1007/s00442-017-3938-9
van Heerwaarden LM, Toet S, Aerts R (2003) Nitrogen and phosphorus resorption efficiency and proficiency in six sub-arctic bog species after 4 years of nitrogen fertilization. J Ecol 91:1060–1070. https://doi.org/10.1046/j.1365-2745.2003.00828.x
Vergutz L, Mazoni S, Porporato A, Novais RF, Jackson RB (2012) Global resorption efficiencies and concentrations of carbon and nutrients in leaves of terrestrial plants. Ecology 82:205–220. https://doi.org/10.1890/11-0416.1
Wang M, Murphy MT, Moore TR (2014) Nutrient resorption of two evergreen shrubs in response to long-term fertilization in a bog. Oecologia 174:365–377. https://doi.org/10.1007/s00442-013-2784-7
Xu JW, Lin G, Liu B, Mao R (2020) Linking leaf nutrient resorption and litter decomposition to plant mycorrhizal associations in boreal peatlands. Plant Soil 448:413–424. https://doi.org/10.1007/s11104-020-04449-9
You C, Wu F, Yang W, Xu Z, Tan B, Zhang L, Yue K, Ni X, Li H, Chang C, Fu K (2018) Does foliar nutrient resorption regulate the coupled relationship between nitrogen and phosphorus in plant leaves in response to nitrogen deposition? Sci Total Environ 645:733–742. https://doi.org/10.1016/j.scitotenv.2018.07.186
Yu ZC (2011) Holocene carbon flux histories of the world’s peatlands: global carbon cycle implications. Holocene 21:761–774. https://doi.org/10.1177/0959683610386982
Yu LL, Zhang XH, Shi FX, Mao R (2022a) Effect of shrub encroachment on leaf nutrient resorption in temperate wetlands in the Sanjiang Plain of Northeast China. Ecol Processes 11:67. https://doi.org/10.1186/s13717-022-00413-w
Yu L, Huang Z, Li Z, Korpelainen H, Li C (2022b) Sex-specific strategies of nutrient resorption associated with leaf economics in Populus euphratica. J Ecol 110:2062–2073. https://doi.org/10.1111/1365-2745.13952
Yuan ZY, Chen HYH (2015) Negative effects of fertilization on plant nutrient resorption. Ecology 96:373–380. https://doi.org/10.1890/14-0140.1
Zhan XY, Yu GR, He NP, Fang HJ, Jia BR, Zhou M, Wang C, Zhang J, Zhao G, Wang S, Liu Y, Yan J (2014) Nitrogen deposition and its spatial pattern in main forest ecosystems along north-south transect of eastern China. Chin Geogra Sci 24:137–146. https://doi.org/10.1007/s11769-013-0650-5
Zhang YJ, Yang QY, Lee DW, Goldstein G, Cao KF (2013) Extended leaf senescence promotes carbon gain and nutrient resorption: importance of maintaining winter photosynthesis in subtropical forests. Oecologia 173:721–730. https://doi.org/10.1007/s00442-013-2672-1
Zhang Y, Yang GS, Shi FX, Mao R (2021) Biomass allocation between leaf and stem regulates community-level plant nutrient resorption efficiency response to nitrogen and phosphorus additions in a temperate wetland of Northeast China. J Plant Ecol 14:58–66. https://doi.org/10.1093/jpe/rtaa077
Zhang JL, Zhang SB, Chen YJ, Zhang YP, Poorter L (2015) Nutrient resorption is associated with leaf vein density and growth performance of dipterocarp tree species. J Ecol 103:541–549. https://doi.org/10.1111/1365-2745.12392
Acknowledgements
We thank Dr. Hui-Min Chen, Ya-Lin Hu, Gui-Gang Lin, and Xin-Hou Zhang for the field sampling and chemical analyses, and the editor and reviewers for the constructive suggestions. This study was financially supported by National Natural Science Foundation of China (No. 315704791), the Double Thousand Plan of Jiangxi Province (jxsq2018106044), and the Research Project of Jiangxi Forestry Bureau (No. 202203).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
We declare that we do not have any commercial or associative interest that represents a conflict of interest in connection with the work submitted.
Competing interests
The authors have no relevant financial or non-fnancial interests to disclose.
Additional information
Responsible Editor: Raúl Ochoa-Hueso.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, R., Wu, PP., Peng, C. et al. Shifted plant composition predominantly controls nitrogen addition effect on community-level leaf nutrient resorption in a boreal peatland. Plant Soil 494, 321–331 (2024). https://doi.org/10.1007/s11104-023-06280-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-023-06280-4