Abstract
Purpose
Biotic and abiotic properties of soils can hinder or facilitate ecological restoration, and management practices that impact edaphic factors can strongly influence plant growth and restoration outcomes. Salvaged topsoil is an invaluable resource for mine-site restoration, and a common practice is topsoil transfer from mined areas to restoration sites. However, direct transfer is often not feasible, necessitating storage in stockpiles. We evaluated the effects of topsoil stockpiling on plant performance across diverse ecosystems impacted by mining throughout Western Australia.
Methods
We conducted a bioassay experiment using a widespread native Acacia species to assess how topsoil storage might impact plant growth, physiology, and nodulation by N-fixing bacteria using soils from native reference vegetation and stockpiled soils from six mine sites across Western Australia.
Results
Plant responses varied across mine sites, but overall plants performed better in soils collected from native vegetation, exhibiting greater biomass, more root nodules, and higher water-use efficiency compared to those grown in stockpiled soils. Soil physiochemistry showed few and minor differences between native soils and stockpiles.
Conclusion
Results strongly suggest observed differences in plant performance were biotic in nature. This study highlights the negative effects of topsoil storage on the biological integrity of soil across diverse ecosystems, with important implications for mine-site restoration; our results show that topsoil management can strongly influence plant performance, and stockpiled soils are likely inferior to recently disturbed topsoil for restoration purposes. We also use this study to illustrate the utility of bioassays for assessing soil quality for ecological restoration.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The biological, chemical, and physical properties of soil are critical determinants of plant performance (Gregory and Nortcliff 2013), and soils form the foundation of terrestrial restoration projects (Heneghan et al. 2008; Stanturf and Callaham 2021). Soils disturbed or altered by human activities can severely inhibit the ability of plants to germinate, grow, survive, and reproduce. For example, insufficient nutrient availability, low water-holding capacity, extreme pH, altered soil structure, or high concentrations of toxic contaminants may pose a challenge to plant growth and survival. Plants also depend on a variety of symbiotic and free-living soil microbes and other belowground biotic interactions (Coleman et al. 2017; Van Der Heijden et al. 2008). The success of some plant species, such as those that form associations with specific mycorrhizal fungi or nitrogen- (N) fixing bacteria, may even be entirely dependent on these mutualistic relationships. Conversely, soil pathogens can negatively impact plant performance or even result in plant death. Therefore, the biotic and abiotic properties of soils can both hinder or facilitate the ecological restoration of self-sustaining plant communities (Kardol and Wardle 2010; Stanturf and Callaham 2021).
Considerations of soil properties are important for any plant restoration program (Stanturf and Callaham 2021), but this is especially true for restoration sites impacted by mining operations (Bradshaw 1997; Cooke and Johnson 2002). Mining represents one of the most severe anthropogenic disturbances on ecosystems, often involving the wholesale removal of vegetation and soil (Maus et al. 2020; Walker and Willig 1999). As reservoirs of seeds, soil biota, and nutrients, the value of topsoil for restoration following mine closure is widely appreciated, and a common practice during mining operations is to salvage stripped topsoil and transfer it to recipient sites requiring restoration (Fowler et al. 2015; Koch 2007). Oftentimes, direct transfer of topsoil is not feasible, and instead it is stored in stockpiles to be used for subsequent restoration activities (Strohmayer 1999), with storage times ranging from weeks, to years, to decades.
The act of stripping and stockpiling may negatively impact soils in multiple ways, for example by disturbing soil structure and aggregation, reducing soil organic carbon, increasing compaction, decreasing belowground biodiversity, and altering soil microbial community composition and abundance (Anderson et al. 2008; Birnbaum et al. 2017; Block et al. 2020; Bulot et al. 2017; Harris et al. 1989). Over longer periods of storage, soil biota that depend on plant carbon inputs (e.g., through rhizodeposition or direct interactions with plant roots) may be lost from the system unless stockpiles are revegetated, either actively or passively (Eisenhauer et al. 2017; Rillig 2004; Sheoran et al. 2010). Soil microorganisms play a critical role in soil development and plant nutrition, growth, and stress tolerance, ultimately contributing to plant community diversity and productivity (Rillig 2004; Van Der Heijden et al. 2008). Therefore, if mining activities compromise the biological integrity of topsoils, this could severely limit the potential for successful restoration.
While many different approaches have been proposed to evaluate “soil health” to inform restoration (Hart et al. 2020), including in a mining context (Kumaresan et al. 2017; van der Heyde et al. 2020), a key test of soil quality for restoration purposes is the ability to support plant growth. Long-term monitoring of restored vegetation is necessary for evaluating the ability of soils to support self-sustaining plant communities, and monitoring data from one site can inform future restoration projects. Obviously, however, this approach is unable to provide a priori information on how soils impacted by mining activities, topsoil storage, or various biological, organic, or inorganic amendments will influence plant performance and therefore the potential for successful restoration at a given site. Plant bioassay experiments (also referred to as “phytometer” experiments) are one method employed to gain information on the influence of soil treatments on plant performance (Dietrich et al. 2013). When appropriately designed (Dale and Beyeler 2001; Dietrich et al. 2013), such experiments have the potential to provide key insight into the ability of soils to support plant growth and restoration success (Table 1).
In this study, we utilized a bioassay approach to evaluate the effects of topsoil stockpiling on plant growth and physiological performance using soils collected from intact native vegetation and topsoil stockpiles from diverse ecosystems across Western Australia (Fig. 1). Western Australia is one of the most productive mining areas in the world (Maxwell 2018) as well as a global hotspot of biodiversity and species endemism (Beard et al. 2000; Broadhurst and Coates 2017), necessitating wide-scale ecological restoration. Our goal was to understand how mining activities might compromise the ability of soils to support plant growth and ecological restoration. Essentially, our experiment allowed us to compare the effects of direct topsoil transfer versus stockpile storage on subsequent plant performance. We also evaluated the effect of stockpile age on plant growth and physiology. We hypothesized that soils impacted by storage in stockpiles would be inferior to those from previously undisturbed native reference vegetation, resulting in reduced plant growth and physiological performance. We also use this study to explore how plant bioassay experiments such as this can inform mine-site restoration. We discuss the important limitations of such an approach and provide recommendations for experimental design, mining operations, and restoration practitioners in an effort to improve science-based strategies for topsoil management.
We assessed the biological integrity of topsoil for mine site restoration through a plant bioassay experiment. In a controlled glasshouse experiment (a), we evaluated effects of stockpiled topsoil and soils collected from previously undisturbed native reference vegetation on plant physiological performance (b), above- and belowground measures of plant growth (c), and nodulation by N-fixing rhizobia (d)
Materials and methods
Study sites and soil collection
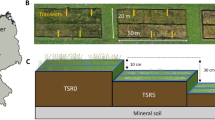
We selected six mine sites from across Western Australia, representing a range of soil types, climates, and ecosystems (Fig. 2). Sites included a sand mine in the Banksia woodlands of the Mediterranean-climate coastal sandplains (Fig. 2a), two iron ore mines in the arid savannas of the Pilbara region (Fig. 2b and c), a copper-cobalt-nickel mine in the Great Western Woodlands (Fig. 2d), a nickel mine in the semi-arid shrublands in the Goldfields region (Fig. 2e), and a mine (anonymity requested – commodity undisclosed) in the Jarrah Forest dominated by Eucalyptus marginata (Fig. 2f). At each mine, we selected three areas of native reference vegetation and four topsoil stockpiles of different ages for sampling. Operating procedures at each mine differed, and there was variation in topsoil management and storage approaches within and across mines (Table S1). Stockpile age ranged from less than one year to 29 years, and the range of storage times differed by site. At all mine sites, stripping depth was restricted to the upper topsoil and did not include subsoil horizons. Storage methods included one of two approaches; soil was stored in either large, continuous flat-topped stockpiles, or in paddock dumps consisting of smaller deposited mounds over a given area. Generally, storage methods were consistent within mines across stockpiles, with some exceptions (Table S1). Stockpile height varied within and across mines and ranged from 1 to 6 m, with most stockpiles 2–3 m. Due to this variation in storage methods, we restricted our soil sampling to the upper horizon of stockpiles (and in native reference vegetation).
Location of study sites where soil were collected throughout Western Australia (left), including a sand mine in the Banksia woodlands of the Mediterranean-climate coastal sandplains (a), two iron ore mines in the arid Pilbara region (b and c), a copper-cobalt-nickel mine in the Great Western Woodlands (d), a nickel mine in the semi-arid shrublands of the Goldfields region (e), and a mine (commodity undisclosed – anonymity requested) in the Jarrah Forest dominated by Eucalyptus marginata (f)
Sampling was completed between December 2018 and February 2019. At each reference site and stockpile, we randomly placed five 5 m × 5 m plots for soil sampling. Within each of these plots, we collected five replicate soil samples from the top 0–20 cm of the soil profile using a 5 cm diameter soil auger or a trowel (depending on ease of coring). The five soil samples collected at each location (reference or stockpile) were pooled and homogenised to produce one composite sample per plot (approximately 500 g), with five replicates (each consisting of these pooled samples) total for each collection location (either native reference site or stockpile). We were unable to include multiple replicate stockpiles of the same age class within each mine. Sample collection equipment was sterilised with bleach solution in between sample collections to prevent cross contamination. Air-dried subsamples of approximately 160 g of soil from each collection plot (either from native reference vegetation or from topsoil stockpiles) were pooled for use in plant growth assays. Soils were stored dry at ~20 °C prior to potting in September 2019.
Soil analysis
Soil samples were analyzed at the CSBP Soil and Plant Analysis Laboratory (Bibra Lake, Western Australia; Rayment and Lyons 2012), for the following: bulk density, pH (in H2O), electrical conductivity, organic carbon (Walkley-Black method; Walkley and Black 1934), potassium (Colwell method; Colwell 1965), phosphorus (Colwell method; Colwell 1965), ammonium (NH4+) and nitrate (NO3−) nitrogen (extracted using 2 M potassium chloride solution), available boron (extracted using 0.01 M calcium chloride), trace elements (DTPA extractable copper, zinc, manganese, and iron), and exchangeable cations (calcium, magnesium, sodium, potassium, aluminium). A total of five replicate samples per stockpile and reference site were characterised for each mine site.
Plant bioassay experiment
We selected the native plant species Acacia saligna (Labill.) H.L.Wendl. (Fabaceae) for our experiment. We chose this species because it is widespread throughout Australia (including across our study sites), it germinates readily and grows rapidly, it is commonly used in restoration projects in the region, and it has been previously used as a focal species in other experiments exploring plant responses to soil stockpiling (Birnbaum et al. 2017; Jasper et al. 1989). This species also forms associations with both arbuscular mycorrhizal fungi and N-fixing bacteria (Birnbaum et al. 2017).
We initiated the plant growth experiment in September 2019. We prepared square pots (5 cm width × 12 cm height) by placing squares of synthetic fabric at the bottom, followed by a layer of steam-pasteurized river sand (to improve drainage) and then field-collected soil samples (~160 g soil from the pooled soil mix from each collection site). An additional layer of pasteurized sand was added to the top of each pot to minimize pot-to-pot cross contamination across soil treatments. For each of the six mine sites, we included five replicate pots for each soil source, which included three reference sites and four stockpiles of different ages.
Seeds of A. saligna were acquired from Nindethana Seed Company, Albany, Western Australia. Prior to sowing, seeds were subjected to a dormancy-breaking treatment by soaking them for 90 s in water just below boiling temperature. We seeded pots with five seeds each, and following germination thinned pots to a final density of one seedling per pot (selecting seedlings of similar size to remain in each pot). Pots were watered daily until seedlings emerged (about one week for all pots) and every 1–3 days thereafter. We grew plants in a controlled glasshouse environment at University of Western Australia’s Plant Growth Facility. Mean daytime and night-time temperatures were approximately 22 °C and 18 °C, respectively. Relative humidity was maintained at approximately 60%. An overhead shade screen (~60% light transmission) automatically deployed if PAR exceeded 1650 μmol m−2 s−1 or if temperature exceeded ~15 °C.
Physiological measurements
Physiological measurements were taken on fully mature phyllodes (leaf-like modified petioles found in many Acacia species, hereafter referred to as “leaves”). After 18 weeks of plant growth, we measured leaf gas exchange (including photosynthetic rate, stomatal conductance, and transpiration) using a LI-6400XT portable open system (LI-COR, Inc.) equipped with a standard leaf chamber with LED light source and CO2 injector system. Measurements were made at PAR of 1500 μmol m−2 s−1 sufficient to saturate leaf photosynthesis and reference CO2 at 400 μmol CO2 mol−1 air, similar to ambient conditions. Measurements were taken at flow rates between 100 to 500 μmol s−1 according to the plants’ physiological state so as to obtain reliable readings. Measurements were made on the youngest fully mature phyllode of each plant, with three replicate plants measured for each treatment group. We also measured plant chlorophyll fluorescence (Fv/Fm), a measure of the efficiency of Photosystem II) using a handheld chlorophyll fluorometer (Pocket PEA, Hansatech Instruments Ltd.). Measurements were taken at PAR 3500 μmol m−2 s−1 from the same phyllodes used for gas-exchange measurements after at least 30 min of dark adaptation in leaf clips.
Plant harvest
We harvested plants in February 2020 after approximately 20 weeks of growth. We clipped stems at the base and separated aboveground biomass into true leaves, phyllodes, and stems. We scanned all leaves and phyllodes using a flatbed scanner, and processed images using ImageJ software (Schneider et al. 2012) to determine leaf area (including true leaves and phyllodes). Roots were carefully removed from soil and washed with tap water. We recorded the number of effective nodules present (based on size and pink pigmentation, which indicates active rhizobia). Following the scanning of leaves and assessment of nodulation, all plant material was dried in an oven for at least 48 h at 70 °C. We then determined the dry weight of leaves, phyllodes, stems, and roots for each plant. From these data, we calculated total, shoot, and root biomass, specific leaf area (SLA; cm2 g−1), and root mass ratio (RMR; g root g−1 total biomass). Single, newly mature whole-leaf samples were ground using a ball mill and then analysed for stable isotope ratios of C and N (δ15N and δ13C) and total C and N concentration using a continuous flow system consisting of a Delta V Plus mass spectrometer connected with a Thermo Flush 1112 via Conflo IV (Thermo-Finnigan). All isotopic analyses were performed by the West Australian Biogeochemistry Centre at the University of Western Australia.
Statistical analyses
We took several statistical approaches to evaluate the effects of topsoil stockpiling on plant performance. All analyses were conducted using R version 3.6.2. Our aims were to (1) compare plant performance in native reference soils versus stockpiled soils and (2) evaluate potential effects of stockpile age.
We first evaluated differences between plants grown in native reference soils and soils from stockpiles (irrespective of age) across all mine sites using linear mixed effect models. We used the ‘lme’ function in the R package ‘nlme’. We ran individual statistical models for each plant growth parameter, including root, shoot, and total biomass, RMR, number of root nodules, leaf area, SLA, leaf N, C/N ratio, δ15N, δ13C, fluorescence (Fv/Fm), photosynthetic rate, stomatal conductance (gs), transpiration (E), and instantaneous water-use efficiency (WUE). For each model, we included sampling plot and mine site as random effects, with plot nested within mine site. We also explored potential differences in soil properties between native reference soils and soils from stockpiles across all mine sites, including soil texture (i.e, percent sand, silt and clay), bulk density, organic C, pH, conductivity, soil nutrients, and trace elements, using the same approach. To test the effects of stockpile age on plant performance, we ran additional linear mixed effect models for each measure of plant performance, using data only from plants grown in soil stockpiles. We log- or square root-transformed data as needed and ensured that residuals were normally distributed for each model.
We then explored these relationships separately for each mine site, as our analyses indicated that plant performance varied greatly by mine site, and the results of these analyses are especially relevant for topsoil management practices at each individual mine. To evaluate differences among plants grown in soil collected from native reference vegetation and topsoil stockpiles of different ages, we employed one-way analysis of variance (ANOVA) using the ‘aov’ function in the R package ‘agricolae’. In light of unequal sample sizes across treatments, we specified a Type III Sums of Squares. For each mine, we analysed the effect of soil source (i.e. soil from different reference sites and stockpiles) on measures of plant performance individually. We used log and square root transformations as needed to meet assumptions of ANOVA. Data for RMR was arcsine transformed. Where ANOVA models indicated a significant effect of soil source on a given parameter, we used Tukey’s Honestly Significant Difference (HSD) test for post-hoc mean comparisons using the ‘HSD.test’ function. Next, we used linear regression to evaluate the potential effect of stockpile age on different measures of plant performance using the ‘lm’ function. For these analyses, we included only data from plants grown in soils from stockpiles.
Our analyses revealed significant differences in plant WUE and the number of root nodules in plant grown in native reference soils versus those grown in stockpiled soils. Both of these parameters could be influenced by plant size; larger plants may have more root biomass for rhizobial infection, and greater shoot biomass may result in higher water demand and therefore greater WUE. Therefore, we ran additional analysis of covariance (ANCOVA) models with soil source (either native reference soils or stockpiled soils) as a main effect, and shoot mass (for plant WUE) and root mass (for nodule count) as covariates.
Given observed differences between plants grown in native reference soils and soils impacted by stockpiling, we further explored differences in overall plant performance using principal components analysis (PCA) of all trait data by each individual mine site. Traits included in these analyses were number of root nodules, root mass, shoot mass, RMR, leaf area, SLA, δ15N, δ13C, leaf C/N ratio, leaf fluorescence, photosynthetic rate, stomatal conductance, transpiration, and WUE. In light of some missing data for individual plants, we used the ‘imputePCA’ function in the package ‘missMDA’ to impute missing values. We ran each PCA using the resulting data matrix with complete observations using the function ‘prcomp’.
Results
While plant performance varied greatly by mine site, we observed several clear patterns of plant growth and performance across mine sites due to soil source (Fig. 3). Across all mine sites, multiple metrics of plant growth and performance showed significant differences between native reference soil and topsoil stockpiles, with plants grown in soil from native vegetation outperforming those grown in stockpiled soil. Specifically, when analysed across all mine sites, plants grown in native soils exhibited greater root, shoot, and total biomass, leaf area, nodule numbers, and leaf WUE compared to plants grown in soils from stockpiles (Table 2). Results from ANCOVA models indicated that soil source (F = 9.10, p = 0.0031) but not shoot mass (F = 0.03, p = 0.8660) had an effect on plant WUE, indicating the negative effect of soil stockpiling on plant WUE was independent of plant size. The number of root nodules per plant was significantly affected by soil source (F = 49.58, p < 0.0001) and root biomass (F = 130.29, p < 0.0001); plants with greater root biomass had a greater number of nodules, but overall plants grown in native soils had more nodules than those grown in stockpiled soils independent of plant size.
We also evaluated potential differences in soil physiochemical properties between soils from native vegetation and stockpiles, with few and minor differences observed. Field-collected soils from stockpiles tended to have slightly higher bulk density (p = 0.0111), more available nitrate (p = 0.0002), and slightly lower concentrations of iron (p = 0.0208).
Across all mine sites, stockpile age had no effect on any of the plant traits measured, except nodule counts, where plants grown in soil from older stockpiles tended to have fewer root nodules (Table 2). Despite these overall patterns across mine sites, plants also exhibited very site-specific responses. For example, in half of the mine sites, plants grew significantly larger in native reference soils than in soil from stockpiles, and in four of the six mine sites plants grown in native soils had a greater number of root nodules compared to those grown in stockpiled soil (Table 3, Fig. 4). Other measures of plant performance showed few differences due to soil source, with no consistent patterns across mine sites (Table 3).
In addition to evaluating differences in plant performance in reference soils versus stockpiled soils more broadly across and within mine sites, we also tested for differences in total plant biomass due to soil source at each mine site (i.e., in soils from the three areas of native vegetation and four stockpiles of different ages at each mine site) using ANOVA. Plant growth showed significant differences due to soil source at four of the six mine sites (Fig. 5; Mines b, c, e, and f). At each of these sites, total plant biomass was greatest in native reference soils. Patterns of plant growth in soil stockpiles differed by mine site, with some observed differences due to stockpile age. For example, at Mine e, the greatest plant biomass was observed in soils from the oldest stockpile.
Total plant biomass of plants grown in native reference soils (three reference sites per mine site) and topsoil stockpiles of different ages (four stockpiles per mine site) from each of six individuals mine sites (a-f) throughout Western Australia. Results of one-way ANOVA models with source plot as a fixed effect are shown, including F statistics and level statistical significance. (N.S. = not significant, *p ≤ 0.05, **p ≤ 0.01, ***p ≤ 0.001). Different letters above bars indicate significant difference from post-hoc mean comparisons from individual Tukey HSD tests. Dashed lines show mean total biomass of all plants grown in native reference soils
Finally, principal components analysis of all plant traits (by mine site) showed clear differences in overall plant performance in those grown in native soils versus those grown in soils impacted by stockpiling at several sites (Fig. 6). Specifically, we observed distinct separation in trait space in plants grown in native reference and stockpiled soils at Mines b, c, and f, and to a lesser extent, mine d.
Principal components analysis of all plant traits (including number of root nodules, root mass, shoot mass, RMR, leaf area, SLA, δ15N, δ13C, leaf C/N ratio, leaf fluorescence, photosynthetic rate, stomatal conductance, transpiration, and WUE) of plants grown in native reference soils and stockpiled topsoil at six mine sites (a-f). The proportion of variance explained by the first and second principal components is shown on each axis
Discussion
Our results highlight the utility of plant bioassays to evaluate the ability of soils to support plant growth and, potentially, ecological restoration efforts. Our aim was to understand if such an approach could detect broadscale impacts of mining activities on the biological integrity of soils across diverse ecosystem types of Western Australia. Consistent with our initial hypothesis, we found soils impacted by stockpiling to be inferior to soils from previously undisturbed native vegetation in their ability to support plant growth; overall, plants grown in stockpiled topsoils were smaller, had fewer root nodules formed by N-fixing bacteria, and exhibited lower WUE than those grown in native soils. These results strongly suggest that direct soil transfer from mined areas to restoration sites is preferable to storage in stockpiles, as shown by improved plant performance in the recently collected native reference soil compared to soils from stockpiles. We also evaluated potential impacts of the duration of soil storage time on plant performance. While we observed no clear patterns in measures of plant growth or physiology due to stockpile age across mine sites, we did detect an overall negative effect of soil storage time on the number of root nodules. These results have important implications for topsoil management and mine-site restoration, but, as we discuss below, there are also key limitations to what information experiments such as this are able to provide practitioners.
Effects of topsoil stockpiling
Our results strongly suggest that the topsoil storage may negatively impact the biological integrity of native topsoil, as evidenced by the reduced plant growth, lower plant WUE, and fewer root nodules (independent of plant size) in soils from stockpiles across all mine sites. Severe soil disturbances, including mining, are known to negatively impact soil biota important for plant growth and stress tolerance, including mycorrhizal fungi and N-fixing bacteria (Gorzelak et al. 2020; Harris et al. 1989, 1993). For example, Jasper et al. (1987) reported reduced mycorrhizal potential in soils disturbed by stockpiling compared to soils from native vegetation. Rhizobia are thought to be more resilient to disturbance than other soil microorganisms (Jasper 2007), but our results clearly showed reduced nodulation in plants grown in stockpiled soil from several of the mine sites evaluated. It is possible that these effects are site- and species-specific and also depend on the depth and age of stockpiles. For example, failure to establish N-fixing symbiosis is not thought to occur in legumes grown in soils impacted by bauxite mining in the jarrah forests of southwestern Australia (Jasper 2007), but negative impacts of stockpiling on rhizobia have been reported at other mine sites (Gorzelak et al. 2020; Jasper 2007). It is possible that soil biota will recover over time in soil stockpiles, particularly if they are revegetated (Banning et al. 2008; Jasper et al. 1987; Jasper 2007; Sheoran et al. 2010). For example, Birnbaum et al. (2017) found that mycorrhizal colonization of A. saligna was greater in soils from older (10 year) stockpiles compared to younger stockpiles. It is worth noting, however, that this greater mycorrhizal colonization did not result in improved plant growth, suggesting other biotic interactions, such as the loss of N-fixing bacteria, may continue to constrain plant growth in stockpiled soils and impede restoration (Birnbaum et al. 2017; Gorzelak et al. 2020).
Physiological parameters
Notably, despite clear differences in growth measures (e.g., biomass, leaf area) and nodulation between plants grown in native versus stockpiled soil, we observed few differences in the measured physiological parameters. This is likely in part due to the fact that plants were well-watered throughout the experiment. Plants therefore experienced only low levels of water stress between watering, which could have influenced the formation of relationships with soil microorganism. Furthermore, single instantaneous measurements (e.g., plant gas exchange), especially those done at times when rates are highest, may not always reveal longer-term differences in plant functioning. Soil biota play an important role in plant stress tolerance (Kim et al. 2012; Valliere et al. 2020), and it is possible that if we had manipulated water availability or other stressors, we would have observed more pronounced differences in physiological traits. Interestingly, one of the few physiological differences we did observe between plants grown in native reference soils and soils from stockpiles was an increase in leaf WUE in native reference soils. Plants grown in native soils were generally larger and had greater leaf area than those grown in stockpiled soils, conceivably resulting in greater water demand and higher WUE. While plants were regularly watered to field capacity, larger plants may have reduced soil water availability more rapidly, resulting in periods of more conservative water use and lower stomatal conductance (and greater leaf WUE). However, we detected a significant effect of soil source on WUE even when accounting for plant size (and shoot mass had no significant effect on plant WUE in ANCOVA models).
Symbiotic soil biota, including both mycorrhizal fungi and N-fixing bacteria are known to increase plant WUE through a variety of direct and indirect mechanisms (Kim et al. 2012; Ruiz-Lozano and Aroca 2010). For example, improved plant nutrition due to soil mutualists could allow for greater photosynthetic rates at the same level of stomatal conductance, resulting in greater WUE. We did not detect any significant differences in leaf N across plants grown in native references soils and soil stockpiles, but it is possible that other nutrients not measured (e.g., phosphorus) or other plant-microbial signalling effects are responsible (Aroca and Ruiz-Lozano 2009), and the underlying mechanisms of this result will be an interesting avenue for future research. Nevertheless, it appears likely that soil biota in native reference soils can improve plant physiological performance, and soil stockpiling may negatively impact these belowground mutualistic relationships, resulting in reduced WUE. This phenomenon could be especially important for dryland restoration, including in Western Australia’s arid and semi-arid ecosystems, where water limitation is a major challenge for restoration and improved drought-tolerance could increase restoration success (Muñoz-Rojas et al. 2016; Valliere et al. 2019).
Soil abiotic properties
Across mine sites, we observed few differences in soil physiochemistry between native reference sites and soil stockpiles, which strongly suggests that observed plant responses in the different soil sources were biotic in nature. In general, soils from stockpiles had a greater bulk density than those from native reference sites. This is likely due to a loss of soil structure and compaction during stripping and stockpiling, which could impede water infiltration and root penetration (Rokich et al. 2001; Strohmayer 1999). It is possible this change in bulk density could impact plant growth, and soil compaction is indeed a challenge for plant establishment during mine site restoration (Duncan et al. 2020; Koch 2007; Rokich et al. 2001). However, it is unlikely that this effect is fully responsible for observed differences in our experiment given that soils were all sieved prior to potting (further disturbing soil structure), plants were continuously well-watered, and we observed no evidence that root penetration was impeded in any of the pots. Soils from stockpiles also tended to have higher available N in the form of soil nitrate, possibly due to N mineralization during the decomposition of organic matter. This higher N availability, however, did not appear to result in improved plant performance, as we found plant growth was consistently lower in soils from stockpiles relative to native reference soils. This could be due in part to less reliance on soil N, as this species derives microbially-fixed N from rhizobial symbiosis.
Effects of stockpile age
We did not observe any clear patterns in plant performance due to topsoil storage time across mine sites, with one exception: stockpile age appeared to have an overall negative effect on the number of root nodules. In addition to impacting the growth and performance of native legumes, reduced rhizobial activity in disturbed soils could have important implications for nutrient cycling and the establishment of other non-legume species in post-mining restoration sites (Koch 2007; Ward 2000). The success of these N-fixing pioneer species, like A. saligna, could therefore be important for facilitating plant community development over time as well as to restore ecosystem services in areas impacted by mining activities (Grant et al. 2007). Others have reported a negative effect of soil storage time on plant growth, likely due to effects of stockpiling on soil biota. For example, in a similar study exploring the effects of stockpiling at a single mine site, Birnbaum et al. (2017) reported a negative effect of storage time on plant biomass, which could be attributed to the absence of Bradyrhizobium in older stockpiles (Gorzelak et al. 2020). Consistent with our results, the authors also found that plants grown in the oldest stockpiles (ten years) had much lower nodule biomass than those grown in younger stockpiles, suggesting that in some contexts, the activity or abundance of N-fixing bacteria may decline over time in soil stockpiles (Birnbaum et al. 2017; Gorzelak et al. 2020). At some mines evaluated in this study, there appeared to be a positive trend of increased plant growth with stockpile age, which could be the result of recovery of some beneficial soil biota not directly assessed here. Such recovery could be the result of either passive or active revegetation of stockpiles over time, and this warrants future investigation to determine best practices for managing topsoils that must be stored prior to the initiation of restoration (as opposed to direct transfer). Finally, it should be noted that we were unable to include multiple stockpiles of the same age from each mine site, and this inevitably limited our evaluation of the effect of stockpile age on plant performance metrics.
Practitioner recommendations
Topsoil stripping and subsequent storage is a necessary and unavoidable component of mining operations (Strohmayer 1999). In order to facilitate science-based, adaptive management for mine closure and restoration, mining operations should keep detailed records on when and how topsoil stripping occurred, when storage began, and how stockpiles were formed. This will enable further studies to evaluate potential deleterious effects of mining operations on soil quality for restoration in order to develop standard practices for stripping and storage. Monitoring stockpiles throughout the storage period to quantify patterns of passive or active plant re-colonization on stockpiles (prior to subsequent soil transfer and restoration activities), including the identity and abundance/cover of plant species and soil properties, will also yield useful data that could inform future management practices. Once restoration activities are initiated following mine closure, records should be kept indicating which soils were deployed at each restoration site. This will allow for future comparisons of restoration trajectories based on the specific history of topsoil management. While these activities will require more careful planning, time, and resources, it is essential for understanding how topsoil handling impacts restoration success and adapting future management practices. Furthermore, the cost of this monitoring and data curation is likely minor relative to the cost of poor restoration outcomes. Sharing of information across mine sites and different mining corporations will be especially useful for the development of standardized best practices for topsoil management, restoration, and monitoring.
A strength of the work presented here is the inclusion of multiple mine sites from diverse ecoregions throughout Western Australia, and we identified some important patterns across mine sites that have implications for topsoil management and restoration. However, a key finding of this study is that plant responses varied greatly across mine sites, suggesting that the impacts of soil stockpiling and topsoil management activities are highly context-dependent and site-specific. Such variation is likely due to differences in soil biology and physiochemistry and environmental conditions across the different biomes explored here. Therefore, topsoil management practices that promote positive restoration outcomes at one site may not be universally effective, and site-specific evaluations of the ability of soils to support plant growth are needed to develop best practices.
Despite the site-specific variation observed here, direct return of topsoil to a site immediately after clearing and stripping is likely the best approach for mine-site restoration. Even short-term storage can result in significant declines in the number of viable propagules within and shifts in species composition of the soil seed bank compared to fresh topsoil (Golos et al. 2016; Koch et al. 1996; Rokich et al. 2000). Our results provide further evidence that direct soil transfer from mined areas to restoration sites is likely the best way to ensure the biological integrity of soils and to support plant growth. It is worth noting that in our experiment, plants grown in the youngest stockpiles (even those stored for less than a year) often exhibited inferior growth and performance compared to those grown in recently collected soil from intact native vegetation. This demonstrates that even short-term storage could negatively impact soil biota that are important for plant growth. While much research has focused on the potential role of soil microbial communities in restoration (Kardol and Wardle 2010), the level to which this compromised biotic activity actually limits restoration remains unknown.
How can practitioners restore the biological integrity of soils for restoration? Reintroducing soil biota through microbial inoculations may be one potential solution. For example, Jasper et al. (1989) found that inoculation with arbuscular mycorrhizal fungi increased plant growth and phosphorus uptake of Australian Acacia species in stockpiled topsoil. While there is growing interest in the use of commercially available inoculants in restoration, these products are likely not as effective as indigenous soil biota (Maltz and Treseder 2015; Moreira-Grez et al. 2019; Valliere et al. 2020), and this is an important avenue for future research.
Limitations: What can’t plant bioassays tell us?
The use of ecological indicators, including plant bioassay experiments, can provide key insights into the ability of soils to support restoration activities (Dale and Beyeler 2001; Dietrich et al. 2013). This approach may identify early warning signs of restoration failure and be useful for diagnosing specific deficiencies in soils. However, these experiments are also limited in what information they can provide regarding how plants will respond in situ. For example, plants in the field will be subject to a variety of biotic and abiotic stressors not accounted for under controlled glasshouse conditions. Conditions experienced by plants in a potted greenhouse study may also not be analogous to field conditions. For example, in our study design, we topped pots with sterile sand to minimize cross contamination across pots. This approach could have interfered with microorganisms found in biological soil crusts, however, which could play an important role in dryland restoration (Antoninka et al. 2020). Furthermore, the timing (e.g., seasonality) and depth of soil collection for such experiments could influence results due to spatiotemporal differences in soil biota activity and abundance. Short-term pot studies may also fail to capture how plants will perform beyond the seedling stage. Plant responses to soil conditions are also very species-specific, and the most powerful experiments will be those that examine the responses of diverse suites of native species and that include a variety of lifeforms and life-stages (Dietrich et al. 2013). Combining potted and greenhouse experiments with field studies will also be useful for evaluating how well plant performance under controlled conditions relates to performance in the field. Finally, an inherent limitation of experiments such as this one is that the identity of the soil biota driving observed responses are concealed by the “black box” (Cortois and De Deyn 2012). For example, the species examined here is known to form relationships with mycorrhizal fungi (Birnbaum et al. 2017), but we did not assess fungal colonization of roots. Advances and greater accessibility to methods in DNA sequencing may also help overcome this challenge, as has been done in similar studies exploring these dynamics (Gorzelak et al. 2020).
Conclusions
This study adds to the body of literature evaluating the deleterious effects of mining activities on the biological integrity of soils. Oftentimes restoration efforts are singularly focused on restoring aboveground (i.e., plant communities) diversity and structure, but a more explicit consideration of soil ecological impacts could improve topsoil management and post-mining restoration success. The use of ecological indicators, such as through plant bioassays, may provide important insight into the ability of disturbed soils to support plant growth and sustainable restoration.
Data availability
Upon publication, data files associated with this project will be uploaded to the Dryad data repository (https://datadryad.org).
Code availability
All R scripts used for data analysis will be made available upon request. All R packages and functions are described in the Methods section of the paper.
References
Anderson JD, Ingram LJ, Stahl PD (2008) Influence of reclamation management practices on microbial biomass carbon and soil organic carbon accumulation in semiarid mined lands of Wyoming. Appl Soil Ecol 40:387–397
Antoninka A, Faist A, Rodriguez-Caballero E, Young KE, Chaudhary VB, Condon LA, Pyke DA (2020) Biological soil crusts in ecological restoration: emerging research and perspectives. Restor Ecol 28:S3–S8
Aroca R, Ruiz-Lozano J (2009) Induction of plant tolerance to semi-arid environments by beneficial soil microorganisms–a review. Climate change, intercropping, pest control and beneficial microorganisms. Springer
Banning N, Grant C, Jones D, Murphy D (2008) Recovery of soil organic matter, organic matter turnover and nitrogen cycling in a post-mining forest rehabilitation chronosequence. Soil Biol Biochem 40:2021–2031
Beard J, Chapman A, Gioia P (2000) Species richness and endemism in the Western Australian flora. J Biogeogr 27:1257–1268
Birnbaum C, Bradshaw LE, Ruthrof KX, Fontaine JB (2017) Topsoil stockpiling in restoration: impact of storage time on plant growth and symbiotic soil biota. Ecol Restor 35:237–245
Block PR, Gasch CK, Limb RF (2020) Biological integrity of mixed-grass prairie topsoils subjected to long-term stockpiling. Appl Soil Ecol 145:103347
Bradshaw A (1997) Restoration of mined lands—using natural processes. Ecol Eng 8:255–269
Broadhurst L, Coates D (2017) Plant conservation in Australia: current directions and future challenges. Plant Divers 39:348–356. https://doi.org/10.1016/j.pld.2017.09.005
Bulot A, Potard K, Bureau F, Bérard A, Dutoit T (2017) Ecological restoration by soil transfer: impacts on restored soil profiles and topsoil functions. Restor Ecol 25:354–366
Coleman DC, Callaham MA, Crossley D Jr (2017) Fundamentals of soil ecology. Academic press
Colwell J (1965) An automatic procedure for the determination of phosphorus in sodium hydrogen carbonate extracts of soils. Chem Industry 22:893–895
Cooke J, Johnson M (2002) Ecological restoration of land with particular reference to the mining of metals and industrial minerals: a review of theory and practice. Environ Rev 10:41–71
Cortois R, De Deyn GB (2012) The curse of the black box. Plant Soil 350:27–33
Dale VH, Beyeler SC (2001) Challenges in the development and use of ecological indicators. Ecol Indic 1:3–10
Dietrich AL, Nilsson C, Jansson R (2013) Phytometers are underutilised for evaluating ecological restoration. Basic Appl Ecol 14:369–377
Duncan C, Good MK, Sluiter I, Cook S, Schultz NL (2020) Soil reconstruction after mining fails to restore soil function in an Australian arid woodland. Restor Ecol 28:A35–A43
Eisenhauer N, Lanoue A, Strecker T, Scheu S, Steinauer K, Thakur MP, Mommer L (2017) Root biomass and exudates link plant diversity with soil bacterial and fungal biomass. Sci Rep 7:1–8
Fowler WM, Fontaine JB, Enright NJ, Veber WP (2015) Evaluating restoration potential of transferred topsoil. Appl Veg Sci 18:379–390
Golos PJ, Dixon KW, Erickson TE (2016) Plant recruitment from the soil seed bank depends on topsoil stockpile age, height, and storage history in an arid environment. Restor Ecol 24:S53–S61. https://doi.org/10.1111/rec.12389
Gorzelak M, McAmmond BM, Van Hamme JD, Birnbaum C, Thomsen C, Hart M (2020) Soil microbial communities in long-term soil storage for sand mine reclamation. Ecol Restor 38:13–23
Grant CD, Ward SC, Morley SC (2007) Return of ecosystem function to restored bauxite mines in Western Australia. Restor Ecol 15:S94–S103
Gregory PJ, Nortcliff S (2013) Soil conditions and plant growth. Wiley Online Library
Harris J, Birch P, Short K (1989) Changes in the microbial community and physico-chemical characteristics of topsoils stockpiled during opencast mining. Soil Use Manag 5:161–168
Harris J, Birch P, Short K (1993) The impact of storage of soils during opencast mining on the microbial community: a strategist theory interpretation. Restor Ecol 1:88–100
Hart MM, Cross AT, D'Agui HM, Dixon KW, Van der Heyde M, Mickan B, Horst C, Grez BM, Valliere JM, Rossel RV (2020) Examining assumptions of soil microbial ecology in the monitoring of ecological restoration. Ecol Solut Evid 1:e12031. https://doi.org/10.1002/2688-8319.12031
Heneghan L, Miller SP, Baer S, Callaham MA Jr, Montgomery J, Pavao-Zuckerman M, Rhoades CC, Richardson S (2008) Integrating soil ecological knowledge into restoration management. Restor Ecol 16:608–617
Jasper DA (2007) Beneficial soil microorganisms of the jarrah forest and their recovery in bauxite mine restoration in southwestern Australia. Restor Ecol 15:S74–S84
Jasper D, Robson A, Abbott L (1987) The effect of surface mining on the infectivity of vesicular-arbuscular mycorrhizal fungi. Aust J Bot 35:641–652
Jasper DA, Abbott LK, Robson AD (1989) Acacias respond to additions of phosphorus and to inoculation with VA mycorrhizal fungi in soils stockpiled during mineral sand mining. Plant Soil 115:99–108. https://doi.org/10.1007/BF02220699
Kardol P, Wardle DA (2010) How understanding aboveground–belowground linkages can assist restoration ecology. Trends Ecol Evol 25:670–679
Kim Y-C, Glick BR, Bashan Y, Ryu C-M (2012) Enhancement of plant drought tolerance by microbes. Plant responses to drought stress. Springer
Koch JM (2007) Restoring a jarrah forest understorey vegetation after bauxite mining in Western Australia. Restor Ecol 15:S26–S39
Koch J, Ward S, Grant C, Ainsworth G (1996) Effects of bauxite mine restoration operations on topsoil seed reserves in the jarrah forest of Western Australia. Restor Ecol 4:368–376
Kumaresan D, Cross AT, Moreira-Grez B, Kariman K, Nevill P, Stevens J, Allcock RJ, O’Donnell AG, Dixon KW, Whiteley AS (2017) Microbial functional capacity is preserved within engineered soil formulations used in mine site restoration. Sci Rep 7:1–9
Maltz MR, Treseder KK (2015) Sources of inocula influence mycorrhizal colonization of plants in restoration projects: a meta-analysis. Restor Ecol 23:625–634
Maus V, Giljum S, Gutschlhofer J, da Silva DM, Probst M, Gass SL, Luckeneder S, Lieber M, McCallum I (2020) A global-scale data set of mining areas. Scientific Data 7:1–13
Maxwell P (2018) The end of the mining boom? A Western Australian perspective. Miner Econ 31:153–170
Moreira-Grez B, Muñoz-Rojas M, Kariman K, Storer P, O’Donnell AG, Kumaresan D, Whiteley AS (2019) Reconditioning degraded mine site soils with exogenous soil microbes: plant fitness and soil microbiome outcomes. Front Microbiol 10:1617
Muñoz-Rojas M, Erickson TE, Martini DC, Dixon KW, Merritt DJ (2016) Climate and soil factors influencing seedling recruitment of plant species used for dryland restoration. Soil 2:287–298
Rayment GE, Lyons DJ (2012) New, comprehensive soil chemical methods book for Australasia. Commun Soil Sci Plant Anal 43:412–418
Rillig MC (2004) Arbuscular mycorrhizae and terrestrial ecosystem processes. Ecol Lett 7:740–754
Rokich DP, Dixon KW, Sivasithamparam K, Meney KA (2000) Topsoil handling and storage effects on woodland restoration in Western Australia. Restor Ecol 8:196–208
Rokich DP, Meney KA, Dixon KW, Sivasithamparam K (2001) The impact of soil disturbance on root development in woodland communities in Western Australia. Aust J Bot 49:169–183
Ruiz-Lozano JM, Aroca R (2010) Host response to osmotic stresses: stomatal behaviour and water use efficiency of arbuscular mycorrhizal plants. Arbuscular mycorrhizas: physiology and function. Springer
Schneider CA, Rasband WS, Eliceiri KW (2012) NIH image to ImageJ: 25 years of image analysis. Nat Methods 9:671–675
Sheoran V, Sheoran A, Poonia P (2010) Soil reclamation of abandoned mine land by revegetation: a review. Int J Soil Sediment Water 3:13
Stanturf JA, Callaham MA (2021) Soils and landscape restoration. Academic Press
Strohmayer P (1999) Soil stockpiling for reclamation and restoration activities after mining and construction. Restor Reclamat Rev 4:1–4
Valliere JM, Zhang J, Sharifi MR, Rundel PW (2019) Can we condition native plants to increase drought tolerance and improve restoration success? Ecol Appl 29:e01863. https://doi.org/10.1002/eap.1863
Valliere JM, Wong WS, Nevill PG, Zhong H, Dixon KW (2020) Preparing for the worst: utilizing stress-tolerant soil microbial communities to aid ecological restoration in the Anthropocene. Ecol Solut Evid 1:e12027
Van Der Heijden MG, Bardgett RD, Van Straalen NM (2008) The unseen majority: soil microbes as drivers of plant diversity and productivity in terrestrial ecosystems. Ecol Lett 11:296–310
van der Heyde M, Bunce M, Dixon K, Wardell-Johnson G, White N, Nevill P (2020) Changes in soil microbial communities in post mine ecological restoration: implications for monitoring using high throughput DNA sequencing. Sci Total Environ 749:142262
Walker LR, Willig MR (1999) An introduction to terrestrial disturbances. Ecosystems World:1–16
Walkley A, Black IA (1934) An examination of the Degtjareff method for determining soil organic matter, and a proposed modification of the chromic acid titration method. Soil Sci 37:29–38
Ward S (2000) Soil development on rehabilitated bauxite mines in south-West Australia. Soil Res 38:453–464
Acknowledgements
We are grateful for support provided by the Plant Growth Facilities at the University of Western Australia. We recognize the traditional owners of the land on which this research was undertaken and pay our respects to Elders past, present and emerging.
Funding
Funding to support this project was received from the Australian Research Council Industrial Transformation Training Centre for Mine Site Restoration (Project Number ICI150100041), the Research Office at Curtin University, Independence Group, Fortescue Metals Group, and a mine in South West Australia that chooses to remain anonymous. In-kind support was provided by BHP, Alcoa, and Tronox.
Author information
Authors and Affiliations
Contributions
JV, HD, KD, PN, and EV conceived of and designed the experiment. JV and HD executed the experiment, and WSW and HZ assisted with data collection and management. JV analyzed the data and wrote the manuscript. All authors provided feedback on the paper.
Corresponding author
Ethics declarations
Conflict of interest
We have no conflicts of interest to report. None of the industry partners that supported this research were involved in research activities beyond facilitating soil collection at each mine site.
Additional information
Responsible Editor: Matthew A. Bowker.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(PDF 27 kb)
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Valliere, J.M., D’Agui, H.M., Dixon, K.W. et al. Stockpiling disrupts the biological integrity of topsoil for ecological restoration. Plant Soil 471, 409–426 (2022). https://doi.org/10.1007/s11104-021-05217-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-021-05217-z