Abstract
Background and aims
Plant nutrient concentrations and their correlations with soil nutrient conditions are regarded as effective tools for exploring plant adaptation and resource utilisation strategies in a severe environment. However, few comparative studies have addressed the nutrient traits of different organs along natural fertility gradients.
Methods
We quantified the nitrogen (N) and phosphorus (P) concentrations and N:P ratios in roots and leaves of 139 plant samples from 14 species on a nutrient-limited alpine steppe on the Tibetan Plateau. Next, we explored the correlation between root and leaf nutrient traits of different plant functional groups as well as soil fertility.
Results
Soil P had a significant impact on plant N:P stoichiometry, whereas soil N had little effect on plant nutrient traits. Leaf P and the N:P ratio of legumes exhibited more sensitive responses to soil P than roots. Among non-legume species, however, root N and P concentrations of Stipa purpurea and Orinus thoroldii (grasses) were more sensitive than N and P concentrations of leaves to variations in soil P availability. In contrast, leaf P and the N:P ratio of Leontopodium nanum, Potentilla bifurca and ect. (forbs) exhibited more sensitive responses to soil P than roots. Both root and leaf nutrient traits of Carex moorcroftii (sedges) were uncorrelated with soil fertility. The N:P stoichiometry of different functional groups showed disparate responses to soil P, and even the roots and leaves of the same functional group exhibited inconsistent correlations with soil nutrients.
Conclusions
The distinct response patterns to soil nutrient conditions across functional groups helped elucidate the diversification of alpine plant adaptations to nutrient-poor environments and offered insights into quantifying the trade-off of different organs in co-existing species between resource use/conservation “strategies” and soil fertility.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Plant nutrient concentrations and their correlation with soil nutrient conditions are regarded as useful tools for exploring plant adaption and resource utilisation strategies in a severe environment (Ordoñez et al. 2009; He et al. 2010; Geng et al. 2011). Acquisitive species tend to have high resource acquisition, high tissue nutrient concentrations, specific leaf area, net photosynthetic rate and specific root length, whereas conservative species have high resource conservation, low tissue nutrient concentrations, high nutrient resorption efficiency and high stress tolerance (Grime 1974; Díaz et al. 2004; Sterck et al. 2006, 2011). Ordoñez et al. (2009) found that soil P had a great influence on leaf nitrogen and phosphorus concentrations, while soil N was only positively correlated with leaf nitrogen concentrations from a global dataset. However, no significantly relationship was observed between soil nitrogen and leaf nitrogen concentrations across Chinese grassland (He et al. 2010). Although correlations between plant functional traits and soil nutrients have been studied extensively (Chapin III 1980; Aerts and Chapin 1999; Ordoñez et al. 2009; Geng et al. 2011), studies in cold and arid areas are lacking.
Recent studies have assumed that dominant and wide-ranging species always have a stronger homeostasis or weak relationship in nutrient concentrations than non-dominant and narrow-ranging species in response to environmental factors (e.g., soil fertility) (Yu et al. 2010; Geng et al. 2011). However, these field studies have primarily focused on leaf nutrient traits. Until recently, relatively little effort has been dedicated to detecting root nutrient concentrations along natural fertility gradients on a large scale. Moreover, few attempts have been made to examine whether nutrients in different organs of the same plant would show a consistent response to soil fertility in a nutrient-limited environment.
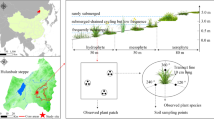
The Tibetan Plateau is considered the “third pole” of the world, which is characterised by low temperature, low precipitation, strong solar radiation, large amount of gale-force winds and extensive permafrost (Fig. 1). Plants on the Tibetan Plateau grow in an extreme environment in nutrient-deficient conditions and are expected to be sensitive to climate change, including global warming and changes in precipitation and N deposition (He et al. 2006; Jiang et al. 2012). Quantifying nutrient utilisation strategies of different species in this area is important for understanding plant adaptation to a harsh environment and future climate changes. Because of the cold and harsh conditions, the species diversity is very low in this alpine steppe. We conducted a systematic survey of the plant N:P stoichiometry of 139 plant samples from 14 species and seven families that covered the common species in the nutrient-limited alpine steppe of the northern Tibetan Plateau of China. We divided the 14 species into two functional groups, which included legumes and non-legumes (three sub-classes groups: grasses, sedges and forbs) (Mamolos et al. 2005; Song et al. 2012) (Table 1). Our objectives were (1) to quantify the root and leaf N and P concentrations and N:P ratios between legumes and non-legumes in the same community and (2) to clarify the correlation between the root and leaf N:P stoichiometry of the different functional groups (sub-classes groups) and soil total nitrogen (STN), soil available nitrogen (SAN), soil total phosphorus (STP) and soil available phosphorus (SAP) in the alpine tundra.
Materials and methods
Site descriptions
The study area was in a nutrient-limited alpine steppe, where Stipa purpurea and Carex moorcroftii were the most dominant and widely distributed species. Thirty-two sites were selected, extending from longitude 80.12 to 91.35 °E (approx. 1300 km long from east to west of the Tibetan Plateau) and latitude 31.23 to 32.31 °N, along with altitudes ranging from 4398 to 5561 m (Fig. 1). The mean annual temperature (MAT) and mean annual precipitation (MAP) ranged from −5.93 to 0.29 °C and 181 to 359 mm, respectively (data obtained from the World Climate web site, www.worldclimate.com).
Plant and soil sampling
Field measurements were conducted in August 2012. To minimise the influences of microtopography and grazing disturbances, all samples were selected on flat terrain and far from human habitats. The dominant species were selected and sampled at each of the 32 sites. However, in some species-poor sites, fewer species (as few as one) were collected (Appendix Table 2). In each site, a 10 * 10 m quadrat was set. The soil core (15 cm in length, 15 cm in width, and 30 cm in depth), which contained a dominant species, was gathered by a spade in the quadrat. To maintain the integrity of the plant, we only collected a dominant individual plant from one soil core. Ten to twenty individuals of each species in each quadrat were collected. Then, the root and leaf samples from the same species were carefully cleaned and sundried in a paper envelope. In all, we investigated 139 plant samples for 14 species over a total of 32 sites (Table 1; Appendix Table 2).
Three soil samples (0–15 cm in depth) were collected from each site. Each sample was thoroughly mixed and air-dried. Roots in the soil were removed by hand and sieved through a 100-mesh sieve. Then, the soil was divided into four subsamples for analysing STN, SAN, STP and SAP.
Sample analysis
All plant samples were divided into roots and leaves (139 * root and 139 * leaf samples), oven-dried at 60 °C to a constant mass and ground into a fine powder in the laboratory. Soil and plant total N concentrations were analysed using the micro-Kjeldahl digestion method (Coombs et al. 1985). Plant P concentrations were analysed using the ammonium molybdate method (Institute of Soil Academia Sinica 1978). Soil total P concentrations were determined using the sodium bicarbonate alkali digestion method and molybdenum antimony colorimetry (Kuo 1996). The soil available N concentration was analysed using the alkaline hydrolysis diffusion method (Institute of Soil Academia Sinica 1978). The available soil P concentration was extracted by sodium bicarbonate and determined using the molybdenum blue method (Institute of Soil Academia Sinica 1978). The nutrient concentrations in plant and soil samples were expressed on a mass basis. We used the mean value of three samples from the same site for the STN, SAN, STP and SAP concentrations of each site.
Statistical analyses
Differences in the root and leaf N and P concentrations and N:P ratios between legumes and non-legumes species was processed using Mann–Whitney U-test. The correlation between plant nutrient traits (N and P concentrations and N:P ratios) and soil fertility (STN, SAN, STP and SAP) was analysed using simple regression analyses. Plant nutrient traits (N, P and N:P ratios) and soil fertility (STN, SAN, STP and SAP) were log-transformed to normalise the statistical distribution where necessary. All statistical analyses were performed using the SPSS version 16.0 software (SPSS Inc., Chicago, IL, USA), and cartograms were plotted using the SigmaPlot 11.0 software (Systat Software, Inc., Richmond, USA).
Results
Patterns of the N and P concentrations and N:P ratios in root and leaf between different plant functional groups
Leaf nitrogen and phosphorus concentrations were nearly double that of root both in legumes and non-legume species (Fig. 2). Mean value of root N and P concentrations of legumes were 19.86 mg g−1 and 0.71 mg g−1. Non-legumes had lower root N concentrations than legumes (mean value for root N: 11.03 mg g−1) (P < 0.05) (Fig. 2). Root N:P of legumes was higher than non-legumes (P < 0.05). The mean value were 33.31 mg g−1, 1.80 mg g−1 and 19.76 for leaf N and P concentrations and N:P ratios of legumes, respectively (Table 1). Legumes had higher leaf N and P concentrations than non-legumes (P < 0.05) (Fig. 2).
N and P concentrations and N:P ratios in the roots and leaves of 14 species and different functional groups on the alpine steppe of the Tibetan Plateau. Legumes included Oxytropis glacialis, Oxytropis microphylla and Oxytropis serioopetala. Non-legumes species included Stipa purpurea, Orinus thoroldii, Carex moorcroftii, Leontopodium nanum, Potentilla bifurca, Youngia simulatrix, Incarvillea compacta, Lagotis brachystachya, Artemisia wellbyi, Artemisia younghusbandii and Saussurea stoliczkai. *, ** indicated significant differences in root (R) and leaf (L) between legumes and non-legumes species at 0.1 and 0.05. NS indicated no significant differences between legumes and non-legumes species
The correlations between the N and P concentrations of different plant functional groups and soil fertility
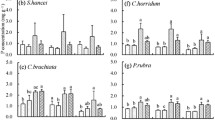
For legumes and non-legumes species, both root and leaf N and P concentrations, as well as the N:P ratios, were uncorrelated with STN and SAN (Figs. 3 and 4, P > 0.05). Positive linear correlations were detected between the leaf P concentrations of legumes and SAP (Fig. 4h, P < 0.05), whereas the N:P ratios were negatively associated with SAP (Fig. 4d, P < 0.05). Weakly positive correlation was detected between the leaf N:P concentrations of non-legumes and SAP (Fig. 4d, P < 0.05). Among non-legume species, however, the root N and P concentrations of Stipa purpurea and Orinus thoroldii (grasses) were positively correlated with STP and SAP (Fig. 3g, h, k, l; P < 0.01 for root N with STP; P < 0.05 for root N with SAP; P < 0.001 for root P with STP; P < 0.01 for root P with SAP). The leaf P concentrations of Leontopodium nanum, Potentilla bifurca and ect. (forbs) were positively correlated with STP and SAP (Fig. 4g, h, P < 0.01 for leaf P with STP; P < 0.001 for leaf P with SAP). The leaf N:P of forbs was negatively associated with STP and SAP (Fig. 4c, d, P < 0.01 for leaf N:P with STP; P < 0.001 for leaf N:P with SAP). All of the nutrient traits of Carex moorcroftii (sedges) were uncorrelated with soil fertility (P > 0.05) (Figs. 3 and 4).
Correlations between nutrient traits (N, P and N:P ratios) of the root and soil nutrients (STN, SAN, STP and SAP) across the different functional groups. Legumes included Oxytropis glacialis (O.g.), Oxytropis microphylla (O.m.) and Oxytropis serioopetala (O.s.). Non-legumes species included three sub-classes groups. Stipa purpurea (S.p.) and Orinus thoroldii (O.t.) belonged to grassses; Carex moorcroftii (C.m.) belonged to sedges; Leontopodium nanum (L.n.), Potentilla bifurca (P.b.), Youngia simulatrix (Y.s.), Incarvillea compacta (I.c.), Lagotis brachystachya (L. b.), Artemisia wellbyi (A.w.), Artemisia younghusbandii (A. y.) and Saussurea stoliczkai (S.s.) belonged to forbs
Correlations between the nutrient traits (N, P and N:P ratios) of leaf and soil nutrients (STN, SAN, STP and SAP) across the different functional groups. Legumes included Oxytropis glacialis (O.g.), Oxytropis microphylla (O.m.) and Oxytropis serioopetala (O.s.). Non-legumes species included three sub-classes groups. Stipa purpurea (S.p.) and Orinus thoroldii (O.t.) belonged to grassses; Carex moorcroftii (C.m.) belonged to sedges; Leontopodium nanum (L.n.), Potentilla bifurca (P.b.), Youngia simulatrix (Y.s.), Incarvillea compacta (I.c.), Lagotis brachystachya (L. b.), Artemisia wellbyi (A.w.), Artemisia younghusbandii (A. y.) and Saussurea stoliczkai (S.s.) belonged to forbs
Discussion
Patterns of the N and P concentrations and N:P ratios in roots and leaves between different functional groups
Legumes have abundant nitrogen-fixing bacteria, which help the plant to use atmospheric-N2 for growth and enable the plants to adapt to barren soil (Vitousek et al. 2002). Thus, leguminous plants always have higher N concentrations than other functional groups. In the present study, legumes also have higher leaf P concentrations; this result was inconsistent with a previous study that demonstrated that there was no significant difference in plant P concentrations between legumes and non-legumes across Chinese grasslands (He et al. 2008). This result was most likely due to the ability of leguminous species to trigger some special physiological mechanisms related to improving their absorptive capacity of P in poor quality soil on alpine steppes to prevent nutritional imbalances (Yadav and Tarafdar 2001; Houlton et al. 2008). Olde Venterink (2011) found that legumes had higher root phosphomonoesterase activity (improving absorptive capacity for P) than non-legumes. The result could help explain why the leguminosae species showed relatively high P concentrations in our study.
The root N and P concentrations were positively correlated with the leaf N and P concentrations across all species (Hong et al. 2014). The consistency of the root-leaf N and P relationship was consistent with a previous meta-analysis (Kerkhoff et al. 2006) and indicated that this general nutrient allocation rule also applies to alpine plants (Geng et al. 2014). In the present study, we took insufficient account of the root length, mycorrhizal hyphal length and root longevity, which are closely linked with the plant N:P stoichiometry (Olde Venterink and Güsewell 2010). These important morphological and physiological ecological characteristics will be considered in our future research.
Influence of soil nutrients on the N and P concentration and N:P ratios among different functional groups
Soil N concentrations have almost no significant effects on plant nutrient traits, whereas soil P concentrations have varying degrees of impact on the plant N and P concentrations and N:P ratios across all functional groups. This stronger influence of soil P on plant nutrients was consistent with previous studies (Elser et al. 2000; Ordoñez et al. 2009; Fujita et al. 2010; Chen et al. 2011). Plants, especially legumes, not only make use of soil available N but also N in the atmosphere with the help of multiple nitrogen-fixing bacteria (Fitter et al. 1998; Aerts and Chapin 1999). In contrast, the soil P for absorbed by plants was primarily derived from rock weathering, and its diffusivity was significantly lower. Therefore, the soil available P supply was strongly influenced by climate variation, and it consequently affects plant P concentrations (Chen et al. 2011). With multiple-channels for obtaining N, plants obtain more stable N than P.
Interestingly, the N:P stoichiometry of different plant functional groups (sub-classes groups) showed disparate responses to soil P, and even organs (root and leaf) of the same functional group appeared to exhibit inconsistent correlations with soil nutrient conditions. Although the N:P stoichiometry represents part of a multivariate aspect of a phenotype that is influenced by complicated factors (biotic and abiotic factors), this difference could provide valuable means and implicit information for linking the perspective of ecology and evolution on plant organs and environmental factors (Elser et al. 2000; Kay et al. 2005). In addition, this pattern might reflect different phenological times between leaf and root growth; this pattern may also reflect a trade-off between resource allocation and investment, which is related to ecological success in a nutrient-limited steppe (Yu et al. 2011). Because roots belong to the “structural” component and leaves belong to the “metabolic” component, the nutrient concentrations of the two organs may be affected by different regulatory mechanisms. For instance, the plant size within species had a marked impact on the nutrient concentration of roots with little or no effect observed among leaves (Kerkhoff et al. 2006).
The leaf P and N:P ratios of legumes displayed more sensitive responses to SAP. The reduced leaf phenotypic variation of legumes may be an important factor, which increases the susceptibility to external environmental variability (Walck et al. 1999; Lloyd et al. 2002). Because of the weak leaf internal self-regulation of multifactorial physiological traits of narrow-ranging, distributed and accompanying species, these plants were more sensitive to environmental variables, particularly soil fertility (Futuyma and Moreno 1988; Sultan et al. 1998; Geng et al. 2011).
Among non-legume species, the root N and P concentrations of Stipa purpurea and Orinus thoroldii (grasses) were positively correlated with soil P in the present study. The increase in the root N concentration with increasing soil P may be because an improvement in the P supply by a different pathway of N-fixation would promote plant N uptake (Ekblad and Huss-Danell 1995; Iversen et al. 2010). Under low temperatures, such as alpine or arctic ecosystems, inorganic P is always released during frozen ground thaws; however, the intensity of the freeze-thaw varies its interannual variability (Øien 2009). In severe and long-lasting frost years, more soil P is released and absorbed by grasses during the growing season (Richardson and Marshall 1986; Moen 1990), and then the plant allocates the extra P to roots (storage strategy) (Øien 2009). The extra nutrients that were stored in roots would reduce the dependence of grasses on soil fertility availability particularly when grasses require a sizeable demand of nutrients for growth (e.g., in early spring) (Lambers et al. 1998). Stipa purpurea and Orinus thoroldii (grasses) have adopted a storage strategy belowground for potentially limited nutrients in infertile environments, which is consistent with other plants that dominate infertile soils (Chapin III 1980). Intense responses of sedges to soil N were observed in alpine grasslands in the Great Pyrenees and Teberda (Sebastià 2006; Onipchenko et al. 2012). However, root and leaf nutrients of Carex moorcroftii had no relationship with soil N in our study. Compared with other study areas and plants, our study was conducted on a dry grassland, in which the roots of Carex moorcroftii were in the upper soil layers; therefore, extensively low surface soil moisture may constrain nitrogen uptake for the sedges. The leaf P and N:P ratios of Leontopodium nanum, Potentilla bifurca and ect. (forbs) displayed more sensitive responses to STP and SAP. The result may derive from the weak leaf internal self-regulation of multifactorial physiological traits of forbs which is the same with other narrow-ranging and accompanying species (e.g. legumes) (Futuyma and Moreno 1988; Sultan et al. 1998; Geng et al. 2011).
Conclusion
The present study is a large-scale field comparative study to explicitly dissect the correlation between nutrient traits in different organs of the same plant group and soil fertility in an alpine tundra. Our results highlight that soil P has a significant impact on the plant N and P concentrations and N:P ratios, whereas soil N has relatively little effect on plant N:P stoichiometry along natural fertility gradients. The differences in the nutrient traits and their correlations with soil fertility among the different functional groups indicate that coexisting species have different nutrient use/conservation strategies in one community. Further studies will focus on combining more global data between root and leaf nutrient concentrations with soil fertility.
References
Aerts R, Chapin FS (1999) The mineral nutrition of wild plants revisited: a re-evaluation of processes and patterns. Adv Ecol Res 30:1–67
Chapin FS III (1980) The mineral nutrition of wild plants. Annu Rev Ecol Syst 11:233–260
Chen Y, Han W, Tang L, Tang Z, Fang J (2011) Leaf nitrogen and phosphorus concentrations of woody plants differ in responses to climate, soil and plant growth form. Ecography 34:1–7
Coombs J, Hind G, Leegood RC, Tieszen LL, Vonshak A (1985) Analytical techniques. In: Coombs J, Hall DO, Long SP, Scurlock JM (eds) Techniques in Bioproductivity and Photosynthesis. Pergamon Press, Oxford, p 298
Díaz S, Hodgson J, Thompson K, Cabido M, Cornelissen J, Jalili A, Montserrat-Martí G, Grime J, Zarrinkamar F, Asri Y (2004) The plant traits that drive ecosystems: evidence from three continents. J Veg Sci 15:295–304
Ekblad A, Huss-Danell K (1995) Nitrogen fixation by Alnus incana and nitrogen transfer from A. incana to Pinus sylvestris influenced by macronutrients and ectomycorrhiza. New Phytol 131:453–459
Elser J, Sterner R, Gorokhova E, Fagan W, Markow T, Cotner J, Harrison J, Hobbie S, Odell G, Weider L (2000) Biological stoichiometry from genes to ecosystems. Ecol Lett 3:540–550
Fitter A, Wright W, Williamson L, Belshaw M, Fairclough J, Meharg A (1998) The phosphorus nutrition of wild plants and the paradox of arsenate tolerance: does leaf phosphate concentration control flowering? Curr Top Plant Physiol 19:39–51
Fujita Y, de Ruiter PC, Wassen MJ, Heil GW (2010) Time-dependent, species-specific effects of N: P stoichiometry on grassland plant growth. Plant Soil 334:99–112
Futuyma DJ, Moreno G (1988) The evolution of ecological specialization. Annu Rev Ecol Syst 19:207–233
Geng Y, Wang Z, Liang C, Fang J, Baumann F, Kühn P, Scholten T, He J (2011) Effect of geographical range size on plant functional traits and the relationships between plant, soil and climate in Chinese grasslands. Global Ecol Biogeogr 21:416–427
Geng Y, Wang L, Jin D, Liu H, He J (2014) Alpine climate alters the relationships between leaf and root morphological traits but not chemical traits. Oecologia 175:445–455
Grime J (1974) Vegetation classification by reference to strategies. Nature 250:26–31
He J, Wang Z, Wang X, Schmid B, Zuo W, Zhou M, Zheng C, Wang M, Fang J (2006) A test of the generality of leaf trait relationships on the Tibetan Plateau. New Phytol 170:835–848
He J, Wang L, Flynn DFB, Wang X, Ma W, Fang J (2008) Leaf nitrogen: phosphorus stoichiometry across Chinese grassland biomes. Oecologia 155:301–310
He J, Wang X, Schmid B, Flynn DF, Li X, Reich PB, Fang J (2010) Taxonomic identity, phylogeny, climate and soil fertility as drivers of leaf traits across Chinese grassland biomes. J Plant Res 123:551–561
Hong J, Wang X, Wu J (2014) Stoichiometry of root and leaf nitrogen and phosphorus in a dry alpine steppe on the northern tibetan plateau. PLoS One 9:e109052
Houlton BZ, Wang YP, Vitousek PM, Field CB (2008) A unifying framework for dinitrogen fixation in the terrestrial biosphere. Nature 454:327–330
Institute of soil academia sinica (1978) Analysis of soil physics and chemistry. Science and Technology of Shanghai Publications, Shanghai (In Chinese)
Iversen CM, Bridgham SD, Kellogg LE (2010) Scaling plant nitrogen use and uptake efficiencies in response to nutrient addition in peatlands. Ecology 91:693–707
Jiang C, Yu G, Li Y, Cao G, Yang Z, Sheng W, Yu W (2012) Nutrient resorption of coexistence species in alpine meadow of the Qinghai-Tibetan Plateau explains plant adaptation to nutrient-poor environment. Ecol Eng 44:1–9
Kay AD, Ashton IW, Gorokhova E, Kerkhoff AJ, Liess A, Litchman E (2005) Toward a stoichiometric framework for evolutionary biology. Oikos 109:6–17
Kerkhoff AJ, Fagan WF, Elser JJ, Enquist BJ (2006) Phylogenetic and growth form variation in the scaling of nitrogen and phosphorus in the seed plants. Am Nat 168:E103–E122
Kuo S (1996) Phosphorus. In: Sparks DL, Page AL, Loeppert PA, Soltanpour PN, Tabatabai MA, Johnston CT et al (eds) Methods of soil analysis part 3: chemical methods. Soil Science Society of America and American Society of Agronomy, Madison, pp 869–920
Lambers H, Chapin FS, Pons TL (1998) Plant physiological ecology. Springer, New York
Lloyd KM, Lee WG, Wilson JB (2002) Competitive abilities of rare and common plants: comparisons using Acaena (Rosaceae) and Chionochloa (Poaceae) from New Zealand. Conserv Biol 16:975–985
Mamolos A, Vasilikos C, Veresoglou D (2005) Vegetation in contrasting soil water sites of upland herbaceous grasslands and N: P ratios as indicators of nutrient limitation. Plant Soil 270:355–369
Moen A (1990) The plant cover of the boreal uplands of Central Norway. I. Vegetation ecology of Sølendet nature reserve; haymaking fens and birch woodlands. Universitetet i Trondheim, Vitenskapsmuseet, Trondheim, NO
Øien DI (2009) Nutrient limitation in boreal rich-fen vegetation: a fertilization experiment. Appl Veg Sci 7:119–132
Olde Venterink H (2011) Legumes have a higher root phosphatase activity than other forbs, particularly under low inorganic P and N supply. Plant Soil 347:137–146
Olde Venterink H, Güsewell S (2010) Competitive interactions between two meadow grasses under nitrogen and phosphorus limitation. Funct Ecol 24:877–886
Onipchenko V, Makarov M, Akhmetzhanova A, Soudzilovskaia N, Aibazova F, Elkanova M, Stogova A, Cornelissen JHC (2012) Alpine plant functional group responses to fertiliser addition depend on abiotic regime and community composition. Plant Soil 357:101–113
Ordoñez JC, Van Bodegom PM, Witte JPM, Wright IJ, Reich PB, Aerts R (2009) A global study of relationships between leaf traits, climate and soil measures of nutrient fertility. Global Ecol Biogeogr 18:137–149
Richardson CJ, Marshall PE (1986) Processes controlling movement, storage, and export of phosphorus in a fen peatland. Ecol Monogr 56:279–302
Sebastià MT (2006) Plant guilds drive biomass response to global warming and water availability in subalpine grassland. J Appl Ecol 44:158–167
Song M, Yu F, Ouyang H, Cao G, Xu X, Cornelissen JHC (2012) Different inter-annual responses to availability and form of nitrogen explain species coexistence in an alpine meadow community after release from grazing. Global Change Biol 18:3100–3111
Sterck F, Poorter L, Schieving F (2006) Leaf traits determine the growth-survival trade-off across rain forest tree species. Am Nat 167:758–765
Sterck F, Markesteijn L, Schieving F, Poorter L (2011) Functional traits determine trade-offs and niches in a tropical forest community. PNAS 108:20627–20632
Sultan S, Wilczek A, Bell D, Hand G (1998) Physiological response to complex environments in annual Polygonum species of contrasting ecological breadth. Oecologia 115:564–578
Vitousek PM, Cassman K, Cleveland C, Crews T, Field CB, Grimm NB, Howarth RW, Marino R, Martinelli L, Rastetter EB (2002) Towards an ecological understanding of biological nitrogen fixation. Biogeochemistry 57:1–45
Walck JL, Baskin JM, Baskin CC (1999) Relative competitive abilities and growth characteristics of a narrowly endemic and a geographically widespread Solidago species (Asteraceae). Am J Bot 86:820–828
Yadav R, Tarafdar J (2001) Influence of organic and inorganic phosphorus supply on the maximum secretion of acid phosphatase by plants. Biol Fert Soils 34:140–143
Yu Q, Chen Q, Elser JJ, He N, Wu H, Zhang G, Wu J, Bai Y, Han X (2010) Linking stoichiometric homoeostasis with ecosystem structure, functioning and stability. Ecol Lett 13:1390–1399
Yu Q, Elser JJ, He N, Wu H, Chen Q, Zhang G, Han X (2011) Stoichiometric homeostasis of vascular plants in the inner Mongolia grassland. Oecologia 166:1–10
Acknowledgments
This study was supported by the Strategic Pilot Science and Technology Projects of the Chinese Academy of Sciences (Grant No. XDB03030505), the Western Action Plan Project of the Chinese Academy of Sciences (Grant No. KZCX2-XB3-08), and the Public Benefit Research Foundation of the Ministry of Environmental Protection (Grant No.201209032). We gratefully acknowledge Yanjiang Cai, Xingxing Ma and Xuyang Lu for their field assistance.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Harry Olde Venterink .
Appendix
Appendix
Rights and permissions
About this article
Cite this article
Hong, J., Wang, X. & Wu, J. Effects of soil fertility on the N:P stoichiometry of herbaceous plants on a nutrient-limited alpine steppe on the northern Tibetan Plateau. Plant Soil 391, 179–194 (2015). https://doi.org/10.1007/s11104-015-2416-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-015-2416-6