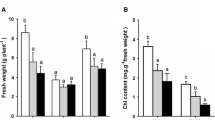
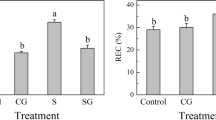
Abstract
Plants are able to acclimate to their growth light environments by utilizing a number of short- and long-term mechanisms. One strategy is to prevent accumulation of excess reactive oxygen species that can lead to photoinhibition of photosynthesis. Ureides, generated from purine degradation, have been proposed as antioxidants and involved in certain abiotic stress responses. Eutrema salsugineum (Thellungiella salsuginea) is an extremophilic plant known to exhibit a high degree of tolerance to a variety of abiotic stresses that invariably generate reactive oxygen species. In the present study we have investigated the possible role of the ureide metabolic pathway during acclimation to growth irradiance and its conference of tolerance to photoinhibition in Eutrema. Ureide accumulation was greater under high light growth which also conferred tolerance to photoinhibition at low temperature as measured by the maximal quantum yield of PSII photochemistry. This may represent an adaptive plastic response contributing to the extreme tolerance exhibited by this plant. Our results would provide evidence that ureide accumulation may be involved in abiotic stress as another defence mechanism in response to oxidative stress.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- ABA:
-
abscisic acid
- ACT7 :
-
actin7
- AAH:
-
allantoate amidohydrolase
- ALN:
-
allantoinase
- Chl:
-
chlorophyll
- FM:
-
fresh mass
- Fv/Fm :
-
maximal quantum yield of PSII photochemistry
- HL:
-
high light
- ML:
-
moderate light
- ROS:
-
reactive oxygen species
- sqPCR:
-
semiquantitative reverse transcriptase-polymerase chain reaction
- UAH:
-
ureidoglycolate amidohydrolase
- UGlyAH:
-
ureidoglycine aminohydrolase
- UO:
-
urate oxidase
- XDH:
-
xanthine dehydrogenase
References
Alamillo J.M., Díaz-Leal J.L., Sánchez-Moran M.A.V., Pineda M.: Molecular analysis of ureide accumulation under drought stress in Phaseolus vulgaris L. — Plant Cell Environ. 33: 1828–1837, 2010.
Aro E.-M., Virgin I., Andersson B.: Photoinhibition of photosystem II. Inactivation, protein damage and turnover. — Biochim. Biophys. Acta 1143: 113–134, 1993.
Amtmann A.: Learning from evolution: Thellungiella generates new knowledge on essential and critical components of abiotic stress tolerance in plants. — Mol. Plant 2: 3–12, 2009.
Anderson J.M.: Photoregulation of the composition, function, and structure of thylakoid membranes. — Annu. Rev. Plant Physio. 37: 93–136, 1986.
Anderson J.M., Chow W.S., Park Y-I.: The grand design of photosynthesis: Acclimation of the photosynthetic apparatus to environmental cues. — Photosynth. Res. 46: 129–139, 1995.
Anderson J.M., Osmond C.B.: Shade-sun responses: compromises between acclimation and photoinhibition. — In: Kyle D.J., Osmond C.B., Arntzen C.J. (ed.): Topics in Photosynthesis, Vol. 9. Photoinhibition. Pp. 1–38. Elsevier, Amsterdam 1987.
Asada K., Takahashi M.: Production and scavenging of active oxygen in photosynthesis. — In: Kyle D.J., Osmond C.B., Arntzen C.J. (ed.): Topics in Photosynthesis, Vol. 9, Photoinhibition. Pp. 227–287. Elsevier, Amsterdam 1987.
Boardman N.K.: Comparative photosynthesis of sun and shade plants. — Annu. Rev. Plant Physio. 28: 355–377, 1977.
Brychkova G., Alikulov Z., Fluhr R., Sagi M.: A critical role for ureides in dark and senescence-induced purine remobilization is unmasked in the Atxdh1 Arabidopsis mutant. — Plant J. 54: 496–509, 2008.
Castro A.H.F., Young M.C.M., de Alvarenga A.A., Alves J.D.: Influence of photoperiod on the accumulation of allantoin in comfrey plants. — R. Bras. Fisiol. Veg. 13: 49–54, 2001.
Chalker-Scott L.: Environmental significance of anthocyanins in plant stress responses. — Photochem. Photobiol. 70: 1–9,1999.
Chytyk C.J., Hucl P.J., Gray G.R.: Leaf photosynthetic properties and biomass accumulation of selected western Canadian spring wheat cultivars. — Can. J. Plant Sci. 91: 305–314, 2011.
Coleto I., Pineda M., Rodiño A.P. et al.: Comparison of inhibition of N2 fixation and ureide accumulation under water deficit in four common bean genotypes of contrasting drought tolerance. — Ann. Bot.-London 113: 1071–1082, 2014.
Demmig-Adams B., Adams W.W. III: Photoprotection and other responses of plants to high light stress. — Annu. Rev. Plant Phys. 43: 599–626, 1992.
Foyer C.H., Noctor G.: Redox regulation in photosynthetic organisms: signaling, acclimation, and practical implications. — Antioxid. Redox Sign. 11: 861–905, 2009.
Gill S.S., Tuteja N.: Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants. — Plant Physiol. Bioch. 48: 909–930, 2010.
Givnish T.J.: Adaptation to sun and shade: A whole-plant perspective. — Aust. J. Plant Physiol. 15: 63–92, 1988.
Gould K.S., Markham K.R., Smith R.H., Goris J.J.: Functional role of anthocyanins in the leaves of Quintinia serrata A. Cunn. — J. Exp. Bot. 51: 1107–1115, 2000.
Gray G.R., Hope B.J., Qin X.Q. et al.: The characterization of photoinhibition and recovery during cold acclimation in Arabidopsis thaliana using chlorophyll fluorescence imaging. — Physiol. Plantarum 119: 365–375, 2003.
Griffith M., Timonin M., Wong A.C. et al.: Thellungiella: An Arabidopsis-related model plant adapted to cold temperatures. — Plant Cell Environ. 30: 529–538, 2007.
Guevara D.R., Champigny M.J., Tattersall A. et al.: Transcriptomic and metabolomic analysis of Yukon Thellungiella plants grown in cabinets and their natural habitat show phenotypic plasticity. — BMC Plant Biol. 12: 175, 2012.
Havaux M., Kloppstech K.: The protective functions of carotenoid and flavonoid pigments against excess visible radiation at chilling temperature investigated in Arabidopsis npq and tt mutants. — Planta 213: 953–966, 2001.
Hesberg C., Hänsch R., Mendel R.R., Bittner F.: Tandem orientation of duplicated xanthine dehydrogenase genes from Arabidopsis thaliana: differential gene expression and enzyme activities. — J. Biol. Chem. 279: 13547–13554, 2004.
Inan G., Zhang Q., Li P.H. et al.: Salt cress. A halophyte and cryophyte Arabidopsis relative model system and its applicability to molecular genetic analyses of growth and development of extremophiles. — Plant Physiol. 135: 1718–1737, 2004.
Järvi S., Suorsa M., Aro E.-M.: Photosystem II repair in plant chloroplasts - regulation, assisting proteins and shared components with photosystem II biogenesis. — Biochim. Biophys. Acta 1847: 900–909, 2015.
Kansal R., Kuhar K., Verma I. et al.: Improved and convenient method of RNA isolation from polyphenols and polysaccharide rich plant tissues. — Indian J. Exp. Biol. 47: 842–845, 2008.
Kant S., Bi Y.M., Weretilnyk E. et al.: The Arabidopsis halophytic relative Thellungiella halophila tolerates nitrogenlimiting conditions by maintaining growth, nitrogen uptake, and assimilation. — Plant Physiol. 147: 1168–1180, 2008.
Karuppanapandian T., Moon J.C., Kim C. et al.: Reactive oxygen species in plants: their generation, signal transduction, and scavenging mechanisms. — Aust. J. Crop Sci. 5: 709–725, 2011.
Kazachkova Y., Batushansky A., Cisneros A. et al.: Growth platform-dependent and -independent phenotypic and metabolic responses of Arabidopsis thaliana and its halophytic relative, Eutrema salsugineum, to salt stress. — Plant Physiol. 162: 1583–1598, 2013.
King C.A., Purcell L.C.: Inhibition of N2 fixation in soybean is associated with elevated ureides and amino acids. — Plant Physiol. 137: 1389–1396, 2005.
Koch M.A., German D.A.: Taxonomy and systematics are key to biological information: Arabidopsis, Eutrema (Thellungiella), Noccaea and Schrenkiella (Brassicaceae) as examples. — Front. Plant Sci. 4: 267, 2013.
Kono M., Terashima I.: Long-term and short-term responses of the photosynthetic electron transport to fluctuating light. — J. Photoch. Photobio. B 137: 89–99, 2014.
Lichtenthaler H.K., Wellburn A.R.: Determination of total carotenoids and chlorophylls a and b of leaf extracts in different solvents. — Biochem. Soc. Trans. 11: 591–592,1983.
Murata N., Allakhverdiev S.I., Nishiyama Y.: The mechanism of photoinhibition in vivo: Re-evaluation of the roles of catalase, α-tocopherol, non-photochemical quenching, and electron transport. — BBA-Bioenergetics 1817: 1127–1133, 2012.
Nakashima K., Yamaguchi-Shinozaki K., Shinozaki K.: The transcriptional regulatory network in the drought response and its crosstalk in abiotic stress responses including drought, cold and heat. — Front. Plant Sci. 5: 170, 2014.
Neill S.O., Gould K.S.: Anthocyanins in leaves: light attenuators or antioxidants? — Funct. Plant Biol. 30: 865–873, 2003.
Oliver M.J., Guo L., Alexander D.C. et al: A sister group contrast using untargeted global metabolomic analysis delineates the biochemical regulation underlying desiccation tolerance in Sporobolus stapfianus. — Plant Cell 23: 1231–1248, 2011.
Powles S.B.: Photoinhibition of photosynthesis induced by visible light. — Annu. Rev. Plant Physio. 35: 15–44, 1984.
Reinbothe H., Mothes K.: Urea, ureides, and guanidines in plants. — Annu. Rev. Plant Phys. 13: 129–150, 1962.
Sagi M., Omarov R.T., Lips S.H.: The Mo-hydroxylases xanthine dehydrogenase and aldehyde oxidase in ryegrass as affected by nitrogen and salinity. — Plant Sci. 135: 125–135, 1998.
Sambrook J., Russell D.W.: Molecular Cloning: A Laboratory Manual. 3rd Edition. Pp. 2100. Cold Spring Harbor Laboratory Press, New York 2001.
Schneider C.A., Rasband W.S., Eliceiri K.W.: NIH Image to ImageJ: 25 years of image analysis. — Nat. Methods 9: 671–675, 2012.
Schubert K.R.: Products of biological nitrogen fixation in higher plants: synthesis, transport, and metabolism. — Annu. Rev. Plant Physio. 37: 539–574, 1986.
Serraj R., Sinclair T.R., Purcell L.C.: Symbiotic N2 fixation response to drought. — J. Exp. Bot. 50: 143–155, 1999.
Stasolla C., Katahira R., Thorpe T.A., Ashihara H.: Purine and pyrimidine nucleotide metabolism in higher plants. — J. Plant Physiol. 160: 1271–1295, 2003.
Stepien P., Johnson G.N.: Contrasting responses of photosynthesis to salt stress in the glycophyte Arabidopsis and halophyte Thellungiella: Role of the plastid terminal oxidase as an alternative electron sink. — Plant Physiol. 149: 1154–1165, 2009.
Sui N., Han G.: Salt-induced photoinhibition of PSII is alleviated in halophyte Thellungiella halophila by increases of unsaturated fatty acids in membrane lipids. — Acta Physiol. Plant. 36: 983–992, 2014.
Suzuki N., Koussevitzky S., Mittler R., Miller G.: ROS and redox signalling in the response of plants to abiotic stress. — Plant Cell Environ. 35: 259–270, 2012.
Takahashi S., Badger M.R.: Photoprotection in plants: a new light on photosystem II damage. — Trends Plant Sci. 16: 53–60, 2011.
Takahashi S., Murata N.: How do environmental stresses accelerate photoinhibition? — Trends Plant Sci. 13: 178–182, 2008.
Todd C.D., Tipton P.A., Blevins D.G. et al.: Update on ureide degradation in legumes. — J. Exp. Bot. 57: 5–12, 2006.
Ventura Y., Wuddineh W.A., Myrzabayeva M. et al.: Effect of seawater concentration on the productivity and nutritional value of annual Salicornia and perennial Sarcocornia halophytes as leafy vegetable crops. — Sci. Hortic.-Amsterdam 128: 189–196, 2011.
Vogels G.D., Van Der Drift C.: Differential analyses of glyoxylate derivatives. — Anal. Biochem. 33: 143–157, 1970.
Walters R.G.: Towards an understanding of photosynthetic acclimation. — J. Exp. Bot. 56: 435–447, 2005.
Wang P., Kong C.H, Sun B., Xu X.H.: Distribution and function of allantoin (5-ureidohydantoin) in rice grains. — J. Agric. Food Chem. 60: 2793–2798, 2012.
Watanabe S., Matsumoto M., Hakomori Y. et al.: The purine metabolite allantoin enhances abiotic stress tolerance through synergistic activation of abscisic acid metabolism. — Plant Cell Environ. 37: 1022–1036, 2014.
Watanabe S., Nakagawa A., Izumi S. et al.: RNA interferencemediated suppression of xanthine dehydrogenase reveals the role of purine metabolism in drought tolerance in Arabidopsis. — FEBS Lett. 584: 1181–1186, 2010.
Werner A.K., Witte C.P.: The biochemistry of nitrogen mobilization: purine ring catabolism. — Trends Plant Sci. 16: 381–387, 2011.
Wong C.E., Li Y., Labbe A. et al.: Transcriptional profiling implicates novel interactions between abiotic stress and hormonal responses in Thellungiella, a close relative of Arabidopsis. — Plant Physiol. 140: 1437–1450, 2006.
Ye J., Coulouris G., Zaretskaya I. et al.: Primer-BLAST: A tool to design target-specific primers for polymerase chain reaction. — BMC Bioinformatics 13: 134, 2012.
Yobi A., Wone B.W., Xu W. et al.: Metabolomic profiling in Selaginella lepidophylla at various hydration states provides new insights into the mechanistic basis of desiccation tolerance. — Mol. Plant 6: 369–385, 2013.
Zrenner R., Stitt M., Sonnewald U., Boldt R.: Pyrimidine and purine biosynthesis and degradation in plants. — Annu. Rev. Plant Biol. 57: 805–836, 2006
Author information
Authors and Affiliations
Corresponding author
Additional information
Acknowledgments: This project was supported from Discovery funding provided by the Natural Sciences and Engineering Research Council (NSERC) of Canada to G.R. Gray and C.D. Todd, and in part, by an Undergraduate Research Grant, Office of the Vice-President Research, University of Saskatchewan to V.M. Malik. We thank Eva-Maria Aro (University of Turku) for useful discussion.
These authors contributed equally to this work.
Rights and permissions
About this article
Cite this article
Malik, V.M., Lobo, J.M., Stewart, C. et al. Growth irradiance affects ureide accumulation and tolerance to photoinhibition in Eutrema salsugineum (Thellungiella salsuginea). Photosynthetica 54, 93–100 (2016). https://doi.org/10.1007/s11099-015-0164-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11099-015-0164-8