Abstract
Background and Aims
Forests play a vital role in regulation of the global carbon cycle. Mechanistically understanding how their ecosystem functioning relates to biodiversity is necessary for predicting the consequences of biodiversity loss and for setting conservation priorities. Here, we test whether carbon stocks in a subtropical evergreen broad-leaved forest in China are more strongly influenced by plant functional diversity (FD), as would be predicted by the ‘niche complementarity hypothesis’, or by community-weighted mean (CWM) functional trait values, as would be predicted by the ‘mass ratio hypothesis’.
Methods
Using data from a 24-ha plot subdivided into 400 m2 quadrats, we determined relationships of aboveground carbon (AGC) and topsoil (1–10 cm) organic carbon (SOC) to topographic variables, stem density, CWM and FD of six functional traits hypothesized to influence carbon stocks.
Results
After accounting for topographic variables and tree stem density, boosted regression tree models revealed that CWMs were the dominant driving factors for both AGC and SOC, whereas FD had negligible effects. AGC and SOC were influenced by different functional traits, with AGC responding most strongly to CWM values for wood density and maximum tree height, and SOC responding most strongly to elevation, indicating that these carbon stocks are shaped by different underlying mechanisms.
Conclusions
Our results support the mass ratio hypothesis but not the niche complementarity hypothesis. Our study implies that, when it comes to maximizing forest carbon storage, conservation priorities should focus on protection of species with traits associated to high carbon stocks.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Anthropogenic activities are extirpating species from large portions of their historical ranges, resulting in reduced biodiversity over much of Earth’s land area and rates of species loss estimated to be 1000-fold greater than natural extinction rates (Sala et al. 2000; Pimm et al. 2014). Experiments using relatively short-lived species such as bacteria, fungi, algae, and grasses have demonstrated that ecosystems with higher biodiversity generally have enhanced ecosystem functioning — i.e., higher carbon (C) stocks, productivity, or resilience — and that biodiversity loss could impair ecosystem functioning (Hooper et al. 2005; Isbell et al. 2011; Cardinale et al. 2012; Tilman et al. 2014). However, there remains debate as to how biodiversity affects ecosystem functioning in more structurally complex natural communities containing long-lived organisms, including forests (e.g., Paquette and Messier 2011; Ruiz-Jaen and Potvin 2011; Conti and Díaz 2013; Cavanaugh et al. 2014; Ruiz-Benito et al. 2014; Finegan et al. 2015; Prado-Junior et al. 2016). Considering the fact that forests cover ~31 % of the global land area, harbor about two thirds of terrestrial biodiversity and play a critical role in global C balance (MEA 2005; FAO 2010), improved understanding of the relationship between biodiversity and C stocks in natural forests is critical.
Biodiversity is mechanistically linked to ecosystem functioning through the functional traits of the species that comprise the community (Petchey and Gaston 2006; Díaz et al. 2007; Cadotte et al. 2011). Two testable, non-mutually exclusive, hypotheses have been put forward to address the mechanisms that potentially underpin relationships between biodiversity and ecosystem functioning: (i) the mass ratio hypothesis, and (ii) the niche complementarity hypothesis. The mass ratio hypothesis posits that ecosystem functioning is determined predominantly by the functional traits of the dominant species in a community (Grime 1998; Mokany et al. 2008). More explicitly, it implies that community-weighted mean (CWM) functional trait values can explain significant variation in ecosystem functioning (Díaz et al. 2007). In forest ecosystems, it has been reported that trait values associated with higher wood volume (e.g., higher maximum potential tree height, Hmax) or higher wood density (WD) lead to higher biomass C stocks (Bunker et al. 2005; Conti and Díaz 2013; Finegan et al. 2015; Lin et al. 2015). Traits associated with high productivity or rapid biomass turnover — e.g., higher leaf nutrient concentrations or specific leaf area (SLA) — may lead to faster tree growth and higher rates of C input to the soil, but may also result in shorter C residence times through higher tree mortality or faster litter decomposition rates (Garnier et al. 2004; Wright et al. 2010; Freschet et al. 2012).
The niche complementarity hypothesis suggests that trait functional diversity (FD) positively influences ecosystem functioning (Hooper and Vitousek 1997; Heemsbergen et al. 2004). The complementarity effect occurs through niche partitioning (i.e., differences in resource-use strategies) and/or interspecific interactions (e.g., facilitation) that allow for more efficient use of available resources by functionally diverse species assemblages (Cardinale et al. 2002; Cardinale 2011). Higher FD in forests would imply that trees exhibit greater resource-use complementarity and/or lower competition among species, leading to greater productivity. In turn, higher woody productivity would lead to higher live biomass if not counterbalanced by higher wood mortality, and higher root productivity or litterfall would lead to higher soil C if not counterbalanced by faster decomposition rates. Recently, a number of studies have examined the role of FD in driving forest productivity or C stocks, with mixed results (e.g., Paquette and Messier 2011; Conti and Díaz 2013; Ruiz-Benito et al. 2014; Finegan et al. 2015; Prado-Junior et al. 2016).
To date, most studies on the relationships between biodiversity and C stocks in forests have been limited to aboveground C (e.g., Ruiz-Jaen and Potvin 2011; Cavanaugh et al. 2014; Ruiz-Benito et al. 2014; Finegan et al. 2015; Prado-Junior et al. 2016). However, forest soil C stocks can rival or exceed aboveground C stocks (Fahey et al. 2010), and it is therefore critical to understand the mechanisms through which plant traits influence soil C. Plant species composition influences soil C through interspecific variation in litter quality (e.g., decomposability) and input rates (De Deyn et al. 2008; Fornara and Tilman 2008; Lange et al. 2015). Plants and the soil biota that metabolize plant C input interactively shape the soil C balance, such that plant C input rates do not necessarily predict soil C stocks (De Deyn et al. 2008; Grigulis et al. 2013; Lange et al. 2015). For instance, higher C inputs to the litter layer in the form of increased litterfall or cellulose (in a controlled experiment) can have a “priming” effect that accelerates the decomposition of soil C, resulting in net soil C loss (Fontaine et al. 2004; Sayer et al. 2011). Thus, the influences of plant functional traits on soil C will be mediated by soil biota over time scales of decades to centuries, resulting in complex linkages between tree functional traits and soil C.
Here, we utilize tree census and soil data from an old-growth subtropical evergreen broad-leaved forest in China to understand how CWM and FD of tree functional traits affects aboveground and soil C stocks on a 20 m spatial scale. We hypothesize that, after accounting for topographic variables and stem density, forest C stocks will be influenced by both CWM and FD of tree functional traits. Specifically, we predict positive effects of CWM of Hmax and WD on aboveground C stock (AGC), owing to potential greater tree biomass, and negative effects of CWM leaf nutrient concentrations and SLA on soil organic C stock (SOC), owing to faster litter decomposition rates. With regards to FD, we expect that quadrats with higher FD will have higher AGC and SOC, owing to more efficient utilization of available resources, thus leading to higher biomass production and greater litter inputs to the soil. We further hypothesize that CWMs will explain more variation in C stocks than FD — the mass ratio hypothesis will be more appropriate than the niche complementarity hypothesis for explaining the variation of C stocks in this old-growth subtropical forest.
Material and methods
Study site
This study was conducted in a natural evergreen broad-leaved forest in the Gutianshan National Nature Reserve in southeast China (29.15° N, 118.07° E). The region has a subtropical monsoon climate, receiving on average 1964 mm of precipitation annually and with a mean annual temperature of 15.3 °C. The bedrock of the mountain range is granite, and the soils are moderately acidic (pH 5.5–6.5). The reserve covers 8100-ha of evergreen broad-leaved mixed forest, with Castanopsis eyrei and Schima superba as dominant tree species (Zhu et al. 2008; Legendre et al. 2009; Lin et al. 2012). A permanent forest dynamics plot with an area of 24-ha (400 × 600 m) belonging to CForBio (www.cfbiodiv.org/english/) and affiliated with the Center for Tropical Forest Science-Forest Global Earth Observatory (CTFS-ForestGEO; www.forestgeo.si.edu) was established in 2005 following CTFS-ForestGEO protocols (Condit 1998), which have been applied at >60 sites globally (Anderson-Teixeira et al. 2015). Specifically, the plot was divided into 600 20 × 20 m quadrats and all stems with diameter at breast height (1.3 m height) ≥1 cm were mapped, tagged, identified to species and measured (Zhu et al. 2008). A total of ~140,700 individuals belonging to 159 species were recorded in the plot (Zhu et al. 2008).
Quantifying C stocks
We quantified C stocks in live trees (aboveground C; AGC) and soil to a depth of 10 cm (soil organic C; SOC) for the entire 24-ha plot. Aboveground biomass was estimated based on tree census data in combination with 14 species-specific allometric equations (covering ~80 % of total basal area of the plot) and a general allometric equation for the remaining species, as detailed in Lin et al. (2012). All allometric equations are given in Lin et al. (2012). AGC was estimated using the IPCC conversion rate of 0.47 for biomass to carbon i.e. biomass × 0.47 (IPCC 2006).
SOC was quantified by collecting soil samples from the surface (0–10 cm) soil (Zhang et al. 2011). Sampling took place in July and August 2007 using a protocol similar to that of John et al. (2007), which has been applied at numerous CTFS-ForestGEO plots (Anderson-Teixeira et al. 2015). Specifically, we sampled soils across a regular grid of points every 30 m using a metal cylinder (Zhang et al. 2011). In order to capture variations in SOC at finer scales, two additional samples were taken at 2, 5, or 15 m in a randomly selected direction (N, NE, E, SE, etc.) from each base point (Zhang et al. 2011). A total of 893 soil samples were taken from the plot. Before C analysis, soil samples were air-dried, macroscopic roots were removed, and samples were passed through a 2 mm sieve. C concentration (g kg−1) was determined by a titrimetric method using a strong oxidizing agent (K2Cr2O7) in the presence of H2SO4 (Walkley-Black method). The bulk density was assessed by means of 100 cm3 steel cores taken from the place near the sampling points. Samples were oven-dried (105 °C for 48 h) and bulk density was estimated as the mass of oven-dry soil divided by the core volume. SOC stock (Mg ha−1) was calculated for each individual soil sample as follows:
where C is organic C concentration (g kg−1), ρ is bulk density (g cm−3), a is volumetric percentage of coarse fragments (> 2 mm) in the soil (%), Δd is the thickness of the soil layer (10 cm here), and the 0.1 multiplier integrates multiple conversions such that units cancel. SOC stock values for 20 × 20 m quadrats were estimated using geostatistical methods described in John et al. (2007). Briefly, data were log-transformed to improve the normality. We then performed a polynomial trend-surface regression, which did not reveal a large spatial trend in the data across the plot. Then, we fitted variogram models to the empirical variograms, and obtained spatial predictions for 5 × 5 m blocks using ordinary kriging. Finally, we transformed the data back to a linear scale. The analysis was conducted in GS + ™ version 9.0 (GeoStatistics for the Environmental Sciences, by Gamma design software). For 20 × 20 m quadrats, the SOC stock was calculated as the mean value of the 16 5 × 5 m blocks within each quadrat.
Environmental factors and stem density
As topography correlates with multiple aspects of the physical environment that may influence C stocks including soil fertility, drainage, and sunlight conditions, we included four topographic variables in our analyses (Table 1): relative elevation, slope, aspect and convexity, calculated as in Legendre et al. (2009). Briefly, relative elevation of a quadrat was calculated as the mean of the relative elevation values at its four corners (the lowest part of the 24-ha plot was set as 0 m). Slope was calculated as the mean angular deviation from horizontal of the four planes formed by connecting three corners of a quadrat at a time. Aspect was measured as the direction to which a slope faces. Finally, convexity was calculated as the relative elevation of a quadrat minus the average relative elevation of all immediate neighbor quadrats, but for the edge quadrats, convexity was the relative elevation of the quadrat minus the mean relative elevation of its four corners.
Some quadrats in the high elevation ridge are extremely high in stem density possibly due to the small scale disturbance in the past (Zhu et al. 2008). So, in addition to the topographic variables, we also considered the tree stem density in the analysis (Table 1).
Quantifying CWM and FD
We selected six plant functional traits that could potentially affect ecosystem C stock and for which published data were available for our study plot (Cao et al. 2013; Liu et al. 2012). These included wood density (WD, the ratio of the oven-dried mass of a wood sample divided by its fresh volume, g cm−3), the maximum height typically attained by mature individuals of a species (Hmax, m), and four leaf traits: leaf area (Aleaf, one-sided area of an individual leaf, cm2), SLA (Aleaf divided by its oven-dry mass, cm2 g−1), leaf nitrogen content (Nleaf, total amounts of nitrogen per unit of dry leaf mass, %); and leaf phosphorus content (Pleaf, total amounts of phosphorus per unit of dry leaf mass, ppm). Measurement methods for these traits are detailed in Cao et al. (2013) and Liu et al. (2012).
CWM of each functional trait was calculated for each quadrat as the averaged trait value in the community weighted by the species basal area (Pla et al. 2012). FD was measured through three complementary multi-trait indices: functional richness (FRic), functional evenness (FEve) and functional divergence (FDiv). FRic shows the amount of multivariate-trait space filled by the community, FEve denotes how species’ basal area is spread over multivariate-trait space, and FDiv indicates the degree of divergence from the center that most dominant species occupy in multivariate-trait space (Villeger et al. 2008). All functional metrics were calculated using the open-access software FDiversity, and in order to give each trait equal weight, standard scores of trait values were used in calculating multi-trait indices (Pla et al. 2012).
Statistical analysis
All analysis were conducted across 20 × 20 m quadrats. Six quadrats with some obviously inaccurate DBH measurements (e.g., understorey species with impossible large DBH) were removed from the analyses. We used boosted regression tree (BRT) analysis to assess the relative importance of topographic variables, stem density, CWM and FD in shaping the spatial pattern of C stocks. BRT is a machine learning-based technique that combines two algorithms: regression trees and boosting, and it has been suggested as one of the best modeling approaches to statistically describe the response of dependent variables to multiple predictors (Elith et al. 2008). BRT has several advantages such as strength in handling nonlinear relationships, robust to the effects of outliers, missing data, autocorrelation and collinearity among variables (De’ath 2007; Elith et al. 2008).
The four main parameters to optimize in BRT models are the learning rate, bag fraction, tree complexity and the number of cross-validation folds (Elith et al. 2008). We fitted all BRT models using the recommended optimal settings for ecological study, i.e. learning rate (0.005), bag fraction (0.6) and 10-folds cross-validation (Elith et al. 2008). For tree complexity, i.e. the number of nodes in each tree, indicated the level of interactions in BRT (e.g., a value of 2 permits up to two-way interactions). We ran alternative models by setting tree complexity as 1, 2, 3, 4, 5 and 6. In order to avoid over-fitting, we selected to report the BRT model with a lower tree complexity if the models with a higher tree complexity did not reduce prediction error considerably (i.e. < 5 %; Table S2). BRT analyses were run in R version 2.14.1 (R Development Core Team 2011) with the gbm packages (Ridgeway 2010) and additional functions provided by Elith et al. (2008). Models were fitted using the “gbm.step” function and a Gaussian distribution of the response variables (C stocks data were square-root transformed to improve normality). We removed CWM.Pleaf from BRT analyses because it shown strong correlation with CWM.Nleaf (Spearman’s ρ = 0.81, P < 0.001; Table S1). Because results from cross-validation can vary according to the bag fraction and depending on the random selection of points for the folds, all of the procedure was repeated 30 times for each model, and overall mean was calculated for the prediction error, the optimal number of trees, R 2 and relative influence of the predictor variables (Table S2 and S3). Moreover, we tested for spatial autocorrelation in the residuals of BRT models using Moran’s I statistic, which did not prove to be significant (Table S4).
The relative influence of predictors were expressed as percentages and the sums equal to 100 %, where a higher number indicates a greater influence of the predictor on the response variable (Elith et al. 2008). Visualization of the fitted functions in BRT models was achieved using partial dependence plots, which show how the model response is affected by focal predictor after accounting for the average effects of all other predictors in the model (Elith et al. 2008).
Results
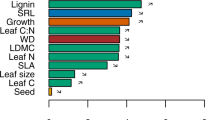
Topographic variables, stem density, CWM and FD jointly accounted for 73 and 76 % of variations in AGC and SOC, respectively (Table S3). The relative influence of the predictors on the two C stocks were different (Fig. 1; Table S3). CWM traits had the highest and second-highest summed relative influence on AGC and SOC, respectively, whereas FD had the lowest relative influence on both AGC and SOC (Fig. 1). CWM.WD and CWM.Hmax were the first two most important predictors for AGC, contributing 28.14 and 18.69 % of the explained variation, respectively (Fig. 1a; Table S3). Relative elevation and CWM.Hmax were the first two most important predictors for SOC, contributing 32.82 and 15.91 % of the explained variation, respectively (Fig. 1b; Table S3).
Relative influence of environmental, CWM and FD variables on (a) aboveground C stocks and (b) topsoil organic C in the boosted regression tree analysis. Pie charts show the summed relative influences of topographic variables and stem density, CWM and FD. Error bars show 95 % confidence intervals obtained from 1000 bootstrap samples of the original dataset (n = 30). Variable abbreviations are given in Table 1 notes
AGC decreased with CWM.WD up to values of ~0.6 g cm−3 and markedly increased with CWM.Hmax between 15 to 28 m (Fig. 2a). AGC decreased slightly with increasing CWM.SLA, while CWM.Aleaf and CWM.Nleaf both contributed very little (<7.5 %) to the explained variation (Fig. 2a; Table S3). AGC had no meaningful relationship to FRic, FEve, or FDiv, each of which contributed <3.5 % of the explained variation (Fig. 3a; Table S3). AGC was at most weakly related to each of the topographic variables, which contributed ≤5.5 % of the explained variation, and to stem density, which contributed 7.8 % of the explained variation (Fig. 4a; Table S3).
Partial dependence plots showing how aboveground C stocks (a) and topsoil organic C (b) depend on CWM variables after accounting for the average effects of the other predictors in boosted regression tree analysis. Each point represents observed value for one quadrat. Variable abbreviations are given in Table 1 notes
Partial dependence plots showing how aboveground C stocks (a) and topsoil organic C (b) depend on FD variables after accounting for the average effects of the other predictors in boosted regression tree analysis. Each point represents observed value for one quadrat. Variable abbreviations are given in Table 1 notes
Partial dependence plots showing how aboveground C stocks (a) and topsoil organic C (b) depend on topographic variables and stem density after accounting for the average effects of the other predictors in boosted regression tree analysis. Each point represents observed value for one quadrat. Variable abbreviations are given in Table 1 notes
In contrast to AGC, SOC increased with CWM.WD until CWM.WD > 0.62 g cm−3 and decreased with CWM.Hmax between 15 and 25 m (Fig. 2b). SOC weakly decreased with CWM.Nleaf and CWM.SLA, and the association with CWM.Aleaf was negligible (Fig. 2b; Table S3). Similar to AGC, SOC was at most weakly associated with FRic, FEve, and FDiv, each of which contributed <5.3 % of the explained variation (Fig. 3b; Table S3). SOC decreased markedly with relative elevation and was only weakly associated with the other three topographic variables (each contributing ≤6.1 % of the explained variation) and with stem density (which contributed 3.2 % of the explained variation; Fig. 4b; Table S3).
Discussion
Our analysis parsed out how different ecological factors shape the spatial pattern of C stocks within a typical subtropical evergreen broad-leaved forest in China, revealing that topographic factors and stem density have a modest influences on AGC and an important role in shaping SOC (Figs. 1 and 4). After controlling for these factors, we found that C stocks were meaningfully influenced by CWM but not by FD (Figs. 1, 2 and 3), supporting the mass ratio hypothesis, but leading no support for the niche complementarity hypothesis in this old-growth natural forest.
CWM effect on C stocks
Consistent with the prediction of mass ratio hypothesis of Grime (1998), which suggesting that ecosystem functioning was driven by the traits of the dominant species in the community, BRT analyses showed that plant functional trait effects were mostly attributed to CWM for both AGC and SOC (Fig. 1). This result is in agreement with findings reported in previous studies that have found the traits of dominant species exerted large effects on various aspects of ecosystem functioning, such as nutrient cycling, biomass production and C stocks (Garnier et al. 2004; Mokany et al. 2008; Conti and Díaz 2013; Grigulis et al. 2013; Cavanaugh et al. 2014; Finegan et al. 2015; Prado-Junior et al. 2016).
CWM.WD and CWM.Hmax were the two most important variables for explaining AGC (Fig. 1a, Table S3). Theoretically, species with higher WD have a higher biomass per unit stem volume, thus leading to higher stand C stock (Bunker et al. 2005; Chave et al. 2009). In contrast to this expectation, we found negative association between CWM.WD and AGC (Fig. 2a), as has also been observed in two tropical rainforest plots (Stegen et al. 2009). The explanation may lie in the fact that lower WD is associated with faster diameter growth rates (e.g., Wright et al. 2010), leading to a negative association between WD and basal area. Indeed, there was a negative relationship between CWM.WD and basal area in our study plot (Spearman’s ρ = − 0.58, P < 0.001). This indicates that quadrats with higher CWM.WD tended to be dominated by smaller trees with higher WD, whereas quadrats including dominant canopy trees had higher biomass and lower CWM.WD. In contrast with our result, Prado-Junior et al. (2016) reported that CWM.WD positively related to aboveground biomass in dry tropical forests, probably because trees with higher WD growth better in the dry environment.
AGC increased markedly with CWM.Hmax (Fig. 2a), suggesting that quadrats with a high percentage of canopy tree species tended to have high AGC. This result agrees with several previous studies conducted in natural forests (Ruiz-Jaen and Potvin 2011; Conti and Díaz 2013; Finegan et al. 2015), and it makes sense that quadrats with trees that can grow tall have higher biomass. Although actual and maximum height differ, mean community height can only be high if canopy species are present.
Consistent with our expectations, SOC was positively associated with CWM.WD and negatively associated with CWM of SLA and Nleaf (Fig. 2b), suggesting that higher SOC was associated with tree community with conservative plant trait values, i.e., low SLA, Nleaf and high WD (Wright et al. 2004; Chave et al. 2009). This is likely the result of slower litter decomposition rates of species with conservative traits. This result is consistent with theoretical predictions that nutrient and organic matter turnover should be faster in communities dominated by exploitative species (high SLA and Nleaf, low WD) and, conversely, slower in communities dominated by conservative species (low SLA and Nleaf, high WD; De Deyn et al. 2008; Freschet et al. 2012). Consistent with our result, in a boreal forest, plant community composition shifts from faster-growing acquisitive species to slower-growing conservative species, leading to greater belowground C stocks (Jonsson and Wardle 2010). Similarly, many studies in grasslands found communities dominated by conservative species contain greater soil C than the communities dominated by exploitative species (e.g., Garnier et al. 2004; Grigulis et al. 2013).
It has been reported that microhabitats dominated by tall plants shed more plentiful litter, thereby tending to have higher SOC (Lavorel et al. 2011; Conti and Díaz 2013). However, in contrast to what would be expected if SOC increased with mean plant height, we found negative association between CWM.Hmax and SOC (Fig. 2b). One potential explanation is that soil microbial activity is stimulated (“primed”) by the addition of large quantities of fresh and easily decomposable organic matter, thus resulting in extra decomposition of soil organic matter (Fontaine et al. 2004; Sayer et al. 2011). Such a phenomenon has been observed in other ecosystems, for example, in a large-scale litter manipulation experiment, increased tree litter input enhanced soil C release in a lowland tropical forest (Sayer et al. 2011).
FD effect on C stocks
Based on the niche complementarity hypothesis, we expected that quadrats with higher FD (higher FRic and FEve, lower FDiv) will have higher AGC and SOC; however, this hypothesis was not supported. Rather, C stocks were minimally influenced by FD variables (Figs. 1 and 3; Table S3). This result is in agreement with previous studies that did not find niche complementarity among tree species promoting C stocks in natural forests (Conti and Díaz 2013; Finegan et al. 2015; Prado-junior et al. 2016).
Previous studies suggest that the mass ratio and niche complementarity hypotheses are not mutually exclusive both can play a role in structuring ecosystem functioning (Schumacher and Roscher 2009; Cavanaugh et al. 2014; Ruiz-Benito et al. 2014). It is possible that we did not observed a niche complementarity effect because functional traits that relate strongly to plant complementary resource use were not included in our analysis. Obviously, detection of niche complementarity effects requires inclusion of functional traits, the identification of which is relatively difficult and strongly dependent on prior knowledge (Petchey and Gaston 2006; Flynn et al. 2011). Further studies could consider incorporating more traits such as tree crown structure and underground root traits (e.g., Brassard et al. 2013; Jucker et al. 2015). Alternatively, phylogenetic diversity could be considered in place of functional traits so long as the traits that important to the focused ecosystem functioning are phylogenetically conserved (Flynn et al. 2011; Srivastava et al. 2012). In addition, although we have considered four topographic variables and stem density in the analyses, we still cannot rule out all possible confounding factors which could possibly mask a relationship between FD and C stocks. Thus, while the existence of a niche complementarity effect in this forest cannot be ruled out, our analysis indicates that FD is unlikely to be a dominant driver of C stocks.
Environmental factors effect on C stocks
We evaluated the role of four topographic variables and stem density in driving forest C stocks. None of these variables influenced ≥10 % of AGC, and only relative elevation had a strong influence on SOC, contributing 33 % of the explained variation (Fig. 1; Table S3). Elevation plays a key role in determining the temperature and moisture regime of any microsite (Griffiths et al. 2009), thereby affecting the SOC by changing the input of litter via primary production and output of organic material through soil mineralization. We found that SOC was most strongly driven by elevation (Fig. 1b; Table S3), decreasing markedly with relative elevation (Fig. 4b). Multiple abiotic variables — including radiation, wind exposure, soil moisture, and temperature — commonly vary with elevation and may influence both plant productivity and decomposition rates, and soil carbon may be increased by either higher productivity or lower decomposition at the lower elevations. In addition, litter and surface soil organic matter transfer from higher to lower elevations through surface erosion and movement of the soil mass could also lead to the negative correlation between SOC and elevation.
Conclusion
In summary, in this natural forests, the mass ratio hypothesis provides a more appropriate framework for explaining how tree functional traits shape C stocks than the niche complementarity hypothesis, for which we found no support. Our study also demonstrates that AGC and SOC are shaped by different factors; plant functional traits that lead to higher AGC do not necessary promote accumulation of C in the soil. It is therefore important to disentangle the separate mechanisms by which plant functional traits driving different ecosystem-level C stocks.
A clear implication of our results is that it is important to consider the traits of dominant species when managing subtropical evergreen broad-leaved forest to maximize C stocks. Specifically, traits associated with high wood volume maximize AGC, while traits associated with conservative growth strategies maximize SOC. These findings may be useful for developing optimally effective strategies to preserve and promote C sequestration in forest ecosystems; however, it is also important that management strategies balance maximization of C stocks against potentially conflicting ecosystem goods and services including protection of biodiversity and resilience.
References
Anderson-Teixeira KJ, Davies SJ, Bennett AC, Gonzalez-Akre EB, Muller-Landau HC, Wright SJ, Abu Salim K, Zambrano AMA, Alonso A, Baltzer JL, Basset Y, Bourg NA, Broadbent EN, Brockelman WY, Bunyavejchewin S, Burslem DFRP, Butt N, Cao M, Cardenas D, Chuyong GB, Clay K, Cordell S, Dattaraja HS, Deng XB, Detto M, XJ D, Duque A, Erikson DL, Ewango CEN, Fischer GA, Fletcher C, Foster RB, Giardina CP, Gilbert GS, Gunatilleke N, Gunatilleke S, Hao ZQ, Hargrove WW, Hart TB, Hau BCH, He FL, Hoffman FM, Howe RW, Hubbell SP, Inman-Narahari FM, Jansen PA, Jiang MX, Johnson DJ, Kanzaki M, Kassim AR, Kenfack D, Kibet S, Kinnaird MF, Korte L, Kral K, Kumar J, Larson AJ, Li YD, Li XK, Liu SR, Lum SKY, Lutz JA, Ma KP, Maddalena DM, Makana JR, Malhi Y, Marthews T, Serudin RM, McMahon SM, McShea WJ, Memiaghe HR, Mi XC, Mizuno T, Morecroft M, Myers JA, Novotny V, de Oliveira AA, Ong PS, Orwig DA, Ostertag R, den Ouden J, Parker GG, Phillips RP, Sack L, Sainge MN, Sang WG, Sri-ngernyuang K, Sukumar R, Sun IF, Sungpalee W, Suresh HS, Tan S, Thomas SC, Thomas DW, Thompson J, Turner BL, Uriarte M, Valencia R, Vallejo MI, Vicentini A, Vrska T, Wang XH, Wang XG, Weiblen G, Wolf A, Xu H, Yap S, Zimmerman J (2015) CTFS-ForestGEO: a worldwide network monitoring forests in an era of global change. Glob Chang Biol 21:528–549. doi:10.1111/gcb.12712.
Brassard BW, Chen HYH, Cavard X, Laganiere J, Reich PB, Bergeron Y, Pare D, Yuan ZY (2013) Tree species diversity increases fine root productivity through increased soil volume filling. J Ecol 101:210–219. doi:10.1111/1365-2745.12023.
Bunker DE, DeClerck F, Bradford JC, Colwell RK, Perfecto I, Phillips OL, Sankaran M, Naeem S (2005) Species loss and aboveground carbon storage in a tropical forest. Science 310: 1029–1031. doi: 10.1126/science.1117682.
Cadotte MW, Carscadden K, Mirotchnick N (2011) Beyond species: functional diversity and the maintenance of ecological processes and services. J Appl Ecol 48:1079–1087. doi:10.1111/j.1365-2664.2011.02048.x.
Cao K, Rao M, Yu J, Liu X, Mi X, Chen J (2013) The phylogenetic signal of functional traits and their effects on community structure in an evergreen broad-leaved forest. Biodivers Sci 21:564–571. doi:10.3724/SP.J.1003.2013.08068. (in Chinese)
Cardinale BJ (2011) Biodiversity improves water quality through niche partitioning. Nature 472:86–U113. doi:10.1038/nature09904.
Cardinale BJ, Palmer MA, Collins SL (2002) Species diversity enhances ecosystem functioning through interspecific facilitation. Nature 415:426–429. doi:10.1038/415426a.
Cardinale BJ, Duffy JE, Gonzalez A, Hooper DU, Perrings C, Venail P, Narwani A, Mace GM, Tilman D, Wardle DA, Kinzig AP, Daily GC, Loreau M, Grace JB, Larigauderie A, Srivastava DS, Naeem S (2012) Biodiversity loss and its impact on humanity. Nature 486:59–67. doi:10.1038/Nature11148.
Cavanaugh KC, Gosnell JS, Davis SL, Ahumada J, Boundja P, Clark DB, Mugerwa B, Jansen PA, O’Brien TG, Rovero F, Sheil D, Vasquez R, Andelman S (2014) Carbon storage in tropical forests correlates with taxonomic diversity and functional dominance on a global scale. Glob Ecol Biogeogr 23:563–573. doi:10.1111/geb.12143.
Chave J, Coomes D, Jansen S, Lewis SL, Swenson NG, Zanne AE (2009) Towards a worldwide wood economics spectrum. Ecol Lett 12:351–366. doi:10.1111/j.1461-0248.2009.01285.x.
Condit R (1998) Tropical forest census plots: methods and results from Barro Colorado Island, Panama, and a comparison with other plots. Springer, New York
Conti G, Díaz S (2013) Plant functional diversity and carbon storage - an empirical test in semi-arid forest ecosystems. J Ecol 101:18–28. doi:10.1111/1365-2745.12012.
De Deyn GB, Cornelissen JHC, Bardgett RD (2008) Plant functional traits and soil carbon sequestration in contrasting biomes. Ecol Lett 11:516–531. doi:10.1111/j.1461-0248.2008.01164.x.
De’ath G (2007) Boosted trees for ecological modeling and prediction. Ecology 88:243–251. doi:10.1890/0012-9658.
Díaz S, Lavorel S, de Bello F, Quetier F, Grigulis K, Robson M (2007) Incorporating plant functional diversity effects in ecosystem service assessments. P Natl Acad Sci USA 104: 20684–20689. doi: 10.1073/pnas.0704716104.
Elith J, Leathwick JR, Hastie T (2008) A working guide to boosted regression trees. J Anim Ecol 77:802–813. doi:10.1111/j.1365-2656.2008.01390.x.
Fahey TJ, Woodbury PB, Battles JJ, Goodale CL, Hamburg SP, Ollinger SV, Woodall CW (2010) Forest carbon storage: ecology, management, and policy. Front Ecol Environ 8:245–252. doi:10.1890/080169.
FAO (Food and Agriculture Organization of the United Nations) (2010) Global Forest resources assessment 2010. Food and Agriculture Organization of the United Nations, Rome
Finegan B, Peña-Claros M, de Oliveira A, Ascarrunz N, Bret-Harte MS, Carreño-Rocabado G, Casanoves F, Díaz S, Eguiguren Velepucha P, Fernandez F, Licona JC, Lorenzo L, Salgado Negret B, Vaz M, Poorter L, Canham C (2015) Does functional trait diversity predict above-ground biomass and productivity of tropical forests? Testing three alternative hypotheses. J Ecol 103:191–201. doi:10.1111/1365-2745.12346.
Flynn DFB, Mirotchnick N, Jain M, Palmer MI, Naeem S (2011) Functional and phylogenetic diversity as predictors of biodiversity-ecosystem-function relationships. Ecology 92:1573–1581. doi:10.1890/10-1245.1
Fontaine S, Bardoux G, Abbadie L, Mariotti A (2004) Carbon input to soil may decrease soil carbon content. Ecol Lett 7:314–320. doi:10.1111/j.1461-0248.2004.00579.x.
Fornara DA, Tilman D (2008) Plant functional composition influences rates of soil carbon and nitrogen accumulation. J Ecol 96:314–322. doi:10.1111/j.1365-2745.2007.01345.x.
Freschet GT, Aerts R, Cornelissen JHC (2012) A plant economics spectrum of litter decomposability. Funct Ecol 26:56–65. doi:10.1111/j.1365-2435.2011.01913.x.
Garnier E, Cortez J, Billes G, Navas ML, Roumet C, Debussche M, Laurent G, Blanchard A, Aubry D, Bellmann A, Neill C, Toussaint JP (2004) Plant functional markers capture ecosystem properties during secondary succession. Ecology 85:2630–2637. doi:10.1890/03-0799.
Griffiths RP, Madritch MD, Swanson AK (2009) The effects of topography on forest soil characteristics in the Oregon Cascade Mountains (USA): Implications for the effects of climate change on soil properties. For Ecol Manag 257:1–7. doi:10.1016/j.foreco.2008.08.010.
Grigulis K, Lavorel S, Krainer U, Legay N, Baxendale C, Dumont M, Kastl E, Arnoldi C, Bardgett RD, Poly F, Pommier T, Schloter M, Tappeiner U, Bahn M, Clément J-C, Hutchings M (2013) Relative contributions of plant traits and soil microbial properties to mountain grassland ecosystem services. J Ecol 101:47–57. doi:10.1111/1365-2745.12014.
Grime JP (1998) Benefits of plant diversity to ecosystems: immediate, filter and founder effects. J Ecol 86:902–910. doi:10.1046/j.1365-2745.1998.00306.x.
Heemsbergen DA, Berg MP, Loreau M, van Haj JR, Faber JH, Verhoef HA (2004) Biodiversity effects on soil processes explained by interspecific functional dissimilarity. Science 306: 1019–1020. doi: 10.1126/science.1101865.
Hooper DU, Vitousek PM (1997) The effects of plant composition and diversity on ecosystem processes. Science 277: 1302–1305. doi: 10.1126/science.277.5330.1302.
Hooper DU, Chapin FS, Ewel JJ, Hector A, Inchausti P, Lavorel S, Lawton JH, Lodge DM, Loreau M, Naeem S, Schmid B, Setala H, Symstad AJ, Vandermeer J, Wardle DA (2005) Effects of biodiversity on ecosystem functioning: A consensus of current knowledge. Ecol Monogr 75:3–35. doi:10.1890/04-0922.
IPCC (Intergovernmental Panel on Climate Change) (2006) Agriculture, forestry and other land use. In: Eggleston S, Buendia L, Miwa K, Ngara T, Tanabe K (eds) IPCC Guidelines for National Greenhouse Gas Inventories. Institute for Global Environmental Strategies, Kanagawa
Isbell F, Calcagno V, Hector A, Connolly J, Harpole WS, Reich PB, Scherer-Lorenzen M, Schmid B, Tilman D, van Ruijven J, Weigelt A, Wilsey BJ, Zavaleta ES, Loreau M (2011) High plant diversity is needed to maintain ecosystem services. Nature 477:199–202. doi:10.1038/nature10282.
John R, Dalling JW, Harms KE, Yavitt JB, Stallard RF, Mirabello M, Hubbell SP, Valencia R, Navarrete H, Vallejo M, Foster RB (2007) Soil nutrients influence spatial distributions of tropical tree species. P Natl Acad Sci USA 104:864–869. doi:10.1073/pnas.0604666104.
Jonsson M, Wardle DA (2010) Structural equation modelling reveals plant-community drivers of carbon storage in boreal forest ecosystems. Biol Lett 6:116–119. doi:10.1098/rsbl.2009.0613.
Jucker T, Bouriaud O, Coomes DA, Baltzer J (2015) Crown plasticity enables trees to optimize canopy packing in mixed-species forests. Funct Ecol 29:1078–1086. doi:10.1111/1365-2435.12428.
Lange M, Eisenhauer N, Sierra CA, Bessler H, Engels C, Griffiths RI, Mellado-Vazquez PG, Malik AA, Roy J, Scheu S, Steinbeiss S, Thomson BC, Trumbore SE, Gleixner G (2015) Plant diversity increases soil microbial activity and soil carbon storage. Nat Commun 6:6707. doi:10.1038/ncomms7707.
Lavorel S, Grigulis K, Lamarque P, Colace MP, Garden D, Girel J, Pellet G, Douzet R (2011) Using plant functional traits to understand the landscape distribution of multiple ecosystem services. J Ecol 99:135–147. doi:10.1111/j.1365-2745.2010.01753.x.
Legendre P, Mi XC, Ren HB, Ma KP, MJ Y, Sun IF, He FL (2009) Partitioning beta diversity in a subtropical broad-leaved forest of China. Ecology 90:663–674. doi:10.1890/07-1880.1.
Lin DM, Lai JS, Muller-Landau HC, Mi XC, Ma KP (2012) Topographic variation in aboveground biomass in a subtropical evergreen broad-leaved forest in China. PLoS One 7:e48244. doi:10.1371/journal.pone.0048244.
Lin DM, Lai JS, Yang B, Song P, Li N, Ren HB, Ma KP (2015) Forest biomass recovery after different anthropogenic disturbances: relative importance of changes in stand structure and wood density. Eur J For Res 134:769–780. doi:10.1007/s10342-015-0888-9.
Liu XJ, Swenson NG, Wright SJ, Zhang LW, Song K, YJ D, Zhang JL, Mi XC, Ren HB, Ma KP (2012) Covariation in plant functional traits and soil fertility within two species-rich forests. PLoS One 7:e34767. doi:10.1371/journal.pone.0034767.
MEA (Millennium Ecosystem Assessment) (2005) Biodiversity: What is it, where is it, and why is it important? Ecosystems and Human Well-Being: Biodiversity Synthesis. World Resources Institute, Washington, DC
Mokany K, Ash J, Roxburgh S (2008) Functional identity is more important than diversity in influencing ecosystem processes in a temperate native grassland. J Ecol 96:884–893. doi:10.1111/j.1365-2745.2008.01395.x.
Paquette A, Messier C (2011) The effect of biodiversity on tree productivity: from temperate to boreal forests. Glob Ecol Biogeogr 20:170–180. doi:10.1111/j.1466-8238.2010.00592.x.
Petchey OL, Gaston KJ (2006) Functional diversity: back to basics and looking forward. Ecol Lett 9:741–758. doi:10.1111/j.1461-0248.2006.00924.x.
Pimm SL, Jenkins CN, Abell R, Brooks TM, Gittleman JL, Joppa LN, Raven PH, Roberts CM, Sexton JO (2014) The biodiversity of species and their rates of extinction, Distribution, and protection. Science 344: 987. doi: 10.1126/Science.1246752.
Pla LE, Casanoves F, Di Rienzo JA (2012) Quantifying functional biodiversity. Springer, Dordrecht
Prado-Junior JA, Schiavini I, Vale VS, Arantes CS, van der Sande MT, Lohbeck M, Poorter L (2016) Conservative species drive biomass productivity in tropical dry forests. J Ecol. doi:10.1111/1365-2745.12543.
R Development Core Team, 2011. R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, ISBN 3–900051–07–0
Ridgeway, G (2010) Gbm: Generalized Boosted Regression Models R Package Version 1.6–3.1.
Ruiz-Benito P, Gomez-Aparicio L, Paquette A, Messier C, Kattge J, Zavala MA (2014) Diversity increases carbon storage and tree productivity in Spanish forests. Glob Ecol Biogeogr 23:311–322. doi:10.1111/geb.12126.
Ruiz-Jaen MC, Potvin C (2011) Can we predict carbon stocks in tropical ecosystems from tree diversity? Comparing species and functional diversity in a plantation and a natural forest. New Phytol 189:978–987. doi:10.1111/j.1469-8137.2010.03501.x.
Sala OE, Chapin FS, Armesto JJ, Berlow E, Bloomfield J, Dirzo R, Huber-Sanwald E, Huenneke LF, Jackson RB, Kinzig A, Leemans R, Lodge DM, Mooney HA, Oesterheld M, Poff NL, Sykes MT, Walker BH, Walker M, Wall DH (2000) Biodiversity - Global biodiversity scenarios for the year 2100. Science 287:1770–1774. doi:10.1126/science.287.5459.1770.
Sayer EJ, Heard MS, Grant HK, Marthews TR, Tanner EVJ (2011) Soil carbon release enhanced by increased tropical forest litterfall. Nat Clim Chang 1:304–307. doi:10.1038/Nclimate1190.
Schumacher J, Roscher C (2009) Differential effects of functional traits on aboveground biomass in semi-natural grasslands. Oikos 118:1659–1668. doi:10.1111/j.1600-0706.2009.17711.x.
Srivastava DS, Cadotte MW, MacDonald AAM, Marushia RG, Mirotchnick N (2012) Phylogenetic diversity and the functioning of ecosystems. Ecol Lett 15:637–648. doi:10.1111/j.1461-0248.2012.01795.x.
Stegen JC, Swenson NG, Valencia R, Enquist BJ, Thompson J (2009) Above-ground forest biomass is not consistently related to wood density in tropical forests. Glob Ecol Biogeogr 18:617–625. doi:10.1111/j.1466-8238.2009.00471.x.
Tilman D, Isbell F, Cowles JM (2014) Biodiversity and ecosystem functioning. Annu Rev Ecol Evo S 45:471–493. doi:10.1146/annurev-ecolsys-120213-091917.
Villeger S, Mason NWH, Mouillot D (2008) New multidimensional functional diversity indices for a multifaceted framework in functional ecology. Ecology 89:2290–2301. doi:10.1890/07-1206.1.
Wright IJ, Reich PB, Westoby M, Ackerly DD, Baruch Z, Bongers F, Cavender-Bares J, Chapin T, Cornelissen JHC, Diemer M, Flexas J, Garnier E, Groom PK, Gulias J, Hikosaka K, Lamont BB, Lee T, Lee W, Lusk C, Midgley JJ, Navas ML, Niinemets U, Oleksyn J, Osada N, Poorter H, Poot P, Prior L, Pyankov VI, Roumet C, Thomas SC, Tjoelker MG, Veneklaas EJ, Villar R (2004) The worldwide leaf economics spectrum. Nature 428:821–827. doi:10.1038/Nature02403.
Wright SJ, Kitajima K, Kraft NJB, Reich PB, Wright IJ, Bunker DE, Condit R, Dalling JW, Davies SJ, Diaz S, Engelbrecht BMJ, Harms KE, Hubbell SP, Marks CO, Ruiz-Jaen MC, Salvador CM, Zanne AE (2010) Functional traits and the growth-mortality trade-off in tropical trees. Ecology 91:3664–3674. doi:10.1890/09-2335.1.
Zhang LW, Mi XC, Shao HB, Ma KP (2011) Strong plant-soil associations in a heterogeneous subtropical broad-leaved forest. Plant Soil 347:211–220. doi:10.1007/s11104-011-0839-2.
Zhu Y, Zhao GF, Zhang LW, Shen GC, Mi XC, Ren HB, Yu MJ, Chen JH, Chen SW, Fang T, Ma KP (2008) Community composition and structure of Gutianshan forest dynamics plot in a mid-subtropical evergreen broad-leaved forest, East China. Chinese J Plant Ecol 32: 262–273. doi: 10.3773/j.issn.1005-264x.2008.02.004. (in Chinese)
Acknowledgments
This work was financially supported by the National Natural Science Foundation of China (NO.31270496), the Fundamental Research Funds for the Central Universities (106112015CDJXY210012) and the 111 Project (B13041).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Responsible Editor: Per Ambus.
Electronic supplementary material
ESM 1
(DOCX 28 kb)
Rights and permissions
About this article
Cite this article
Lin, D., Anderson-Teixeira, K.J., Lai, J. et al. Traits of dominant tree species predict local scale variation in forest aboveground and topsoil carbon stocks. Plant Soil 409, 435–446 (2016). https://doi.org/10.1007/s11104-016-2976-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-016-2976-0