Abstract
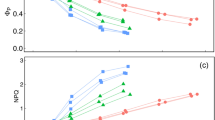
We conducted an experiment to assess the predictive capability of a leaf optical meter for determining leaf pigment status of Acer mono Maxim., A. ginnala Maxim., Quercus mongolica Fisch., and Cornus alba displaying a range of visually different leaf colors during senescence. Concentrations of chlorophyll (Chl) a, Chl b, and total Chl [i.e., Chl (a+b)] decreased while the concentration of carotenoids (Car) remained relatively static for all species as leaf development continued from maturity to senescence. C. alba exhibited the lowest average concentration of Chl (a+b), Chl a, and Car, but the highest relative anthocyanin concentration, while Q. mongolica exhibited the highest Chl (a+b), Chl b, and the lowest relative anthocyanin concentration. A. mono exhibited the highest Chl a and Car concentrations. The relationships between leaf pigments and the values measured by the optical meter generally followed an exponential function. The strongest relationships between leaf pigments and optical measurements were for A. mono, A. ginnala, and Q. mongolica (R 2 ranged from 0.64 to 0.95), and the weakest relationships were for C. alba (R 2 ranged from 0.13 to 0.67). Moreover, optical measurements were more strongly related to Chl a than to Chl b or Chl (a+b). Optical measurements were not related to Car or relative anthocyanin concentrations. We predicted that weak relationships between leaf pigments and optical measurements would occur under very low Chl concentrations or under very high anthocyanin concentrations; however, these factors could not explain the weak relationship between Chl and optical measurements observed in C. alba. Overall, our results indicated that an optical meter can accurately estimate leaf pigment concentrations during leaf senescence — a time when pigment concentrations are dynamically changing — but that the accuracy of the estimate varies across species. Future research should investigate how species-specific leaf traits may influence the accuracy of pigment estimates derived from optical meters.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- Ag:
-
Acer ginnala
- Am :
-
Acer mono
- Ant:
-
anthocyanin
- Ca :
-
Cornus alba
- CAnt :
-
relative anthocyanin concentration
- Car:
-
carotenoids
- Chl:
-
chlorophyll
- DMSO:
-
dimethylsulphoxide
- DOY:
-
day of year
- FM:
-
fresh mass
- MAT:
-
minimum air temperature
- OD:
-
optical density readings
- Q m :
-
Quercus mongolica
- R 2 :
-
coefficient of determination
- SPAD:
-
unitless value obtained with the SPAD502 optical meter
References
Anand M.H., Byju G.: Chlorophyll meter and leaf colour chart to estimate chlorophyll content, leaf colour, and yield of cassava.–Photosynthetica 46: 511–516, 2008.
Ban Y., Mitani N., Hayashi T. et al.: Exploring quantitative trait loci for anthocyanin content in interspecific hybrid grape (Vitis labruscana × Vitis vinifera).–Euphytica 198: 101–114, 2014.
Biber P.D.: Evaluating a chlorophyll content meter on three coastal wetland plant species.–J. Agr. Food Environ. Sci. 1: 1–11, 2007.
Burnham K.P., Anderson D.R.: Model Selection and Inference: A Practical Information-Theoretic Approach. Pp. 487. Springer-Verlag, New York 1998.
Cai Z.Q., Slot M., Fan Z.X.: Leaf development and photosynthetic properties of three tropical tree species with delayed greening.–Photosynthetica 43: 91–98, 2005.
Cate T.M., Perkins T.D.: Chlorophyll content monitoring in sugar maple (Acer saccharum).–Tree Physiol. 23: 1077–1079, 2003.
Chang S.X., Robison D.J.: Nondestructive and rapid estimation of hardwood foliar nitrogen status using the SPAD-502 chlorophyll meter.–Forest Ecol. Manage. 181: 331–338, 2003.
Coste S., Baraloto C., Leroy C. et al.: Assessing foliar chlorophyll contents with the SPAD-502 chlorophyll meter: a calibration test with thirteen tree species of tropical rainforest in French Guiana.–Ann. Forest Sci. 67: 607, 2010.
de Jesus S.V., Marenco R.A.: The SPAD-502 as alternative for determining chlorophyll content in fruit tree species.–Acta Amaz. 38: 815–818, 2008.
Diaz C., Saliba-Colombani V., Loudet O. et al.: Leaf yellowing and anthocyanin accumulation are two genetically independent strategies in response to nitrogen limitation in Arabidopsis thaliana.–Plant Cell Physiol. 47: 74–83, 2006.
Dwyer L.M., Tollenaar M., Houwing L.: A nondestructive method to monitor leaf greenness in corn.–Can. J. Plant Sci. 71: 505–509, 1991.
Fanizza G., della Gatta C., Bagnulo C.: A non-destructive determination of leaf chlorophyll in Vitis vinifera.–Ann. Appl. Biol. 119: 203–205, 1991.
Feild T.S., Lee D.W., Holbrook N.M.: Why leaves turn red in autumn. The role of anthocyanins in senescing leaves of redosier dogwood.–Plant Physiol. 127: 566–574, 2001.
Fu X.Y., Zhou L.Y., Huang J.B. et al.: Relating photosynthetic performance to leaf greenness in litchi: A comparison among genotypes.–Sci. Hortic.-Amsterdam 152: 16–25, 2013.
Fuleki T., Francis F.J.: Quantitative methods for anthocyanins. 1. Extraction and determination of total anthocyanin in cranberries.–J. Food Sci. 33: 72–77, 1968.
Gitelson A.A., Viña A., Verma S.B. et al.: Relationship between gross primary production and chlorophyll content in crops: Implications for the synoptic monitoring of vegetation productivity.–J. Geophys. Res. 111: 1–15, 2006.
Hawkins T.S., Gardiner E.S., Comer G.S.: Modeling the relationship between extractable chlorophyll and SPAD-502 readings for endangered plant species research.–J. Nat. Conserv. 17: 123–127, 2009.
Hlavinka J., Nauš J., Špundová M.: Anthocyanin contribution to chlorophyll meter readings and its correction.–Photosynth. Res. 118: 277–295, 2013.
Hosseinian F.S., Li W.D., Beta T.: Measurement of anthocyanins and other phytochemicals in purple wheat.–Food Chem. 109: 916–924, 2008.
Jifon J.L., Syvertsen J.P., Whaley E.: Growth environment and leaf anatomy affect nondestructive estimates of chlorophyll and nitrogen in Citrus sp. leaves.–J. Am. Soc. Hortic. Sci. 130: 152–158, 2005.
Kaakeh W., Pfeiffer D.G., Marini R.P.: Combined effects of Spirea aphid (Homoptera: Aphididae) and nitrogen fertilization on net photosynthesis, total chlorophyll content, and greenness of apple leaves.–J. Econ. Entomol. 85: 939–946, 1992.
Kenward M.G., Roger J.H.: Small sample inference for fixed effects from restricted maximum likelihood.–Biometrics 53: 983–997, 1997.
Knapp A.K., Carter G.A.: Variability in leaf optical properties among 26 species from a broad range of habitats.–Am. J. Bot. 85: 940–946, 1998.
Landi M., Tattini M., Gould K.S.: Multiple functional roles of anthocyanins in plant-environment interactions.–Environ. Exp. Bot. 119: 4–17, 2015.
Lazcano C.A., Yoo K.S., Pike L.M.: A method for measuring anthocyanins after removing carotenes in purple colored carrots.–Sci. Hortic.-Amsterdam 90: 321–324, 2001.
Lee J., Rennaker C., Wrolstad R.E.: Correlation of two anthocyanin quantification methods: HPLC and spectrophotometric methods.–Food Chem. 110: 782–786, 2008.
Ling Q.H., Huang W.H., Jarvis P.: Use of a SPAD-502 meter to measure leaf chlorophyll concentration in Arabidopsis thaliana.–Photosynth. Res. 107: 209–214, 2011.
Mancinelli A.L.: Photoregulation of anthocyanin synthesis. VIII. Effect of light pre-treatments.–Plant Physiol. 75: 447–453, 1984.
Manetas Y., Grammatikopoulos G., Kyparissis A.: The use of the portable, non-destructive, SPAD-502 (Minolta) chlorophyll meter with leaves of varying trichome density and anthocyanin content.–J. Plant Physiol. 153: 513–516, 1998.
Marenco R.A., Antezana-Vera S.A., Nascimento H.C.S.: Relationship between specific leaf area, leaf thickness, leaf water content and SPAD-502 readings in six Amazonian tree species.–Photosynthetica 47: 184–190, 2009.
Markwell J., Osterman J.C., Mitchell J.L.: Calibration of the Minolta SPAD-502 leaf chlorophyll meter.–Photosynth. Res. 46: 467–472, 1995.
Martínez D.E., Guiamet J.J.: Distortion of the SPAD 502 chlorophyll meter readings by changes in irradiance and leaf water status.–Agronomie 24: 41–46, 2004.
Mielke M.S., Schaffer B., Li C.: Use of a SPAD meter to estimate chlorophyll content in Eugenia uniflora L. leaves as affected by contrasting light environments and soil flooding.–Photosynthetica 48: 332–338, 2010.
Minocha R., Martinez G., Lyons B. et al.: Development of a standardized methodology for quantifying total chlorophyll and carotenoids from foliage of hardwood and conifer tree species.–Can. J. Forest Res. 39: 849–861, 2009.
Monje O.A., Bugbee B.: Inherent limitations of nondestructive chlorophyll meters: A comparison of two types of meters.–HortScience 27: 69–71, 1992.
Moy A., Le S., Verhoeven A.: Different strategies for photoprotection during autumn senescence in maple and oak.–Physiol. Plantarum 155: 205–216, 2015.
Murray J.R., Hackett W.P.: Dihydroflavonol reductase activity in relation to differential anthocyanin accumulation in juvenile and mature phase Hedera helix L.–Plant Physiol. 97: 343–351, 1991.
Nascimento H.C.S., Marenco R.A.: SPAD-502 readings in response to photon fluence in leaves with different chlorophyll content.–Rev. Ceres. 57: 614–620, 2010.
Neufeld H.S., Chappelka A.H., Somers G.L. et al.: Visible foliar injury caused by ozone alters the relationship between SPAD meter readings and chlorophyll concentrations in cutleaf coneflower.–Photosynth. Res. 87: 281–286, 2006.
Novak A.B., Short F.T.: Leaf reddening in the seagrass Thalassia testudinum in relation to anthocyanins, seagrass physiology and morphology, and plant protection.–Mar. Biol. 158: 1403–1416, 2011.
Pang Q.Y., Zhuo L.H.: [Spatial difference in physiological indexes of autumn-colored maple leaves.]–J. Northeast Forest. Univ. 35: 16–18, 2007. [In Chinese]
Pelletier V., Caron E., Caron J. et al.: Impact of irrigation thresholds on total anthocyanin content in cranberries.–Commun. Soil Sci. Plan. 46: 2095–2099, 2015.
Percival G.C., Keary I. P., Noviss K.: The potential of a chlorophyll content SPAD meter to quantify nutrient stress in foliar tissue of sycamore (Acer pseudoplatanus), English oak (Quercus robur), and European beech (Fagus sylvatica).–Arbor. Urban Forest. 34: 89–100, 2008.
Pinkard E.A., Patel V., Mohammed C.: Chlorophyll and nitrogen determination for plantation-grown Eucalyptus nitens and E. globulus using a non-destructive meter.–Forest Ecol. Manage. 223: 211–217, 2006.
Pirie A., Mullins M.G.: Changes in anthocyanin and phenolics content of grapevine leaf and fruit tissues treated with sucrose, nitrate, and abscisic acid.–Plant Physiol. 58: 468–472, 1976.
Reis A.R., Favarin J.L., Malavolta E. et al.: Photosynthesis, chlorophylls, and SPAD readings in coffee leaves in relation to nitrogen supply.–Commun. Soil Sci. Plan. 40: 1512–1528, 2009.
Richardson A.D., Duigan S.P., Berlyn G.P.: An evaluation of noninvasive methods to estimate foliar chlorophyll content.–New Phytol. 153: 185–194, 2002.
Samsone I., Andersone U., Vikmane M. et al.: Nondestructive methods in plant biology: an accurate measurement of chlorophyll content by a chlorophyll meter.–Acta U. Latviensis 723: 145–154, 2007.
Schaper H., Chacko E.K.: Relation between extractable chlorophyll and portable chlorophyll meter readings in leaves of eight tropical and subtropical fruit-tree species.–J. Plant Physiol. 138: 674–677, 1991.
Shiraishi M., Yamada M., Mitani N. et al.: A rapid determination method for anthocyanin profiling in grape genetic resources.–J. Jpn. Soc. Hortic. Sci. 76: 28–35, 2007.
Silla F., González-Gil A., Gonzalez-Molina M.E. et al.: Estimation of chlorophyll in Quercus leaves using a portable chlorophyll meter: effects of species and leaf age.–Ann. Forest Sci. 67: 108, 2010.
Sudahono Byrne D.H., Rouse R.E.: Greenhouse screening of citrus rootstock for tolerance to bicarbonate-induced iron chlorosis.–HortScience 29: 113–116, 1994.
Tang Q.R., Chen D.F., Chen Y.Y. et al.: [Changes of physiology and biochemistry during leafcolor transformation in Loropetalum chinense var. rubrum.]–Sci. Silvae Sin. 42: 111–115, 2006. [In Chinese]
Torres Netto A., Campostrini E., de Oliveira J.G. et al.: Portable chlorophyll meter for the quantification of photosynthetic pigments, nitrogen and the possible use for assessment of the photochemical process in Carica papaya L.–Braz. J. Plant Physiol. 14: 203–210, 2002.
Torres Netto A., Campostrini E., de Oliveira J.G. et al.: Photosynthetic pigments, nitrogen, chlorophyll a fluorescence and SPAD-502 readings in coffee leaves.–Sci. Hortic.-Amsterdam 104: 199–209, 2005.
Uddling J., Gelang-Alfredsson J., Piikki K. et al.: Evaluating the relationship between leaf chlorophyll concentration and SPAD-502 chlorophyll meter readings.–Photosynth. Res. 91: 37–46, 2007.
van den Berg A.K., Perkins T.D.: Evaluation of a portable chlorophyll meter to estimate chlorophyll and nitrogen contents in sugar maple (Acer saccharum Marsh.) leaves.–Forest Ecol. Manage. 200: 113–117, 2004.
Wang Q.B., Chen J.J., Stamps R.H. et al.: Correlation of visual quality grading and SPAD reading of green-leaved foliage Plants.–J. Plant Nutr. 28: 1215–1225, 2005.
Wellburn A.R.: The spectral determination of chlorophylls a and b, as well as total carotenoids, using various solvents with spectrophotometers of different resolution.–J. Plant Physiol. 144: 307–313, 1994.
Wen L.Y., Chen T., Zhang M.X. et al.: Seasonal changes in anthocyanin contents and in activities of xanthophyll and ascorbate-glutathione cycles in Sabina species derived from different environments.–Acta Physiol. Plant. 32: 801–808, 2010.
Yang Y., Wang G.X., Yang L.D. et al.: Physiological responses of Kobresia pygmaea to warming in Qinghai-Tibetan Plateau permafrost region.–Acta Oecol. 39: 109–116, 2012.
Zhang K.M., Yu H.J., Shi K. et al.: Photoprotective roles of anthocyanins in Begonia semperflorens.–Plant Sci. 179: 202–208, 2010.
Acknowledgments
The authors gratefully acknowledge the support of the National Science Foundation of China (No. 31300507), Scientific Research Foundation for Returned Overseas Chinese Scholars, State Education Ministry, and National Science & Technology Pillar Program during the Twelfth Five-year Plan Period (No. 2011BAD37B0101). This work is based upon work supported by the Department of Energy under Award Number DE-EM0004391 to the University of Georgia Research Foundation. We also thank the two anonymous reviewers for their valuable comments.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Li, G., Aubrey, D. & Sun, H. Predictive capability of a leaf optical meter for determining leaf pigment status during senescence. Photosynthetica 55, 543–552 (2017). https://doi.org/10.1007/s11099-016-0678-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11099-016-0678-8