Abstract
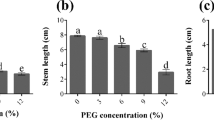
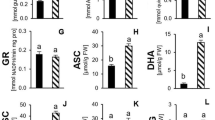
Six genotypes of taro (Colocasia esculenta L. Schott) were evaluated under in vitro and in vivo polyethylene glycol (PEG–6000)-mediated osmotic stress conditions. A significant variation in growth response was observed among the taro genotypes under in vitro-induced stress conditions. In vivo results indicated a significant effect of osmotic stress on photosynthetic parameters, such as net photosynthetic rate, transpiration rate, stomatal conductance, stomatal resistance, internal CO2 concentration, carboxylation efficiency, and transpiration efficiency on the tested genotypes at the tuberization stage. Lesser variations in photosynthesis and higher accumulation of proline, phenols, and antioxidative enzymes, namely, superoxide dismutase and guaiacol peroxidase, were associated with yield maintenance under osmotic stress conditions. The genotypes DP–89, IGCOL–4, and Ramhipur showed a higher degree of tolerance towards osmotic stress with a minimum variation in the studied parameters. These genotypes could be lines of interest for intensification of breeding strategies to develop drought-tolerant plants.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- BA:
-
6-benzyladenine
- C i :
-
intercellular CO2 concentration
- CE:
-
carboxylation efficiency
- Chl:
-
chlorophyll
- CSI:
-
chlorophyll stability index
- E :
-
transpiration rate
- EDTA:
-
ethylene diamine tetraacetic acid
- GA3 :
-
gibberellic acid
- GPX:
-
guaiacol peroxidase
- gs:
-
stomatal conductance
- NAA:
-
α–naphthalene acetic acid
- NBT:
-
nitroblue tetrazolium
- PAGE:
-
polyacrylamide gel electrophoresis
- P N :
-
photosynthetic rate
- ROS:
-
reactive oxygen species
- RS:
-
reducing sugar
- R s :
-
stomatal resistance
- RWC:
-
relative water content
- SOD:
-
superoxide dismutase
- TE:
-
transpiration efficiency
- TSS:
-
total soluble sugar
References
Albiski F., Najla S., Sanoubar R. et al.: In vitro screening of potato lines for drought tolerance.–Physiol. Mol. Biol. Plants 18: 315–321, 2012.
Anjum S.A., Xie X., Wang L. et al.: Morphological, physiological and biochemical responses of plants to drought stress.–Afr. J. Agric. Res. 6: 2026–2032, 2011.
Bates L.S., Waldren R.P., Teare I.D.: Rapid determination of free proline for water stress studies.–Plant Soil 39: 205–207, 1973.
Biradar R.S., Venkateswaralu T., Hrishi N.: Leaf area estimation in Colocasia.–J. Root Crops. 4: 51–53, 1978.
Blum A.: Drought resistance, water-use efficiency, and yield potential–are they compatible, dissonant, or mutually exclusive?–Aust. J. Agric. Res. 56: 1159–1168, 2005.
Blum A.: Effective use of water (EUW) and not water–use efficiency (WUE) is the target of crop yield improvement under drought stress.–Field Crops Res. 112: 119–123, 2009.
Bradford M.M.: A rapid and sensitive method for the quantification of microgram quantities of proteins utilizing the principle of protein–dye binding.–Anal. Biochem. 72: 248–254, 1976.
Chakraborty U., Pradhan B.: Oxidative stress in five wheat varieties (Triticum aestivum L.) exposed to water stress and study of their antioxidant enzyme defense system, water stress responsive metabolites and H2O2 accumulation.–Braz. J. Plant Physiol. 24: 117–130, 2012.
Chandra A., Pathak P.S., Bhatt R.K. et al.: Variation in drought tolerance of different Stylosanthes accessions.–Biol. Plantarum 48: 457–460, 2004.
Chaves M.M., Maroco J.P., Pereira J.S.: Understanding plant responses to drought–from genes to the whole plant.–Funct. Plant Biol. 30: 239–264, 2003
Chen K, Arora R.: Priming memory invokes seed stresstolerance.–Environ. Exp. Bot. 94: 33–45, 2013.
Chen K., Fessehaie A., Arora R.: Selection of reference genes for normalizing gene expression during seed priming and germination using qPCR in Zea mays and Spinacia oleracea.–Plant Mol. Biol. Rep. 30: 478–487, 2012.
Dasgupta M., Sahoo M.R., Kole P.C. et al.: Evaluation of orangefleshed sweet potato (Ipomoea batatas L.) genotypes for salt tolerance through shoot apex culture under in vitro NaCl mediated salinity stress conditions.–Plant Cell Tiss. Org. 94: 161, 2008.
Farooq M., Wahid A., Kobayashi N. et al.: Plant drought stress: effects, mechanisms and management.–Agron. Sustain. Dev. 29: 185–212, 2009.
Flexas J., Bota J., Escalona J.M. et al.: Effects of drought on photosynthesis in grapevines under field conditions: an evaluation of stomatal and mesophyll limitations.–Funct. Plant Biol. 29: 461–471, 2002.
Gharineh M.H., Karmollachaab A.: Effect of silicon on physiological characteristics wheat growth under water-deficit stress induced by PEG.–Int. J. Agron. Plant Prod. 4: 1543–1548, 2013.
Ghorbanli M., Gafarabad M., Amirkian T. et al.: Investigation of proline, total protein, chlorophyll, ascorbate and dehydro–ascorbate changes under drought stress in Akria and Mobil tomato cultivars.–Iran J. Plant Physiol. 3: 651–658, 2012.
Giannopolitis C.N., Ries S.K.: Superoxide dismutases. I. Occurrence in higher plants.–Plant Physiol. 59: 309–314, 1977.
Gill S.S., Tuteja N.: Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants.–Plant Physiol. Bioch. 48: 909–930, 2010.
Gomez J.M., Jimenez A., Olmos E. et al.: Location and effects of long–term NaCl stress on superoxide dismutase and ascorbate peroxidase isoenzymes of pea (Pisum sativum cv. Puget) chloroplasts.–J. Exp. Bot. 55: 119–130, 2004.
Hayat S., Hayat Q., Alyemeni M.N. et al.: Role of proline under changing environments.–Plant Signal Behav. 7: 1456–1466, 2012.
Hussain M., Malik M.A., Farooq M. et al.: Improving drought tolerance by exogenous application of glycinebetaine and salicylic acid in sunflower.–J. Agron. Crop Sci. 194: 193–199, 2008.
Khanna-Chopra R., Selote D.S.: Acclimation to drought stress generates oxidative stress tolerance in drought-resistant than -susceptible wheat cultivar under field conditions.–Environ. Exp. Bot. 60: 276–283, 2007.
Kumar R.R., Karajol K., Naik G.R.: Effect of polyethylene glycol induced water stress on physiological and biochemical responses in Pigeonpea (Cajanus cajan L. Mill sp.).–Rec. Res. Sci. Technol. 3: 148–152, 2011.
Laemmli U.K.: Cleavage of structural proteins during the assembly of the head of bacteriophage T4.–Nature 227: 680–685, 1970.
Legay S., Lefèvre I., Lamoureux D. et al.: Carbohydrate metabolism and cell protection mechanisms differentiate drought tolerance and sensitivity in advanced potato clones (Solanum tuberosum L.).–Funct. Integr. Genomic. 11: 275–291, 2011.
Levitt J.: Responses of Plants to Environmental Stresses, Vol. II. Water, Radiation, Salt and Others Stresses. Pp. 395–434. Academic Press, New York 1980.
Li X., Liu F.: Drought stress memory and drought stress tolerance in plants: biochemical and molecular basis.–In: Hossain M.A., Wani S.H., Bhattacharjee S. et al. (ed.): Drought Stress Tolerance in Plants, vol. 1. Physiology and Biochemistry. Pp. 17–44. Springer, Basel 2016.
Li X., Topbjerg H.B., Jiang D. et al.: Drought priming at vegetative stage improves the antioxidant capacity and photosynthesis performance of wheat exposed to a short-term low temperature stress at jointing stage.–Plant Soil 393: 307–318, 2015.
Liu F.L., Jensen C.R., Shahanzari A. et al.: ABA regulated stomatal control and photosynthetic water use efficiency of potato (Solanum tuberosum L.) during progressive soil drying.–Plant Sci. 168: 831–836, 2005.
Luo H.H., Zhang Y.L., Zhang W.F.: Effect of water stress and re watering on photosynthesis, root activity, and yield of cotton with drip irrigation under mulch.–Photosynthetica 54: 65–73, 2016.
Mabhaudhi T., Modi A.T., Beletse Y.G.: Response of taro (Colocasia esculenta L. Schott) landraces to varying water regimes under a rain shelter.–Agr. Water Manage. 121: 102–112, 2013.
Mabhaudhi T., Modi A.T.: Drought tolerance of selected South African taro (Colocasia esculents L. Schott) landraces.–Exp. Agr. 51: 451–466, 2015.
Mohan M.M., Narayanan S.L., Ibrahim S.M.: Chlorophyll stability index (CSI): its impact on salt tolerance in rice.–Int. Rice Res. Notes 25: 38–39, 2000.
Murashige T., Skoog F.: A revised medium for rapid growth and bioassay with tobacco tissue culture.–Physiol. Plantarum 15: 473–497, 1962.
Muscolo A., Junker A., Klukas C. et al.: Phenotypic and metabolic responses to drought and salinity of four contrasting lentil accessions.–J. Exp. Bot. 66: 5467–5480, 2015.
Nelson N.: A photometric adaptation of the Somogyi’s method for the determination of glucose.–J. Biol. Chem. 153: 375–380, 1944.
Obidiegwu J.E., Bryan G.J., Jones H.G. et al.: Coping with drought: stress and adaptive responses in potato and perspectives for improvement.–Front. Plant Sci. 6: 542, 2015.
Perez N.C.M., Espinosa R.G., Castaneda C.L. et al.: Water relations, histopathology and growth of common bean (Phaseolus vulgaris L.) during pathogenesis of Macrophomina phaseolina under drought stress.–Physiol. Mol. Plant P. 60: 185–195, 2002.
Pérez-Clemente R.M., Vives V., Zandalinas S.I. et al.: Biotechnological approaches to study plant responses to stress.–Biomed. Res. Int. 2013: 654120, 2013.
Ramírez D.A., Rolando J.L., Yactayo W. et al.: Improving potato drought tolerance through the induction of long-term water stress memory.–Plant Sci. 238: 26–32, 2015.
Ravi V., Chowdhury S.R.: Physiological response of taro to moisture stress.–J. Root Crops 23: 63–66, 1997.
Roe J.H.: The determination of sugar in blood and spinal fluid with anthrone reagent.–J. Biol. Chem. 212: 335–343, 1955.
Sahoo M.R., Dasgupta M., Kole P.C. et al.: Antioxidative enzymes and isoenzymes analysis of taro genotypes and their implications in Phytophthora blight disease resistance.–Mycopathologia 163: 241–248, 2007.
Sahoo M.R., Dasgupta M., Mukherjee A.: Effect of in vitro and in vivo induction of polyethylene glycol-mediated osmotic stress on hybrid taro (Colocasia esculenta L. Schott.).–Ann. Trop. Res. 28: 1–11, 2006b.
Sahoo M.R., Kole P.C., Dasgupta M. et al.: Screening and selection of Colocasia for leaf blight, drought and salinity.–Curr. Agr. Res. 19: 14–19, 2006a.
Singh D.P.: Water Deficit Stress in Stress Physiology. Pp. 64–79. New Age Publishers, New Delhi 2003.
Sourour A., Afef O., Mounir R. et al.: A review: morphological, physiological, biochemical and molecular plant responses to water deficit stress.–Int. J. Eng. Sci. 6: 1–4, 2017.
Sunitha S., Ravi V., George J. et al.: Aroids and water relations: An overview.–J. Root Crops 39: 10–21, 2013.
Suseela V., Tharayil N., Xing B. et al.: Warming and drought differentially influence the production and resorption of elemental and metabolic nitrogen pools in Quercus rubra.–Glob. Change Biol. 21: 4177–4195, 2015.
Tombesi S., Nardini A., Frioni T. et al.: Stomatal closure is induced by hydraulic signals and maintained by ABA in drought-stressed grapevine.–Sci. Rep. 5: 12449, 2015.
Tukey J.W.: Comparing individual means in the analysis of variance.–Biometrics 5: 99–114, 1949.
Urbanek H., Kuzniak-Gebarowska E., Herka K.: Elicitation of defense responses in bean leaves by Botrytis cinerea polygalacturonase.–Acta Physiol. Plant. 13: 43–50, 1991.
Wei L., Wang L., Yang Y. et al.: Abscisic acid enhances tolerance of wheat seedlings to drought and regulates transcript levels of genes encoding ascorbate–glutathione biosynthesis.–Front. Plant Sci. 30: 458, 2015.
Yue B., Xue W., Xiong L. et al.: Genetic basis of drought resistance at reproductive stage in rice: separation of drought tolerance from drought avoidance.–Genetics 172: 1213–1228, 2006.
Zieslin N., Ben-Zaken R.: Peroxidase activity and presence of phenolic substances in peduncles of rose flowers.–Plant Physiol. Bioch. 31: 239–247, 1993.
Author information
Authors and Affiliations
Corresponding author
Additional information
Acknowledgements: The work was supported by Indian Council of Agricultural Research, New Delhi, India. The authors are also thankful to the Director and Head, Central Tuber Crops Research Institute, Regional Centre, Bhubaneswar, India for infrastructure facilities.
Rights and permissions
About this article
Cite this article
Sahoo, M.R., Dasgupta, M., Kole, P.C. et al. Photosynthetic, physiological and biochemical events associated with polyethylene glycol-mediated osmotic stress tolerance in taro (Colocasia esculenta L. Schott). Photosynthetica 56, 1069–1080 (2018). https://doi.org/10.1007/s11099-018-0819-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11099-018-0819-3