Abstract
Aims
Soils of northern latitude tundra ecosystems have accumulated large amounts of carbon that might be released as CO2 when temperature rises and the tree-line moves north. We aim to investigate the potential CO2 flux changes at a subarctic tundra heath under changing climate.
Methods
We measured daytime ecosystem respiration and photosynthesis at a subarctic heath over a full year under ambient conditions and in factorial long-term (13 years) increased summer temperature and leaf litter addition plots, and in additional short-term (2 years) summer warming plots.
Results
Under ambient conditions the ecosystem was a daytime sink of CO2 in the five warmest months, but a net daytime source in the cold season. Thirteen years of summer warming by 1 °C at soil surface increased CO2 emissions, as daytime respiration increased by 37% and photosynthesis by 29% over the year. Short-term warming likewise increased fluxes. Litter addition also increased the emission of CO2 as ecosystem respiration rose by 21% but photosynthesis remained unchanged. Both warming and litter addition significantly enhanced the amount of green biomass.
Conclusions
This study suggests that in a changed climate subarctic ecosystems will act as a positive feedback source of atmospheric CO2. It shows the significance of CO2 fluxes outside the growing season and demonstrates a cold-season long- but not short-term legacy effect of increased summer warming on CO2 emission.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Projected global warming is anticipated to be most pronounced at high latitudes (Collins et al. 2013). Predictions imply an annual mean temperature rise of 5–7 °C in the Arctic before 2100, if the Paris Agreement’s aim of limiting the increase of the global temperature to 1.5 °C is met (Seneviratne et al. 2018). This will be accompanied by reduced ice- and snow-cover, a prolonged growing season, increased cloud-formation and winter precipitation (Collins et al. 2013; Serreze et al. 2000) and a northward range expansion of subarctic and low arctic trees and shrubs (Elmendorf et al. 2012; Post et al. 2009).
Even though arctic and subarctic ecosystems have low-statured vegetation and low annual net production, the constraints on decomposition imposed by the low temperatures provides northern biomes with a high capacity for soil carbon accumulation. High-latitude ecosystems currently store 44% of the soil C-pool of the Earth (Hugelius et al. 2014). The response of polar ecosystems to global change is hence a key to the understanding and prediction of future global atmospheric CO2 patterns, and is therefore essential to the understanding of future challenges due to a changed climate (Crowther et al. 2016).
Whether an ecosystem acts as net carbon source or sink is a delicate balance between ecosystem respiration (ER = autotrophic + heterotrophic respiration) and gross ecosystem production (GEP = photosynthesis). Even small changes in gas flux components may have critical implications for the ecosystem carbon balance. If global warming stimulates the breakdown of soil organic matter and hereby CO2 release to the atmosphere, the extensive arctic carbon-pool has the potential to provide a significant positive feedback to climate change (Arndal et al. 2009; Crowther et al. 2016; Grogan and Chapin 1999; Larsen et al. 2007b; Schuur et al. 2015; Welker et al. 2004). Plant growth in the Arctic is temperature and nutrient limited (Campioli et al. 2012; Shaver and Chapin 1980; Street et al. 2007; Sistla et al. 2013). In a warmer climate, enhanced decomposition could increase nutrient availability and together with a prolonged snow-free season stimulate plant growth and CO2 sequestration, and hence result in a negative feedback on atmospheric CO2 concentrations. The various impacts of climate change on ecosystem respiration (ER) and photosynthesis (GEP) hereby determines the future direction of net ecosystem production, NEP, and its feedback to the global climate (Arndal et al. 2009; Crowther et al. 2016; Oberbauer et al. 2007; Welker et al. 2004).
The CO2 fluxes in the Arctic and Subarctic undergo significant seasonal variation (Grogan and Jonasson 2006; Moore et al. 2006) and considerable biological activity has been demonstrated outside the growing season (Brooks et al. 2011; Christiansen et al. 2012; Grogan et al. 2001; Larsen et al. 2007c; Webb et al. 2016). In spring, substantial ecosystem respiration and photosynthesis may occur even underneath snow (Grogan et al. 2001; Larsen et al. 2007c; Moore et al. 2006; Starr and Oberbauer 2003). In subarctic tundra Larsen et al. (2007c) found that GEP in April and May account for 14% of the yearly carbon sequestration and that ER in May can reach half of the yearly maximum. While ER by far exceeds GEP during winter, Grogan and Jonasson (2006) found GEP in May to outbalance ER at a subarctic heath. A description of arctic carbon exchange solely based upon growing season fluxes is hence inadequate. While current literature shows an increasing body of studies in shoulder season and winter, there are to our knowledge currently very few studies that provide plot-level measurements of CO2 fluxes in subarctic or arctic ecosystems over the entire year in experiments with long-term warming (Belshe et al. 2013; Mauritz et al. 2017; Natali et al. 2014; Webb et al. 2016), and none that include litter addition. Litter addition simulates the increased litter-fall in tundra subjected to shrubification (Myers-Smith et al. 2011; Hicks-Pries et al. 2017), and leads to enhanced soil nutrient and labile carbon availability and potential priming of organic matter turnover (Bengtson et al. 2012; Phillips et al. 2019). Hence, litter availability is an important component of anticipated future climate change because the shrub expansion might be stimulated further due to a faster internal nutrient cycling, when leaf production is stimulated (DeMarco et al. 2014a).
To better understand the full year carbon balance and the long-term implications of changes in temperature and litter input, the aim of this study is to investigate gross ecosystem production, ecosystem respiration and net ecosystem production at a subarctic heath tundra in northern Sweden across all seasons. Measurements were undertaken in plots subjected to 13 years of leaf litter addition and summer warming with open-top chambers with previously observed vegetation changes (Lett and Michelsen 2014). To investigate warming effects without anticipated vegetation changes, measurements were furthermore conducted in additional plots only subjected to 2 years of warming. Measurements were done with high resolution sampling (3–6 day interval) from snowmelt into late growing season, and with additional sample dates in both autumn and winter.
We expect ambient CO2 fluxes to peak in mid-summer due to dependence on trends in temperature and light intensity. We expect to detect ecosystem respiration across the year and photosynthesis underneath the snow in late spring.
We hypothesize that 1) warming and litter addition will increase ER as result of direct warming as well as increased substrate availability and indirect changes as increased green biomass (measured as NDVI). The treatment effects are expected to be additive. We also hypothesize 2) that increased plant biomass and changes in vegetation composition due to the treatments will increase daytime GEP, and the treatment effects are expected to be additive. We also expect 3) more pronounced effects after 13 than after 2 years of warming and based on results from another wet tundra (Welker et al. 2004) and a nearby heath (Illeris et al. 2004b) we expect 4) stronger warming effect on ER compared to GEP resulting in a reduction of NEP.
Materials and methods
Study site
Measurements were conducted near Abisko, Northern Sweden (68°21’N, 18°49′E). Climate in the region is subarctic, with a mean annual temperature of 0.2 °C and an annual precipitation of 337 mm. The warmest and coldest months, July and February, have average temperatures of 11.9 °C and − 10.0 °C, respectively (30 year mean 1986–2015). The snow-covered season usually lasts from early October to mid-May (Abisko Scientific Research Station 2016) and the growing season is from early June to the end of August.
The field site is located on a wet dwarf-shrub dominated gently sloping, NE facing heath at 400 m a.s.l. Vegetation is dominated by the dwarf shrub Andromeda polifolia (18%), the low shrubs Empetrum hermaphroditium (29%) and Vaccinium uliginosum (17%) together with the sedges Carex vaginata (28%) and Carex rupestris (17%). Mosses (dominated by Hylocomium splendens, Sphagnum warnstorfii and Tomentypnum nitens) cover about 60% of the ground and lichens about 30% (Sorensen and Michelsen 2011). The highly organic soil that overlays the bedrock has a pH of around 6.9 and a well-developed humus-layer of 20 cm, with a SOM-content of 80–95% (Phillips et al. 2019; Rinnan et al. 2008).
Experimental design
To simulate direct and indirect long-term effects of expected climate change, summer temperatures and litter quantity were manipulated since 1999. Manipulations included warming with open-top chambers (OTCs) made of polythene sheets, addition of litter and the combination of these treatments. In sum, the treatments were: control (C), litter addition (L), warming (T), the combination of litter and warming (TL), and an additional short-term warming treatment (Tshort), carried out for two years. The treatments were replicated across six randomized blocks (n = 6), making all together 30 plots of 120 × 120 cm. Each year from May/early June and until late August/early September, OTCs were erected on the site. According to 24 h continuous measurements the OTCs increase the air temperature by 2–3 °C and ground surface temperature by 1.1 °C (less at night and greater at day) from 1st of June to 31st of August (Lett and Michelsen 2014). In 2011 the OTCs were erected on May 12th and dismantled on September 3rd. Every autumn in early September (in 2011 on September 3rd) 90 g (d.w.) litter of Betula pubescens ssp. tortuosa was added per square meter, corresponding to the annual litter fall in the nearby open birch forest. The litter contains approx. 450 mg g−1 d.w. C, 9.8 mg g−1 d.w. N and 1.2 mg g−1 d.w. P (Rinnan et al. 2008). Hence a total of 526.5 g C, 11.5 g N and 1.4 g P has been added per m2 during the 13 years of litter addition. Shrubification and expansion of forest are expected due to a warmer climate and will impact the ecosystem as a consequence of snow accumulation, albedo feedback and changes of the nutrient cycling (Myers-Smith et al. 2011; Sturm et al. 2005). Litter addition attempts to give insights into effects related to the latter as a higher amount and/or a change in litter-quality are also expected to follow from a higher proportion of deciduous plants (Lett and Michelsen 2014). Litter addition furthermore causes a shading effect, which is realistic both following shrub and tree encroachment and with increased cloudiness in the future.
CO2 exchange measurements
In a continuous campaign between April 23rd and September 12th 2011 CO2-fluxes were measured in all 30 plots a total of 26 times (generally with 3–6 days interval between measurements, maximally 9 days). Measurement were done as daytime fluxes spanning across midday. Additional cold season measurements (7 times) were conducted in December 2011, January 2012, March 2012 and October/November 2012. Measurements of all plots were performed at the same day in block-wise cycles, except on a few of the winter dates where less replicates (n = 4) were measured due to technical difficulties in the cold and the lack of daylight.
To detect changes in the CO2-concentration permanent aluminium bases with dimensions of 21.4 × 21.4 cm were inserted about 10 cm into the ground at each plot in 1999, enclosing soil and vegetation. During measurements a 13.5 L transparent polycarbonate chamber was placed in a groove along the upper rim of the base and the groove filled with water or snow to tightly seal the chamber. CO2 fluxes were measured with EGM-4 Environmental Gas Monitor (PP Systems, Hitching, United Kingdom) connected to an Environmental Monitor Sensor Probe Type 3 (PP Systems, Hitching, United Kingdom) mounted inside the chamber, as described in Christiansen et al. (2012). The chamber was fitted with a circulation fan to provide proper air-mixing. The EGM-4 logged CO2-concentration for five consecutive minutes in the warm-season and for 8-min intervals on April 23rd and 24th and for 10-min intervals in December, January and March, due to low exchange rates. At each plot, a full measurement consisted of a measurement in light and in darkness. The light period measured net ecosystem production (NEP), equal to the carbon sequestration by photosynthesis minus carbon release from plant and soil microbial respiration. After aeration of the chamber, the measurement were re-taken with two layers of dark plastic covering the chamber, with the dark period established to measure the ecosystem (plant plus soil) respiration (ER). Whenever snow was present, the groove was gently cleared of snow and it was possible to measure the gas flux without having to remove the snow cover of the plot itself, a procedure that could have initiated an unnatural CO2 pulse (Grogan et al. 2001).
Environmental measurements
While quantifying CO2-concentrations the EGM-4 simultaneously logged incoming photosynthetically active radiation (PAR), air temperature and relative humidity inside the chamber. Soil temperature in 2 cm depth was measured during flux measurements with a hand-held, digital thermometer (T Handle Lab Thermometer DT520TH) at three positions in each plot, adjacent to the chamber. Furthermore temperature was continuously recorded with Tinytags TGP-4520 (Gemini Data Loggers (UK) Ltd.) in soil and at the soil surface in control, OTC, litter and OTC-and-litter plots (n = 3 per treatment). When the soil was not frozen, volumetric soil moisture content was also recorded during flux measurements at three positions in each plot using a Theta Probe ML2x (Delta-T Devices Ltd., Cambridge, UK). Precipitation was logged at the nearby (1 km distance) Abisko Scientific Research Station (2016).
Green biomass was non-destructively measured by the normalized difference vegetation index (NDVI) with a SKR 110 sensor (Skye Instruments, Powys, UK) with narrow band interference filters centred at 660 and 730 nm. Measurements were made eight cm above the vegetation inside the chamber-base. The NDVI-index utilizes the differential absorption properties of leaves (reflectance of 730 and 660 nm respectively). Soil in 0–5 and 5–10 cm depth was sampled from each plot with a soil auger (Ø: 3.9 cm) on the 12th of July. Roots were removed and two subsamples of 5 g soil were taken; one subsample was suspended right away in 25 ml 0.5 M K2SO4 for one hour and filtered (Whatman GF/D). These extracts were analyzed for dissolved organic C (DOC) with Shimadzu TOC analyzer, and ammonium (NH4+-N), nitrate (NO3−-N), phosphate (PO43−-P), and dissolved organic N (DON) using flow injection analysis (FIAstar 5000 Analyzer). The second soil subsample from each plot was incubated for 24 h in vacuum with chloroform before extraction and filtration, and DOC and flow injection analysis was used to determine the amount of microbial biomass C, N, and P. The SOM content was estimated as loss on ignition. On 3rd September we additionally sampled two separate subsamples of soil in each plot to 20 cm depth and sorted roots off to analyze the soil N stock in 5 cm depth intervals using an Eurovector elemental analyser.
Data processing
Calculations of NEP and ER were completed as follows:
μmol CO2 m−2 s−1 = \( \frac{dc\ \mu mol}{dT\ mol\ s}\times \frac{P}{1013}\times \frac{1\ mol}{22.414\ L}\times \frac{273}{273+{T}_{air}} \times \frac{V\ {m}^3}{A\ {m}^2}\times \frac{10^3L}{m^3} \), where \( \frac{dc}{dT} \) is the measured rate of change in CO2-concentration in ppm (μmol mol−1), \( \frac{P}{1013} \) is the correction for barometric pressure with P measured in mbar by the EGM-4, \( \frac{1\ mol}{22.414\ L} \) is the molar volume of an ideal gas, corrected for the actual temperature by \( \frac{273}{273+{T}_{air}\ } \) which is the correction for air temperature with Tair as the temperature inside the chamber in °C, V is the chamber volume in m3, and A is the area in m2 covered by the chamber (PP Systems 2018). To convert the unit of the CO2 flux from μmol CO2 m−2 s−1 to mg CO2 m−2 h−1 we multiplied with 158.4 (3600 s h−1 × 44.009 g / 106 μmol × 103 mg g−1) (PP Systems 2018). The detection limit of ER derived by linear regression (ER r2 > 0.9; p < 0.05) was ±5.0 mg CO2 m−2 h−1 (= 0.033 g CO2-C m−2 day−1), slightly higher than that of Grogan and Jonasson (2005). Linear regression with lower r2 than 0.9 or p > 0.05 lead to exclusion of 6 erroneous ER measurements of a total of 890 measurements. For gross ecosystem production (GEP), which is determined as GEP = NEP + ER and hence has twice the potential uncertainty of each of the component fluxes, we only accepted fluxes equal to or exceeding ±10 mg CO2 m−2 h−1, and consequently GEP was not detectable in winter. No pulse was observed associated with the shoulder season measurements.
The ecosystem-atmosphere-system is in this study perceived from an ecosystem point of view; hence C-fluxes into the ecosystem (GEP) were positive and C-fluxes out of the system (ER) negative.
Statistics
Treatment effects on fluxes were examined by three-way repeated measures mixed model ANOVA with block as random factor, OTC (two levels: OTC, no OTC) and litter (two levels: litter, no litter) as fixed factors and with the interaction between OTC and litter included. Treatment responses in short-term warming (Tshort) plots were moreover tested against control plots by two-way repeated measurements ANOVA (incl. block) followed by Tukey’s studentized range test (HSD). Data were tested across the whole study period (measurement campaigns, m = 33) and separately into four seasons as follows: snow melt: with average daily soil temperature between 0 °C and 7 °C and before OTCs were erected (April 20th-May 11th, m = 5); spring+summer: with average daily soil temperature exceeding 7 °C and OTCs on (May 12th-September 3rd, m = 19); fall: from when the NDVI dropped markedly in most plots and the OTCs were dismantled and until soil freezing (September 4th-October 14th, m = 2) and winter: from when soil temperature dropped below 0 °C (October 15th-April 19th, m = 7) (Fig. 1). Soil temperature, water content and NDVI from all treatments (except Tshort tested with two-way ANOVA and Tukey’s test) were tested by three-way ANOVAs as repeated measurements as described for fluxes, repeated for the whole period and each season. Additional tests for each individual day were conducted for all variables. Pearson correlations were performed between CO2 fluxes and the abiotic factors.
Environmental variables and CO2-fluxes during 2011–2012 at control plots at Abisko, northern Sweden. a Mean photosynthetically active radiation (PAR) during flux-measurements (filled diamonds, n = 6 ± SE). Average snow depth at control-plots at dates of flux-measurements (open triangles, n = 6). b Half hourly soil temperature in 2 cm depth (black line). Daily precipitation reported at Abisko meteorological station (bars). c) Gross ecosystem production (GEP) at control plots (filled triangles, n = 6 ± SE). Ecosystem respiration (ER) in control plots (open circles, n = 6 ± SE). Net ecosystem production (NEP) in control plots (filled circles n = 6 ± SE). Between October 1st and November 1st, data are from 2012, otherwise, data from April until January is from 2011
An α value of 0.05 was used to indicate statistical significance, and tendencies toward significance are reported for 0.05 < p < 0.1. Data were investigated for homogeneity of variance using Levenes’s test and inspection of residual plots, and data were log transformed if required. All statistic tests were performed using SAS Enterprise Guide 7.1, SAS Institute Inc., Cary, NC, USA.
Results
Environmental parameters
During the measuring campaign in spring and summer 2011 the monthly air temperature was averaging from 1.3 °C in April to 12.3 °C in July. Summer precipitation during this period was close to the 30-yr average (data 2011 vs. 1986–2015, Abisko Scientific Research Station meteorological station) except for June where precipitation was almost twice that of the period 1981–2015, as a result of a major rain-event on June 21st of 43 mm rainfall (Fig. 1).
Monthly averages of air temperature during the 2011–2012 cold season (October – March) spanned from −0.5 °C to −11.6 °C with a maximum in October and a minimum in February. During the growing season average photosynthetically active radiation (PAR) depended a lot on the cloud cover (and there were no effects of treatment). Snow was present from October 15th through winter and melted between April 23rd and 29th, and in late April soil temperature in 2 cm depth was above 0 °C (Fig. 1).
Treatment effects on environmental parameters
Although OTCs did not significantly increase soil temperature at 2 cm depth across the whole treatment period (spring+summer), a significant warming effect of 1–2 °C was found in the beginning of this period, from May 15th to June 18th (p < 0.05). This is the period from when the OTCs were placed on the plots and before plant cover (measured as NDVI) reached its maximum (Table 1, Online resource 1, 2, 3). Furthermore, from early June to late August, soil surface temperature was increased by 1.14 ± 0.05 (mean ± SE) °C (p < 0.01) in OTC plots compared to controls (average 13.8 and 14.9 °C in control and OTC plots, respectively), while litter addition reduced surface temperature with 0.9 ± 0.20 °C (p < 0.05) (Online resource 3). Addition of litter significantly decreased soil temperature in summer and across the whole sampling period (p < 0.01). OTC and litter addition interacted significantly as OTCs alone increased soil temperature while the combined treatment reduced soil temperature (Online resource 1, 2, 3).
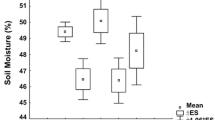
Across the spring and summer campaign the soil water content was 30–50% in control plots, highest in July and lowest in August (Fig. 2). Addition of litter significantly decreased soil water content in spring+summer and across the entire period (p < 0.001), but OTC and litter addition interacted as OTCs alone tended to increase soil moisture (Table 1). Likewise, the short-term warming treatment had higher water content than control plots.
Soil water content at 0–6 cm depth during flux-measurements in 2011 and 2012 (n = 6, mean ± SE). Treatments are: Control (C, filled circles), Litter addition (L, open circles), OTC (T, filled triangles), OTC and Litter addition (TL, open triangles), Short-term OTC treatment (TShort, squares). Results of three-way ANOVA for the treatments C,L,T,TL with factors long-term warming and litter addition plus block, and two-way ANOVA with factors short-term warming plus block supplemented by Tukey’s for C,T,TShort for each day separately are shown as letters above bars, †p < 0.1, *p < 0.05, **p < 0.01, ***p < 0.001, n.s. non significant. Day was significant (P < 0.001) for entire period, summer and fall
Vegetation changes
Normalized differential vegetation index (NDVI), a proxy for green biomass, increased through the spring and early summer (p < 0.001) until reaching maximum values in the beginning of July (Fig. 3). From 10th July and until 2nd September, NDVI declined slowly, after which the decrease was more pronounced. In spring, OTC treatment led to an earlier onset of leaf expansion, with higher NDVI on May 28th. Across the entire period, and in spring+summer, litter and OTC treatments significantly increased NDVI (both p < 0.001) and the effect was additive, causing OTC-and-litter plots to consistently show the highest NDVI while the control plots generally showed the lowest NDVI (Table 1, Fig. 3). Vegetation analysis showed a warming-induced increase of moss, deciduous shrub and total plant cover in early June (Lett and Michelsen 2014), but in early August the increase in NDVI was only reflected in the total vegetation coverage (Online resource 4). However, the dominant evergreen shrub Andromeda polifolia showed a doubling of coverage due to long-term warming. This was the only individual species that showed a response to treatments.
Normalized difference vegetation index (NDVI) during flux-measurements (n = 6, mean ± SE). Treatments are: Control (C, filled circles), Litter addition (L, open circles), OTC (T, filled triangles), OTC and Litter addition (TL, open triangles), Short-term OTC treatment (Tshort, squares). Results of three-way ANOVA for the treatments C,L,T,TL with factors long-term warming and litter addition plus block, and two-way ANOVA with factors short-term warming plus block supplemented by Tukey’s for C,T,TShort for each day separately are shown as letters above bars, †p < 0.1, *p < 0.05, **p < 0.01, ***p < 0.001. Day was significant (p < 0.001) in all periods except fall (p < 0.1) and winter (no data)
Ambient daytime ecosystem CO2 fluxes
Gross ecosystem production in late April 2011, under 2–4 cm snow, at ambient conditions was around 150 mg CO2 m−2 h−1 and steadily increased to 500 mg CO2 m−2 h−1 on June 6th, with the exception of a drop on May 9th caused by cold, cloudy weather (Fig. 1). From June 10th and onwards photosynthesis was 1000–1500 mg CO2 m−2 h−1 with highest monthly average in July (1426 mg CO2 m−2 h−1), and decreased from early August. Photosynthesis (GEP) was non-detectable from October to March (flux <10 mg CO2 m−2 h−1).
Ecosystem respiration increased at roughly the same rate as GEP in spring reaching maximum flux in July (1450 mg CO2 m−2 h−1) and declining below 500 mg CO2 m−2 h−1 in September (Fig. 1). In October, under more than 10 cm of snow, ER rates of 50–100 mg CO2 m−2 h−1 were measured and low but detectable rates (15–35 mg CO2 m−2 h−1) were also measured November to March.
Net ecosystem production in April and May fluctuated at ±100 mg CO2 m−2 h−1 (Fig. 1). Throughout the summer (June, July, August) NEP was generally positive in control plots, which was also reflected in net C influx as monthly averages from June to September, with the highest net ecosystem carbon assimilation in July (326 mg C m−2 h−1). Under ambient conditions NEP showed minor but negative rates (i.e. losses) during the cold season.
Treatment effects on CO2 fluxes
Both long- and short-term OTC treatment significantly enhanced photosynthesis (GEP) across the entire measuring period (T: p < 0.01, Tshort: p < 0.05). Short-term treatment increased GEP slightly more than long-term (40% and 29% respectively, Figs. 4 and 5; Table 2). Long-term warming also increased photosynthesis in spring+summer when OTCs were erected (T: p < 0.05) and tended to do so in the fall (T: p < 0.1). Litter addition alone did not significantly affect GEP, even though fluxes on 21 out of 26 measuring dates were higher in litter-treated than in control plots (Fig. 4; Online resource 5). The combined treatment effect tended to be less than additive compared to OTC and litter treatments separately (T × L: p < 0.1).
Seasonal average gross ecosystem production (GEP) and ecosystem respiration (ER) during 2011 and 2012. Efflux from ecosystem to the atmosphere is represented by negative values. Bars show GEP and ER for each of five treatments (mean ± 1 SE, n = (3-)6 plots per treatment per day). Treatments are: Control (C), Litter addition (L), OTC (T), OTC and Litter addition (TL), Short-term OTC treatment (Tshort). Snow melt: April 20rd – May 11th, spring+summer: May 12th – September 3rd, fall: September 4th – October 14th, winter: October 15th – April 19th. In winter, under snow cover, GEP was not detectable October–March. Results of repeated measures mixed model analyses with significant main factor effects and interactions (three-way ANOVA for treatments C,L,T,TL with factors long-term warming and litter addition plus block, and two-way ANOVA with factors short-term warming plus block supplemented by Tukey’s for C,T,TShort) are shown as letters above bars, †p < 0.1, *p < 0.05, **p < 0.01, ***p < 0.001, n.s. non-significant
Monthly average gross ecosystem production (GEP) and ecosystem respiration (ER) during 2011 and 2012 (January, March, October and November). Efflux from ecosystem to the atmosphere is represented by negative values. Bars show GEP and ER for each of five treatments (m = number of measurement days in the month, mean ± 1 SE, n = (3-)6 plots per treatment per day). Treatments are: Control (C), Litter addition (L), OTC (T), OTC and Litter addition (TL), Short-term OTC treatment (Tshort). Snow melt: April 20rd – May 11th, spring+summer: May 12th – September 3rd, fall: September 4th – October 14th, winter: October 15th – April 19th. In winter, under snow cover, GEP was not detectable October–March. Results of repeated measures mixed model analyses with significant main factor effects and interactions are shown as letters above bars, †p < 0.1, *p < 0.05, **p < 0.01, ***p < 0.001, n.s. non-significant, and effect of short-term OTC were tested by two-way ANOVA and Tukey’s test. Day was significant (p < 0.001) in all periods except ER in spring (p < 0.1) and GEP in winter (no GEP)
Two and 13 years of OTC treatment increased ER with 63% and 37% respectively and litter addition also increased ER with 21% compared to controls across the whole period (Tshort: p < 0.05, T: p < 0.001, L: p < 0.01). The effect was also significant in the spring+summer period (Figs. 4 and 5; Table 2). Furthermore long-term summer warming caused a significant 123% higher ER compared to controls in November though the OTCs were not erected, while ER was unaffected by the legacy of short-term summer warming (Fig. 5, Online resource 6). Both long- and short-term OTC-treatments significantly reduced (i.e. less uptake or higher emission of CO2) the net ecosystem production (NEP) across the whole period (T: p < 0.001, Tshort: p < 0.05) and changed the ecosystem from a daytime CO2 sink to a source (Fig. 4). This was also evident within the spring+summer treatment-period (T: p < 0.001, Tshort: p < 0.05), specifically in May, June and August (Fig. 6), but not in July, when photosynthesis peaked. Litter too significantly decreased NEP in the spring+summer and fall and through the entire period (p < 0.01; p < 0.001; p < 0.001) (Figs. 4 and 6; Online resource 7).
Monthly average net ecosystem production (NEP) during 2011 and 2012 (January, March, October and November). Efflux from ecosystem to the atmosphere is represented by negative values. Bars show NEP for each of five treatments (m = number of measurement days in the month, mean ± 1 SE, n = (3-)6 plots per treatment and day). Treatments are: Control (C), Litter addition (L), OTC (T), OTC and Litter addition (TL), Short-term OTC treatment (Tshort). Wintertime NEP is based on ER as GEP was not detectable in during winter. Snow melt: April 20rd – May 11th, spring+summer: May 12th – September 3rd, fall: September 4th – October 14th, winter: October 15th – April 19th. Results of repeated measures mixed model analyses with significant main factor effects and interactions are shown as letters above bars, †p < 0.1, *p < 0.05, **p < 0.01, ***p < 0.001 and effect of short-term OTC were tested by two-way ANOVA and Tukey’s test
Correlations
Soil temperature in 2 cm depth had the greatest explanatory power over ER across the whole period (p < 0.001, r2 = 0.67), and also correlated strongly with GEP (p < 0.001, r2 = 0.53) (Table 3; Online resource 8). NDVI also correlated with ER (p < 0.001, r2 = 0.57) and GEP (p < 0.001, r2 = 0.59) and soil water content slightly so with NEP (p < 0.001, r2 = 0.02). ER, GEP and NEP were correlated with PAR, though not strongly (p < 0.001; r2 = 0.13; r2 = 0.13; r2 = 0.09, respectively).
Treatment effects on soil nutrient availability and microbial biomass
While warming had no significant effects on soil nutrients or microbial biomass, litter addition increased the DOC concentration and tended to increase the phosphate concentration in 0–5 cm depth. Litter addition furthermore increased the SOM concentration and soil microbial N and P in 5–10 cm depth (Online resource 9). Microbial C and inorganic N was unaffected by treatments. Neither long-term warming nor litter addition changed the soil N stock significantly (Online resource 10).
Discussion
To the best of our knowledge, this is the first study to investigate short and long-term impact of warming and litter addition on carbon dioxide fluxes through warm and cold season in subarctic tundra. We found that short-term warming (2 years), long-term warming (13 years) and litter addition stimulated ER in spring and summer, while GEP in this period only responded positively to long-term warming. Our experiment hence demonstrated that increased litterfall, which is one of the consequences of shrubification, did not impact the photosynthetic capacity of subarctic tundra on its own even after decade-long perturbations, but increase the net carbon emission to the atmosphere through enhanced ER. This increase of ER might be due to dryer soil conditions at this otherwise wet heath, to the increased plant biomass and to the direct effect of added substrate, with increased release of nutrients and labile carbon. Furthermore we observed a legacy effect of long-term summer warming increasing ER in the cold season (November), while short-term warming did not stimulate winter ER. This likely reflects the larger plant biomass in long-term warmed plots fueling respiration, and demonstrates that short-term responses in carbon fluxes differ from long-term (decade-long) responses because slow-growing arctic plants build up biomass over decades.
Ambient CO2 fluxes
When soil temperatures were below 0 °C we did not detect any GEP. We conclude that photosynthesis was not occurring, or occurring at insignificantly low rates, and hence NEP is similar to ER alone between late October and March. While no GEP was detected in winter, significant GEP was as expected observed already in late April when a thin snow cover of 2–4 cm was still present and the soil temperature was slightly above 0 °C. Respiration was detectable throughout the cold season. That both photosynthesis and respiration gather pace when the soil is close to 0 °C support the finding of arctic plants and microbes being capable of reaching high levels of activity within short time upon thaw (Arndal et al. 2009; Fahnestock et al. 1998; Grogan et al. 2001; Larsen et al. 2007c; Natali et al. 2011; Welker et al. 2004). In May, GEP increased to a magnitude of one-third of July fluxes. The highest flux rates, 1200–1500 mg CO2 m−2 h−1 (GEP) and 1000–1400 mg CO2 m−2 h−1 (ER), were as expected measured in July when green biomass (NDVI) and temperature peaked, and these rates are high compared to other studies of subarctic heaths nearby (Illeris et al. 2004a), and of other locations in the arctic region e.g. Toolik Lake, Alaska (Welker et al. 1999), Svalbard and North East Greenland (Lund et al. 2012). This may partly be due to high soil water and nutrient supply throughout the growing season at this foothill heath and underlines how magnitudes of CO2 fluxes are highly dependent on both latitude (and therefore temperature), water regime, topography, vegetation type and cover (Lafleur et al. 2012) and stresses the importance of measurements at a diversity of locations (Metcalfe et al. 2018; Virkkala et al. 2018).
In winter (November–March) the ecosystem release of CO2 at midday was between 15 and 45 mg CO2 m−2 h−1. This was of the same magnitude as mid-winter fluxes reported for nearby heaths (Grogan et al. 2001; Grogan and Jonasson 2005). Larsen et al. (2007c) reported winter-respiration (including April and May) to account for 22% of the annual respiration at a nearby mesic heath. NEP approached zero in May and from June and until early fall the ecosystem was a daytime carbon-sink with an uptake of 100–300 mg CO2 m−2 h−1 from the atmosphere. Preceding studies have likewise demonstrated positive day-time NEP (net C uptake) at this or nearby heaths during the growing season 2003–2004, 2006 and 2007 (Larsen et al. 2007c; Tiiva et al. 2008).
Treatment effect on ecosystem respiration
In accordance with hypothesis 1, short- as well as long-term summer warming increased ER across the entire period and during the spring+summer when OTCs were erected. Warming-induced increase of ER is often reported as a direct result of temperature increasing decomposition (Davidson and Janssens 2006; Larsen et al. 2007c; Natali et al. 2011; Voigt et al. 2016). In our study OTCs increased surface temperature by more than 1 °C and soil temperature in 2 cm depth explained 67% of the variation in ER over the entire period. However, after 13 years of treatment with OTCs, we did not find any effect of OTCs on soil temperature in 2 cm depth with the exception of spring before NDVI reached its maximum. This is probably because the soil-warming effect is partially counteracted by the cooling effect of shading by the increased plant biomass (Lett and Michelsen 2014), as indicated by NDVI-measurements and demonstrated as an increase in total plant cover by 28% (Lett and Michelsen 2014). Hence, the marked increase of ER after 13 years of treatment is partially an indirect effect due to increased plant biomass. This led to increased plant respiration, as observed by Hicks-Pries et al. (2015) as increased autotrophic contribution to ecosystem respiration in a wetter, bryophyte-dominated peatland. Another contributing factor to the ER increase after 13 years of treatment is most likely the enhanced input of organic matter as litter and root exudates, which increase soil respiration, and might lead to priming of soil organic matter decomposition (Bengtson et al. 2012; Larsen et al. 2007a; Ravn et al. 2017). In contrast to our third hypothesis stimulation of ER was higher in short- compared to long-term summer warmed plots and an increase of NDVI was already evident after two years of summer warming, likely suggesting that a higher biomass in short-term warming plots probably increased plant respiration to a higher degree than expected. The NDVI response in short term warmed plots was likely due to a collective response of many plant functional groups and species, and over multiple dates of NDVI measurements, as it was not yet evident in the vegetation analyses after only two years of warming.
Our first hypothesis was also confirmed in regard to litter addition as addition of birch-litter increased ER across the entire period, during the spring+summer and in fall. This is probably a result of increased plant respiration following higher plant biomass in litter-treated plots (measured as increased NDVI). Furthermore the higher soil DOC and phosphate concentration together with elevated soil microbial biomass N and P in litter amended plots coincided with higher ER, suggesting that microbial activity stimulated turnover of organic matter, including most of the added litter. As neither N stocks (Online resource 10) nor C stocks (Ravn et al. 2017) changed significantly by litter addition, the stimulation of ER seems to predominantly originate from turnover of the added litter itself.
As litter addition decreased soil temperature, likely due to a shading effect on the soil, the increase of ER is not controlled by temperature but rather a consequence of higher substrate and nutrient input and/or improved litter quality. In the highly organic heath soil in our experiment, litter addition persistently decreased soil water content, likely because litter may increase interception of rainfall and subsequent evaporation, and because litter impacts the quality and structure of the upper organic soil layers, thereby altering soil aeration and permeability. Dryer soil conditions stimulating decomposition in an otherwise wet heath may further increase ER in litter treated plots (Natali et al. 2015). Increased permeability with added litter might also explain why the difference in water content is especially pronounced after snow melt (from April 29th), due to faster drainage of the soil in litter amended plots with more fluffy soil. The less pronounced effect before snowmelt might be because of cold and partly frozen soil. Incidents of high precipitation input, like on June 21st, leads to recharge both in plots with and without litter amendment, but with higher impact on moisture conditions in plots without added litter. This may also contribute to the observed increase of ER. The combined effect of litter and OTC warming on ER was lower than expected from the single treatments, leading to significant interaction over the entire period. This is likely caused by the cooling effect of litter on soil temperature, counteracting the increasing temperature caused by the OTCs (Pedersen et al. 2017).
We have not found treatment effects on coarse or fine root biomass in the experiment (Ravn et al. 2017), and effects of warming and litter on ER are hence due to both stimulated soil and aboveground (but not belowground) plant respiration, caused by the higher plant cover (NDVI). This study is among the first to measure on long-term OTC and litter manipulation effects on CO2 fluxes across the whole year. Though no significant effect of OTCs where found on fluxes across winter, in response to 13 years of summer warming we demonstrate increased ER in November, when soil temperature was around −1 °C. In spring, on April 29th and May 9th and still outside the treatment period, long-term OTC plots also tended to show higher ER compared to control plots. This suggests that long-term, but not short-term, increase of summer temperature impact the CO2 flux from the ecosystem even in the cold period. Outside the treatment period, when there is no direct effect of warming, the higher ER might be due to higher plant respiration and substrate input as a legacy effect of the higher plant biomass in long-term summer warmed plots (Lett and Michelsen 2014).
Although Grogan et al. (2001) found that recently fixed plant-C, i.e. litter, fine roots and their exudates are the major contributors to winter respiration, litter treatment alone did not alter ER during the cold season. To our knowledge, the present study is the first to investigate long-term effect of experimental litter inputs on CO2 flux during the cold season for tundra ecosystems, and the pronounced effect a few days after litter addition on September 3rd indicates that the leaching of labile carbon from litter is a fairly rapid process under early fall conditions, and that this component of the litter impact on ER may not linger into winter. This is in line with findings of a high degradation-rate just after litter fall/addition observed by Hobbie (1996).
Treatment effect on photosynthesis
Across the entire period and during the spring+summer period we observe a stimulation of GEP by 13 years of OTC summer warming. This is in line with hypothesis 2 and with similar observations (Leffler et al. 2016; Natali et al. 2011; Welker et al. 2004) and is probably due to direct stimulation of photosynthesis by warming, and the indirect effects as increased nutrient availability and increased plant biomass (Boelman et al. 2003; McMichael et al. 1999). Both long-term and short-term warming increased NDVI but in contrast to our hypothesis 3, GEP was increased most by the short-term warming across the entire period. This was despite a lower NDVI increase in short-term compared to long-term warmed plots. The effect of short-term warming on GEP is likely reflecting the direct effect of warming on photosynthesis rate but probably also vegetation changes, and while mosses seemed to respond positively to short-term warming, and mosses are major contributors to GEP in many arctic ecosystem types (Arndal et al. 2009), the cover of deciduous shrubs was increased only in the long-term warmed plots (Lett and Michelsen 2014). It should be noted, that while NDVI is strongly correlated with leaf area index in tundra (Street et al. 2007) it does not fully capture woody biomass responses. However, the detailed measurements of NDVI show how warming not only increases green plant cover but also results in a faster achievement of growing season peak in biomass. This might contribute to the increase of GEP.
The increased moss coverage in warmed plots (Lett and Michelsen 2014) is probably also why warming seems to lead to slightly higher soil moisture in this wet heath type, as mosses stimulate water retention. However, photosynthesis is very sensitive towards low soil moisture conditions. For instance, in August the soil moisture content was only 50% of that in July, and NDVI (greenness) declined, leading to lower photosynthesis, and less pronounced photosynthesis responses to treatments.
As for warming, litter addition also increased NDVI but in contrast to hypothesis 2 the addition of litter did not significantly alter GEP. This is likely because the modest increase in green biomass was insufficient to influence the photosynthesis of the whole ecosystem. The combined effect of litter and OTC warming on GEP was not different from the warming effect alone, leading to a tendency towards a significant interaction over the entire period. We speculate that the small positive effect of litter addition of nutrient availability may be less important under conditions of potentially increased nutrient availability in response to warming.
In the cold season from late October until late March, we did not detect any GEP in this tundra heath and hence no detectable difference on winter GEP of treatments. However, as it is known that tundra plants are able to perform photosynthesis under snow in spring (Larsen et al. 2007c; Starr and Oberbauer 2003) and at cold conditions in autumn (Christiansen et al. 2012), we still consider the attempt to measure GEP in winter important in order to exclude any effects of treatment on cold season GEP.
Treatment effect on net ecosystem production
Short- as well as long-term OTC treatment caused a higher loss of CO2 from the ecosystem during the snow-free season, as ER increased more than GEP, in accordance with hypothesis 4. This observation is consistent with other CO2 flux studies from arctic heaths (Illeris et al. 2004b; Oberbauer et al. 2007; Welker et al. 2004), and with later observations of reduced soil C stocks in OTC plots (but no change in litter amended plots) in our study site (Phillips et al. 2019). Hence, increased temperature has the potential to increase the emission of CO2 to the atmosphere after short as well as long time exposure. Likewise, litter addition decreased NEP as the treatment stimulated ER more than GEP. The combined effects of litter and OTCs were additive and resulted in the most pronounced negative effect on NEP (i.e. more pronounced CO2 release) through the measuring period. This strong and long lasting effect of warming and enhanced litter input on NEP could result in a positive feedback on climate change (Crowther et al. 2016; Schuur et al. 2015) and adds supports to the notion (Hartley et al. 2012) that shrub encroachment and forest expansion may lead to lower soil C stocks in the Subarctic. However, it should be noted that increased C assimilation and increased shrub nutrient demand are other important components of expansion of woody plants in tundra, and that this will affect the carbon balance and increase C stocks in the woody species (DeMarco et al. 2014b).
Conclusion
The current study presents whole year CO2 flux data and shows noteworthy ecosystem respiration throughout the late fall and winter. In late April during snowmelt both photosynthesis and respiration were substantial.
We found that warming increased the efflux of CO2 in the growing season when OTCs were applied but also in the shoulder seasons when there was no direct effect of warming. The increased efflux occurred despite increased GEP, as ER was stimulated even more both after two as well as after 13 years of treatment. Furthermore we report a legacy effect of 13 years of summer warming on ER in November during the cold season when GEP was not detectable. This emphasizes that off-growing season and spring fluxes must be taken into account in the effort to describe the carbon balance of northern ecosystems and the response to climate change.
Our flux measurements suggest that this subarctic wet heath ecosystem currently is a net daytime sink of carbon under ambient conditions during spring, summer and fall. Under future warmer conditions, the ecosystem sink capacity is reduced due to summer warming, both in the short and long term. Furthermore when the OTCs is combined with litter addition the ecosystem was a net daytime C source in all seasons. This suggests that if a warmer climate provides conditions for shrub expansion the combination of increased litter input and higher temperatures will enhance the efflux further. However, carbon uptake by expanding shrubs may partially mitigate this effect.
References
Abisko Scientific Research Station (2016) Temperature and precipitation data 1913–2015. Available from: http://polar.se/abisko. Accessed 13 Nov 2012
Arndal MF, Illeris L, Michelsen A, Albert K, Tamstorf M, Hansen BU (2009) Seasonal variation in gross ecosystem production, plant biomass, and carbon and nitrogen pools in five high arctic vegetation types. Arct Antarct Alp Res 41:164–173
Belshe EF, Schuur EG, Bolker BM (2013) Tundra ecosystems observed to be CO2 sources due to differential amplification of the carbon cycle. Ecol Lett 16:1307–1315
Bengtson P, Barker J, Grayston SJ (2012) Evidence of a strong coupling between root exudation, C and N availability, and stimulated SOM decomposition caused by rhizosphere priming effects. Ecol Evol 2:1843–1852
Boelman NT, Stieglitz M, Rueth HM, Sommerkorn M, Griffin KL, Shaver GR, Gamon JA (2003) Response of NDVI, biomass, and ecosystem gas exchange to long-term warming and fertilization in wet sedge tundra. Oecologia 135:414–421
Brooks PD, Grogan P, Templer PH, Groffman P, Öquist MG, Schimel J (2011) Carbon and nitrogen cycling in snow-covered environments. Geogr Compass 5:682–699
Campioli M, Leblans N, Michelsen A (2012) Twenty-two years of warming, fertilisation and shading of subarctic heath shrubs promote secondary growth and plasticity but not primary growth. PLoS One 7(4):e34842
Christiansen CT, Schmidt NM, Michelsen A (2012) High arctic dry heath CO2 exchange during the early cold season. Ecosystems 15:1083–1092
Collins M, Knutti R, Arblaster J (2013) Long-term climate change: projections, commitents and irreversibility Climate Change 2013: The Physical Science Basis Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change (eds Stocker TF, Qin D, Plattner G-K, Tignor M, Allen SK, Boschung J, Nauels A, Xia Y, Bex V, Midgley PM). Cambridge University Press, Cambridge , United Kingdom and New York, NY, USA
Crowther TW, Todd-Brown KEO, Rowe CW, Wieder WR, Carey JC, Machmuller MB, Snoek BL, Fang S, Zhou G, Allison SD, Blair JM, Bridgham SD, Burton AJ, Carrillo Y, Reich PB, Clark JS, Classen AT, Dijkstra FA, Elberling B, Emmett BA, Estiarte M, Frey SD, Guo J, Harte J, Jiang L, Johnson BR, Kröel-Dulay G, Larsen KS, Laudon H, Lavallee JM, Luo Y, Lupascu M, Ma LN, Marhan S, Michelsen A, Mohan J, Niu S, Pendall E, Peñuelas J, Pfeifer-Meister L, Poll C, Reinsch S, Reynolds LL, Schmidt IK, Sistla S, Sokol NW, Templer PH, Treseder KK, Welker JM, Bradford MA (2016) Quantifying global soil carbon losses in response to warming. Nature 540:104–108
Davidson EA, Janssens IA (2006) Temperature sensitivity of soil carbon decomposition and feedbacks to climate change. Nature 440:165–173
DeMarco J, Mack MC, Bret-Harte MS (2014a) Effects of arctic shrub expansion on biophysical vs. biogeochemical drivers of litter decomposition. Ecology 95:1861–1875
DeMarco J, Mack MC, Bret-Harte MS, Burton M, Shaver GR (2014b) Long-term experimental warming and nutrient additions increase productivity in tall deciduous shrub tundra. Ecosphere 5:72
Elmendorf SC, Henry GHR, Hollister RD, Björk RG, Boulanger-Lapointe N, Cooper EJ, Cornelissen JHC, Day TA, Dorrepaal E, Elumeeva TG, Gill M, Gould WA, Harte J, Hik DS, Hofgaard A, Johnson DR, Johnstone JF, Jónsdóttir IS, Jorgenson JC, Klanderud K, Klein JA, Koh S, Kudo G, Lara M, Lévesque E, Magnússon B, May JL, Mercado-Dı´az JA, Michelsen A, Molau U, Myers-Smith IH, Oberbauer SF, Onipchenko VG, Rixen C, Martin Schmidt N, Shaver GR, Spasojevic MJ, Þórhallsdóttir ÞE, Tolvanen A, Troxler T, Tweedie CE, Villareal S, Wahren CH, Walker X, Webber PJ, Welker JM, Wipf S (2012) Plot-scale evidence of tundra vegetation change and links to recent summer warming. Nat Clim Chang 2:453–457
Fahnestock JT, Jones MH, Brooks PD, Walker DA, Welker JM (1998) Winter and early spring CO2 efflux from tundra communities of northern Alaska. J Geophys Res-Atmos 103:29023–29027
Grogan P, Chapin FS (1999) Arctic soil respiration: effects of climate and vegetation depend on season. Ecosystems 2:451–459
Grogan P, Jonasson S (2005) Temperature and substrate controls on intra-annual variation in ecosystem respiration in two subarctic vegetation types. Glob Chang Biol 11:465–475
Grogan P, Jonasson S (2006) Ecosystem CO2 production during winter in a Swedish subarctic region: the relative importance of climate and vegetation type. Glob Chang Biol 12:1479–1495
Grogan P, Illeris L, Michelsen A, Jonasson S (2001) Respiration of recently-fixed plant carbon dominates mid-winter ecosystem CO2 production in sub-arctic heath tundra. Clim Chang 50:129–142
Hartley IP, Garnett M, Sommerkorn M, Hopkins DW, Fletcher BJ, Sloan VL, Wookey PA (2012) A potential loss of carbon associated with greater plant growth in the European Arctic. Nat Clim Chang 2:875–879
Hicks-Pries CE, van Logtestijn RSP, Schuur EAG, Natali SM, Cornelissen JHC, Aerts R, Dorrepaal E (2015) Decadal warming causes a consistent and persistent shift from heterotrophic to autotrophic respiration in contrasting permafrost ecosystems. Glob Chang Biol 21:4508–4519
Hicks-Pries CE, Bird JA, Castanha C, Hatton PJ, Torn MS (2017) Long term decomposition: the influence of litter type and soil horizon on retention of plant carbon and nitrogen in soils. Biogeochemistry 134:5–16
Hobbie SE (1996) Temperature and plant species control over litter decomposition in Alaskan tundra. Ecol Monogr 66:503–522
Hugelius G, Strauss J, Zubrzycki S, Harden JW, Schuur EAG, Ping CL, Schirrmeister L, Grosse G, Michaelson GJ, Koven CD, O'Donnell JA, Elberling B, Mishra U, Camill P, Yu Z, Palmtag J, Kuhry P (2014) Estimated stocks of circumpolar permafrost carbon with quantified uncertainty ranges and identified data gaps. Biogeosciences 11:6573–6593
Illeris L, Christensen TR, Mastepanov M (2004a) Moisture effects on temperature sensitivity of CO2 exchange in a subarctic heath ecosystem. Biogeochemistry 70:315–330
Illeris L, Konig SM, Grogan P, Jonasson S, Michelsen A, Ro-Poulsen H (2004b) Growing-season carbon dioxide flux in a dry subarctic heath: responses to long-term manipulations. Arct Antarct Alp Res 36:456–463
Lafleur PM, Humphreys ER, St Louis VL, Myklebust MC, Papakyriakou T, Poissant L, Barker JD, Pilote M, Swystun KA (2012) Variation in peak growing season net ecosystem production across the Canadian Arctic. Environ Sci Technol 46:7971–7977
Larsen KS, Grogan P, Jonasson S, Michelsen A (2007a) Respiration and microbial dynamics in two subarctic ecosystems during winter and spring thaw: effects of increased snow depth. Arct Antarct Alp Res 39:268–276
Larsen KS, Ibrom A, Beier C, Jonasson S, Michelsen A (2007b) Ecosystem respiration depends strongly on photosynthesis in a temperate heath. Biogeochemistry 85:201–213
Larsen KS, Ibrom A, Jonasson S, Michelsen A, Beier C (2007c) Significance of cold-season respiration and photosynthesis in a subarctic heath ecosystem in northern Sweden. Glob Chang Biol 13:1498–1508
Leffler AJ, Klein ES, Oberbauer SF, Welker JM (2016) Coupled long-term summer warming and deeper snow alters species composition and stimulates gross primary productivity in tussock tundra. Oecologia 181:287–297
Lett S, Michelsen A (2014) Seasonal variation in nitrogen fixation and effects of climate change in a subarctic heath. Plant Soil 379:193–204
Lund M, Falk JM, Friborg T, Mbufong HN, Sigsgaard C, Soegaard H, Tamstorf MP (2012) Trends in CO2 exchange in a high Arctic tundra heath, 2000-2010. J Geophys Res Biogeosci 117:G02001
Mauritz M, Bracho R, Celis G, Hutchings J, Natali SM, Pegoraro E, Salmon VG, Schädel C, Webb EE, Schuur EAG (2017) Nonlinear CO2 flux response to 7 years of experimentally induced permafrost thaw. Glob Chang Biol 23:3646–3666
McMichael CE, Hope AS, Stow DA, Fleming JB, Vourlitis G, Oechel W (1999) Estimating CO2 exchange at two sites in Arctic tundra ecosystems during the growing season using a spectral vegetation index. Int J Remote Sens 20:683–698
Metcalfe DB, Hermans TDG, Ahlstrand J, Becker M, Berggren M, Bjork RG, Bjorkman MP, Blok D, Chaudhary N, Chisholm C, Classen AT, Hasselquist NJ, Jonsson M, Kristensen JA, Kumordzi BB, Lee H, Mayor JR, Prevey J, Pantazatou K, Rousk J, Sponseller RA, Sundqvist MK, Tang J, Uddling J, Wallin G, Zhang WX, Ahlstrom A, Tenenbaum DE, Abdi AM (2018) Patchy field sampling biases understanding of climate change impacts across the Arctic. Nat Ecol Evol 2:1443–1448
Moore TR, Lafleur PM, Poon DMI, Heumann BW, Seaquist JW, Roulet NT (2006) Spring photosynthesis in a cool temperate bog. Glob Chang Biol 12:2323–2335
Myers-Smith IH, Forbes BC, Wilmking M, Hallinger M, Lantz T, Blok D, Tape KD, Macias-Fauria M, Sass-Klaassen U, Lévesque E, Boudreau S, Ropars P, Hermanutz L, Trant A, Collier LS, Weijers S, Rozema J, Rayback SA, Schmidt NM, Schaepman-Strub G, Wipf S, Rixen C, Ménard CB, Venn S, Goetz S, Andreu-Hayles L, Elmendorf S, Ravolainen V, Welker J, Grogan P, Epstein HE, Hik DS (2011) Shrub expansion in tundra ecosystems: dynamics, impacts and research priorities. Environ Res Lett 6:045509
Natali SM, Schuur EAG, Trucco C, Hicks-Pries CE, Crummer KG, Lopez AFB (2011) Effects of experimental warming of air, soil and permafrost on carbon balance in Alaskan tundra. Glob Chang Biol 17:1394–1407
Natali SM, Schuur EG, Webb EE, Hicks-Pries CE, Crummer KG (2014) Permafrost degradation stimulates carbon loss from experimentally warmed tundra. Ecology 95:602–608
Natali SM, Schuur EAG, Mauritz M, Schade JD, Celis G, Crummer KG, Johnston C, Krapek J, Pegoraro E, Salmon VG, Webb EE (2015) Permafrost thaw and soil moisture driving CO2 and CH4 release from upland tundra. J Geophys Res Biogeosci 120:525–537
Oberbauer SF, Tweedie CE, Welker JM, Fahnestock JT, Henry GHR, Webber PJ, Hollister RD, Walker MD, Kuchy A, Elmore E, Starr G (2007) Tundra CO2 fluxes in response to experimental warming across latitudinal and moisture gradients. Ecol Monogr 77:221–238
Pedersen EP, Elberling B, Michelsen A (2017) Seasonal variations in methane fluxes in response to summer warming and leaf litter addition in a subarctic heath ecosystem. J Geophys Res Biogeosci 122:2137–2153
Phillips CA, Elberling B, Michelsen A (2019) Soil carbon and nitrogen stocks and turnover following 16 years of warming and litter addition. Ecosystems 22:110–124
Post E, Forchhammer MC, Bret-Harte MS, Callaghan TV, Christensen TR, Elberling B, Fox AD, Gilg O, Hik DS, Hoye TT, Ims RA, Jeppesen E, Klein DR, Madsen J, McGuire AD, Rysgaard S, Schindler DE, Stirling I, Tamstorf MP, Tyler NJC, van der Wal R, Welker J, Wookey PA, Schmidt NM, Aastrup P (2009) Ecological dynamics across the Arctic associated with recent climate change. Science 325:1355–1358
PP Systems (2018) EGM-5 portable CO2 gas analyzer operation manual, version 1.03, Amesbury, MA 01913 U.S.A.; www.ppsystems.com
Ravn NR, Ambus P, Michelsen A (2017) Impact of decade-long warming, nutrient addition and shading on emission and carbon isotopic composition of CO2 from two subarctic dwarf shrub heaths. Soil Biol Biochem 111:15–24
Rinnan R, Michelsen A, Jonasson S (2008) Effects of litter addition and warming on soil carbon, nutrient pools and microbial communities in a subarctic heath ecosystem. Appl Soil Ecol 39:271–281
Schuur EAG, McGuire AD, Schadel C, Grosse G, Harden JW, Hayes DJ, Hugelius G, Koven CD, Kuhry P, Lawrence DM, Natali SM, Olefeldt D, Romanovsky VE, Schaefer K, Turetsky MR, Treat CC, Vonk JE (2015) Climate change and the permafrost carbon feedback. Nature 520:171–179
Seneviratne SI, Rogelj J, Seferian R, Wartenburger R, Allen MR, Cain M, Millar RJ, Ebi KL, Ellis N, Hoegh-Guldberg O, Payne AJ, Schleussner CF, Tschakert P, Warren RF (2018) The many possible climates from the Paris Agreement's aim of 1.5 degrees C warming. Nature 558:41–49
Serreze MC, Walsh JE, Chapin FS, Osterkamp T, Dyurgerov M, Romanovsky V, Oechel WC, Morison J, Zhang T, Barry RG (2000) Observational evidence of recent change in the northern high-latitude environment. Clim Chang 46:159–207
Shaver GR, Chapin FS III (1980) Response to fertilization by various plant growth forms in an Alaskan tundra. Ecology 61:662–675
Sistla SA, Moore JC, Simpson RT, Gough L, Shaver GR, Schimel JP (2013) Long-term warming restructures Arctic tundra without changing net soil carbon storage. Nature 497:615–618
Sorensen PL, Michelsen A (2011) Long-term warming and litter addition affects nitrogen fixation in a subarctic heath. Glob Chang Biol 17:528–537
Starr G, Oberbauer SF (2003) Photosynthesis of arctic evergreens under snow: implications for tundra ecosystem carbon balance. Ecology 84:1415–1420
Street LE, Shaver GR, Willliams M, van Wijk MT (2007) What is the relationship between changes in canopy leaf area and changes in photosynthetic CO2 flux in arctic ecosystems? J Ecol 95:139–150
Sturm M, Schimel J, Michaelson G, Welker JM, Oberbauer SF, Liston GE, Fahnestock J, Romanovsky VE (2005) Winter biological processes could help convert arctic tundra to shrubland. Bioscience 55:17–26
Tiiva P, Faubert P, Michelsen A, Holopainen T, Holopainen JK, Rinnan R (2008) Climatic warming increases isoprene emission from a subarctic heath. New Phytol 180:853–863
Virkkala AM, Virtanen T, Lehtonen A, Rinne J, Luoto M (2018) The current state of CO2 flux chamber studies in the Arctic tundra: a review. Progress in Physical Geography-Earth and Environment 42:162–184
Voigt C, Lamprecht RE, Marushchak ME, Lind SE, Novakovskiy A, Aurela M, Martikainen PJ, Biasi C (2016) Warming of subarctic tundra increases emissions of all three important greenhouse gases - carbon dioxide, methane and nitrous oxide. Glob Chang Biol 43:4566–4575
Webb EE, Schuur EAG, Natali SM, Oken KL, Bracho R, Krapek JP, Risk D, Nickerson NR (2016) Increased wintertime CO2 loss as a result of sustained tundra warming. J Geophys Res Biogeosci 121:249–265
Welker JM, Brown KB, Fahnestock JT (1999) CO2 flux in Arctic and alpine dry tundra: comparative field responses under ambient and experimentally warmed conditions. Arct Antarct Alp Res 31:272–277
Welker JM, Fahnestock JT, Henry GHR, O'dea KW, Chimner RA (2004) CO2 exchange in three Canadian high Arctic ecosystems: response to long-term experimental warming. Glob Chang Biol 10:1981–1995
Acknowledgements
We acknowledge The Danish Council for Independent Research and The Danish National Research Foundation (CENPERM DNRF100) for financial support. The research has additionally received funding from INTERACT (grant agreement No 262693) under the European Community’s Seventh Framework Programme. We thank Marie Glahn for field assistance, Abisko Scientific Research Station for logistic support and access to climatic data and Casper T. Christiansen for internal revision of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Luca Bragazza.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(PDF 742 kb)
Rights and permissions
About this article
Cite this article
Finderup Nielsen, T., Ravn, N.R. & Michelsen, A. Increased CO2 efflux due to long-term experimental summer warming and litter input in subarctic tundra – CO2 fluxes at snowmelt, in growing season, fall and winter. Plant Soil 444, 365–382 (2019). https://doi.org/10.1007/s11104-019-04282-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-019-04282-9