Abstract
Nutrition issues and the increase in food allergy diseases experienced in recent years have encouraged part of the worldwide population to seek foods that, in addition to nourishing, can benefit their health, such as foods abundant in bioactive compounds. In this context, our objective was to conduct a review to emphasize the richness of the biodiversity of the Brazilian flora concerning the presence of bioactive compounds, their pharmacological applications, and their extraction characteristics. This narrative review screened the databases and identified 120 research articles published on the topic. These articles were analyzed to examine associated parameters such as the availability and applications of these bioactive compounds. These compounds exhibit therapeutic properties, such as antioxidant, antitumor, and anti-inflammatory effects, with multiple benefits to health and treating illnesses. In addition, the methods for extracting bioactive compounds must be constantly improved to increase their purity and yield because these metabolites are present in complex media. Addressing these issues is essential for improving the overall experience of health-conscious individuals.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The Brazilian flora is highly diversified. Approximately 52,598 species are cataloged, including several plants, fungi, and algae (Flora e Funga do Brasil, 2024). The wide variety of species supports many studies targeting the Brazilian flora, emphasizing bioactive compounds with high biological potential and extensive use (Silva et al. 2020). Bioactive compounds from Brazilian plants and fruits with antioxidant, antitumor, and anti-inflammatory properties are therapeutically and economically relevant. Understanding the mechanisms of action of these compounds and their interactions with the human body is relevant due to their multiple health benefits (Barbosa-Pereira et al. 2014).
Healthy eating is gaining the attention of the public and business sectors and supports research to meet new food trends (Cadario and Chandon 2019). The scarcity of nutrients throughout life is a global public health issue. Therefore, the current supplementation alternatives must be evaluated and examined individually (Maggini et al. 2018). Few studies have conclusively demonstrated the application or incorporation of bioactive compounds into diets and their use as medicines. Although bioactive compounds have been widely researched, the complexity of cell environments can interfere with their activities due to synergistic interactions and consequent stabilization (Giaconia et al. 2020).
Improving bioactive compound extraction methods is a recurrent research approach in which new techniques that enhance the production yield have been identified (Sosa-Hernández et al. 2018). In addition, developing new separation techniques associated with strategic sets of purification processes favors the expansion of knowledge about the mechanisms for obtaining bioactive compounds (Ramirez-Estrada et al. 2016).
This narrative review presents the potential of bioactive compounds (flavonoids and carotenoids) from Brazilian flora species for therapeutic use and their extraction mechanisms. Such a subject favors the knowledge of new compounds to formulate foods and drugs and contributes to the discussion on preserving Brazilian flora biodiversity. Therefore, the main objective of this narrative review is to explore the dietary and pharmaceutical applications and extraction methods of flavonoids and carotenoids found in the Brazilian flora. The aim is for the reader to feel invited to navigate this world of possibilities. It is a topic with great possibilities; therefore, focusing only on a plant, disease, or biome fails to present these possibilities.
Methods
A search was conducted for full-text articles to support the development of a narrative review. Thus, the selected articles were written in English, and most of them were published in recent years. These articles used specific keyword combinations of Brazilian flora, bioactive compounds, health, extraction mechanisms, Brazil, flavonoids, and carotenoids in the well-known international databases Scopus, National Center for Biotechnology Information (NCBI)/PubMed, PubMed Central (PMC), Elsevier, Web of Science, and Google Scholar. Other inclusion criteria were information on the extraction and use of flavonoids and carotenoids from fruits of Brazilian biomes. The selected articles were thoroughly reviewed, and relevant data were extracted for analysis.
Brazilian flora biodiversity
The scientific community has not agreed on the planet's biodiversity, but the latest estimate predicts approximately 8.7 million eukaryotic species, 14.22% of which are cataloged worldwide. Among these, 314,600 species are plants, and 71.28% have already been cataloged (Mora et al. 2011). However, only approximately 20% of these flora species have been evaluated pharmacologically, and only 6% are associated with patents (Simmonds et al. 2020), exploiting the existence of vast unexplored flora species. In this scenario, Brazil is a hotspot with a high concentration of endemic plant and fungal species (Antonelli et al. 2020).
Brazil is the country with the greatest plant genetic biodiversity on the planet (MMA, 2017; CBD 2020), with 54,278 species of flora (Flora do Brasil, 2024) and 116,839 species of fauna (MMA, 2020 cited by Wosnick et al., 2021). The country is estimated to be home to approximately 20% of the world's biodiversity and is the territory with the greatest number of plant species. Over 50% of cases are endemic (MMA, 2017); they occur only in Brazil. The country has continental proportions, i.e., 8.5 million km2, covering different climatic regions (Fig. 1). These climate zones generated six different biomes: the Amazon, Cerrado, Caatinga, Atlantic Forest, Pampa, and Pantanal (Table 1). Each biome is a biotic unit with a dominant vegetation type, characteristic living organisms, and peculiarities due to its biological diversity. It should be emphasized that 20% of the world's freshwater flows into Brazilian territory (Coutinho 2006; IBGE, 2019).
The Amazon is the largest biome in Brazil, covering an area of approximately 5 million km2, which represents 5% of the Earth's surface (IBGE, 2019). This biome contains approximately 30,000 plant species (Wittmann 2011), approximately 10% of known plant species.
The second-largest biome in Brazil is the Cerrado (MMA, 2020). This biome contains approximately 4000 fruit varieties that are attractive to wildlife and 5% of the planet's biodiversity (MMA, 2020; Kuhlmann and Ribeiro 2016). It is the world's largest and most biodiverse savanna and has the richest vascular plant flora (Colli et al. 2020). Approximately 35% of the trees and 70% of the shrubs and herbaceous plants found in the Cerrado are endemic (Fiaschi and Pirani 2009). Its flora is similar to that of the Amazon biome flora.
The Pantanal biome has a dry winter and a hot and rainy summer, which are responsible for its flooding. These floods can last up to eight months in the lower regions (Embrapa 2020). The climatic instability of the Pantanal causes an annual biogeochemical cycle that feeds great biological diversity (Alho et al. 2019). Hence, its flora range from hydrophilic (aquatic) to xerophilic (adapted to soils with low humidity) plants. The Pantanal biome is one of the most extensive flooded plains on the planet (MMA, 2020).
The Caatinga biome is restricted to the Brazilian territory (IBGE, 2019). Its vegetation is adapted to the hot and dry climate, which varies little throughout the year. This biome is the most biodiverse semiarid region worldwide, with an endemism rate above 50%. It is considered the foremost biological area of seasonally dry tropical forests and is classified by UNESCO as a significant reserve (Apgaua et al. 2014).
The Pampa biome has a rainy climate without a dry period, with an average temperature between 13 °C and 17 °C. However, it can reach negative temperatures in winter (IBGE, 2024). This biome is home to forests and cacti (70 types), but its primary landscape is formed by extensive grassy fields (Embrapa 2020). In just one area of 1 m2 of this field, 56 different plant species have already been found, a national record (Menezes et al. 2018).
The Atlantic Forest biome currently comprises more than 20% of the original area (IBGE, 2024). Nonetheless, despite being reduced and fragmented, this biome still sustains one of the highest quantities of species per square meter of the planet. Its flora includes almost 18,000 species with an endemism of 59%. Studies have identified more than 450 tree species in one hectare of forest, another record (Flora e Funga do Brasil, 2024).
The coastal marine system of Brazil is a set of multiple and varied environments, each with specific characteristics that justify being treated as a single system despite not being recognized as biomes (IBGE, 2019). The Brazilian coastal marine system occupies nearly 4.5 million km2 and includes coral reefs, dunes, mangroves, lagoons, estuaries, and swamps (ICMBIO 2020). Thus, this system represents a vast area with high biodiversity and suggests high endemism, even though little has been studied (Couto et al. 2003).
Since 2008, Brazil has cataloged the greatest number of new plant species, corresponding to approximately 200 species per year, or 10% of the global total (Antonelli et al. 2020). This occurrence demonstrates the biodiversity of the Brazilian flora and reinforces the need to increase taxonomic and pharmacological research to maximize the profit from the multiplicity of tropical flora species. Furthermore, there is an urgent need to preserve endangered species of Brazilian flora.
Bioactive compounds from Brazilian flora
The use of native plants to develop drugs is based on aspects such as biodiversity, acceptability, and economic markets. Several studies of disease spread suggest that a high intake of specific plant products can reduce some chronic diseases because of bioactive compounds. These metabolites stimulate the immune system, reduce platelet aggregation, and exert antioxidant, antibacterial, and antiviral effects. The main vegetable bioactive compounds are pigments, such as carotenoids, and polyphenols, such as flavonoids.
Carotenoids
Carotenoids are fat-soluble pigments, yellow, orange, and red, present in numerous fruits and vegetables of the Brazilian flora. The structural representations of the most frequent plant carotenoids are shown in Fig. 2. These compounds include β-carotene, bixin, capsanthin, lycopene, lutein, and zeaxanthin. The chemical structures were obtained from PubChem (2024) at the National Institutes of Health (NIH).
Chemical structure of the most common plant carotenoids: a β-carotene; b bixin; c capsanthin; d lycopene; e lutein; and f zeaxanthin (Pubchem. (2024)
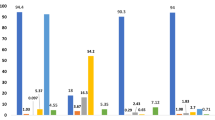
β-Carotene is considered the most abundant carotenoid in food. It has provitamin A activity and can be converted into retinol because of a β-ionone ring in its structure. Anunciação et al. (2019) and De Souza et al. (2020) detected β-carotene in native Amazonian fruits, such as marirana (Couepia subcordata; 6,331 ± 410 μg/100 g), inajá (Maximiliana maripa; 1,371 ± 370 μg/100 g), caranan (Mauritiella armata; 373 ± 80 μg/100 g) (Fig. 3), moriche palm (Mauritia flexuosa; 21.6 mg/100 g), and macauba palm (Acrocomia aculeata; 56.0 mg/100 g). Cardoso et al. (2011) showed that β-carotene is the main carotenoid in Cagaita (Eugenia dysenterica) fruit (50.8%). The consumption of fruits rich in β-carotene is associated with a lower incidence of cancer, cardiovascular diseases, age-related macular degeneration, and less cataract formation (Meyers et al. 2014; Sharoni et al. 2012).

Source: Anunciação et al. (2019)
(A) Marirana (Couepia subcordata Benth.), (B) inajá (Maximiliana maripa), and (C) caranan (Mauritiella armata) of the Brazilian Amazon biome.
Lutein and zeaxanthin are produced from the hydroxylation of α-carotene and β-carotene, respectively. They are present in various food sources, such as dark green leafy vegetables, spinach, kale, and corn. These pigments have provitamin A activity and antioxidant, anti-inflammatory, photoprotective, and anticarcinogenic properties, improving skin elasticity (Woodside et al. 2015). These macular pigments in the human retina are responsible for two functions: protecting the macula against oxidative stress and filtering high-energy blue light, improving visual acuity (Santocono et al. 2007; Traversa et al. 2012).
Lutein is a carotenoid present in camu-camu (Myrciaria dubia), a native fruit of the Amazon region, and comprises 45% to 55% of the total carotenoids (Zanatta and Mercadante 2007); leaves of Pariri (Arrabidaea chica), a medicinal plant found in the Amazon, at 204.28 μg/g (dry mass) (Siqueira et al. 2019); spinach, kale, arugula, sorrel, chard, watercress, mustard, and broccoli; and some nonconventional vegetables consumed in Brazil, such as Lobrobo or equally Ora-pro-nobis (Pereskia aculeata) (78.45%), Serralha (Sonchus oleraceus) (58.30%), Almeirão (Cichorium intybus subsp. intybus) (88.77%) and Taioba (Xanthosoma sagittifolium) (61.05%) (Nachtigall et al. 2007).
Lycopene is a β-carotene isomer with an acyclic structure without provitamin A activity. It is a natural fat-soluble pigment from some red or orange fruits and vegetables. Red guava (Psidium guajava), a tropical Brazilian fruit, contains lycopene as the primary carotenoid (83 ± 7 μg/g) (Rodriguez-Amaya et al. 2008). Tomatoes are a primary source of lycopene (80–90% total carotenoids) (Vitale et al. 2010). Other sources of this carotenoid include watermelon, red pepper, and papaya. The presence of many conjugated diene bonds makes lycopene a natural carotenoid with a powerful capacity to absorb oxygen and act as an antioxidant agent. Lycopene extract from red guavas decreased the viability of human breast adenocarcinoma cells and had significant effects on normal cells (Dos Santos et al. 2018). In addition, at physiological concentrations, lycopene can prevent prostate cancer mutagenesis and the growth of human cancer cells without evidence of toxic effects or cellular apoptosis (Scolastici et al. 2007; Blum et al. 2005).
Capsanthin is a natural lipophilic carotenoid with a red color. This pigment can represent up to 50% of the total carotenoids of fruits and vegetables during the ripening stage, such as the skin of red bell peppers (Capsicum annuum Group) cultivated in Brazil. Zoccali et al. (2021) and Lekala et al. (2019) reported the presence of capsanthin in pepper (Capsicum annuum) and red sweet pepper cultivars, respectively. Despite not having provitamin A activity, capsanthin is classified as a functional compound due to its high antioxidant activity, which is greater than that of β-carotene (Gómez-García and Ochoa-Alejo 2013).
Polyphenols
Polyphenols are a class of bioactive compounds that include flavonoids, tannins, and lignans. Figure 4 lists the chemical structures of some polyphenols found in species of Brazilian flora. The chemical structures were obtained from PubChem (2024) at the National Institutes of Health (NIH).
Chemical structures of several common plant polyphenols: a quercetin, b myricetin, c lignan, and d tannin. Pubchem. (2024)
Flavonoids represent the largest class of natural polyphenols and act differently in the human body (Povh et al. 2012). They are found in seeds, fruits, flowers, vegetables, tree bark, roots, stalks, and their derived products, such as wines and teas.
Povh et al. (2012) detected high levels of flavonoids in four Cerrado species of the genus Hyptis (Hyptis marrubioides Epling, H. microphylla Pohl ex Benth, H. lantanifolia Poit, and H. suaveolens Poit), indicating possible antioxidant potential. Silva et al. (2016) observed a high content of flavonoids (85.71 ± 0.30 mg/100 g) in Amazon hibiscus (Fig. 5).

Source: Silva et al. (2016)
(A) Hibiscus rosa-sinensis L. (B) Hibiscus syriacus L.' Totus Albus'.
Flavonols and flavones are the most common flavonoids in tropical, subtropical, and temperate vegetables. Most flavonols in some Brazilian fruits were quercetin in acerola (4.1 to 5.3 mg/100 g) and apple (3.7 to 7.5 mg/100 g) and myricetin in cashew nuts (2.0 mg/100 g). Only quercetin flavonol was found in fig, guava, jabuticaba, and orange (Hoffmann-Ribani et al. 2009).
Anthocyanins are flavonoids present as pigments in dark-colored fruits. Red araçá (Psidium cattleianum Sabine), a member of Myrtaceae from southern Brazil, had a greater concentration of anthocyanins (36.12 ± 5.56) than did yellow Araçá (10.69 ± 9.49) and pear Araçá (10.41 ± 1.66). The concentrations are in mg equivalents of cyanidin-3-glycoside/100 g fresh samples. Red araçá exhibited a high content of phenolic compounds (668.63 ± 41.32 mg/100 g) with great antioxidant activity. Thus, such pigments can capture free reactive oxygen and free radical species, exerting antioxidant activity and preventing disease (Verma et al. 2013).
Lignans are products of the conversion of lignin into phenolic compounds, which are metabolized in the human intestine. They exhibit antimitotic, antifungal, antioxidant, and anticarcinogenic properties (Brzezinski and Debi 1999). Lignans are found mainly in cereals, fruits, and vegetables. Brown linseed is cultivated in Brazil and has antioxidant properties, probably from phenolic compounds such as lignans. According to Kinniry et al. (2006), the antioxidant activity of flaxseed lignans inhibits lipid peroxidation and the production of reactive oxygen species by white blood cells.
Tannins are polyphenols found primarily in green fruits and Leguminosae family plants. These compounds are classified according to their molecular structure. Condensed tannins are mostly found in trees, shrubs, and forage plants (Barry and Mcnabb 1999).
In addition to carotenoids and polyphenols, many Brazilian flora substances have demonstrated human health benefits. Studies have shown that it is possible to apply these substances in the prevention and treatment of diseases such as Alzheimer's disease, leishmaniasis, rheumatoid arthritis, depression, diabetes, cardiovascular diseases, metabolic syndrome, viruses, and even cancer, as shown in Table 2.
Techniques for extracting carotenoids and tocopherols from oils and biomasses of Brazilian flora species
Selecting suitable extraction methods is crucial for accurately purifying bioactive compounds from the flora of different biomes, adding value to their production chain and making subsequent use easier in other procedures. Most separation methods are based on the extractive power of solvents, which must be carefully selected considering the specificity of the metabolite to be extracted (Azmir et al. 2013; Belwal et al. 2018). Similarly, the process yield, final product quality, and cost of operations influence the choice of method. Conventional extraction methods, such as Soxhlet extraction (Krumreich et al. 2018), maceration (Santana and Macedo 2019), and hydrodistillation (Moura et al. 2016), are still suggested for biocompound separation (Azmir et al. 2013).
Although traditional extraction methods are simple and accessible, some disadvantages can affect the quality of the extracted product. The toxicity of solvents, high temperatures, and prolonged extraction time can degrade some bioactive compounds and even lead to the loss of volatile substances. In addition, the difficulty of separating certain solvents from the final product often leaves unwanted residues in the extracts (Mejri et al. 2018). In this scenario, alternative extraction methods have become attractive because of their more economical and sustainable characteristics, the possibility of extracting bioactive compounds in less time, and a greater yield than conventional methods (Barba et al. 2016). Examples of alternative methods include supercritical fluid extraction (Goyeneche et al. 2020), microwave-assisted extraction (Ruiz-Aceituno et al. 2016), and pulsed electric field methods (Plazzotta et al., 2020). The present review addresses both conventional and alternative methods of bioactive extraction.
Soxhlet extraction
The Soxhlet extraction method is widely used to obtain lipids from solids and essential oils. The Soxhlet cartridge containing the ground sample was placed inside the Soxhlet equipment. After coming into contact with the sample, the extraction solvent penetrates the plant tissue; the oil-soluble plant constituents are transferred from the sample to the solvent. The solvent contacts the sample successively due to siphoning. Subsequently, the heated solvent is condensed inside the balloon at the device's base. The processes are physical because the oil transferred to the solvent is recovered without any chemical reaction (De Castro and Priego-Capote 2010; Azwanida 2015).
Through a relatively simple method, this extraction method makes contact between the sample and a pure solvent possible, favoring the displacement of the chemical balance. However, the long extraction time and the large quantity of discarded solvents are obstacles to environmental adaptations (Azwanida 2015).
Krumreich et al. (2018) evaluated the quality of avocado oil (Persea americana Mill.) extracted using the Soxhlet method and cold pressing. Although carotenoids, phenolic compounds, and tocopherols were better preserved in the cold-pressing process, Soxhlet extraction with petroleum ether generated an oil with better conservation of properties and nutritional value. P. americana Mill. is found in the Cerrado.
Maceration + liquid extraction
Maceration is a straightforward method widely used in homemade preparations. The raw materials were first ground to increase their surface area, followed by the addition of solvent in a closed container where they remained at room temperature for at least three days. Occasionally, shaking this container increases the diffusion rate and allows the pure solvent to contact the solid. Next, the saturated solution was removed. After the contact time, the liquid is separated by filtration, and the residues are pressed to collect large amounts of the desired substance (Azmir et al. 2013; Azwanida 2015).
Santana and Macedo (2019) separated catechin and methylxanthine bioactive compounds from guarana (Paullinia cupana) seeds using hydroalcoholic maceration under hot, cold, and enzyme-assisted operation conditions. Guarana is an Amazon biome plant. The highest levels of catechins (80.87 g/100 g of extract) and methylxanthines (53.01 g of caffeine/100 g of extract) were detected using hot hydroalcoholic maceration at 60 °C. Furthermore, the pectinase enzyme facilitated biocompound extraction under the most studied operating conditions.
Silva et al. (2019a) determined the optimal conditions for obtaining anthocyanins from açaí (Euterpe oleracea), a prevalent plant in the Amazon region. The authors evaluated the effects of the maceration time and the ethanol (92% purity) and acetic acid volumes on the total solid contents and anthocyanin extraction indices. The volume of acetic acid was the most influential factor. Higher fractions of acetic acid increased the amount of anthocyanin extracted. On the other hand, ethanol volume and maceration time had opposite effects. Under the optimized conditions, the anthocyanin content of the dry extract was 61.75 mg/L.
Ferioli et al. (2020) analyzed the effects of maceration and high-pressure extraction in obtaining organosulfur compounds from garlic (Allium sativum L.) present in Brazilian biomes. The targeted substances were more concentrated in bulbil extracts than in plant stems. However, the type of separated compound varied according to the extraction method; maceration favored obtaining lipophilic compounds and high-pressure extraction of hydrophilic compounds.
Hydrodistillation
Hydrodistillation is another conventional extraction method in which an aqueous mixture containing the raw material is evaporated to separate essential oils from aqueous vapor (steam). Steam is condensed and transferred to a decanter to separate the essential oils and the water solvent. Hydrodistillation can occur in three different ways: by immersion of the raw material in water, by direct injection of steam, or by simultaneous use of these options. The extraction time may vary according to the material extracted (Aziz et al. 2018; Rassem et al. 2016).
Oliveira et al. (2017) analyzed the effects of hydrodistillation time on the yield and oil composition of Gabiroba do Campo (Campomanesia adamantium) leaves, a characteristic Cerrado plant. After one hour of hydrodistillation, 36 distinct compounds were identified. At longer time intervals, the number of compounds decreased, indicating the degradation of the molecules. However, as different sesquiterpenes, some substances were obtained in higher concentrations at longer extraction times. The authors reported stabilizing the extract composition after two hours of hydrodistillation.
Moura et al. (2016) produced leaf extracts of two species of Angicos, red (Parapiptadenia rigida) and white (Piptadenia gonoacantha), which are found in the Caatinga, Cerrado, and Atlantic Forest regions. Bioactive compounds were separated from leaf extracts using hydrodistillation and CO2 supercritical fluid extraction methods. The extract compositions of both methods presented differences. Hydrodistillation afforded compounds with lower molecular masses, such as m-cumenol, cis-pulegol, and ionones; supercritical extraction produced compounds such as 3-methyl-5-propylnonane, n-tetradecane, and n-octadecane; and benzenesulfonamide was present in the oil from both methods and provided antifungal activity.
Supercritical extraction
An extraction system reaches the supercritical state at specific temperature and pressure conditions. It presents gas properties, such as diffusivity, viscosity, and surface tension, and liquid properties, such as density and solvation power. The extractor is filled with plant parts and inert porous material. After reaching the operating temperature, the supercritical fluid is pumped at a defined flow rate under controlled pressure (Azmir et al. 2013; Mejri et al. 2018). Carbon dioxide is the solvent most commonly used in supercritical fluid extraction due to its critical temperature close to room temperature and moderate pressure (74 bars), making the process highly applicable and preventing the degradation of thermosensitive molecules. Although the low polarity of this fluid can make the extraction process difficult in some cases, this problem can be solved through small additions of solvents, such as ethanol and methanol, called cosolvents (Azmir et al. 2013).
Goyeneche et al. (2020) obtained beetroot (Beta vulgaris L.) extracts rich in polyphenols using CO2 supercritical fluid and ethanol as a cosolvent. The best conditions for polyphenol extraction occurred at 35 °C and 400 bar. The antioxidant capacities were 1454.0 µg TE (Trolox equivalent)/g of dry matter and 3370.8 µg of total phenolics/g of dry matter. An increase in polyphenol content was associated with increased antioxidant capacity. Beetroot is found in diverse Brazilian biomes. Cardenas-Toro et al. (2014) compared the performance of supercritical fluid extraction and subcritical hydrolysis for oil separation from pressed palm fiber (Opuntia ficus-indica), a typical Caatinga plant. The supercritical fluid method at 45 °C and 15 MPa led to an oil fraction rich in carotenoids with 0.81 mg of β-carotene/g of extract.
On the other hand, subcritical hydrolysis produced extracts with a high content of fermentable sugars. Benito-Román et al. (2019) used CO2 supercritical fluid as a solvent and ethanol as a cosolvent to separate oil from rice bran (Oryza sativa), a plant of the Pampa biome. The quality of the oil obtained with the CO2 solvent was superior to that obtained using the Soxhlet method concerning the antioxidant activity, fatty acid profile, and bioactive compound composition. However, the bioactive extraction yield was lower than that of the Soxhlet technique. The best conditions for extracting flavonoids, tocopherols, and γ-oryzanol phenolic compounds were 40 MPa, 40 °C, and 5 to 10% ethanol.
Microwave-assisted extraction
Heat is supplied to polar compounds through ionic conduction and dipole rotation mechanisms in microwave-assisted extraction. The synergistic effect between the transfer of heat and mass accelerates the extraction, increases the extract yield, and reduces the thermal degradation of the material. First, the solutes present in the active sites of the sample are separated under increasing temperature and pressure. Then, solute diffusion occurs across the matrix, and finally, the solute is released into the solvent (Barba et al. 2016; Zhang et al. 2018).
Pongmalai et al. (2015) evaluated the separation yield of bioactive compounds from cabbage (Brassica oleracea var. capitata L.) using the Soxhlet extraction method, assisted by microwaves and ultrasound, and simultaneously assisted by microwaves and ultrasound. Soxhlet extraction assisted by microwaves significantly reduced the extraction time and the amount of solvent and showed the highest energy efficiency compared to the other methods. Combining microwaves and ultrasound techniques led to greater damage to the cabbage leaves. The Soxhlet method, without assistance, extracted more bioactive compounds, even though it had a considerably lower extraction rate due to the high time needed.
Ruiz-Aceituno et al. (2016) compared microwave-assisted extraction with the pressurized liquid method to separate the bioactive carbohydrates inulin and inositol from the outer bracts of artichoke (Cynara scolymus L.) found in the Atlantic Forest. Extraction with pressurized liquid provided the highest inulin content, 185.4 mg inulin/g dry sample. However, the microwave-assisted technique promoted the collection of more inositol, reaching 11.6 mg inositol/g dry sample, which required less time to extract the bioactive compound—just 3 min. Nevertheless, the 27 min required for pressurized liquid extraction is acceptable. Backes et al. (2018) compared the efficiency of maceration, microwave, and ultrasound extraction methods in obtaining anthocyanins from fig (Ficus carica L.) pell found in the Atlantic Forest and Cerrado. The optimized conditions produced 5.78, 7.43, and 9.01 g anthocyanin/g residues using maceration, microwave, and ultrasound techniques, respectively. Although ultrasound-assisted extraction is slightly more efficient, the other methods performed similarly.
Pulsed electric field extraction and ultrasound-assisted extraction
Pulsed electric field (PEF) treatment increases the extraction yield by breaking down plant cell membranes. The starting material is placed between two electrodes and subjected to an electrical pulse of less than 1 s. Due to their dipole nature, this potential electrical causes the membrane molecules to separate according to their charge and form pores that make the structure more permeable. In addition to increasing yield and decreasing extraction time, this method favors the obtainment of heat-sensitive compounds, as occurs at temperatures close to ambient temperature (Azmir et al. 2013; Barba et al. 2016; Zhang et al. 2018).
Rajha et al. (2019) evaluated the separation of polyphenols from pomegranate peel (Punica granatum L.) from the Atlantic Forest and Caatinga using conventional extraction, extraction assisted by infrared irradiation, ultrasound, PEF, and high-voltage electrical discharge (HVED) methods. These last two methods proved to be highly efficient: PEF selectively extracted ellagic acid, reaching approximately 740 µg/g of dry matter, and HVED stimulated the selective obtainment of gallic acid, reaching values close to 345 µg/g of dry matter. Although PEF was less effective, it was more selective than HVED. Plazzotta et al. (2020) studied the recovery of bioactive compounds from peach (Prunus persica) bagasse found in the Atlantic Forest and Pampa biomes by applying PEF and conventional heating. The authors proposed a kinetic model for PEF extraction and reported a significant reduction in the PEF extraction time of only microseconds. The thermal treatment required 40 min to extract the target biocompounds. However, optimization is needed to increase the preservation of the obtained anthocyanins, flavonoids, and other compounds.
Medina-Meza and Barbosa-Cánovas (2015) used water extraction assisted by ultrasound and PEF to separate bioactive compounds from plum and grape skins (Prunus domestica var. Casselman and Vitis vinifera L.) from the Pampa region. The extraction yield was greater when using the two-assisted methods than when using water extraction at 70 °C for both fruits. However, each method extracted different compounds. Ultrasound-assisted extraction was notorious for extracting anthocyanins and flavonoids; PEF, in turn, stood out in the extraction of phenols. These techniques increased the extraction of anthocyanins and phenols from plums, and the flavonoid content increased for grapes.
Challenges and perspectives
Some species of Brazilian flora have potential unexplored novel bioactive compounds because they are known or used only by specific human groups and are underused in many other regions, as is the case for unconventional food plants (UFPs). Bezerra and Brito (2020) described UFP species with high levels of flavonoids, phenolic compounds, and tocopherols that could be studied and have their benefits disseminated in the scientific world. Among the many plants mentioned by the authors, Spondias purpurea (Jocote) and Opuntia ficus indica (Palm) are found in Caatinga, and Eugenia stipitate (Araçá-boi) is found in the Cerrado due to its high flavonoid content.
Gonçalves et al. (2019) evaluated the levels of carotenoids, phenolic compounds, flavonoids, and anthocyanins in the edible flowers of five plant species (Fig. 6) in the Atlantic Forest and Cerrado biomes. Purple cauliflower (Brassica oleracea var. botrytis) and green cauliflower (Brassica oleracea var. italica) are conventional food plants. The UPPs include yellow-ipê (Tabebuia serratifolia), pansy Rosalyn (Viola x wittrockiana), nasturtium (red and orange, Tropaeolum quinquelobum), and Ora-pro-nobis (Pereskia grandifolia) flowers. The yellow-ipê flowers stood out for their total carotenoid content (1,443.3 µg/100 g fresh sample), and the pansy and orange–yellow flowers exhibited higher levels of phenolic compounds (2.9 and 3.2 g gallic acid equivalents/100 g fresh sample), flavonoids (294.2 and 106.4 mg/100 g fresh sample) and anthocyanins (45.1 and 58.9 mg/100 g fresh sample).

Source: Gonçalves et al. (2019)
(a) Purple cauliflower flowers, (b) green romance cauliflower, (c) pansy, (d) red and orange nasturtium, (e) yellow ipe, and (f) ora-pro-nobis.
The diversity of the types of fruits and vegetables and the type and levels of their bioactive compounds are unexplored when considering the great promise of Brazilian flora (Brasil 2016). Several fruits from Brazilian biomes are slightly diffuse and disseminated, and their potential for application in the food and medicine sectors is regionalized. In the Cerrado biome, fruits such as Marolo (Annona crassiflora), hog plum (Spondias mombin), Cagaita (Eugenia dysenterica), Moriche palm (Mauritia flexuosa), and Araçá (Psidium Cattleianum Sabine) fruits are well known. Nevertheless, few studies have shown their potential in food and medicine.
Schiassi et al. (2018) characterized some Brazilian Cerrado fruits concerning their bioactive compounds and antioxidant capacity, as determined by DPPH (2,2-diphenyl-1-picryl-hydrazine) values. The highest DPPH value of 1,310.23 g fresh weight/g DPPH was observed for the hog plum (Spondias mombin). The ascorbic acid content of the hog plum was 42.96 mg/100 g fresh weight, which was greater than that of fruits such as oranges, which are known as a vitamin C source. Marolo (Annona crassiflora), also known as Panan or Araticum, presented a high content of total phenolics (728.17 mg GAEs (gallic acid equivalents)/100 g fresh weight). Nascimento et al. (2020) chemically characterized Araticum (Annona crassiflora), moriche palm (Mauritia flexuosa), jelly palm (Butia capitata), Cagaita (Eugenia dysenterica), and hog plum (Spondias mombin) from the Cerrado. The antioxidant potential and micromineral profile of the pulp, such as calcium and magnesium levels, were analyzed. All analyzed extracts of the fruits showed great potential for mineral nutrients and bioactive compounds. However, araticum and jelly palm had the highest percentages of phenolic compounds, with values of 433.80 and 173.5 mg GAE (gallic acid equivalent)/g, respectively.
Biazotto et al. (2019) highlighted the lack of research on the bioactive compounds of some Brazilian species. Fruits such as jatobá (Hymenae coubaril), Brazilian grapetree (Plinia cauliflora), Cambuci (Campomanesia courbaril), and Araçá (Psidium cattleianum) are excellent sources of carotenoids and phenolic compounds that could gain prominence in national consumption. Araçá was also reported by Denardin et al. (2015) to be a little-explored variety that contains 660.19 mg GAE/100 g fresh weight of total phenolic compounds and has a carotenoid content of 6.27 µg β-carotene/g fresh weight.
The high nutritional levels of bioactives do not favor society in some Brazilian biomes. For example, people frequently associate carotenoid sources with carrots containing 572.7 mg of carotenoid/L fresh juice (Stinco et al. 2019) and not with native Atlantic Forest species, such as Uvaia (Eugenia pyriformis), because some cultivars contain 4.41 mg of carotenoids/g fresh weight (Silva et al. 2019b), which is 55 times greater than the carrot value. The same is true for vitamin C; citrus fruits such as orange and lemon are commonly considered primary sources of vitamin C, containing 58.30 mg/100 g fresh sample and 43.96 mg/100 g fresh sample, respectively (Fatin and Azrina 2017). Camu-camu (Myrciaria dubia), a member of the Brazilian fruit flora of the Amazon region, produces 1,882 to 4,752 mg of vitamin C/100 g fresh weight, depending on its origin (Neri-Numa et al. 2018).
Waste generated from agro-industrial processes can disturb environmental equilibrium if it is incorrectly disposed of or handled. Moreover, if these residues contain high-value nutrients, their discarding will represent a loss of biomass with valuable nutrients. Therefore, waste reuse is an alternative for obtaining nutraceutical compounds. Shirahigue and Ceccato-Antonini (2020) evaluated the potential of agro-industrial residues as natural antioxidant sources and their applications in the food and fermentation industries. The authors highlighted the identification of baking bioactive compounds from acerola bagasse (Malpighia emarginata), such as quercetin, p-coumaric acid, gallic acid, epigallocatechin gallate, catechin, syringic acid, and epicatechin. Different uses of agro-industrial residues from the seeds and peels of mango (Mangifera indica) and avocado (Persea americana Mill) have been suggested. They are rich in phenolic compounds and can be applied to inhibit microbial activity. Therefore, Brazilian native biological diversity is neglected regarding nonnative species, causing their devaluation or loss of species. Thus, the discovery of food ingredients and drugs would contribute to species preservation by revealing their nutritional potential and the possibility of their use on scientific and commercial scales.
Concluding remarks
The Brazilian flora is a rich source of bioactive compounds, such as flavonoids and carotenoids, which have not been explored for their potential to produce differentiated food ingredients, cosmetics, pharmaceuticals, and medicines to prevent and/or treat various diseases. It should be emphasized that most species of the Brazilian flora have not yet been evaluated for their technical and economic capabilities due to their enormous biodiversity. The processing and application of Brazilian flora can boost the development of diverse industries. However, methods for plant processing should also consider the nature of the raw material, the process yield, the quality of the final product, and the operating costs to guarantee that the technological impact of using such flora should be accurately evaluated to prevent adverse impacts on environmental biomes. Several actions for species preservation can be intensified if society claims to use original biodiversity resources to safeguard natural biomes in an environmentally friendly way. Species preservation is essential for keeping our planet's ecosystems in equilibrium.
References
Agra MF, Freitas PF, Barbosa-Filho JM (2007) Synopsis of the plants known as medicinal and poisonous in northeast of Brazil. Brazilian J Pharmacogn 17:114–140. https://doi.org/10.1590/S0102-695X2007000100021
Alho CJR, Mamede SB, Benites M, Andrade BS, Sepúlveda JJO (2019) Threats to the biodiversity of the Brazilian Pantanal due to land use and occupation. Environ Soc 22:1–22. https://doi.org/10.1590/1809-4422asoc201701891vu2019L3AO
Alves JSF et al (2020) In vivo antidepressant effect of Passiflora edulis f. flavicarpa into cationic nanoparticles: Improving bioactivity and safety. Pharmaceutics 12:383. https://doi.org/10.3390/pharmaceutics12040383
Antonelli, A. et al. (2020). State of the World's Plants and Fungi 2020. Royal Botanic Gardens, Kew. K. Royal Botanic Gardens (Ed.) Royal Botanic Gardens, Kew. https://doi.org/10.34885/172
Anunciação PC, Giuffrida D, Murador DC, De Paula Filho GX, DugoPinheiro-Sant’ana GHM (2019) Identification and quantification of the native carotenoid composition in fruits from the Brazilian Amazon by HPLC–DAD–APCI/MS. J Food Compos Anal 83:1–5. https://doi.org/10.1016/j.jfca.2019.103296
Apgaua DMG, Dos Santos RM, Pereira DGS, De Oliveira Menino GC, Pires GG, Fontes MAL, Tng DYP (2014) Beta diversity in seasonally dry tropical forests (SDTF) in the caatinga biogeographic domain, Brazil, and its implications for conservation. Biodivers Conserv 23:217–232. https://doi.org/10.1007/s10531-013-0599-9
Aziz ZAA et al (2018) Essential oils: extraction techniques, pharmaceutical and therapeutic potential—A review. Curr Drug Metab 19:1100–1110. https://doi.org/10.2174/1389200219666180723144850
Azmir J et al (2013) Techniques for extraction of bioactive compounds from plant materials: a review. J Food Eng 117:426–436. https://doi.org/10.1016/j.jfoodeng.2013.01.014
Azwanida NN (2015) A review on the extraction methods use in medicinal plants, principle, strength and limitation. Med & Aromat Plants 4:3–8. https://doi.org/10.4172/2167-0412.1000196
Backes E et al (2018) Recovery of bioactive anthocyanin pigments from Ficus carica Peel by heat, microwave, and ultrasound-based extraction techniques. Food Res Int 113:197–209. https://doi.org/10.1016/j.foodres.2018.07.016
Barba FJ et al (2016) Green alternative methods for the extraction of antioxidant bioactive compounds from winery wastes and by-products: a review. Trends Food Sci Technol 49:96–109. https://doi.org/10.1016/j.tifs.2016.01.006
Barbosa DC et al (2020) Chemical composition and acetylcholinesterase inhibitory potential, in silico, of Myrciaria floribunda (h. West ex willd.) O. Berg fruit peel essential oil. Ind Crops Prod 151:1–10. https://doi.org/10.1016/j.indcrop.2020.112372
Barbosa-Pereira L et al (2014) Brewery waste as a potential source of phenolic compounds: optimization of the extraction process and evaluation of antioxidant and antimicrobial activities. Food Chem 45:191–197. https://doi.org/10.1016/j.foodchem.2013.08.033
Barry TN, Mcnabb WC (1999) The implication of condensed tannins on the nutritive value of temperature forages fed to ruminants. Br J Nutr 81:263–272. https://doi.org/10.1017/S0007114599000501
Belwal T et al (2018) A critical analysis of extraction techniques used for botanicals: trends, priorities, industrial uses and optimization strategies. Trac—Trends in Anal Chem 100:82–102. https://doi.org/10.1016/j.trac.2017.12.018
Benito-Román O et al (2019) Valorization of rice bran: modified supercritical co2 extraction of bioactive compounds. J Ind Eng Chem 80:273–282. https://doi.org/10.1016/j.jiec.2019.08.005
Bezerra JA, De BMM (2020) Nutritional and antioxidant potential of unconventional food plants and their use in food: Review. Res Soc Dev 9:369997159. https://doi.org/10.33448/rsd-v9i9.7159
Biazotto KR et al (2019) Brazilian biodiversity fruits: discovering bioactive compounds from underexplored sources. J Agric Food Chem 7:1860–1876. https://doi.org/10.1021/acs.jafc.8b05815
Blum A, Monir M, Wirsansky I, Ben-Azir S (2005) The beneficial effects of tomatoes. Eur J Intern Med 1:402–404. https://doi.org/10.1016/j.ejim.2005.02.017
Brasil. (2016). Ministério do Meio Ambiente. Espécies nativas da flora brasileira de valor econômico atual ou potencial: Plantas para o futuro: Região centro-oeste/Ministério do Meio Ambiente. Secretaria de biodiversidade, Roberto Fontes Vieira (ed.). Julcéia Camillo (ed.). Lidio Coradin (ed.). Brasília, DF, MMA.
Brzezinski A, Debi A (1999) Phytoestrogens: The “natural” selective estrogen receptor modulators? Eur J Obstet Gynecol Reproduct Biol 85(47):51. https://doi.org/10.1016/s0301-2115(98)00281-4
Cabral FD et al (2020) Bioactivities of essential oils from different parts of spiranthera odoratissima (rutaceae). Rodriguésia 71:1–8. https://doi.org/10.1590/2175-7860202071050
Cadario R, Chandon P (2019) Viewpoint: effectiveness or consumer acceptance? Tradeoffs in selecting healthy eating nudges. Food Policy 85:1–6. https://doi.org/10.1016/j.foodpol.2019.04.002
Cardenas-Toro FP et al (2014) Integrated supercritical fluid extraction and subcritical water hydrolysis for the recovery of bioactive compounds from pressed palm fiber. J Supercrit Fluids 93:42–48. https://doi.org/10.1016/j.supflu.2014.02.009
CBD. (2020). Convention on biological diversity. Available at: https://www.cbd.int/countries/?country=br. Access on: 7 out. 2020.
Cechinel-Zanchett CC, Tenfen A, Siebert DA, Micke G, Vitali L, Cechinel-Filho V, de Souza P (2019) Bauhinia forficata link, a Brazilian medicinal plant traditionally used to treat cardiovascular disorders, exerts endothelium-dependent and independent vasorelaxation in thoracic aorta of normotensive and hypertensive rats. J Ethnopharmacol 243:112118
Ceole LF, Companhoni MVP, Lopes SMS, De Oliveira AJB, Gonçalves RAC, Filho BPD, Nakamura CV, Ueda-Nakamura T (2018) Anti-herpes activity of polysaccharide fractions from Stevia rebaudiana leaves. Nat Prod Res 34:1–111. https://doi.org/10.1080/14786419.2018.1516662
Colli GR, Vieira CR, Dianese JC (2020) Biodiversity and conservation of the Cerrado: recent advances and old challenges. Biodivers Conserv 29:1465–1475. https://doi.org/10.1007/s10531-020-01967-x
Conegundes JLM, da Silva JM, de Freitas Mendes R, Fernandes MF, Pinto NDCC, de Almeida MA, Scio E (2021) Anti-inflammatory and antinociceptive activity of Siparuna guianensis aublet, an Amazonian plant traditionally used by indigenous communities. J Ethnopharmacol 265:113344
Coutinho LM (2006) The biome concept. Acta Botanica Brasilica 20(1):13–23
Couto ECG, Da Silveira FL, Rocha GRA (2003) Marine biodiversity in Brazil: Estado actual del conocimiento. Gayana (concepción) 67(2):327–340
Da Silva APG et al (2019a) Chemical composition, nutritional value and bioactive compounds in six uvaia accessions. Food Chem 294:547–556. https://doi.org/10.1016/j.foodchem.2019.04.121
Da Silva HR et al (2019b) Obtaining and characterization of anthocyanins from euterpe oleracea (açaí) dry extract for nutraceutical and food preparations. Braz J Pharmacogn 29:677–685. https://doi.org/10.1016/j.bjp.2019.03.004
da Silva Menezes L, Vogel Ely C, Lucas DB, Minervini Silva GH, Boldrini II, Overbeck GE (2018) Plant species richness record in Brazilian pampa grasslands and implications. Braz J Botany 41:817–823
Dalmagro AP, Camargo A, Rodrigues ALS, Zeni ALB (2019) Involvement of PI3K/AKT/GSK-3β signaling pathway in the antidepressant-like and neuroprotective effects of Morus nigra and its major phenolic, syringic acid. Chem Biol Interact 314:1–11. https://doi.org/10.1016/j.cbi.2019.108843
De Castro MDL, Priego-Capote F (2010) Soxhlet extraction: Past and present panacea. J Chromatogr A 1217:2383–2389. https://doi.org/10.1016/j.chroma.2009.11.027
De Faveri A, De Faveri R, Broering MF, Bousfield IT, Goss MJ, Muller SP, Antin JR (2020) Effects of passion fruit peel flour (Passiflora edulis f. flavicarpa O. Deg.) in cafeteria diet-induced metabolic disorders. J Ethnopharmacol 250:1–9. https://doi.org/10.1016/j.jep.2019.112482
De Oliveira JD et al (2017) Chemical composition of essential oil extracted from leaves of Campomanesia adamantium subjected to different hydrodistillation times. Rural Sci 47:1–7. https://doi.org/10.1590/0103-8478cr20151131
De Siqueira FC et al (2019) Profile of phenolic compounds and carotenoids of Arrabidaea chica leaves and the in vitro singlet oxygen quenching capacity of their hydrophilic extract. Food Res Int 126:1–8. https://doi.org/10.1016/j.foodres.2019.108597
De Souza FG, De Araújo FF, De Paulo Farias D, Zanotto AW, Neri-Numa IA, Pastore GM (2020) Brazilian fruits of arecaceae family: an overview of some representatives with promising food, therapeutic and industrial applications. Food Res Int 138:1–11. https://doi.org/10.1016/j.foodres.2020.109690
de Morais SM, Alves DR, Frota LS, Pinheiro SDOP, Silva AC, da Silva WMB (2020) Atividades antioxidantes e anticolinesterásicas do extrato das folhas de Jaramataia (Vitex gardneriana Schauer). Braz J Dev 6(5):28802–28810
de Morais Cardoso L, Martino HSD, Moreira AVB, Ribeiro SMR, Pinheiro-Sant’Ana HM (2011) Cagaita (Eugenia dysenterica DC.) of the Cerrado of Minas Gerais, Brazil: Physical and chemical characterization, carotenoids and vitamins. Food Res Int 44(7):2151–2154
Denardin CC et al (2015) Antioxidant capacity and bioactive compounds of four Brazilian native fruits. J Food Drug Anal 23:387–398. https://doi.org/10.1016/j.jfda.2015.01.006
Dos Santos RC, Ombredane AS, Souza JMT, Vasconcelos AG, Plácido A, Das GNN, Amorim A, Arcanjo DD (2018) Lycopene-rich extract from red guava (Psidium guajava l.) Displays cytotoxic effect against human breast adenocarcinoma cell line MCF-7 via an apoptotic-like pathway. Food Res Int 105:184–196. https://doi.org/10.1016/j.foodres.2017.10.045
Embrapa. (2020). Biomas. Embrapa. Available at: https://www.embrapa.br/contando-ciencia/bioma-amazonia. Access on: 9 out. 2020.
Fatin RN, Azrina A (2017) Comparison of vitamin C content in citrus fruits by titration and high-performance liquid chromatography (HPLC) methods. Int Food Res J 24:726–733
Ferioli F et al (2020) Comparison of two extraction methods (high-pressure extraction vs. Maceration) for the total and relative amount of hydrophilic and lipophilic organosulfur compounds in garlic cloves and stems. An application to the Italian ecotype “aglio rosso di sulmona.” Food Chem 312:1–11. https://doi.org/10.1016/j.foodchem.2019.126086
Fiaschi P, Pirani JR (2009) Review of plant biogeographic studies in Brazil. J Syst Evol 47:477–496. https://doi.org/10.1111/j.1759-6831.2009.00046.x
Flora e Funga do Brasil. Jardim Botânico do Rio de Janeiro. Disponível em: < http://floradobrasil.jbrj.gov.br/ >. Acesso em: 01 Jul 2024
Gasca CA, Moereira NCS, De Almeida FC, Dutra-Gomes JV, Castillo WO, Fagg CW, Silveira D (2020) Acetylcholinesterase inhibitory activity, anti-inflammatory, and neuroprotective potential of Hippeastrum psittacinum (ker gawl.) Herb (amaryllidaceae). Food Chem Toxicol 145:1–12. https://doi.org/10.1016/j.fct.2020.111703
Giaconia MA et al (2020) Overcoming restrictions of bioactive compounds biological effects in food using nanometer-sized structures. Food Hydrocolloids 107:1–12. https://doi.org/10.1016/j.foodhyd.2020.105939
Girardelo JR et al (2020) Bioactive compounds, antioxidant capacity and antitumoral activity of ethanolic extracts from fruits and seeds of eugenia involucrata DC. Food Res Int 137:1–11. https://doi.org/10.1016/j.foodres.2020.109615
Gómez-García MR, Ochoa-Alejo N (2013) Biochemistry and molecular biology of carotenoid biosynthesis in chili peppers (capsicum spp.). Int J Mol Sci 14:19025–19053. https://doi.org/10.3390/ijms140919025
Gonçalves J, Silva GCO, Carlos LA (2019) Compostos bioativos em flores comestíveis. Perspectivas Online: Biológicas & Saúde 9:11–20. https://doi.org/10.25242/886892920191719
Goyeneche R et al (2020) Recovery of bioactive compounds from beetroot leaves by supercritical CO2 extraction as a promising bioresource. J Supercrit Fluids 155:1–5. https://doi.org/10.1016/j.supflu.2019.104658
Hoffmann-Ribani R, Huber LS, Rodriguez-Amaya DB (2009) Flavonols in fresh and processed Brazilian fruits. J Food Compos Anal 22:263–268. https://doi.org/10.1016/j.jfca.2008.12.004
ICMBIO. (2020). Instituto Chico Mendes de Conservação da Biodiversidade. Available at: https://www.icmbio.gov.br/portal/unidadesdeconservacao/biomas-brasileiros/marinho. Access on: 27 out. 2020.
Instituto Brasileiro de Geografia e Estatisticas. IBGE. (2019). Biomas e sistema costeiro-marinho do brasil: compatível com a escala 1:250 000, instituto brasileiro de geografia e estatística (IBGE), isbn: 9788524045103.
Instituto Brasileiro de Geografia e Estatisticas. IBGE. (2024). IBGE educa jovens - biomas brasileiros. IBGE Educa Jovens. Available at: https://educa.IBGE.gov.br/jovens/conheca-o-brasil/territorio/18307-biomas-brasileiros.html. Access on: 01 jul. 2024.
Jiménez VM, Gruschwitz M, Schweiggert RM, Carle R, Esquivel P (2014) Identification of phenolic compounds in soursop (Annona muricata) pulp by high-performance liquid chromatography with diode array and electrospray ionization mass spectrometric detection. Food Res Int 65:42–46. https://doi.org/10.1016/j.foodres.2014.05.051
Justino AB, De Moura FRB, Franco RR, Espindola FS (2020) α-Glucosidase and non-enzymatic glycation inhibitory potential of Eugenia dysenterica fruit pulp extracts. Food Biosci 35:1–11. https://doi.org/10.1016/j.fbio.2020.100573
Kinniry P et al (2006) Dietary flaxseed supplementation ameliorates inflammation and oxidative tissue damage in experimental models of acute lung injury in mice. J Nutr 136:1545–1551. https://doi.org/10.1093/jn/136.6.1545
Krumreich FD et al (2018) Bioactive compounds and quality parameters of avocado oil obtained by different processes. Food Chem 257:376–381. https://doi.org/10.1016/j.foodchem.2018.03.048
Kuhlmann M, Ribeiro JF (2016) Fruits and frugivores of the Brazilian Cerrado: ecological and phylogenetic considerations. Acta Botanica Brasilica 30:495–507. https://doi.org/10.1590/0102-33062016abb0192
Lekala CS, Madani KSH, Phan ADT, Maboko MM, Fotouo H, Soundy P, Sultanbawa Y, Sivakumar D (2019) Cultivar-specific responses in red sweet peppers grown under shade nets and controlled-temperature plastic tunnel environment on antioxidant constituents at harvest. Food Chem 275:85–94. https://doi.org/10.1016/j.foodchem.2018.09.097
Lima TC, Matos SS, Carvalho TF, Silveira-Filho AJ, Couto LPSM, Quintans-Júnior LJ, Silva FA (2020) Evidence for the involvement of il-1β and TNF-α in anti-inflammatory effect and antioxidative stress profile of the standardized dried extract from Miconia albicans sw (Triana) Leaves (Melastomataceae). J Ethnopharmacol 259:1–11. https://doi.org/10.1016/j.jep.2020.112908
Maggini S, Pierre A, Calder PC (2018) Immune function and micronutrient requirements change over the life course. Nutrients 10:1–8. https://doi.org/10.3390/nu10101531
Marinho DG, Alviano DS, Matheus ME, Alviano CS, Fernandes PD (2011) The latex obtained from hancornia speciosa gomes possesses anti-inflammatory activity. J Ethnopharmacol 135:530–537. https://doi.org/10.1016/j.jep.2011.03.059
Medina-Meza IG, Barbosa-Cánovas GV (2015) Assisted extraction of bioactive compounds from plum and grape peels by ultrasonics and pulsed electric fields. J Food Eng 166:268–275. https://doi.org/10.1016/j.jfoodeng.2015.06.012
Mejri J et al (2018) Emerging extraction processes of essential oils: A review. Asian J Green Chem 2:246–267. https://doi.org/10.22034/AJGC.2018.61443
Meyers KJ, Mares JA, Igo RP, Truitt B, Liu Z, Millen AE, Blodi B (2014) Genetic evidence for role of carotenoids in age-related macular degeneration in the carotenoids in age-related eye disease study (careds). Invest Ophthalmol vis Sci 55:587–599. https://doi.org/10.1167/iovs.13-13216
Ministério do Meio Ambiente. MMA. (2017). Estratégia e plano de ação nacionais para a biodiversidade: 2016–2020. Brasília: Brazil. Ministério do Meio Ambiente, out.
Ministério do Meio Ambiente. MMA. (2020). Biomas. Ministério do Meio Ambiente. Available at: https://antigo.mma.gov.br/epanb/item/15012-esp%C3%A9cies-brasileiras.html
Mora C, Tittensor DP, Adl S, Simpson AGB, Worm B (2011) How many species are there on earth and in the ocean? PLoS Biol 9:E1001127. https://doi.org/10.1371/journal.pbio.1001127
Moura BS et al (2016) Evaluation of bioactive extracts of Piptadenia gonoacantha and Piptadenia rigida using supercritical CO2. Revista Fitos 10:199–212. https://doi.org/10.5935/2446-4775.20160016
Nabavi SF et al (2015) Curcumin: a natural product for diabetes and its complications. Curr Top Med Chem 15:2445–2455. https://doi.org/10.2174/1568026615666150619142519
Nachtigall AM, Stringheta PC, Fidelis PC, Nachtigall FM (2007) Determinação do teor de luteína em hortaliças. Ceppa 25:181–192
Nascimento ALAA, Brandi IV, Durães CAF, Lima JP, Soares SB, Mesquita BMAC (2020) Chemical characterization and antioxidant potential of native fruits of the Cerrado of northern Minas Gerais. Braz J Food Technol 23:e2019296. https://doi.org/10.1590/1981-6723.29619
Neri-Numa IA et al (2018) Small Brazilian wild fruits: nutrients, bioactive compounds, health-promotion properties and commercial interest. Food Res Int 103:345–360. https://doi.org/10.1016/j.foodres.2017.10.053
Nugraha AS, Haritakun R, Lambert JM, Dillon CT, Keller PA (2019) Alkaloids from the root of Indonesian annonamuricata l. Nat Prod Res 35:481–489. https://doi.org/10.1080/14786419.2019.1638380
Ombredane AS et al (2020) Nanoemulsion-based systems as a promising approach for enhancingthe antitumoral activity of pequi oil (caryocar brasilense cambess), In breastcancercells. J Drug Deliv Sci Technol 58:1–9. https://doi.org/10.1016/j.jddst.2020.101819
Pereira AC et al (2015) Hancornia speciosa Gomes (Apocynaceae) as a potential anti-diabetic drug. J Ethnopharmacol 161:30–35. https://doi.org/10.1016/j.jep.2014.11.050
Pereira IAG, Mendonça DVC, Tavares GSV, Lage DP, Ramos FF, Oliveira DA, Silva JA, Gonçalves DU (2020) Parasitological and immunological evaluation of a novel chemotherapeutic agent against visceral leishmaniasis. Parasite Immunol 42:1–8. https://doi.org/10.1111/pim.12784
Plazzotta S et al (2020) Modelling the recovery of biocompounds from peach waste assisted by pulsed electric fields or thermal treatment. J Food Eng 290:1–8. https://doi.org/10.1016/j.jfoodeng.2020.110196
Pongmalai P, Devahastin S, Chiewchan N, Soponronnarit S (2015) Enhancement of microwave-assisted extraction of bioactive compounds from cabbage outer leaves via the application of ultrasonic pretreatment. Sep Purif Technol 144:37–45. https://doi.org/10.1016/j.seppur.2015.02.010
Povh JA, Santos FB, Silva KR (2012) Total phenols and flavonoids in four species of the genus hyptis jacq. occurring in cerrado. Braz Geogr J: Geosci Humanit Res Medium 3:520–528
Pubchem. (2024). National Institutes of Health. The National Center for Biotechnology Information. United States government. Acessed on March 20, 2024. https://pubchem.ncbi.nlm.nih.gov/
Quintão NLM, Pastor MVD, De Souza Antonialli C, Da Silva GF, Rocha LW, Berté TE, Filho VC (2019) Aleurites moluccanus and its main active constituent, the flavonoid 2 ″-o–rhamnosylswertisin, in experimental model of rheumatoid arthritis. J Ethnopharmacol 235:248–254. https://doi.org/10.1016/j.jep.2019.02.014
Rajha HN et al (2019) Comparison of aqueous extraction efficiency and biological activities of polyphenols from pomegranate peels assisted by infrared, ultrasound, pulsed electric fields and high-voltage electrical discharges. Innov Food Sci Emerg Technol 58:1–9. https://doi.org/10.1016/j.ifset.2019.102212
Ramirez-Estrada K et al (2016) Elicitation, an effective strategy for the biotechnological production of bioactive high-added value compounds in plant cell factories. Molecules 21:1–4. https://doi.org/10.3390/molecules21020182
Rassem HH, Nour AH, Yunus RM (2016) Techniques for extraction of essential oils from plants: a review. Aust J Basic Appl Sci 10(16):117–127
Regginato A et al (2020) Antidiabetic and hypolipidemic potential of Campomanesia xanthocarpa seed extract obtained by supercritical CO2. Braz J Biol 81:621–631. https://doi.org/10.1590/1519-6984.227388
Reis ACC, Silva BM, De Moura HMM, Pereira GR, Brandão GC (2020) Anti-zika virus activity and chemical characterization by ultra-high performance liquid chromatography (UPLC-DAD-UV-MS) of ethanol extracts in tecoma species. BMC Complement Med Ther 20:1–15. https://doi.org/10.1186/s12906-020-03040-0
Rodriguez-Amaya DB et al (2008) Updated Brazilian database on food carotenoids: factors affecting carotenoid composition. J Food Compos Anal 21:445–463. https://doi.org/10.1016/j.jfca.2008.04.001
Ruiz-Aceituno, l. et al (2016) Extraction of bioactive carbohydrates from artichoke (Cynara scolymus l.) External bracts using microwave assisted extraction and pressurized liquid extraction. Food Chem 196:1156–1162. https://doi.org/10.1016/j.foodchem.2015.10.046
Sahebkar A (2017) Effects of quercetin supplementation on lipid profile: a systematic review and meta-analysis of randomized controlled trials. Crit Rev Food Sci Nutr 57:666–676. https://doi.org/10.1080/10408398.2014.948609
Santana ÁL, Macedo GA (2019) Effects of hydroalcoholic and enzyme-assisted extraction processes on the recovery of catechins and methylxanthines from crude and waste seeds of guarana (Paullinia cupana). Food Chem 281:222–230. https://doi.org/10.1016/j.foodchem.2018.12.091
Santocono M, Zurria M, Berrettini M, Fedeli D, Falcioni G (2007) Lutein, zeaxanthin and astaxanthin protect against DNA damage in Sk-N-Sh human neuroblastoma cells induced by reactive nitrogen species. J Photochem Photobiol, B 88:1–10. https://doi.org/10.1016/j.jphotobiol.2007.04.007
Schiassi MCEV, Souza VR, Lago AMT, Campos LG, Queiroz F (2018) Fruits from the Brazilian Cerrado region: Physico-chemical characterization, bioactive compounds, antioxidant activities, and sensory evaluation. Food Chem 245:305–311. https://doi.org/10.1016/j.foodchem.2017.10.104
Scolastici C, Lima RO, Barbisan LF, Ferreira AL, Ribeiro DA, Salvadori DM (2007) Lycopene activity against chemically induced DNA damage in Chinese hamster ovary cells. Toxicol in Vitro 21:840–845. https://doi.org/10.1016/j.tiv.2007.01.020
Sharoni Y, Linnewiel-Hermoni K, Khanin M, Salman H, Veprik A, Danilenko M, Levy J (2012) Carotenoids and apocarotenoids in cellular signaling related to cancer: a review. Mol Nutr Food Res 56:259–269. https://doi.org/10.1002/mnfr.201100311
Shirahigue LD, Ceccato-Antonini SR (2020) Agro-industrial wastes as sources of bioactive compounds for food and fermentation industries. Rural Sci 50:1–17. https://doi.org/10.1590/0103-8478cr20190857
Silva ABD, Wiest JM, Carvalho HHC (2016) Chemicals and antioxidant activity analisys in Hibiscus rosa-sinensis L. (mimo-de-venus) and Hibiscus syriacus L. (hibiscus-the-syrian). Braz J Food Technol 19:1–9. https://doi.org/10.1590/1981-6723.7415
Silva LE, Confortin C, Alberton MD, Siebert DA, Paganelli CJ (2020) Enzyme Inhibitory Potentials from Brazilian Flora. In: Swamy M (ed) Plant-derived Bioactives. Springer, Singapore, pp 383–393
Simmonds MSJ, Fang R, Wyatt L, Bell E, Allkin B, Forest F, Wynberg R, Silva M, Zhang BG, Shi Liu J, Dong Qi Y, Demissew S (2020) Biodiversity and patents: overview of plants and fungi covered by patents. Plants, People, Planet 2:546–556. https://doi.org/10.1002/ppp3.10144
Sosa-Hernández JE et al (2018) State-of-the-art extraction methodologies for bioactive compounds from algal biome to meet bio-economy challenges and opportunities. Molecules 23:1–28. https://doi.org/10.3390/molecules23112953
Souza RCZ, Zandonadi FS, Freitas DP, Tófoli LFF, Sussulini A (2019) Validation of an analytical method for the determination of the main ayahuasca active compounds and application to real ayahuasca samples from brazil. J Chromatogr B 1124:197–203. https://doi.org/10.1016/j.jchromb.2019.06.014
Stinco CM et al (2019) Effect of high-pressure processing on carotenoids profile, colour, microbial and enzymatic stability of cloudy carrot juice. Food Chem 299:1–7. https://doi.org/10.1016/j.foodchem.2019.125112
Traversa E, Ruberto G, Guagliano R, Barillà D, Piccinini P, Suzani M, Stronati M (2012) Funzionalità visiva nel prematuro alimentato con infant formulae, con e senza integrazione di luteina. Bollettino Della Società Medico Chirurgica Di Pavia 125:153–159. https://doi.org/10.6092/2039-1404.125.1045
Vechi G, Tnfen A, Capusiri ES, Gimenez A, Cechinel-Filho V (2020) antiparasitic activity of two Brazilian plants: Eugenia mattosii and Marlierea eugeniopsoides. Nat Prod Res 35:1–8. https://doi.org/10.1080/14786419.2020.1739676
Verma AK, Rajkumar V, Banerjee R, Biswas S, Das AK (2013) Guava (psidium guajava l.) Powder as an antioxidant dietary fiber in sheep meat nuggets. Asian Australas J Anim Sci 26:886–895. https://doi.org/10.5713/ajas.2012.12671
Vilas Boas GR, Stefanello Da Silveira AP, Feitosa Farinelli BC, Lima Cardoso CA, Arce E, Oesterreich SA (2018) The ethanolic extract obtained from Campomanesia pubescens (dc) O.BERG fruits exerts anxiolytic and antidepressant effects on chronic mild stress model and on anxiety models in Wistar rats: Behavioral evidence. Nutr Neurosci 23:1–11. https://doi.org/10.1080/1028415X.2018.1466513
Vitale A, Bernatene E, Pomilio A (2010) Carotenoids in chemoprevention: lycopene. Acta Bioquimica Clinica Latinoamericana 44:195–238
Warthen D, Gooden EL, Jacobson M (1969) Tumor inhibitors: Liriodenine, a cytotoxic alkaloid from Annona glabra. J Pharm Sci 58:637–638
Wittmann F (2011) Tree species composition and diversity in Brazilian freshwater floodplains. Mycorrhiza Occur Nat Restored Environ. 1:223–263
Woodside JV, Mcgrath AJ, Lyner N, Mckinley MC (2015) Carotenoids and health in older people. Maturitas 80:63–68. https://doi.org/10.1016/j.maturitas.2014.10.012
Wosnick N, Leite RD, Giareta EP, Nunes AROP, Nunes JLS, Charvet P, Monteiro-Filho ELA (2021) Evaluating conservation status and governmental efforts toward regional flagship species in Brazil. J Environ Manag 292:1–12. https://doi.org/10.1016/j.jenvman.2021.112732
Zanatta CF, Mercadante AZ (2007) Carotenoid composition from the Brazilian tropical fruit Camu–camu (Myrciaria dubia). Food Chem 101:1526–1532. https://doi.org/10.1016/j.foodchem.2006.04.004
Zhang QW, Lin LG, Ye WC (2018) Techniques for extraction and isolation of natural products: a comprehensive review. Chinese Medicine 13:1–26. https://doi.org/10.1186/s13020-018-0177-x
Zoccali M, Giuffrida D, Salafia F, Rigano F, Dugo P, Casale M, Mondello L (2021) Apocarotenoids profiling in different Capsicum species. Food Chem 334:127595. https://doi.org/10.1016/j.foodchem.2020.127595
Funding
This work was supported by Brazilian agencies, the National Council for Scientific and Technological Development (CNPq), the Coordination of Higher Education Personnel Improvement (CAPES), and the Minas Gerais State Foundation of Support to the Research (FAPEMIG).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Andrade, L.M., de Vasconcelos, R.A., Santos, A.O. et al. Flavonoids and carotenoids from Brazilian flora: food and pharmaceutical applications and their extraction features. Phytochem Rev (2024). https://doi.org/10.1007/s11101-024-09998-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11101-024-09998-w